Chinese Journal of Tissue Engineering Research ›› 2017, Vol. 21 ›› Issue (20): 3248-3254.doi: 10.3969/j.issn.2095-4344.2017.20.021
Previous Articles Next Articles
MG53 protein protects against multiorgan ischemia/reperfusion injury: present and future
Liu Teng-fei1, Zhou Jian-kang1, Huang Tuan-jie1, Xing Qu1, Cheng Kang1, Li Peng1, Li Dong-peng2, Yang Bo2, Ma Shan-shan1, Guan Fang-xia1
- 1School of Life Sciences, Zhengzhou University, Zhengzhou 450001, Henan Province, China; 2the First Affiliated Hospital of Zhengzhou University, Zhengzhou 450052, Henan Province, China
-
Revised:2017-05-09Online:2017-07-18Published:2017-07-28 -
Contact:Ma Shan-shan, Associate professor, Master’s supervisor, School of Life Sciences, Zhengzhou University, Zhengzhou 450001, Henan Province, China; Guan Fang-xia, Professor, Doctoral supervisor, School of Life Sciences, Zhengzhou University, Zhengzhou 450001, Henan Province, China -
About author:Liu Teng-fei, Studying for master’s degree, School of Life Sciences, Zhengzhou University, Zhengzhou 450001, Henan Province, China -
Supported by:The National Natural Science Foundation of China, No. 81601078 and 81471306; the Science & Technology Innovation Foundation for Higher Education Institutes of Henan Province of China, No. 15IRTSTHN022; the Plan for Scientific Innovation Talent of Henan Province, No. 154200510008; the International Cooperation Project of Henan Province of China, No. 2016GH03 and 2016GH15
CLC Number:
Cite this article
Liu Teng-fei, Zhou Jian-kang, Huang Tuan-jie, Xing Qu, Cheng Kang, Li Peng, Li Dong-peng, Yang Bo, Ma Shan-shan, Guan Fang-xia . MG53 protein protects against multiorgan ischemia/reperfusion injury: present and future[J]. Chinese Journal of Tissue Engineering Research, 2017, 21(20): 3248-3254.
share this article
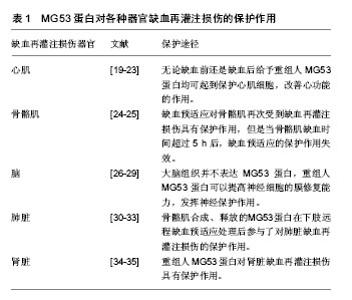
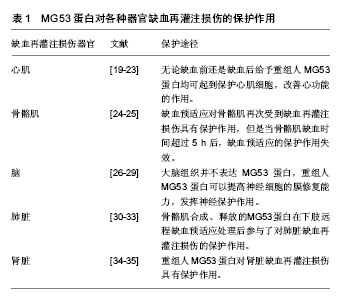
其中SPRY 结构域位于羧基端,该结构域由1个α螺旋、13个β折叠片和1个310螺旋组成;而另外的3种结构域为TRIM蛋白家族成员所共有[2-4]。TRIM蛋白家族的功能有些还不是很清楚,有研究报道其参与细胞内重要生化过程如阻止H1V感染[5]、细胞周期调控[6]、细胞凋亡和肿瘤形成等[7-10]。 2.2 MG53蛋白对器官缺血再灌注损伤的保护作用 在MG53蛋白对器官缺血再灌注损伤的保护作用研究中,心肌缺血再灌注损伤是一个主要的研究对象。心肌和骨骼肌都可以通过缺血预适应和缺血后适应这两种机制内生大量的MG53蛋白[11-12];而脑、肺脏和肾脏等器官通过缺血预适应和缺血后适应却几乎不产生MG53蛋白,但对这些器官的缺血再灌注损伤来说缺血预适应和缺血后适应依然是两种很强的保护机制[13-18]。目前研究MG53蛋白对器官缺血再灌注损伤起保护作用的方法除了缺血预适应和缺血后适应外,还有MG53蛋白基因敲除或基因沉默、MG53蛋白或基因过表达、注射重组人MG53蛋白以及RNA干扰技术所致的MG53蛋白表达下调等,见表1。"
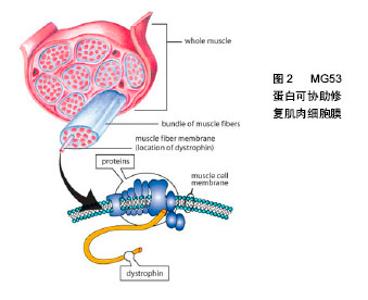
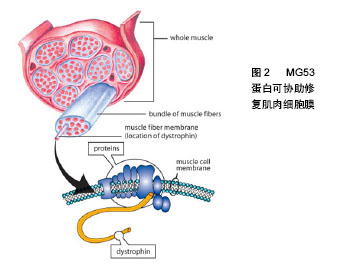
