Chinese Journal of Tissue Engineering Research ›› 2019, Vol. 23 ›› Issue (1): 110-117.doi: 10.3969/j.issn.2095-4344.1531
Previous Articles Next Articles
Physical stimulation methods promote myocardial differentiation and maturation of stem cells
Zhang Zeqian1, 2, Wu Jiaqi1, 2, Fan Yubo1, 2, Zheng Lisha1, 2
- 1School of Biological Science and Medical Engineering, Beihang University, Beijing 100083, China; 2Beijing Advanced Innovation Center for Biomedical Engineering, Beihang University, Beijing 100083, China
-
Revised:2018-09-22Online:2019-01-08Published:2018-11-28 -
Contact:Zheng Lisha, PhD, Associate professor, Master’s supervisor, School of Biological Science and Medical Engineering, Beihang University, Beijing 100083, China; Beijing Advanced Innovation Center for Biomedical Engineering, Beihang University, Beijing 100083, China -
About author:Zhang Zeqian, School of Biological Science and Medical Engineering, Beihang University, Beijing 100083, China; Beijing Advanced Innovation Center for Biomedical Engineering, Beihang University, Beijing 100083, China. Wu Jiaqi, School of Biological Science and Medical Engineering, Beihang University, Beijing 100083, China; Beijing Advanced Innovation Center for Biomedical Engineering, Beihang University, Beijing 100083, China. Zhang Zeqian and Wu Jiaqi contributed equally to this work. -
Supported by:the National Key Research and Development Program of the Ministry of Science and Technology of China, No. 2017YFC0108505 (to ZLS); the National Natural Science Foundation of China, No. 11572030 (to ZLS), 11120101001 (to FYB), 11421202 (to FYB), 11827803 (to FYB); the Fundamental Research Funds for the Central Universities (111 Plan), No. B13003 (to ZLS)
CLC Number:
Cite this article
Zhang Zeqian, Wu Jiaqi, Fan Yubo, Zheng Lisha. Physical stimulation methods promote myocardial differentiation and maturation of stem cells[J]. Chinese Journal of Tissue Engineering Research, 2019, 23(1): 110-117.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
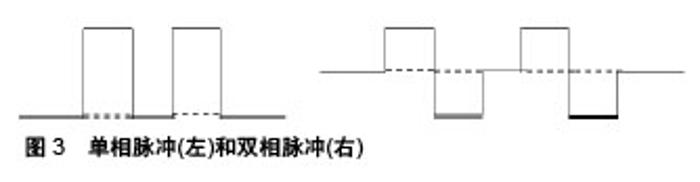
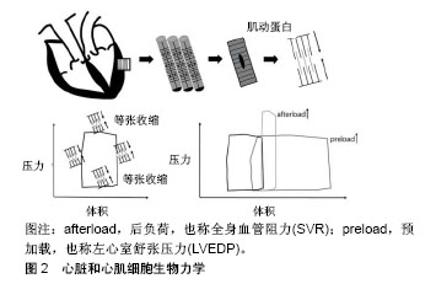
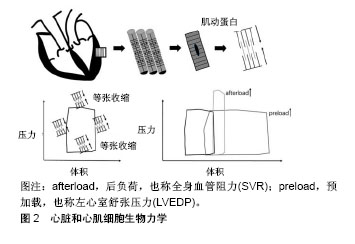
Frank-Starling机制表明,对于天然心肌,其收缩力的增加与舒张期心肌纤维的拉伸程度呈正相关[18]。此外,细胞伸展调节了细胞间隙连接蛋白的取向,这促进了细胞之间的连接,有利于细胞通讯[19]。在正常发育和衰老过程中,心脏必须持续运输足够的血流量,以维持人正常的生理功能。因此随着心脏的发育成熟,血液流动会提供更大的驱动力,使得心肌细胞具有更好的收缩能力和完善的生理功能。Hove等[20]使用斑马鱼量化胚胎发育过程中心脏的血流动力学。定量分析表明,受精后37 h,心脏内皮细胞受到的剪切应力为0.25 Pa;受精后4.5 d,剪切应力增加为7.6 Pa。研究证明,力学刺激对心脏发育成熟起重要作用。因此,对心肌细胞施加力学刺激,是促进干细胞心肌向分化成熟的重要方法。 2.1.2 机械刺激促进细胞心肌向成熟 在提高心肌细胞成熟度的相关研究中,研究人员主要通过对细胞施加牵张力或流动剪切应力,来模拟心脏搏动时加载力和卸载力的生理学原理。目前已经研究了两种不同的牵张力加载模式:静态拉伸和循环拉伸。 静态拉伸是使用静态支架进行稳定的牵张力加载。一般使用聚二甲基硅氧烷等材料,制作各种形状的弹性支柱等成型体,以施加牵张力[21]。Ruan等[22]使用4%的静态拉伸力,拉伸心肌组织2周后,工程心肌的收缩性增加[由(0.055±0.009) mN/mm2增加至(0.63±0.10) mN/mm2],表明其接近天然心肌的基本特征。Leonard等[23]使用4种不同的负荷:K1=0.09 μN/μm,K2=0.45 μN/μm,K3=1.2 μN/μm,K4=9.2 μN/μm,施加在干细胞分化的心肌细胞上,结果表明,增加血管阻力有利于成熟心脏基因的表达。 循环拉伸需要根据工程组织的内源跳动频率来调整拉伸周期的长度。循环机械拉伸可以改善心脏贴片在收缩性、细胞排列、心脏基因表达和内皮细胞网络形成等方面的心血管特性[24]。Zhang等[25]研究表明,与静态拉伸和未拉伸对照组相比,复杂的循环拉伸使工程化心脏组织在收缩力、弹性模量和肌节长度等功能参数方面具有较高的成熟度。Mihic等[26]使用12%的牵张力,1.25 Hz的频率(75次循环/min)进行周期性拉伸,培养72 h后,细胞内与心脏成熟相关的基因以及编码心脏离子通道的基因mRNA表达增强。Qi等[27]研究发现,2 h的单轴循环拉伸(20%,1 Hz)促进细胞TRPV4的表达,进而引起了细胞内Ca2+浓度上升,促进细胞的重新排列。在心脏的发育过程中,机械拉伸是心肌细胞生长和成熟的主要影响因素之一。这种刺激有助于确保心脏的尺寸和性能,可以适应越来越多的血液流动所引起的额外的血流动力学作用。 流体剪切应力,指流体沿表面移动产生的摩擦力。目前关于流体剪切应力如何影响干细胞心肌向分化成熟的信息有限,且相关研究多集中于小鼠的胚胎干细胞。单层培养的小鼠胚胎干细胞在流动剪切应力下(1 Pa)进行30 min至8 h不同时间的培养[28]。培养1 h后,中胚层细胞的心肌细胞标志物,包括血管内皮生长因子受体2(VEGFR-2)和肌细胞增强因子2C(MEF2C)的表达增加,表明在适当条件下,流体剪切应力可以促进胚胎干细胞的心肌向分化成熟。 牵张力和流动剪切应力是比较常见的机械应变形式,近期有研究表明,磁性力介导的应变也可以诱导干细胞的心肌向分化成熟。Geuss等[29]使用永久性钕磁体吸引类胚体内的细胞。在细胞聚集过程中,精氨酸-甘氨酸-天冬氨酸(RGD)共轭磁珠与类胚体细胞结合,使用不同的磁场强度刺激类胚体1 h。