Chinese Journal of Tissue Engineering Research ›› 2016, Vol. 20 ›› Issue (49): 7437-7422.doi: 10.3969/j.issn.2095-4344.2016.49.021
Previous Articles Next Articles
Intra-patient variability of tacrolimus concentration in transplant recipients: a prognostic predictor post transplantation
Li Ling1, Li Ning1, Ai Zi-ye1, Yao Ya-jun1, Wei Wan-hui1, He Wei-yang1, Wang Yan-feng1, Ye Qi-fa1, 2
- 1Zhongnan Hospital of Wuhan University, Institute of Hepatobiliary Diseases of Wuhan University, Transplant Center of Wuhan University, Hubei Key Laboratory of Medical Technology on Transplantation, Wuhan 430071, Hubei Province, China; 2the Third Xiangya Hospital of Central South University, Research Center of National Health Ministry on Transplantation Medicine Engineering and Technology, Changsha 410013, Hunan Province, China
-
Received:2016-09-21Online:2016-11-30Published:2016-11-30 -
Contact:Ye Qi-fa, Professor, Zhongnan Hospital of Wuhan University, Institute of Hepatobiliary Diseases of Wuhan University, Transplant Center of Wuhan University, Hubei Key Laboratory of Medical Technology on Transplantation, Wuhan 430071, Hubei Province, China; the Third Xiangya Hospital of Central South University, Research Center of National Health Ministry on Transplantation Medicine Engineering and Technology, Changsha 410013, Hunan Province, China -
About author:Li Ling, Pharmacist, Zhongnan Hospital of Wuhan University, Institute of Hepatobiliary Diseases of Wuhan University, Transplant Center of Wuhan University, Hubei Key Laboratory of Medical Technology on Transplantation, Wuhan 430071, Hubei Province, China -
Supported by:the Natural Science Foundation of Hubei Province, No. 2015CFA018
CLC Number:
Cite this article
Li Ling, Li Ning, Ai Zi-ye, Yao Ya-jun, Wei Wan-hui, He Wei-yang, Wang Yan-feng, Ye Qi-fa. Intra-patient variability of tacrolimus concentration in transplant recipients: a prognostic predictor post transplantation[J]. Chinese Journal of Tissue Engineering Research, 2016, 20(49): 7437-7422.
share this article
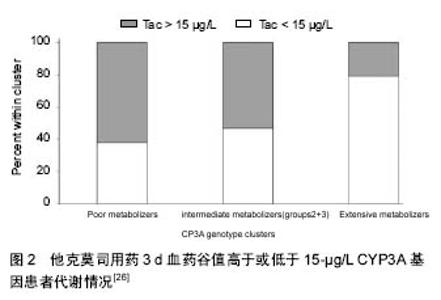
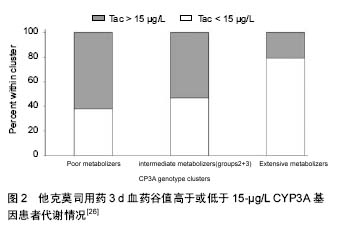
2.1 他克莫司浓度的个体内变异性 他克莫司是一个药代动力学高度个体差异、量效关系明确、治疗窗窄的临界剂量药物。大量研究报道,他克莫司浓度体内变异高是急性排斥反应和肾移植长期不良预后的高危险因素[5]。浓度低可能导致免疫抑制低效和急性排斥反应,而浓度高则使患者承担药物中毒风险。他克莫司浓度的个体内变异性,是指一段时间内,尽管单个患者给药剂量不变,各时间点他克莫司血药浓度值的变异程度。 2.2 他克莫司浓度个体内变异性的决定因素 诸多因素可影响他克莫司药代动力学参数及他克莫司浓度个体内变异性。具体如下: 2.2.1 他克莫司浓度检测分析方法 目前,全球有多种分析方法应用于他克莫司的分析检测。免疫学方法检测他克莫司药物浓度经常偏高,主要与他克莫司与其代谢产物之间存在交叉反应性有关。另有研究报道,当药物浓度低于检测限时,免疫学方法缺乏足够灵敏度,其变异区间相当大(他克莫司药物浓度小于5.0 μg/L时,CV%为18%),可信度较低[6]。尽管目前免疫评价仪器在不断优化与改进,但高效液相色谱法串联质谱法(High performance liquid chromatography tandem mass spectrometry, HPLC-MS/MS)灵敏度、选择性和精密度高,仍是他克莫司药物浓度监测的“金标准”[7]。近十年,他克莫司药物浓度检测已逐渐从免疫评价方法向HPLC- MS/MS检测转变,但由于后种技术操作要求高、前处理程序复杂,导致无法广泛使用和推广。 2.2.2 食物 他克莫司口服生物利用度与进食脂肪含量与进食时间长短有关[8]。Bekersky等[9]发现,与禁食状态相比,高脂饮食可显著降低他克莫司的生物利用度、减慢药物吸收,但不影响药物半衰期。禁食状态下服用他克莫司比与食物同服或者早餐后1-1.5 h服用他克莫司表现出更高的生物利用度[10]。 食物影响CYP3A和或者ABCB1的活性,从而改变他克莫司药物浓度。西柚(Citrus paradisi)可以与很多药物产生相互作用,包括钙调磷酸酶抑制剂[11]。西柚汁抑制CYP3A和ABCB1活性,同样下调肠道CYP3A4蛋白表达,导致他克莫司浓度增加[12]。Egashira等[13]报道,柚子含呋喃香豆素被成分,可以抑制CYP3A活性,肾移植患者进食柚子后他克莫司药物浓度翻倍。预服用100%柚子汁大鼠他克莫司浓度是对照组大鼠的两倍。姜黄和生姜也具有类似效果。姜黄可影响ABCB1和CYP3A4的功能和表达,但生姜仅改变了二者的活性[14]。 2.2.3 腹泻 大量研究报道,腹泻导致他克莫司的体内浓度增加[8]。Maes等[15]证明,在26例肾移植受者中,尽管服用同样剂量他克莫司,严重腹泻(每日多于3次稀便)可引起他克莫司药物浓度显著增加。腹泻状态下,为保证理想的他克莫司浓度,给药剂量必须减少30%。研究证明,腹泻患者他克莫司浓度增加,主要与肠黏膜损伤降低肠道ABCB1活性,增加口服生物利用度有关[16]。缩短肠道转运时间,使更多他克莫司转运至低表达CYP3A4的回肠,也是恶性腹泻患者发作期他克莫司药物浓度增高的原因[17]。然而,轻微腹泻并不引起他克莫司浓度增加[18]。 