2.2.1 MG53蛋白对心肌缺血再灌注损伤的保护作用冠心病是严重危害人类健康的心血管疾病,冠状动脉血管的动脉粥样硬化病变会使血管腔变得狭窄或堵塞,从而导致心肌缺血,心肌缺血又会导致心肌梗死。对缺血心肌适时的再灌注是保持心肌细胞活力的惟一方法;但再灌注又会引起心律失常等一些更为严重的损害。研究发现MG53蛋白在心肌缺血再灌注损伤中起到保护作 用[19-21];但心肌缺血再灌注损伤本身又会降低内源性MG53蛋白的表达[19]。研究发现,在心肌组织中,MG53蛋白存在两种内生机制,即缺血预适应和缺血后适应。对心肌缺血再灌注损伤来说缺血预适应和缺血后适应是两种非常强大的内源性保护机制。Cao[19]和Zhang 等[21]通过动物实验分别证实了缺血预适应和缺血后适应对心肌缺血再灌注损伤的保护作用。另外,当内源性MG53蛋白不能满足心肌缺血再灌注损伤的需要时,给予重组人MG53蛋白就成为了一种新的治疗方式[2]。在心肌缺血再灌注损伤模型中无论缺血前还是缺血后给予重组人MG53蛋白均可起到保护心肌细胞,改善心功能的作用[22-23]。 2.2.2 MG53蛋白对骨骼肌缺血再灌注损伤的保护作用 机体大负荷运动会引起骨骼肌的剧烈收缩,肌肉收缩压迫血管,长时间运动导致血管对骨骼肌的供血不足,骨骼肌出现缺血缺氧的情况。运动后休息期间骨骼肌的供血逐渐恢复正常,缺血缺氧得到缓解。该过程类似于临床上的缺血再灌注过程,这种缺血再灌注也会引起骨骼肌损伤,即骨骼肌缺血再灌注损伤。Lorenz等[24]证实了缺血预适应对骨骼肌再次受到缺血再灌注损伤具有保护作用,但是当骨骼肌缺血时间超过5 h后,缺血预适应的保护作用失效。研究发现,在小鼠骨骼肌缺血再灌注损伤模型中,MG53蛋白基因敲除小鼠更易受到损伤,而野生型小鼠相对不容易受到损伤,证明了MG53蛋白对受损肌肉组织发挥着重要的保护作用[25]。这些研究提示了MG53蛋白可能是治疗骨骼肌缺血再灌注损伤的一个新思路。 2.2.3 MG53蛋白对脑缺血再灌注损伤的保护作用 脑血管病是世界上发病率、死亡率和致残率很高的疾病之一。国内脑血管病中约70%为缺血性脑血管病,主要是由大脑动脉尤其大脑中动脉闭塞引起[26]。脑缺血后梗死组织周边存在的缺血半暗带恢复再通是缺血性脑血管疾病治疗的基础。但缺血区恢复再灌注后会加重脑组织和细胞损伤,即脑缺血再灌注损伤。实验证明,大脑组织并不表达MG53蛋白[22]。Moloughney等[27]发现重组MG53蛋白可以提高神经细胞的膜修复能力,发挥神经保护作用。由此,研究者采用大鼠脑缺血再灌注损伤模型,观察无MG53蛋白表达的器官是否能够在重组人MG53蛋白的治疗下得到改善。研究发现,MG53蛋白能透过血脑屏障[28];在术前30 min及术后 0,1,2,4 h分别给予重组人MG53蛋白,均能使神经系统症状得到改善,降低神经系统Zea-Longa评分,减轻脑梗死面积以及坏死程度,且重组人MG53蛋白的保护作用呈时间相关性,有效时间窗为4 h。这一研究为临床治疗缺血性脑血管病提供了新的方法[29]。 2.2.4 MG53蛋白对肺脏缺血再灌注损伤的保护作用在临床上心肺移植手术、心脏动脉搭桥术、心肺脑复苏以及休克等均会导致肺缺血再灌注损伤。肺缺血再灌注损伤已成为肺移植术后重要的死因之一[30-31]。在肺缺血再灌注损伤模型中,重组人MG53蛋白能降低肺脏湿干比例以及炎性介质的浸润,提高小鼠的生存率[1,32]。给予急性肺损伤的实验动物静脉注射或吸入重组人MG53蛋白,可有效减轻肺损伤的症状,重复使用重组人MG53蛋白可改善慢性损伤对肺部结构的改变[32]。另外在肺脏缺血再灌注损伤前提前给予大鼠下肢远程缺血预适应,可以使肺脏中的MG53蛋白表达量显著增加,这提示骨骼肌合成、释放的MG53蛋白在下肢远程缺血预适应处理后参与了对肺脏缺血再灌注损伤的保护作用[33]。 2.2.5 MG53蛋白对肾脏缺血再灌注损伤的保护作用肾脏缺血再灌注损伤是一种常见的临床病理生理过程,多见于严重烧伤、低血容量性休克、肾动脉阻断以及肾脏移植等情况,可以造成肾衰竭或肾功能障碍。近年来发现,重组人MG53蛋白对肾脏缺血再灌注损伤具有保护作用[1,34]。肾脏为高灌注器官,对缺血缺氧较敏感。例如在严重烧伤后,尤其是大面积烧伤后,机体体液快速丢失,导致急性的肾缺血缺氧。肾脏随之会发生一系列显著的形态改变,而这些形态改变就与肾功能障碍的发生、发展密切相关。另外在小鼠30%Ⅲ度烫伤模型中,给予重组人MG53蛋白静脉注射,观察其对烧伤引起的肾脏缺血再灌注损伤的保护作用。研究发现重组人MG53蛋白治疗组的小鼠生存率高于对照组,并且观察到治疗组肾小管空泡的变性和坏死程度均比对照组有明显的减轻[35]。 2.3 MG53蛋白对器官缺血再灌注损伤的保护机制 在MG53蛋白对器官缺血再灌注损伤的保护机制研究中,心肌缺血再灌注损伤也是一个主要的研究对象。研究发现,心肌缺血预适应和缺血后适应或MG53蛋白均能激活RISK信号通路[19,21]。研究发现,心肌缺血预适应和缺血后适应虽然也能激活生存活化因子增强(survivor activating factor enhancement,SAFE)信号通路,但并不能通过MG53蛋白来触发STAT磷酸化,所以说MG53蛋白不参与SAFE信号通路[36-37]。另外,MG53蛋白介导的膜修复过程也是MG53蛋白对器官缺血再灌注损伤起保护作用的一个重要机制。除此之外,MG53蛋白对器官缺血再灌注损伤的保护机制还可能有:MG53蛋白抑制Cx43蛋白的去磷酸化和重分布、MG53蛋白上调KChIP2蛋白的表达等。总之,MG53蛋白对器官缺血再灌注损伤的保护机制研究仍需要进一步的深入。 2.3.1 RISK信号通路 缺血预适应和缺血后适应在心肌中可内生出大量的MG53蛋白,而MG53蛋白参与缺血预适应和缺血后适应的心肌保护作用是通过激活RISK信号通路实现的[19,21]。