结果表明,在0.2 T的磁场中培养1 h后,蛋白激酶A和α-肌动蛋白的表达量提高,心肌细胞收缩性能增加,心肌细胞占百分比加倍。这项实验设计为今后的磁性力诱导干细胞心肌向分化的深入研究奠定了基础。 2.2 电刺激 心肌细胞之间通过闰盘结构形成间隙连接,有利于细胞间的电兴奋传导并根据电信号在节律上同步收缩,因此有功能合胞体之称[30]。在心脏发育过程中直流电信号主要指导细胞的增殖迁移和形态变化,而脉冲信号则涉及心脏合胞体的形成[31]。Radisic等[32]首先在体外应用脉冲电刺激来增强心肌细胞分化和工程化组织的形成,仅8 d,受刺激的构建体在超微结构水平表现出类似于天然心肌的显著分化,收缩幅度比未刺激组增加了7倍。但是体外培养的干细胞源心肌细胞表型类似于胚胎心肌,其电生理学特征与成体心肌细胞相比显示出更大的异质性和不成熟性[33]。为了促进干细胞心肌向分化,在应用脉冲电刺激来模拟心肌发育电生理信号的过程中,调节所施加电场刺激的参数例如脉冲方向、电场强度与持续时间、起始作用时间、频率是至关重要的。 2.2.1 脉冲方向 根据电流方向是否改变可分为单相脉冲和双相脉冲,在单相脉冲中电流方向不发生改变,在双相脉冲中电流会反向,单个波形周期内正负脉冲相伴出现[34],见图3。"
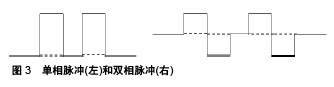
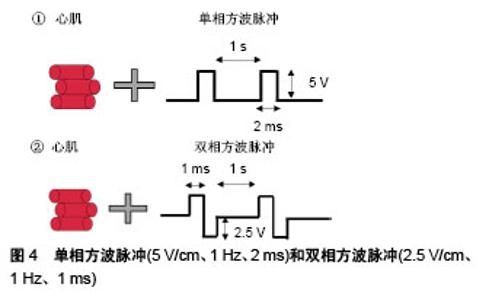
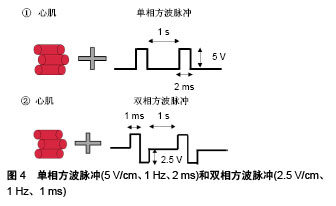
单相脉冲波形简单,能有效地刺激心肌细胞产生动作电位,而在双相脉冲中次级脉冲超级化可能会干扰动作电位的启动。Baumgartner等[35]对胎鼠心肌细胞连续6 d施加振幅1 V、脉宽5 ms、频率10 Hz的单相脉冲,电刺激促进了细胞形态伸长和平行排列以及连接蛋白43的表达。但是连续的单相脉冲会引起电极直流极化进而产生电化学副作用,容易对细胞造成损伤并腐蚀电极,使用双相脉冲则可以避免这些不利影响。Chan等[36]用电场强度6.6 V/cm、脉宽2 ms、频率1 Hz的双相脉冲刺激人胚胎干细胞源心肌细胞,培养4 d后发现细胞心脏特异性基因表达和分化率增加,钙处理得到改善。为了比较单相和双相电场刺激对体外培养心肌细胞结构和功能的影响,Chiu等[37]对新生大鼠心肌细胞分别施加电场强度2.5 V/cm、频率1 Hz、脉宽1 ms的双相方波脉冲以及具有相同总幅度和持续时间的单相方波脉冲(5 V/cm、1 Hz、2 ms),见图4。他们发现双相脉冲刺激组有更高的细胞密度、同步收缩率和更高表达水平的Cx-43,双相脉冲更有效地促进了心肌成熟。Pietronave等[38]用同样的单相和双相电刺激作用于人心脏祖细胞,得到的结论与Chiu一致,双相电刺激的效果更好并且随着刺激时间的增加而增强。此外,与单相脉冲相比,双相脉冲可改善除颤功效,因此双相电场刺激可能对工程心肌组织的构建更有利。"
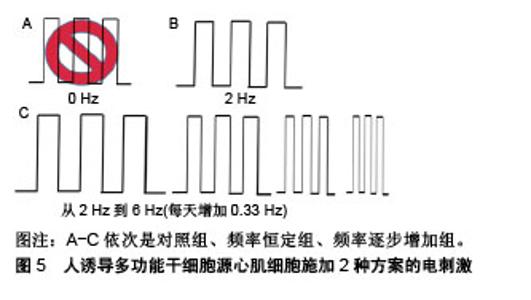
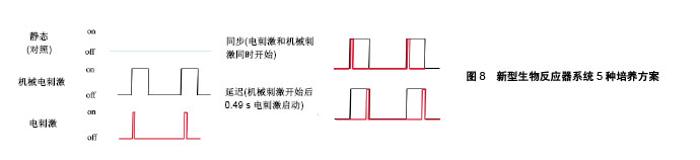
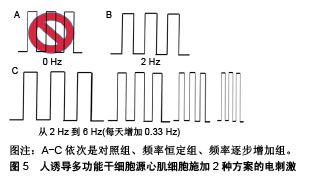
2.2.2 电场强度与持续时间 目前终止心律失常的唯一有效疗法是向心脏组织提供高强度电场(HEF),但它可能会对心脏细胞造成伤害。对于相同的强度,当脉冲持续时间较长时,细胞凋亡率更高[39]。因此在体外对细胞施加电刺激时,协调其电场强度和持续时间是十分重要的。当所用电场强度低时需要增加作用时间以获得更好的刺激效果。用低强度电刺激(25 mV/cm、频率1 Hz、脉宽2 ms)的单相方波脉冲,对人心肌祖细胞分别作用7 d和14 d。相比于7 d组,所研究的所有心脏标志物在刺激14 d后均增加其表达[40]。为了探究短暂电刺激对人诱导多功能干细胞心肌向分化的影响,Hernandez等[41]向拟胚体施加强度为65 mV/mm和200 mV/mm、频率1 Hz、脉宽1 ms电荷平衡双相脉冲,并分别持续1.5,5,10,15 min。结果发现5 min,200 mV/mm组使心肌分化效率提高了2倍,心脏特异性基因表达显著增加。Sauer等[42]对4 d龄小鼠胚胎干细胞形成的拟胚体施加强度较高的500 V/m持续90 s的直流电场,Serena等[43]对4 d龄人胚胎干细胞形成的拟胚体施加强度更高的1 V/mm持续90 s的直流电场,二者都显著促进了心肌向分化。在这4项研究中随着所用的电场强度增加,刺激时间也缩短。 2.2.3 电刺激起始作用时间 电刺激的影响很大程度上取决于其施加时间。如果施加时间太早,电刺激会抑制心肌蛋白的聚集并产生不良的收缩行为。如果施加时间太晚,电刺激不再有助于细胞的功能组装[31]。Serena等[43]分别对4 d龄和8 d龄人胚胎干细胞形成的拟胚体施加电刺激,结果表明4 d龄拟胚体具有更高的心肌分化率。Ronaldson-Bouchard等[44]对体外培养的人多功能干细胞源心肌细胞早期(第一次自发收缩后,第12天)和晚期(第28天)施加单相方波脉冲,结果发现早期人多功能干细胞源心肌细胞显示出明显的可塑性。这2项研究都说明电刺激作用对早期发育阶段的干细胞源心肌细胞更有效。为了探究局部电刺激对不同分化阶段的胚胎干细胞的影响,Chen等[45]分别对4 d龄、7 d龄和12 d龄的拟胚体施加电流强度为10,30,60 μA、频率1 Hz、脉宽10 ms的双相电刺激培养4 d,分别对应于干细胞分化的早期、中期、末期3个阶段。作者选择β-MHC和肌钙蛋白T作为检测的心肌标志物。低强度电刺激对早期分化阶段的干细胞作用不大,但在60 μA电刺激下,其β-MHC水平显著增加。对于中期分化阶段的干细胞而言,β-MHC和肌钙蛋白T在30 μA刺激时表达增加,而在60 μA时表达降低。在末期阶段,分化成心肌细胞的干细胞数目与刺激幅度成正比。这说明不同分化阶段的胚胎干细胞对电刺激有不同程度的敏感性,因此根据施加电刺激的时间来调整其参数是十分重要的。 2.2.4 频率 刺激频率可以影响心肌细胞的收缩和搏动行为。频率为1.0-2.0 Hz的电刺激可以诱导细胞内Ca2+水平瞬时激增,同时促进肌节组装和成熟,而较高频率(10 Hz)未能在肌管中引发这种效应[46]。Eng等[47]发现电刺激可以促进干细胞源心肌细胞的成熟并改变其内在搏动特性。当移除对人干细胞源心肌细胞的电刺激后,其自发搏动频率会调整到与此前施加的电刺激频率(1 Hz和2 Hz)一致。这种适应频率的搏动行为可以持续2周,此特性还可以转移到周围的心肌细胞。Tandon等[48]对新生大鼠心肌细胞的组织工程心脏构建体施加3 V/cm、脉宽2 ms、频率分别为1,3,5 Hz的单相方波脉冲。研究表明3 Hz组有最高浓度的心肌肌钙蛋白和连接蛋白43以及最好的收缩行为。Hirt等[49]设计了2种电刺激方案:频率0.5 Hz和第1周2 Hz随后1.5 Hz,场强2 V/cm,脉宽4 ms的双相脉冲,分别作用于人诱导多功能干细胞源心肌细胞,第2种方案对促进心肌成熟的效果更好。Ronaldson-Bouchard等[44]也对人诱导多功能干细胞源心肌细胞施加了2种方案的电刺激,4.5 V/cm、2 ms单相方波脉冲:①2 Hz刺激3周;②2周内频率从2 Hz到6 Hz(每天增加0.33 Hz)然后2 Hz培养1周,如图5所示,同样是第2种方案刺激效果更好。"
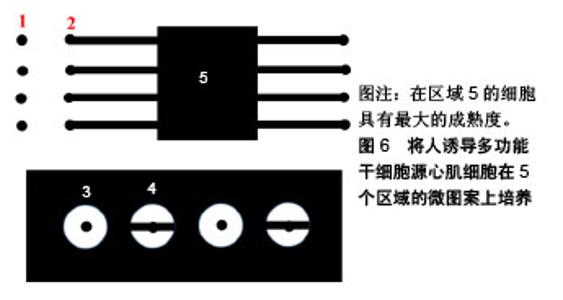
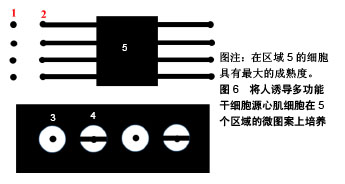