2.2.4 药物-药物相互作用 他克莫司与许多药物都存在相互作用,其主要机制是抑制或者诱导肠道或肝脏CYP3A系统[19]。这些药物包括糖皮质激素、钙通道抑制剂、利托那韦、氮唑类抗真菌药、利福平和抗癫痫药物等[20-21]。另外,非处方药和草药亦能与他克莫司发生相互作用。用于治疗抑郁、焦虑和睡眠障碍的圣约翰麦芽汁可以诱导肝脏及肠道CYP3A4和肠道ABCB1,但不影响CYP3A5表达[22]。Mai等[23]证明圣约翰麦芽汁降低他克莫司的生物利用度。连续14 d以600 mg/d剂量给予圣约翰麦芽汁,可以使他克莫司剂量矫正的AUC0-12降低57.8%。为维持治疗剂量他克莫司浓度,在服用圣约翰麦芽茶时,需要将他克莫司给药剂量从基线值4.5 mg/d调整为8.0 mg/d。 2.2.5 遗传因素 个体自身的遗传差异可部分解释他克莫司浓度个体内变异性。CYP3A5表达型(携带有CYP3A5*1/*3或者CYP3A5*1/*1的个体)需要比CYP3A5不表达型(携带有CYP3A5*3/*3个体)高出约50%的初试给药剂量[24-25]。相反,研究发现,CYP3A5不表达型比CYP3A5表达型患者他克莫司的Cmax和AUC0-last个体内变异性高出52%-41%[26]。猜想CYP3A5不表达型患者他克莫司代谢可能主要受CYP3A4活性控制,而CYP3A4易于被其他药物或食物抑制或诱导,该类患者药代动力学波动机会更大(图2)。然而,在249、209和118例稳定肾移植受者人群中研究发现,他克莫司个体内变异性和CYP3A5基因型没有明显关系[27]。导致结果完全矛盾的原因尚不清楚,猜想这可能与样本量、研究人群选择不同或与实验设计不同有关。"
| [1] Matas AJ, Smith JM, Skeans MA, et al. OPTN/SRTR 2011 annual data report: kidney. Am J Transplant. 2013;13:11-46. [2] Nankivell BJ, Kuypers DR.Diagnosis and prevention of chronic kidney allograft loss. Lancet.2011;378:1428-1437. [3] Jardine AG, Gaston RS, Fellstrom BC, et al. Prevention of cardiovascular disease in adult recipients of kidney transplants. Lancet.2011;378:1419-1427. [4] Farrugia D, Mahboob S, Cheshire J, et al. Malignancy- related mortality following kidney transplantation is common. Kidney Int.2014;85:1395-1403. [5] Saint-Marcoux F, Woillard JB, Monchaud C, et al. How to handle missed or delayed doses of tacrolimus in renal transplant recipients? A pharmacokinetic investigation. Pharmacol Res.2015;100:281-287. [6] Saitman A, Metushi IG, Mason DS,et al. Evaluation of the Waters MassTrak LC-MS/MS Assay for Tacrolimus and a Comparison to the Abbott Architect Immunoassay. Ther Drug Monit. Ther Drug Monit. 2016;38(3):300-304. [7] Ansermot N, Fathi M, Veuthey JL, et al. Quantification of cyclosporine and tacrolimus in whole blood. Comparison of liquid chromatography-electrospray mass spectrometry with the enzyme multiplied immunoassay technique. Clin Biochem.2008;41(10-11): 910-913. [8] Vanhove T, Annaert P, Kuypers DR,et al.Clinical determinants of calcineurin inhibitor disposition: a mechanistic review. Drug Metab Rev. 2016;48(1):88-112. [9] Bekersky I, Dressler D, Mekki QA. Effect of low- and high-fat meals on tacrolimus absorption following 5 mg single oral doses to healthy human subjects.J Clin Pharmacol 2001;41:176-182. [10] Bekersky I, Dressler D, Mekki Q. Effect of time ofmeal consumption on bioavailability of a single oral 5 mg tacrolimus dose. J Clin Pharmacol. 2001;41:289–297. [11] Hanley MJ, Cancalon P, Widmer WW, et al. Program in Pharmacology and Experimental Therapeutics Tufts University School of Medicine,Boston, Massachusetts 02111, United States of America. Expert Opin Drug Metab Toxicol, 2011; 7(3): 267-286. [12] Uno T, Yasui-Furukori N. Effect of grapefruit juice in relation to human pharmacokinetic study. Curr Clin Pharmacol.2006;1:157-161. [13] Egashira K,SasakiH, HiguchiH,Ieiri I. Food–drug interaction of tacrolimuswith pomelo, ginger, and turmeric juice in rats. Drug Metab Pharmacokinet. 2012;27:242-247. [14] Zhang W,Lim LY.Effects of spice constituents on P-glycoprotein-mediated transport and CYP3A4-mediated metabolism in vitro. Drug Metab Dispos. 2008;36:1283-1290. [15] Maes BD, Lemahieu W, Kuypers D, et al. Differential effect of diarrhea on FK506 versus cyclosporine A trough levels and resultant prevention of allograft rejection in renal transplant recipients. Am J Transplant. 