RISK信号通路主要包括PI3K-Akt-糖原合酶激酶3β(PI3K-Akt-glycogen synthase kinase-3β,PI3K-Akt-GSK3β)和细胞外信号调节激酶1/2(extracellular signal-regulated kinase 1/2,ERK1/2)信号通路。Cao等[19]证明了MG53蛋白参与缺血预适应的心肌保护与PI3K、Akt、GSK和ERK1/2磷酸化水平有关,最终证实MG53蛋白在RISK信号通路上发挥着至关重要的作用。研究还发现MG53蛋白与微囊蛋白3形成复合物后再与PI3K的P85亚基结合从而激活RISK信号通路[19]。MG53蛋白的N端和C端能分别与微囊蛋白3和p85亚基结合[21]。PI3K-Akt-GSK3β信号通路的激活会引起Akt磷酸化水平的升高,而PI3K-Akt-GSK3β信号通路的终末效应器是GSK3β,最终实现GSK3β的磷酸化。另外MG53蛋白也能提高ERK1/2的磷酸化水平,已证实激活ERK1/2信号通路同样可使其下游的GSK-3β发生磷酸化[38]。在这里值得一提的是,缺血再灌注损伤的一个重要途径就是开放线粒体通透性转换通道通透性的增高,导致开放线粒体通透性转换通道的开放,从而促进促凋亡蛋白从线粒体中释放到细胞浆中,引起细胞凋亡或坏死[39-40]。进一步研究发现,GSK3β被RISK信号通路磷酸化修饰之后失去了开放线粒体通透性转换通道的作用,从而减少了线粒体膜电位崩解和线粒体途径的细胞凋亡,发挥心肌保护作用[41]。 2.3.2 MG53蛋白介导的膜修复机制 器官缺血再灌注损伤通常伴随着细胞膜损伤的发生。内源性MG53蛋白作为一种肌特异性膜修复蛋白,能修复心肌和骨骼肌损伤的细胞膜。在MG53蛋白介导的膜修复过程中,存在着多种膜修复因子如微囊蛋白3、Dysferlin和转录释放因子等,其中微囊蛋白3是一个调节因子,能够稳定由MG53蛋白介导的膜修复过程;Dysferlin能促进膜修复过程中囊泡的融合;转录释放因子是一个停靠蛋白,在损伤部位,绑定MG53蛋白寡聚体于暴露的膜胆固醇上。研究表明,暴露的膜胆固醇是MG53蛋白介导的膜修复的始发因子[20]。外界氧化物的流入细胞内后,MG53蛋白被氧化并在卷曲螺旋结构域亮氨酸拉链的帮助下促成MG53蛋白的寡聚化,起到了稳定MG53蛋白修复的作用[22,42]。MG53蛋白寡聚体与细胞内囊泡关联,招募其他膜修复因子如微囊蛋白3和Dysferlin等,从而形成一个修复复合体[43-45]。同时MG53蛋白通过识别囊泡和质膜上的磷脂酰丝氨酸并在驱动蛋白和肌球蛋白的帮助下将膜修复复合体转运至膜损伤部位[22,46-47]。然后转录释放因子作为中介,绑定MG53蛋白寡聚体于细胞内囊泡和质膜内部的胆固醇上。最后在Ca2+依赖的Dysferlin的作用下,使囊泡与质膜发生融合,完成修复过程[48-49],见图2。"
| [1] Zhang Y, Wu HK, Lv FX, et al. MG53 is a double-edged sword for human diseases. Sheng Li Xue Bao. 2016;68(4): 505-516.[2] Weisleder N, Takizawa N, Lin P, et al. Recombinant MG53 protein modulates therapeutic cell membrane repair in treatment of muscular dystrophy. Sci Trans Med. 2012; 4(139): 139-185.[3] He B, Tang RH, Weisleder N, et al. Enhancing muscle membrane repair by gene delivery of MG53 ameliorates muscular dystrophy and heart failure in delta-Sarcoglycan- deficient hamsters. Mol Ther. 2012;20(4): 727-735.[4] Park EY, Kwon OB, Jeong BC, et al. Crystal structure of PRY-SPRY domain of human TRIM72. Proteins. 2010;78(3): 790-795.[5] Nisole S, Stoye JP, Saib A. TRIM family proteins: retroviral restriction and antiviral defence [J]. Nat Rev Microbiol. 2005;3(10): 799-808.[6] Cammas F, Khetchoumian K, Chambon P, et al. TRIM involvement in transcriptional regulation. Adv Exp Med Biol. 2012;770: 59-76.[7] Caratozzolo MF, Micale L, Turturo MG, et al. TRIM8 modulates p53 activity to dictate cell cycle arrest. Cell Cycle. 2012;11(3): 511-523.[8] Zaman MMU, Nomura T, Takagi T, et al. Ubiquitination- deubiquitination by the TRIM27-USP7 complex regulates tumor necrosis factor alpha-induced apoptosis. Mol Cell Biol. 2013;33(24): 4971-4984.