Nunes等[50-51]对人类多功能干细胞源心肌细胞施加7 d的电刺激,电场强度为5 V/cm、脉宽1 ms、频率从1 Hz开始逐步增加到3 Hz或6 Hz。他们发现与1-3 Hz的低频增加方式相比,1周内1-6 Hz的起搏频率可进一步增强工程心肌组织的结构和电生理功能。 2.3 基质 干细胞生长的物理微环境对细胞形状、大小、排列方向、分化方向有重要影响。在体内,通过细胞-细胞和细胞-细胞外基质相互作用产生于细胞表面的力学刺激会转换为细胞内信号进而引起表型变化[52]。心肌组织细胞骨架的成熟具有力依赖性,肌原纤维的结构成熟和收缩性能与细胞外基质有关[53]。因此有必要研究培养基底硬度和表面形貌对干细胞心肌向分化的影响。 2.3.1 基质硬度 在心肌细胞发育过程中其生长环境呈动态变化,例如从胚胎发育开始一直到出生后几周内心肌组织的胶原不断积累,导致心肌组织弹性模量增加:从小鼠胚胎到新生阶段增加3倍,从新生到成体增加2倍[54]。2006年Adam Engler及其合作者发现不同硬度的基底可以诱导间充质干细胞的谱系分化[55]。从此,许多研究纷纷展开,基质刚度在调节细胞行为包括参与组织和器官发育、组织修复和疾病进展等方面有显著作用。为了研究底物硬度对心肌分化的影响,Jacot等[56]在刚度分别为1,5,10,25,50 kPa的聚丙烯酰胺凝胶上接种人胚胎干细胞源心肌细胞。在10 kPa底物上的细胞产生最大的钙瞬变,呈现出清晰、有序排列的肌节,而更坚硬的底物如50 kPa组会抑制肌原纤维的形成。Bajaj等[57]研究了底物硬度对心肌跳动的影响,他们将小鸡胚胎的心肌细胞接种在具有不同刚度(1,18,50 kPa)的聚丙烯酰胺凝胶基质上。培养5 d后,心肌在18 kPa底物上搏动最快,但之后所有基质上细胞的跳动频率都收敛到60-80次/min,与刚度无关,此为细胞固有的自然搏动频率。此外,Hazeltine等[58]发现,当在柔性基底上培养时,人多能干细胞源心肌细胞的收缩力随着基质硬度增加而增加。McCain等[59]发现收缩最佳的肌细胞的形状受基质刚度的调节,产生最佳收缩的心肌细胞在中等硬度(13 kPa)凝胶中具有中等长宽比(约为7∶1),然而在更坚硬(90 kPa)的凝胶上呈现出低纵横比(约为2∶1)。Hazeltine等[60]探究了基质刚度对心肌分化的时间效应,他们发现在中胚层诱导早期,人胚胎干细胞心肌向分化对基质刚度的刺激最为敏感,因此正确应用底物机制可以增加干细胞分化为心肌细胞的成功率。 2.3.2 基底表面形貌 成人心肌是各向异性、互相平行排列的,有许多研究通过构建空间图案来模拟心肌生长微环境的微尺度拓扑形貌[61]。常用的技术有拓扑结构表面图案化和化学表面图案化[62]。拓扑结构表面图案化旨在改变基底的表面纹理,如通过微加工技术制作有序排列的突起或凹陷即沟槽结构,可以调节细胞骨架的定向排列和细胞形状。例如,有研究将大鼠心肌细胞接种在宽度450 nm、深度为100 nm或350 nm的聚氨酯凹槽上,心肌细胞的取向和伸长随着凹槽深度的增加而增加[63]。这可能是由于深度增加时,深宽比也增加,细胞的丝状伪足不能渗入沟槽并感应到沟槽的底部而被迫锚定在脊上,导致细胞沿凹槽和脊的方向对齐[61]。Carson等[64]研究了基质凹槽宽度对心肌成熟的影响,他们将人诱导多功能干细胞源心肌细胞接种到宽度范围350-2 000 nm的聚氨酯丙烯酸酯凹槽上,在700-1 000 nm宽度范围凹槽上的细胞有更好的排列和形态以及增加的肌节长度,并在800 nm宽度凹槽上细胞结构的成熟度达到峰值。化学图案化过程将生物分子固定在基底表面,形成尺寸和位置可控的细胞易黏附区,这些生物分子一般是某些细胞外基质蛋白如纤粘连蛋白或者结合肽。这些图案可以通过微接触印刷和光刻技术制作。Salick等[65]发现宽度在30-80 μm的矩形条带微图案显著促进了人胚胎干细胞源心肌细胞的定向排列和肌节形成,且与条带长度无关。Werley等[66]设计了边长为50,100,250,500,1 000 μm的正方形微图案,人诱导多功能干细胞源心肌细胞在较大尺寸的微图案上显示出更高的电生理学和钙动力学功能成熟度。此外,他们进一步研究了不同几何形状的微图案对人诱导多功能干细胞源心肌细胞成熟度的影响,见图6。结果表明在区域5的细胞具有最大的成熟度。"

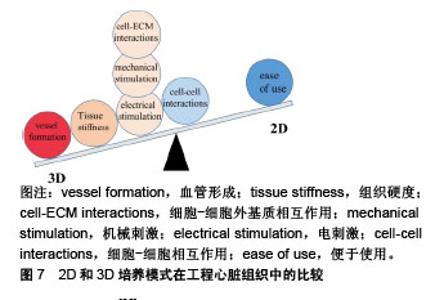
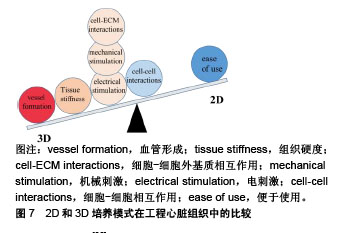
Lundy等[68]通过2D培养发现与早期阶段(体外分化和培养20-40 d)的多功能干细胞源心肌细胞相比,培养80-120 d的细胞才表现出更高级的发育成熟状态。相比之下,用3D水凝胶心脏组织贴片培养的小鼠胚胎干细胞源心肌细胞在21 d后表现出显著的结构和功能分化特征[69]。Zhang等[70]研究了2D单层和3D贴片培养对人胚胎干细胞源心肌细胞成熟的影响,他们发现3D贴片中的心肌细胞表现出更大的动作电位传导速度、更长的肌节,且参与心脏收缩功能的基因表达增强。在3D培养中,对作为基质的生物材料有一定要求,在机械性能方面它要适配心肌的力学性能:①刚性和非弹性贴片会阻碍收缩,太软达不到机械增强的效果;②应变要能够达到15%;③各向异性[71]。合成材料如聚己内酯和聚L-乳酸具有可调控的力学性能,天然材料如胶原蛋白和明胶、藻酸盐可以降低排斥反应,具有良好的生物相容性,因此可以将天然材料和合成材料合成共聚物来改善支架性能。例如,Alperin等[72]发现小鼠胚胎干细胞源心肌细胞在Ⅳ型胶原包被的聚氨酯薄膜上有更好的黏附和收缩性能。LaNasa等[73]分别将Ⅰ型胶原蛋白和层粘连蛋白共价结合到水凝胶上,它们有效地促进了心肌细胞与水凝胶的相互作用,有利于心肌细胞的成熟。由于心肌组织细胞外基质结构复杂,用生物材料构建类似的3D支架是十分具有挑战性的,因此许多研究人员将心脏脱细胞化作为心肌组织工程的理想支架,细胞将生长在与天然心肌相同的微环境中[74]。Ott等[75]在2008年制作出第一个脱细胞心脏支架,并在体外接种心肌细胞和血管内皮细胞,这些构建体能成功执行泵功能。Fong等[76]研究表明,与2D培养相比,接种在牛脱细胞心脏支架上的人诱导多功能干细胞源心肌细胞的成熟度显著提高。另外可以通过生物打印精确控制细胞和细胞外基质成分的密度和分布来构建支架模拟心肌组织结构和功能特征[67]。Gao等[77]用多光子激发的三维印刷(MPE-3DP)制作具有亚微米分辨率的天然样细胞外基质支架,并以2∶1∶1的比例接种人诱导多功能干细胞分化的心肌细胞、平滑肌细胞和内皮细胞,然后将构建体植入心肌梗死小鼠模型中进行测试,结果表明小鼠心肌损伤恢复,心脏功能明显改善。 2.5 联合刺激 为了进一步促进体外干细胞源心肌细胞的成熟,可以联合机械刺激、电刺激、细胞-细胞外基质相互作用、细胞-细胞相互作用来模拟心肌在发育过程中的自然生理条件[78],见表1。"

| [1] Benjamin EJ, Virani SS, Callaway CW, et al. Heart Disease and Stroke Statistics-2018 Update: A Report From the American Heart Association. Circulation. 2018;137(12):e67-e492.[2] Yancy CW, Jessup M, Bozkurt B, et al. 2017 ACC/AHA/HFSA Focused Update of the 2013 ACCF/AHA Guideline for the Management of Heart Failure: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines and the Heart Failure Society of America.Circulation. 2017;136(6):e137-e161.[3] Silva Dos Santos D, Brasil GV, Ramos IPR, et al. Embryonic stem cell-derived cardiomyocytes for the treatment of doxorubicin-induced cardiomyopathy.Stem Cell Res Ther. 2018;9(1):30.[4] Natsumeda M, Florea V, Rieger AC, et al. A Combination of Allogeneic Stem Cells Promotes Cardiac Regeneration.J Am Coll Cardiol. 2017; 70(20):2504-2515.[5] Ellison GM, Vicinanza C, Smith AJ, et al. Adult c-kit(pos) cardiac stem cells are necessary and sufficient for functional cardiac regeneration and repair.Cell. 2013;154(4):827-842.