2002;2:989-992. [16] Kawauchi S, Nakamura T, Yasui H, et al. Intestinal and hepatic expression of cytochrome P450s and mdr1a in rats with indomethacin-induced small intestinal ulcers. Int J Med Sci.2014;11(12):1208-1217. [17] Thorn M, Finnstrom N, Lundgren S,et al. Cytochromes P450 and MDR1 mRNA expression along the human gastrointestinal tract. Br J Clin Pharmacol. 2005;60: 54-60. [18] van Boekel GA, Aarnoutse RE, van der Heijden JJ,et al. Effect of mild diarrhea on tacrolimus exposure. Transplantation.2012;94:763–767. [19] Pallet N, Etienne I, Buchler M, et al. Long-term clinical impact of adaptation of initial tacrolimus dosing to CYP3A5 genotype. Am J Transplant. 2016 Mar 17. doi: 10.1111/ajt.13788. [20] vanMaarseveen EM, Rogers CC, Trofe-Clark J,et al. Drug-drug interactions between antiretroviral and immunosuppressive agents in HIV-infected patients after solid organ transplantation: a review. AIDS Patient Care STDS.2012;26: 568-581. [21] vanMaarseveen EM, Crommelin HA, Mudrikova T,et al. Pretransplantation pharmacokinetic curves of tacrolimus in HIV-infected patients on ritonavir-containing cART: a pilot study. Transplantation 2013;95:397-402. [22] Jackson A, D'Avolio A, Moyle G,et al. Pharmacokinetics of the co-administration of boceprevir and St John's wort to male and female healthy volunteers.J Antimicrob Chemother.2014;69(7):1911-1915. [23] Mai I, Stormer E, Bauer S, et al. Impact of St John's wort treatment on the pharmacokinetics of tacrolimus and mycophenolic acid in renal transplant patients. Nephrol Dial Transplant.2003;18:819-822. [24] Hesselink DA, Bouamar R, Elens L,et al. The role of pharmacogenetics in the disposition of and response to tacrolimus in solid organ transplantation. Clin Pharmacokinet.2014;53:123-139. [25] van Gelder T, van Schaik RH, Hesselink DA. Pharmacogenetics and immunosuppressive drugs in solid organ transplantation.Nat Rev Nephrol. 2014; 10:725-731. [26] Elens L, Bouamar R, Hesselink DA, et al. A new functional CYP3A4 intron 6 polymorphism significantly affects tacrolimus pharmacokinetics in kidney transplant recipients. Clin Chem.2011;57:1574-1583. [27] Ro H, Min SI, Yang J, et al. Impact of tacrolimus intraindividual variability and CYP3A5 genetic polymorphism on acute rejection in kidney transplantation. Ther Drug Monit.2012;34:680-685. [28] Borra LC, Roodnat JI, Kal JA,et al. Highwithin- patientvariability in the clearance of tacrolimus is a risk factor for poor long-term outcome after kidney transplantation. Nephrol DialTransplant. 2010;25: 2757-2763. [29] van Gelder T, Gabardi S. Methods, strengths, weaknesses, and limitations of bioequivalence tests with special regard to immunosuppressive drugs. Transpl Int.2013;26:771-777. [30] van Gelder T,ESOT Advisory Committee on Generic Substitution.European Society for Organ Transplantation Advisory Committee recommendations on generic substitution of immunosuppressive drugs. Transpl Int.2011;24:1135-1141. [31] Kahan BD, Welsh M, Schoenberg L, et al. Variable oral absorption of cyclosporine. A biopharmaceutical risk factor for chronic renal allograft rejection. Transplantation.1996;62:599-606. [32] Stoves J, Newstead CG. Variability of cyclosporine exposure and its relevance to chronic allograft nephropathy: a case–control study. Transplantation. 