[9] Hatakeyama S. TRIM proteins and cancer. Nat Rev Cancer. 2011;11(11): 792-804.[10] Cambiaghi V, Giuliani V, Lombardi S, et al. TRIM proteins in cancer. Adv Exp Med Biol. 2012;770: 77-91.[11] Azra M, Muhammad A, Khan, Shaheen NK, et al. Diazoxide preconditioning of endothelial progenitor cells improves their ability to repair the infarcted myocardium. Cell Biol Int. 2015; 39(11): 1251-1263.[12] Zhao ZQ, Corvera JS, Halkos ME, et al. Inhibition of myocardial injury by ischemic PostC onditioning during reperfusion: comparison with ischemic preconditioning. Am J Phys. 2003;54(2): H579.[13] Mu D, Chang YS, Vexler ZS, et al. Hypoxia-inducible factor 1α and erythropoietin upregulation with deferoxamine salvage after neonatal stroke. Exp Neur. 2005;195(2): 407-415.[14] Zhao H. Ischemic PostC onditioning as a novel avenue to protect against brain injury after stroke. J Cerebral Blood Flow Metabol. 2009;29(5):873-885.[15] Liu KX, Li YS, Huang WQ, et al. Immediate but not delayed PostC onditioning during reperfusion attenuates acute lung injury induced by intestinal ischemia/reperfusion in rats: comparison with ischemic preconditioning. J Surg Res. 2009; 157(1): e55-e62.[16] Patschan D, Krupincza K, Patschan S, et al. Dynamics of mobilization and homing of endothelial progen Ito r cells after acute renal ischemia: modulation by ischemic preconditioning. Am J Phys Renal Phys. 2006;291(1): F176-F185.[17] Jiang B, Liu X, Chen H, et al. Ischemic PostC onditioning attenuates renal ischemic/reperfusion injury in mongrel dogs. Urology. 2010;76(6): 1519.e1-1519.e7.[18] Zhou W, Zeng D, Chen R, et al. Limb ischemic preconditioning reduces heart and lung injury after an open heart operation in infants. Pediatr Cardiol. 2010; 31(1): 22-29.[19] Cao CM, Zhang Y, Weisleder N, et al. MG53 constitutes a primary determinant of cardiac ischemic preconditioning. Circulation. 2010;121(23): 2565-2574.[20] Wang X, Xie W, Zhang Y, et al. Cardioprotection of ischemia/reperfusion injury by cholesterol-dependent MG53-mediated membrane repair. Circ Res. 2010;107(1): 76-83.[21] Zhang Y, Lv F, Jin L, et al. MG53 participates in ischaemic PostC onditioning through the RISK signalling pathway. Cardiovasc Res. 2011;91(1): 108-115.[22] Cai C, Masumiya H, Weisleder N, et al. MG53 nucleates assembly of cell membrane repair machinery. Nature Cell Biol. 2009;11(1): 56-64.