[6] Ben-Ari M, Naor S, Zeevi-Levin N, et al. Developmental changes in electrophysiological characteristics of human-induced pluripotent stem cell-derived cardiomyocytes.Heart Rhythm. 2016; 13(12):2379-2387.[7] Gorospe G, Zhu R, Millrod MA, et al. Automated grouping of action potentials of human embryonic stem cell-derived cardiomyocytes.IEEE Trans Biomed Eng. 2014;61(9):2389-2395.[8] Gherghiceanu M, Barad L, Novak A, et al. Cardiomyocytes derived from human embryonic and induced pluripotent stem cells: comparative ultrastructure.J Cell Mol Med. 2011;15(11):2539-2551.[9] Lieu DK, Liu J, Siu CW, et al. Absence of transverse tubules contributes to non-uniform Ca(2+) wavefronts in mouse and human embryonic stem cell-derived cardiomyocytes.Stem Cells Dev. 2009;18(10):1493-1500.[10] Kim C, Wong J, Wen J, et al. Studying arrhythmogenic right ventricular dysplasia with patient-specific iPSCs.Nature. 2013;494(7435):105-110.[11] Sheehy SP, Pasqualini F, Grosberg A, et al. Quality metrics for stem cell-derived cardiac myocytes.Stem Cell Reports. 2014;2(3):282-294.[12] Liu Z, Tang Y, Lü S, et al. The tumourigenicity of iPS cells and their differentiated derivates.J Cell Mol Med. 2013;17(6):782-791.[13] Gonzalez R, Lee JW, Schultz PG.Stepwise chemically induced cardiomyocyte specification of human embryonic stem cells.Angew Chem Int Ed Engl. 2011;50(47):11181-11185.[14] Zhao XL, Yang B, Ma LN, et al. MicroRNA-1 effectively induces differentiation of myocardial cells from mouse bone marrow mesenchymal stem cells.Artif Cells Nanomed Biotechnol. 2016;44(7):1665-1670.[15] Karakikes I, Senyei GD, Hansen J, et al. Small molecule-mediated directed differentiation of human embryonic stem cells toward ventricular cardiomyocytes.Stem Cells Transl Med. 2014;3(1):18-31.[16] Jacot JG, Martin JC, Hunt DL.Mechanobiology of cardiomyocyte development.J Biomech. 2010;43(1):93-98.[17] Bharadwaj KN, Spitz C, Shekhar A, et al. Computational fluid dynamics of developing avian outflow tract heart valves.Ann Biomed Eng. 2012; 40(10):2212-2227.[18] Tulloch NL, Muskheli V, Razumova MV, et al. Growth of engineered human myocardium with mechanical loading and vascular coculture. Circ Res. 2011;109(1):47-59.[19] Salameh A, Wustmann A, Karl S, et al. Cyclic mechanical stretch induces cardiomyocyte orientation and polarization of the gap junction protein connexin43.Circ Res. 2010;106(10):1592-1602.[20] Hove JR, Köster RW, Forouhar AS, et al. Intracardiac fluid forces are an essential epigenetic factor for embryonic cardiogenesis.Nature. 2003;421(6919):172-177.[21] Abilez OJ, Tzatzalos E, Yang H, et al. Passive Stretch Induces Structural and Functional Maturation of Engineered Heart Muscle as Predicted by Computational Modeling.Stem Cells. 2018;36(2):265-277.[22] Ruan JL, Tulloch NL, Razumova MV, et al. Mechanical Stress Conditioning and Electrical Stimulation Promote Contractility and Force Maturation of Induced Pluripotent Stem Cell-Derived Human Cardiac Tissue.Circulation. 2016;134(20):1557-1567.[23] Leonard A, Bertero A, Powers JD, et al. Afterload promotes maturation of human induced pluripotent stem cell derived cardiomyocytes in engineered heart tissues.J Mol Cell Cardiol. 2018;118:147-158.[24] Lux M, Andrée B, Horvath T, et al. In vitro maturation of large-scale cardiac patches based on a perfusable starter matrix by cyclic mechanical stimulation.Acta Biomater. 2016;30:177-187.[25] Zhang W, Kong CW, Tong MH, et al. Maturation of human embryonic stem cell-derived cardiomyocytes (hESC-CMs) in 3D collagen matrix: Effects of niche cell supplementation and mechanical stimulation.Acta Biomater. 