2002;74:1794-1797. [33] van Gelder T. Within-patient variability in immunosuppressive drug exposure as a predictor for poor outcome after transplantation. Kidney Int. 2014; 85:1267-1268. [34] Sapir-Pichhadze R, Wang Y, Famure O, et al. Time-dependent variability in tacrolimus trough blood levels is a risk factor for late kidney transplant failure. Kidney Int.2014;85:1404-1411. [35] Pollock-Barziv SM, Finkelstein Y,Manlhiot C, et al. Variability in tacrolimus blood levels increases the risk of late rejection and graft loss after solid organ transplantation in older children. Pediatr Transplant 2010;14:968-975. [36] Hsiau M, Fernandez HE, Gjertson D,et al.Monitoring nonadherence and acute rejection with variation in blood immunosuppressant levels in pediatric renal transplantation. Transplantation.2011;92:918–922. [37] Prytula AA, Bouts AH, Mathot RA, et al. Intra-patient variability in tacrolimus trough concentrations and renal function decline in pediatric renal transplant recipients. Pediatr Transplant.2012;16:613-618. [38] Stifft F, Stolk LM, Undre N, et al. Lower variability in 24-hour exposure during once-daily compared to twice-daily tacrolimus formulation in kidney transplantation.Transplantation.2014;97(7):775-780. |
| [1] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [2] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [3] | Yang Xin, Jin Zhe, Feng Xu, Lu Bing. The current situation of knowledge and attitudes towards organ, eye tissue, body donation of residents in Shenyang [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 779-784. |
| [4] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [5] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [6] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| [7] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [8] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| [9] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| [10] | Chen Jiana, Qiu Yanling, Nie Minhai, Liu Xuqian. Tissue engineering scaffolds in repairing oral and maxillofacial soft tissue defects [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 644-650. |
| [11] | Xing Hao, Zhang Yonghong, Wang Dong. Advantages and disadvantages of repairing large-segment bone defect [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 426-430. |
| [12] | Chen Siqi, Xian Debin, Xu Rongsheng, Qin Zhongjie, Zhang Lei, Xia Delin. Effects of bone marrow mesenchymal stem cells and human umbilical vein endothelial cells combined with hydroxyapatite-tricalcium phosphate scaffolds on early angiogenesis in skull defect repair in rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3458-3465. |
| [13] | Wang Hao, Chen Mingxue, Li Junkang, Luo Xujiang, Peng Liqing, Li Huo, Huang Bo, Tian Guangzhao, Liu Shuyun, Sui Xiang, Huang Jingxiang, Guo Quanyi, Lu Xiaobo. Decellularized porcine skin matrix for tissue-engineered meniscus scaffold [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3473-3478. |
| [14] | Mo Jianling, He Shaoru, Feng Bowen, Jian Minqiao, Zhang Xiaohui, Liu Caisheng, Liang Yijing, Liu Yumei, Chen Liang, Zhou Haiyu, Liu Yanhui. Forming prevascularized cell sheets and the expression of angiogenesis-related factors [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3479-3486. |
| [15] | Liu Chang, Li Datong, Liu Yuan, Kong Lingbo, Guo Rui, Yang Lixue, Hao Dingjun, He Baorong. Poor efficacy after vertebral augmentation surgery of acute symptomatic thoracolumbar osteoporotic compression fracture: relationship with bone cement, bone mineral density, and adjacent fractures [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3510-3516. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||