Chinese Journal of Tissue Engineering Research ›› 2022, Vol. 26 ›› Issue (4): 604-609.doi: 10.12307/2022.099
Previous Articles Next Articles
Application of graphene-based nanomaterials in stem cells
Shen Jiahua, Fu Yong
- Children’s Hospital of Zhejiang University School of Medicine, Hangzhou 310052, Zhejiang Province, China
-
Received:2020-06-20Revised:2020-06-30Accepted:2020-11-09Online:2022-02-08Published:2021-12-06 -
Contact:Fu Yong, MD, Chief physician, Children’s Hospital of Zhejiang University School of Medicine, Hangzhou 310052, Zhejiang Province, China -
About author:Shen Jiahua, Master candidate, Children’s Hospital of Zhejiang University School of Medicine, Hangzhou 310052, Zhejiang Province, China
CLC Number:
Cite this article
Shen Jiahua, Fu Yong. Application of graphene-based nanomaterials in stem cells[J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 604-609.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks

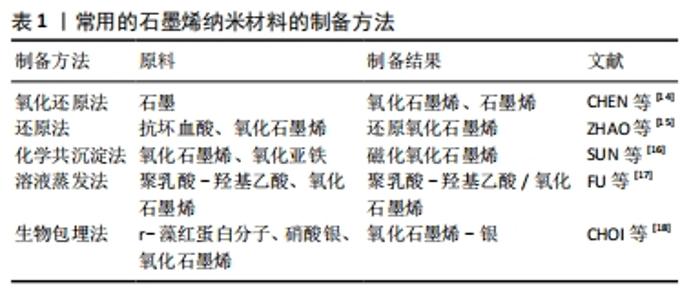
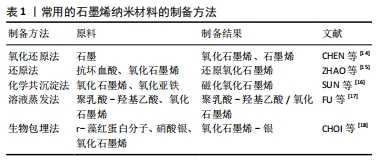
2.2 基于石墨烯的纳米材料对干细胞生物学行为的影响 2.2.1 胚胎干细胞 胚胎干细胞是从早期胚胎中分离出来的一类细胞,具有多能性,能够分化为3种胚胎胚层中的所有细胞类型,进而分化出成体动物所有的组织和器官。胚胎干细胞具有无限的增殖和分化能力,已被广泛应用于组织工程和再生医学领域[7]。 ZHAO等[15]报道还原氧化石墨烯能代替滋养细胞、细胞外基质作为培养基底,能让小鼠胚胎干细胞在其表面上快速黏附和增殖,同时通过比较孔径大小发现,30 μm孔径的还原氧化石墨烯能显著促进和维持胚胎干细胞的自我更新能力并维持多能性。 GARCIA-ALEGRIA等[19]研究发现,氧化石墨烯涂层覆盖的玻璃器皿表面能促进小鼠和人的胚胎干细胞向造血祖细胞的分化。YANG等[20]研究了小鼠胚胎干细胞分别在碳纳米管、石墨烯和氧化石墨烯上的分化,结果表明,只有氧化石墨烯能有效促进胚胎干细胞向多巴胺神经元的分化,并进一步增强多巴胺神经元相关基因的表达。 2.2.2 诱导多能干细胞 诱导多能干细胞是一种通过4种转录因子(Oct4、Klf4、Sox2、c-Myc)的过表达将体细胞重新编程至多能状态的干细胞,这种干细胞与胚胎干细胞具有相似的分化能力,几乎可以分化成3胚层中所有类型的细胞,还能克服使用胚胎干细胞的伦理问题,允许进行基因定制,为组织工程和再生医学提供了新的机遇[7,21] 。 YANG等[20]使用石墨烯和氧化石墨烯作为底物培养诱导多能干细胞,发现相比于玻璃表面,石墨烯表面培养的诱导多能干细胞具有与之相似的细胞黏附和增殖程度,而氧化石墨烯表面的诱导多能干细胞的黏附和增殖速度更快。氧化石墨烯能加速诱导多能干细胞的分化,而石墨烯能较好地保持诱导多能干细胞的未分化状态。石墨烯和氧化石墨烯表面培养的诱导多能干细胞能自发分化为外胚层和中胚层细胞,但石墨烯抑制诱导多能干细胞向内胚层谱系的分化,氧化石墨烯能增强诱导多能干细胞向内胚层谱系的分化,这证明了石墨烯和氧化石墨烯的不同表面性质对诱导多能干细胞的行为有不同的影响。 SABURI等[22]将诱导多能干细胞分别在静电纺丝法制备的聚乙二烯-氧化石墨烯和聚乙二烯表面上培养,比较培养出的细胞在碱性磷酸酶活性和钙含量、Runx2、骨钙素、骨连接基因表达的不同,证实了聚乙二烯-氧化石墨烯表面能增强诱导多能干细胞体外成骨分化的潜能。 