[23] Masumiya H, Asaumi Y, Nishi M, et al. Mitsugumin 53-mediated maintenance of K+ currents in cardiac myocytes. Channels. 2009;3(1): 56-64.[24] Lorenz G, Axel M. Ischemic preconditioning improves post-ischemic skeletal muscle function. Am Surg.1996;62(5): 391-394.[25] Zhu H, Hou J, Roe JL, et al. Amelioration of ischemia-reperfusion-induced muscle injury by the recombinant human MG53 protein. Muscle Nerve. 2015;52(5): 852-858.[26] Go AS, Mozaffarian D, Roger VL, et al. Heart disease and stroke statistics--2013 update: a report from the American Heart Association. Circulation. 2013;127(1): e6-e245.[27] Moloughney JG, Alloush J, Le-Hoang O, et al. Membrane repair protein Mitsugumin 53 as a potential therapeutic for the treatment of traumatic brain injury. FASEB J. 2013; 27(Meeting Abstracts).[28] Yao Y, Zhang B, Zhu H, et al. MG53 permeates through blood-brain barrier to protect ischemic brain injury. Oncotarget. 2016;7(16): 22474-22485.[29] Yao Y, Zhang M, Li M, et al. Protective effect of rhMG53 protein on a focal cerebral ischemia/reperfusion in a rat model. Med J Chin People's Liberat Army. 2014;39(6): 439-443.[30] Lv X, Tan J, Liu D, et al. Intratracheal administration of p38alpha short-hairpin RNA plasmid ameliorates lung ischemia-reperfusion injury in rats. J Heart Lung Trans. 2012; 31(6): 655-662.[31] Chiang CH, Chuang CH, Liu SL. Apocynin attenuates ischemia-reperfusion lung injury in an isolated and perfused rat lung model. Trans Res. 2011;158(1): 17-29.[32] Jia Y, Chen K, Lin P, et al. Treatment of acute lung injury by targeting MG53-mediated cell membrane repair. Nat Commun. 2014;5: 4387.[33] 李郁,陈垦,徐在成,等. MG53在下肢远程缺血预适应保护肺缺血再灌注损伤中的作用[J]. 第三军医大学学报, 2015, 37(5): 409-413.[34] Duann P, Li H, Lin P, et al. MG53-mediated cell membrane repair protects against acute kidney injury. Sci Trans Med. 2015;7(279): 279ra36.[35] Wu Y, Huang J, Liu D, et al. Mitsugumin 53 protects the kidney from severe burn injury in mice. Burns Trauma. 2013; 1(3):128-133.[36] Lacerda L, Somers S, Opie LH, et al. Ischaemic PostC onditioning protects against reperfusion injury via the SAFE pathway]. Cardiovasc Res. 2009;84(2): 201-208.[37] Goodman MD, Koch SE, Fuller-Bicer GA, et al. Regulating RISK: a role for JAK-STAT signaling in PostC onditioning? Am J Phys. 2008;295(4): H1649-H1656.[38] Tsang A, Hausenloy DJ, Yellon DM. Myocardial PostC onditioning: reperfusion injury revisited. Am J Phys Heart Circ Phys. 2005;289(1): H2-H7.