2017;49:204-217. [26] Mihic A, Li J, Miyagi Y, et al. The effect of cyclic stretch on maturation and 3D tissue formation of human embryonic stem cell-derived cardiomyocytes.Biomaterials. 2014;35(9):2798-2808.[27] Qi Y, Li Z, Kong CW, et al. Uniaxial cyclic stretch stimulates TRPV4 to induce realignment of human embryonic stem cell-derived cardiomyocytes.J Mol Cell Cardiol. 2015;87:65-73.[28] Illi B, Scopece A, Nanni S, et al. Epigenetic histone modification and cardiovascular lineage programming in mouse embryonic stem cells exposed to laminar shear stress.Circ Res. 2005;96(5):501-508.[29] Geuss LR, Wu DC, Ramamoorthy D, et al. Paramagnetic beads and magnetically mediated strain enhance cardiomyogenesis in mouse embryoid bodies.PLoS One. 2014;9(12):e113982.[30] Thrivikraman G, Boda SK, Basu B.Unraveling the mechanistic effects of electric field stimulation towards directing stem cell fate and function: A tissue engineering perspective.Biomaterials. 2018;150:60-86.[31] Tandon N, Cannizzaro C, Chao PH, et al. Electrical stimulation systems for cardiac tissue engineering.Nat Protoc. 2009;4(2):155-173.[32] Radisic M, Park H, Shing H, et al. Functional assembly of engineered myocardium by electrical stimulation of cardiac myocytes cultured on scaffolds.Proc Natl Acad Sci U S A. 2004;101(52):18129-18134.[33] Liu J, Laksman Z, Backx PH.The electrophysiological development of cardiomyocytes.Adv Drug Deliv Rev. 2016;96:253-273.[34] Merrill DR, Bikson M, Jefferys JG.Electrical stimulation of excitable tissue: design of efficacious and safe protocols.J Neurosci Methods. 2005;141(2):171-198.[35] Baumgartner S, Halbach M, Krausgrill B, et al. Electrophysiological and morphological maturation of murine fetal cardiomyocytes during electrical stimulation in vitro.J Cardiovasc Pharmacol Ther. 2015;20(1):104-112.[36] Chan YC, Ting S, Lee YK, et al. Electrical stimulation promotes maturation of cardiomyocytes derived from human embryonic stem cells.J Cardiovasc Transl Res. 2013;6(6):989-999.[37] Chiu LL, Iyer RK, King JP, et al. Biphasic electrical field stimulation aids in tissue engineering of multicell-type cardiac organoids.Tissue Eng Part A. 2011;17(11-12):1465-1477.[38] Pietronave S, Zamperone A, Oltolina F, et al. Monophasic and biphasic electrical stimulation induces a precardiac differentiation in progenitor cells isolated from human heart.Stem Cells Dev. 2014;23(8):888-898.[39] Prado LN, Goulart JT, Zoccoler M, et al. Ventricular myocyte injury by high-intensity electric field: Effect of pulse duration.Gen Physiol Biophys. 2016;35(2):121-130.[40] Llucià-Valldeperas A, Sanchez B, Soler-Botija C, et al. Physiological conditioning by electric field stimulation promotes cardiomyogenic gene expression in human cardiomyocyte progenitor cells.Stem Cell Res Ther. 2014;5(4):93.[41] Hernández D, Millard R, Sivakumaran P, et al. Electrical Stimulation Promotes Cardiac Differentiation of Human Induced Pluripotent Stem Cells.Stem Cells Int. 2016;2016:1718041.[42] Sauer H, Rahimi G, Hescheler J, et al. Effects of electrical fields on cardiomyocyte differentiation of embryonic stem cells.J Cell Biochem. 1999;75(4):710-723.[43] Serena E, Figallo E, Tandon N, et al. Electrical stimulation of human embryonic stem cells: cardiac differentiation and the generation of reactive oxygen species.Exp Cell Res. 2009;315(20):3611-3619.[44] Ronaldson-Bouchard K, Ma SP, Yeager K, et al. Advanced maturation of human cardiac tissue grown from pluripotent stem cells.Nature. 2018;556(7700):239-243.[45] Chen MQ, Xie X, Wilson KD, et al. Current-Controlled Electrical Point-Source Stimulation of Embryonic Stem Cells.Cell Mol Bioeng. 2009;2(4):625-635.