2.2.3 间充质干细胞 间充质干细胞是一种多能性细胞,存在于多种组织如骨髓、脐血和脐带组织、胎盘组织、脂肪组织中,具有自我更新和分化为多种特定细胞的潜能,可以诱导分化为成骨细胞、脂肪细胞、成肌细胞、成软骨细胞和神经元等,其中骨髓间充质干细胞具有免疫调节功能,可以避免移植后的免疫排斥反应得到较多研究[23-24]。目前已有多项研究发现,基于石墨烯的纳米材料能够促进间充质干细胞的增殖、黏附及控制间充质干细胞向成骨细胞、脂肪细胞、成软骨细胞和神经元分化[7,25]。 HE等[26]研究证明,磁性氧化石墨烯在低浓度下具有生物活性,可以显著加速人骨髓间充质干细胞的成骨分化,并证明人骨髓间充质干细胞在磁性氧化石墨烯上的成骨分化能力优于石墨烯。DELIORMANLI[27]研究发现,小鼠骨髓间充质干细胞在网格状石墨烯/聚己内酯复合支架上良好附着和增殖,并在缺乏生长因子的情况下表现出软骨分化趋势。 VERRE等[28]报道了氧化石墨烯及还原氧化石墨烯上培养的脂肪间充质干细胞能分化为类许旺细胞,这种类许旺细胞能表达胶质纤维酸性蛋白、S100和p75等胶质细胞标志物,从而推测氧化石墨烯及还原氧化石墨烯可以促进脂肪间充质干细胞向神经胶质细胞分化。 SEONWOO等[29]通过静电纺丝技术制备了还原氧化石墨烯-聚己内酯复合电纺丝,并将其用于培养人牙髓干细胞,证实了还原氧化石墨烯-聚己内酯复合电纺丝能促进人牙髓干细胞向神经分化的潜能。 2.2.4 神经干细胞 神经干细胞是存在于神经系统中的多能干细胞,具有分化为神经元、星形胶质细胞和少突胶质细胞的能力[30]。众所周知,神经是一种对电刺激作出反应的电活动组织,电场可以加强干细胞分化过程中的神经发生[31-32]。近年来,利用具有电导率性质的纳米材料来传输电场发出的电信号已经得到了广泛应用。 KIM等[33]将氧化石墨烯纳米颗粒添加于人胚胎神经干细胞培养基,发现与未添加纳米颗粒相比,人胚胎神经干细胞表现出更强的自我更新能力和更快的分化速度。 FU等[17]将神经干细胞置于导电聚乳酸-羟基乙酸/氧化石墨烯纳米复合膜上培养,并联合电刺激,发现神经干细胞能向神经元分化,同时发现神经干细胞纳米复合膜和电刺激单独都能促进神经干细胞向神经元分化,但是联合电刺激的神经干细胞纳米复合膜上培养的神经干细胞相较于单独氧化石墨烯膜上培养的神经干细胞更少地分化为星形胶质细胞。 TANG等[34]对神经干细胞在石墨烯基质上分化的转录组进行了分析,发现石墨烯基质上培养的神经干细胞可以分化为神经元和星形胶质细胞,同时通过比较聚苯乙烯上培养的神经干细胞中神经元标记物Tuj-1的表达,发现石墨烯基质上分化Tuj-1阳性细胞数量远高于聚苯乙烯组,由此证明石墨烯基质促进神经干细胞向神经元分化。 2.2.5 肿瘤干细胞 肿瘤干细胞是肿瘤中具有自我更新能力并能产生异质性肿瘤细胞的一类干细胞[35]。残余肿瘤干细胞的存活被认为是驱动肿瘤复发、远处转移和耐药的原因,很难用传统的癌症治疗方法如化疗和放疗来根除[36]。因此,促进肿瘤干细胞的分化可能是一种有效治疗肿瘤的新途径。 FIORILLO等[37]研究发现,氧化石墨烯可以有效抑制6种不同类型癌症(乳腺癌、卵巢癌、前列腺癌、肺癌、胰腺癌和胶质母细胞瘤)的多种细胞系中肿瘤球的形成,意味着氧化石墨烯可以抑制这些肿瘤干细胞的克隆扩增,而且氧化石墨烯可以通过几个关键的信号通路(Wnt、Notch和实体信号转导)促进肿瘤干细胞向肿瘤细胞的分化。 CHOI等[18]用r-藻红蛋白分子介导合成还原氧化石墨烯-银纳米复合材料,将之用于孵育人卵巢癌细胞和卵巢癌干细胞,发现r-藻红蛋白分子介导合成还原氧化石墨烯-银纳米复合材料通过介导活性氧的产生、线粒体膜电位的降低、凋亡基因的表达增强等途径降低细胞活力,可能引起细胞凋亡。同时发现,r-藻红蛋白分子介导合成还原氧化石墨烯-银纳米复合材料联合诱导癌细胞凋亡水平是单独使用沙利霉素的5倍。 基于石墨烯的纳米材料对干细胞生物行为的影响,见表2。"


2.3 潜在调控机制的研究 基于石墨烯的纳米材料在干细胞的黏附、增殖、生长和分化上都取得了不同进展,目前关于其调控机制方面也有了不同的猜想,但许多仍未得到准确证实。 YANG等[20]研究发现,氧化石墨烯有显著的剂量依赖性,更高剂量的氧化石墨烯对增强细胞黏附、增殖和分化具有更高的促进作用,可能是由于氧化石墨烯表面具有大量含氧基团、亲水性更强。而且氧化石墨烯能上调多巴胺神经元标记基因TH的表达来促进胚胎干细胞向多巴胺神经元分化。ZHAO等[15]研究发现,还原氧化石墨烯不仅能深化E-钙黏蛋白转导和信息交互,还能激活β-钙黏蛋白的表达,通过DKK1修饰还原氧化石墨烯底物并构建转染E-钙黏蛋白的细胞系,首次证明还原氧化石墨烯通过调节E-钙黏蛋白/Wnt信号通路保持胚胎干细胞的多能性,同时保持胚胎干细胞的自我更新能力和多能性。 WEI等[38]提出,氧化石墨烯纳米薄片可以通过激活Wnt/B-catenin信号通路促进间充质干细胞成骨分化。