[39] Giampietri C, Paone A, D'Alessio A. Cell death. Int J Cell Biol. 2014;2014: 864062.[40] Cohen MV, Yang XM, Downey JM. The pH hypothesis of PostC onditioning: staccato reperfusion reintroduces oxygen and perpetuates myocardial acidosis. Circulation. 2007; 115(14): 1895-1903.[41] Vuure DV, Lochner A. Ischaemic PostC onditioning_ from bench to bedside. Cardiovasc J Afr. 2008;19(6): 311-320.[42] Hwang M, Ko J, Weisleder N, et al. Redox-dependent oligomerization through a leucine zipper motif is essential for MG53-mediated cell membrane repair. Am J Phys. 2011; 301(1): C106.[43] Levy JR, Campbell KP, Glass DJ. MG53's new identity. Skeletal Muscle. 2013.[44] Cai C, Masumiya H, Weisleder N, et al. MG53 regulates membrane budding and exocytosis in muscle cells. J Biol Chem. 2009;284(5): 3314-3322.[45] Cai C, Weisleder N, Ko JK, et al. Membrane repair defects in muscular dystrophy are linked to altered interaction between MG53, caveolin-3, and dysferlin. J Biol Chem. 2009;284(23): 15894-15902.[46] Bi GQ, Morris RL, Liao G, et al. Kinesin- and myosin-driven steps of vesicle recruitment for Ca2+ -regulated exocytosis. J Cell Biol. 1997;138(5): 999-1008.[47] Lin P, Zhu H, Cai C, et al. Nonmuscle myosin IIA facilitates vesicle trafficking for MG53-mediated cell membrane repair. FASEB J. 2012;26(5): 1875-1883.[48] McNeil P. Membrane repair redux: redox of MG53. Nature Cell Biol. 2009;11(1): 7-9.[49] Glover L, Brown RH Jr. Dysferlin in membrane trafficking and patch repair. Traffic. 2007;8(7): 785-794.[50] Figueroa XF, Duling BR. Gap junctions in the control of vascular function. Antiox Redox Signal. 2009;11(2): 251-266.[51] Boulaksil M, Winckels SK, Engelen MA, et al. Heterogeneous Connexin43 distribution in heart failure is associated with dispersed conduction and enhanced susceptibility to ventricular arrhythmias. Eur J Heart Fail.2010;12(9): 913-921.[52] Sato T, Ohkusa T, Honjo H, et al. Altered expression of connexin43 contributes to the arrhythmogenic substrate during the development of heart failure in cardiomyopathic hamster. Am J Phys Heart Circ Phys. 2008;294(3): H1164-H1173.[53] Zeghichi-Hamri S, de Lorgeril M, Salen P, et al. Protective effect of dietary n-3 polyunsaturated fatty acids on myocardial resistance to ischemia-reperfusion injury in rats. Nutr Res. 2010;30(12): 849-857.[54] Oei GT, Aslami H, Kerindongo RP, et al. Prolonged helium PostC onditioning protocols during early reperfusion do not induce cardioprotection in the rat heart in vivo: role of inflammatory cytokines. J Immunol Res. 2015;2015: 216798.