[46] Fujita H, Nedachi T, Kanzaki M.Accelerated de novo sarcomere assembly by electric pulse stimulation in C2C12 myotubes.Exp Cell Res. 2007;313(9):1853-1865.[47] Eng G Lee BW, Protas L, et al. Autonomous beating rate adaptation in human stem cell-derived cardiomyocytes.Nat Commun. 2016;7:10312.[48] Tandon N, Marsano A, Maidhof R, et al. Optimization of electrical stimulation parameters for cardiac tissue engineering.J Tissue Eng Regen Med. 2011;5(6):e115-125.[49] Hirt MN, Boeddinghaus J, Mitchell A, et al. Functional improvement and maturation of rat and human engineered heart tissue by chronic electrical stimulation.J Mol Cell Cardiol. 2014;74:151-161.[50] Nunes SS, Miklas JW, Liu J, et al. Biowire: a platform for maturation of human pluripotent stem cell-derived cardiomyocytes.Nat Methods. 2013;10(8):781-787.[51] Sun X, Nunes SS.Biowire platform for maturation of human pluripotent stem cell-derived cardiomyocytes.Methods. 2016;101:21-26.[52] Zhu R, Blazeski A, Poon E, et al. Physical developmental cues for the maturation of human pluripotent stem cell-derived cardiomyocytes. Stem Cell Res Ther. 2014;5(5):117.[53] Besser RR, Ishahak M, Mayo V, et al. Engineered Microenvironments for Maturation of Stem Cell Derived Cardiac Myocytes.Theranostics. 2018;8(1):124-140.[54] Yang X, Pabon L, Murry CE.Engineering adolescence: maturation of human pluripotent stem cell-derived cardiomyocytes.Circ Res. 2014; 114(3):511-523.[55] Engler AJ, Sen S, Sweeney HL, et al. Matrix elasticity directs stem cell lineage specification.Cell. 2006;126(4):677-689.[56] Jacot JG, Kita-Matsuo H, Wei KA, et al. Cardiac myocyte force development during differentiation and maturation.Ann N Y Acad Sci. 2010;1188:121-127.[57] Bajaj P, Tang X, Saif TA, et al. Stiffness of the substrate influences the phenotype of embryonic chicken cardiac myocytes.J Biomed Mater Res A. 2010;95(4):1261-1269.[58] Hazeltine LB, Simmons CS, Salick MR, et al. Effects of substrate mechanics on contractility of cardiomyocytes generated from human pluripotent stem cells.Int J Cell Biol. 2012;2012:508294.[59] McCain ML, Yuan H, Pasqualini FS, et al. Matrix elasticity regulates the optimal cardiac myocyte shape for contractility.Am J Physiol Heart Circ Physiol. 2014;306(11):H1525-1539.[60] Hazeltine LB, Badur MG, Lian X, et al. Temporal impact of substrate mechanics on differentiation of human embryonic stem cells to cardiomyocytes.Acta Biomater. 2014;10(2):604-612.[61] Wang PY, Thissen H, Kingshott P.Modulation of human multipotent and pluripotent stem cells using surface nanotopographies and surface- immobilised bioactive signals: A review.Acta Biomater. 2016;45:31-59.[62] Wanjare M, Huang NF.Regulation of the microenvironment for cardiac tissue engineering.Regen Med. 2017;12(2):187-201.[63] Wang PY, Yu J, Lin JH, et al. Modulation of alignment, elongation and contraction of cardiomyocytes through a combination of nanotopography and rigidity of substrates.Acta Biomater. 2011;7(9):3285-3293.[64] Carson D, Hnilova M, Yang X, et al. Nanotopography-Induced Structural Anisotropy and Sarcomere Development in Human Cardiomyocytes Derived from Induced Pluripotent Stem Cells.ACS Appl Mater Interfaces. 2016;8(34):21923-21932.[65] Salick MR, Napiwocki BN, Sha J, et al. Micropattern width dependent sarcomere development in human ESC-derived cardiomyocytes. Biomaterials. 2014;35(15):4454-4464.[66] Werley CA, Chien MP, Gaublomme J, et al. Geometry-dependent functional changes in iPSC-derived cardiomyocytes probed by functional imaging and RNA sequencing.PLoS One. 2017;12(3):e0172671.