SHUAI等[39]研究发现,家蚕丝素蛋白/氧化石墨烯10通过驱动F-肌动蛋白组装增加细胞扩散面积,具有较强的促进间充质干细胞黏附的能力。 TANG等[34]通过对神经干细胞在石墨烯上分化的转录基因分析发现,石墨烯上调神经干细胞中Cfap44、Dnah5、Dnah11、Ccdc108这些运动蛋白基因来促进神经干细胞向神经元分化。其中Dnah11是dynein重链家族的一分子,具有诱导迁移和神经突生长的能力。 2.4 基于石墨烯的纳米材料的生物毒性 石墨烯作为一种化学物质对生物具有天然毒性,基于其开发的纳米材料在用于生物医学应用时必须进行体外细胞毒性和体内生物相容性评估[40]。该文主要讨论基于石墨烯的纳米材料在干细胞领域的应用,较少涉及生物体内研究,故现在只讨论其体外细胞毒性。 2.4.1 毒性机制 石墨烯与细胞的相互作用受各种物理化学性质(例如形状、大小、官能团密度和电荷转移能力)的影响,其毒性主要与细胞内活性氧损伤有关[41-42]。研究阐明了石墨烯介导的活性氧损伤的两种机制:氧化石墨烯内化后干扰电子传输系统,诱导H2O2和羟基自由基的过度产生,触发cytc的释放,从而诱导钙从内质网释放并激活caspase3和7导致细胞死亡[43];氧化石墨烯诱导MAPK和TGF-β信号通路的激活,从而导致Bcl-2蛋白激活,激活线粒体诱导的细胞凋亡[44]。除了活性氧损伤机制外,氧化石墨烯可以激活toll样受体并通过炎症途径诱导自噬[45]。 同时,数项研究报道了石墨烯材料尖锐的边缘通过直接接触细胞膜使细胞膜失去稳定性,以破坏细胞完整性[43,46]。TALUKDAR等[47]研究发现,单层氧化石墨烯纳米带由于细胞与纳米带锋利边缘之间相互作用表现出比氧化石墨烯纳米片和石墨烯纳米球更大的细胞和基因毒性,可引起DNA断裂和染色体畸变。LI等[48]研究表明,石墨烯微片通过在角点和边缘凹凸处的自发膜穿透进入细胞。TU等[49]发现石墨烯与脂质之间存在强烈的相互作用,可以渗透细胞膜并从中提取出大量磷脂,进而导致细胞毒性。 2.4.2 生物毒性的主要影响因素 尺寸大小:CHANG等[50]研究了各种尺寸的氧化石墨烯[(160±90),(430±300),(780±410 ) nm]对A549(人肺腺癌)细胞的毒性大小,发现尺寸为(780±410) nm的氧化石墨烯比(160± 90) nm和(430±300) nm的氧化石墨烯生成的活性氧高50%以上,表明氧化石墨烯的细胞活力和活性氧生成潜力取决于石墨烯片的尺寸。YUE等[51]的报告也指出,氧化石墨烯的细胞内化和细胞应答的调节直接取决于其尺寸大小,微米大小的氧化石墨烯可引起更强的炎症反应和细胞因子释放。 剂量、时间和形态:ZHANG等[52]研究了石墨烯与大鼠嗜铬细胞瘤PC12细胞的相互作用,发现0.01-10 mg/L石墨烯中没有细胞死亡,而100 mg/L石墨烯处理后可观察到近15%-20%的细胞死亡,与此同时观察到100 mg/L单壁碳纳米管处理后有超过70%的细胞死亡,表明纳米材料的细胞毒性取决于材料的形态与剂量。LV等[53]研究发现,人神经母细胞瘤SH-SY5Y细胞在低质量浓度(<80 mg/L)氧化石墨烯条件下不会发生细胞凋亡,反而增强了视黄酸诱导的SH-SY5Y细胞分化,增加神经突长度和MAP2(神经标记)的表达。 功能化基团:SASIDHARAN等[54]将猴肾细胞用原始石墨烯和羧化氧化石墨烯处理,发现原始的石墨烯积聚在细胞膜上引起氧化应激,导致细胞凋亡,而羧基官能化的亲水性石墨烯被细胞内化而不引起任何毒性[55]。他的另一个研究也发现,只有4%经功能化石墨烯处理的猴肾细胞在功能化质量浓度高达75 mg/L时表现出活性氧生成,但没有表现出毒性作用,表明原始石墨烯的表面功能化可以防止其大部分毒性。 表面活性剂:WOJTONISZAK等[56]研究了3种不同的表面活性剂(聚乙二醇、聚乙二醇-聚丙二醇-聚乙二醇和脱氧胆酸钠)中分散的氧化石墨烯和还原氧化石墨烯对小鼠成纤维细胞的细胞毒性,发现分散在聚乙二醇中的氧化石墨烯表现出最佳的细胞相容性。HU等[57]用胎牛血清包被氧化石墨烯处理A549细胞,发现胎牛血清涂层的氧化石墨烯不会引起任何膜损伤,氧化石墨烯的细胞毒性可以通过胎牛血清覆盖来减少。 "