[55] 蒋鑫,周平,杨小利,等. MG53预处理改善离体大鼠心脏缺血再灌注心律失常[J].第三军医大学学报,2015,37(1): 22-25.[56] Gregory RB, Hughes R, Riley AM, et al. Inos Ito l trisphosphate analogues selective for types I and II inos Ito l trisphosphate receptors exert differential effects on vasopressin-stimulated Ca2+ inflow and Ca2+ release from intracellular stores in rat hepatocytes. Biochem J. 2004;381: 519-526.[57] Namura S, Ooboshi D, Liu JL, et al. Neuroprotection after cerebral ischemia. Ann New York Acad Sci. 2013;1278(1): 25-32.[58] Ivanics T, Miklós Z, Ruttner Z, et al. Ischemia/reperfusion- induced changes in intracellular free Ca2+ levels in rat skeletal muscle fibers – an in vivo study. Pflüg Arch Eur J Phys. 2000; 440(2): 302-308.[59] Panama BK, Latour-Villamil D, Farman GP, et al. Nuclear factor kappaB downregulates the transient outward potassium current I(to,f) through control of KChIP2 expression. Circ Res. 2011;108(5): 537-543.[60] Liu W, Zhang C, Deng J, et al. Abstract-17484: MG53 modulates KChIP2 expression through regulation of NF-κB activity. Circulation. 2014;130(Suppl 2): A17484.[61] Li X, Xiao Y, Cui Y, et al. Cell membrane damage is involved in the impaired survival of bone marrow stem cells by oxidized low-density lipoprotein. J Cell Mol Med. 2014;18(12): 2445-2453. |
| [1] | Yao Xiaoling, Peng Jiancheng, Xu Yuerong, Yang Zhidong, Zhang Shuncong. Variable-angle zero-notch anterior interbody fusion system in the treatment of cervical spondylotic myelopathy: 30-month follow-up [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1377-1382. |
| [2] | Wang Shuo, Liu Wenying, Lü Chaofan, Li Jiacong, Geng Yi, Zhao Yungang. Cardioprotective effect of 3-nitro-N-methyl salicylamide on the isolated rat heart under cold ischemia preservation [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1194-1201. |
| [3] | Li Zhiyi, He Pengcheng, Bian Tianyue, Xiao Yuxia, Gao Lu, Liu Huasheng. Bibliometric and visualized analysis of ferroptosis mechanism research [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1202-1209. |
| [4] | Yang Shenglin, Pu Xingwei, Luo Chunshan, Yang Jianwen. Neuroprotective effects of tetrandrine preconditioning in rabbits with spinal cord ischemia-reperfusion injury [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1223-1227. |
| [5] | An Weizheng, He Xiao, Ren Shuai, Liu Jianyu. Potential of muscle-derived stem cells in peripheral nerve regeneration [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1130-1136. |