[67] Borovjagin AV, Ogle BM, Berry JL, et al. From Microscale Devices to 3D Printing: Advances in Fabrication of 3D Cardiovascular Tissues. Circ Res. 2017;120(1):150-165.[68] Lundy SD, Zhu WZ, Regnier M, et al. Structural and functional maturation of cardiomyocytes derived from human pluripotent stem cells.Stem Cells Dev. 2013;22(14):1991-2002.[69] Liau B, Christoforou N, Leong KW, et al. Pluripotent stem cell-derived cardiac tissue patch with advanced structure and function.Biomaterials. 2011;32(35):9180-8187.[70] Zhang D, Shadrin IY, Lam J, et al. Tissue-engineered cardiac patch for advanced functional maturation of human ESC-derived cardiomyocytes. Biomaterials. 2013;34(23):5813-5820.[71] Huyer LD, Montgomery M, Zhao Y, et al. Biomaterial based cardiac tissue engineering and its applications.Biomed Mater. 2015;10(3):034004.[72] Alperin C, Zandstra PW, Woodhouse KA.Polyurethane films seeded with embryonic stem cell-derived cardiomyocytes for use in cardiac tissue engineering applications.Biomaterials. 2005;26(35):7377-7386.[73] LaNasa SM, Bryant SJ.Influence of ECM proteins and their analogs on cells cultured on 2-D hydrogels for cardiac muscle tissue engineering. Acta Biomater. 2009;5(8):2929-2938.[74] Reis LA, Chiu LL, Feric N, et al. Biomaterials in myocardial tissue engineering.J Tissue Eng Regen Med. 2016;10(1):11-28.[75] Ott HC, Matthiesen TS, Goh SK, et al. Perfusion-decellularized matrix: using nature's platform to engineer a bioartificial heart.Nat Med. 2008; 14(2):213-221.[76] Fong AH, Romero-López M, Heylman CM, et al. Three-Dimensional Adult Cardiac Extracellular Matrix Promotes Maturation of Human Induced Pluripotent Stem Cell-Derived Cardiomyocytes.Tissue Eng Part A. 2016;22(15-16):1016-1025.[77] Gao L, Kupfer ME, Jung JP, et al. Myocardial Tissue Engineering With Cells Derived From Human-Induced Pluripotent Stem Cells and a Native-Like, High-Resolution, 3-Dimensionally Printed Scaffold.Circ Res. 2017;120(8):1318-1325.[78] Scuderi GJ, Butcher J.Naturally Engineered Maturation of Cardiomyocytes. Front Cell Dev Biol. 2017;5:50.[79] Nguyen MD, Tinney JP, Ye F, et al. Effects of physiologic mechanical stimulation on embryonic chick cardiomyocytes using a microfluidic cardiac cell culture model.Anal Chem. 2015;87(4):2107-2113.[80] Maidhof R, Tandon N, Lee EJ, et al. Biomimetic perfusion and electrical stimulation applied in concert improved the assembly of engineered cardiac tissue.J Tissue Eng Regen Med. 2012;6(10):e12-23.[81] Thavandiran N, Dubois N, Mikryukov A, et al. Design and formulation of functional pluripotent stem cell-derived cardiac microtissues.Proc Natl Acad Sci U S A. 2013;110(49):E4698-4707.[82] Shachar M, Benishti N, Cohen S.Effects of mechanical stimulation induced by compression and medium perfusion on cardiac tissue engineering.Biotechnol Prog. 2012;28(6):1551-1559.[83] Morgan KY, Black LD 3rd.Mimicking isovolumic contraction with combined electromechanical stimulation improves the development of engineered cardiac constructs.Tissue Eng Part A. 2014;20(11-12):1654-1667.[84] Smith AS, Macadangdang J, Leung W, et al. Human iPSC-derived cardiomyocytes and tissue engineering strategies for disease modeling and drug screening.Biotechnol Adv. 2017;35(1):77-94.[85] Korolj A, Wang EY, Civitarese RA, et al. Biophysical stimulation for in vitro engineering of functional cardiac tissues.Clin Sci (Lond). 2017; 131(13):1393-1404.[86] Moretti A, Bellin M, Welling A, et al. Patient-specific induced pluripotent stem-cell models for long-QT syndrome.N Engl J Med. 2010;363(15): 1397-1409. |
| [1] | Pu Rui, Chen Ziyang, Yuan Lingyan. Characteristics and effects of exosomes from different cell sources in cardioprotection [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-. |