| [6] | Xuan Juanjuan, Bai Hongtai, Zhang Jixiang, Wang Yaoquan, Chen Guoyong, Wei Sidong. Role of regulatory T cell subsets in liver transplantation and progress in clinical application [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1143-1148. |
| [7] | Zhang Jinglin, Leng Min, Zhu Boheng, Wang Hong. Mechanism and application of stem cell-derived exosomes in promoting diabetic wound healing [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1113-1118. |
| [8] | Dong Miaomiao, Lai Han, Li Manling, Xu Xiuhong, Luo Meng, Wang Wenhao, Zhou Guoping. Effect of electroacupuncture on expression of nucleotide binding oligomerization domain-like receptor protein 3/cysteinyl aspartate specific proteinase 1 in rats with cerebral ischemia/reperfusion injury [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(5): 749-755. |
| [9] | He Yunying, Li Lingjie, Zhang Shuqi, Li Yuzhou, Yang Sheng, Ji Ping. Method of constructing cell spheroids based on agarose and polyacrylic molds [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 553-559. |
| [10] | He Guanyu, Xu Baoshan, Du Lilong, Zhang Tongxing, Huo Zhenxin, Shen Li. Biomimetic orientated microchannel annulus fibrosus scaffold constructed by silk fibroin [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 560-566. |
| [11] | Chen Xiaoxu, Luo Yaxin, Bi Haoran, Yang Kun. Preparation and application of acellular scaffold in tissue engineering and regenerative medicine [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 591-596. |
| [12] | Kang Kunlong, Wang Xintao. Research hotspot of biological scaffold materials promoting osteogenic differentiation of bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 597-603. |
| [13] | Shen Jiahua, Fu Yong. Application of graphene-based nanomaterials in stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 604-609. |
| [14] | Zhang Tong, Cai Jinchi, Yuan Zhifa, Zhao Haiyan, Han Xingwen, Wang Wenji. Hyaluronic acid-based composite hydrogel in cartilage injury caused by osteoarthritis: application and mechanism [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 617-625. |
| [15] | Li Hui, Chen Lianglong. Application and characteristics of bone graft materials in the treatment of spinal tuberculosis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 626-630. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||