| [2] | Lin Qingfan, Xie Yixin, Chen Wanqing, Ye Zhenzhong, Chen Youfang. Human placenta-derived mesenchymal stem cell conditioned medium can upregulate BeWo cell viability and zonula occludens expression under hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 4970-4975. |
| [3] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [4] | Zhang Xiumei, Zhai Yunkai, Zhao Jie, Zhao Meng. Research hotspots of organoid models in recent 10 years: a search in domestic and foreign databases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1249-1255. |
| [5] | Hou Jingying, Yu Menglei, Guo Tianzhu, Long Huibao, Wu Hao. Hypoxia preconditioning promotes bone marrow mesenchymal stem cells survival and vascularization through the activation of HIF-1α/MALAT1/VEGFA pathway [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 985-990. |
| [6] | Shi Yangyang, Qin Yingfei, Wu Fuling, He Xiao, Zhang Xuejing. Pretreatment of placental mesenchymal stem cells to prevent bronchiolitis in mice [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 991-995. |
| [7] | Liang Xueqi, Guo Lijiao, Chen Hejie, Wu Jie, Sun Yaqi, Xing Zhikun, Zou Hailiang, Chen Xueling, Wu Xiangwei. Alveolar echinococcosis protoscolices inhibits the differentiation of bone marrow mesenchymal stem cells into fibroblasts [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 996-1001. |
| [8] | Fan Quanbao, Luo Huina, Wang Bingyun, Chen Shengfeng, Cui Lianxu, Jiang Wenkang, Zhao Mingming, Wang Jingjing, Luo Dongzhang, Chen Zhisheng, Bai Yinshan, Liu Canying, Zhang Hui. Biological characteristics of canine adipose-derived mesenchymal stem cells cultured in hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1002-1007. |
| [9] | Geng Yao, Yin Zhiliang, Li Xingping, Xiao Dongqin, Hou Weiguang. Role of hsa-miRNA-223-3p in regulating osteogenic differentiation of human bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1008-1013. |
| [10] | Lun Zhigang, Jin Jing, Wang Tianyan, Li Aimin. Effect of peroxiredoxin 6 on proliferation and differentiation of bone marrow mesenchymal stem cells into neural lineage in vitro [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1014-1018. |
| [11] | Zhu Xuefen, Huang Cheng, Ding Jian, Dai Yongping, Liu Yuanbing, Le Lixiang, Wang Liangliang, Yang Jiandong. Mechanism of bone marrow mesenchymal stem cells differentiation into functional neurons induced by glial cell line derived neurotrophic factor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1019-1025. |
| [12] | Duan Liyun, Cao Xiaocang. Human placenta mesenchymal stem cells-derived extracellular vesicles regulate collagen deposition in intestinal mucosa of mice with colitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1026-1031. |
| [13] | Pei Lili, Sun Guicai, Wang Di. Salvianolic acid B inhibits oxidative damage of bone marrow mesenchymal stem cells and promotes differentiation into cardiomyocytes [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1032-1036. |
| [14] | Guan Qian, Luan Zuo, Ye Dou, Yang Yinxiang, Wang Zhaoyan, Wang Qian, Yao Ruiqin. Morphological changes in human oligodendrocyte progenitor cells during passage [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1045-1049. |
| [15] | Wang Zhengdong, Huang Na, Chen Jingxian, Zheng Zuobing, Hu Xinyu, Li Mei, Su Xiao, Su Xuesen, Yan Nan. Inhibitory effects of sodium butyrate on microglial activation and expression of inflammatory factors induced by fluorosis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1075-1080. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||