| [1] HALIM A, LUO Q, JU Y, et al. A Mini Review Focused on the Recent Applications of Graphene Oxide in Stem Cell Growth and Differentiation. Nanomaterials (Basel). 2018;8(9):736. [2] NOVOSELOV KS, GEIM AK, MOROZOV SV, et al. Electric field effect in atomically thin carbon films. Science. 2004;306(5696):666-669. [3] SANCHEZ VC, JACHAK A, HURT RH, et al. Biological interactions of graphene-family nanomaterials: an interdisciplinary review. Chem Res Toxicol. 2012;25(1):15-34. [4] JASTRZĘBSKA AM, KURTYCZ P, OLSZYNA AR. Recent advances in graphene family materials toxicity investigations. J Nanopart Res. 2012;14(12):1320. [5] YAN L, ZHENG YB, ZHAO F, et al. Chemistry and physics of a single atomic layer: strategies and challenges for functionalization of graphene and graphene-based materials. Chem Soc Rev. 2012;41(1):97-114. [6] GEETHA BAI R, MUTHOOSAMY K, MANICKAM S, et al. Graphene-based 3D scaffolds in tissue engineering: fabrication, applications, and future scope in liver tissue engineering. Int J Nanomedicine. 2019;14:5753-5783. [7] 王立宾,祝贺,郝捷,等.干细胞与再生医学研究进展[J].生物工程学报,2015,31(6):871-879. [8] 周琪,任小波,杨旭,等.面向未来的新一轮医疗技术革命——干细胞与再生医学研究战略性先导科技专项进展[J].中国科学院院刊, 2015,30(2):262-271. [9] MENAA F, ABDELGHANI A, MENAA B. Graphene nanomaterials as biocompatible and conductive scaffolds for stem cells: impact for tissue engineering and regenerative medicine. Tissue Eng Regen Med. 2015;9(12):1321-1338. [10] HAN S, SUN J, HE S, et al. The application of graphene-based biomaterials in biomedicine. Am J Transl Res. 2019;11(6):3246-3260. [11] GUO X, MEI N. Assessment of the toxic potential of graphene family nanomaterials. Food Drug Anal. 2014;22(1):105-115. [12] ZHAO D, LI Z, LIU L, et al. Progress of Preparation and Application of Graphene/Carbon Nanotube Composite Materials. Acta Chimica Sinica. 2014;72(2):185. [13] CHEN J, HONG F, ZHANG C, et al. Differentiation and transplantation of human induced pluripotent stem cell-derived otic epithelial progenitors in mouse cochlea. Stem Cell Res Ther. 2018;9(1):230. [14] CHEN GY, PANG DW, HWANG SM, et al. A graphene-based platform for induced pluripotent stem cells culture and differentiation. Biomaterials. 2012;33(2):418-427. [15] ZHAO J, TANG M, CAO J, et al. Structurally Tunable Reduced Graphene Oxide Substrate Maintains Mouse Embryonic Stem Cell Pluripotency. Advanced Science. 2019;6(12):1802136. [16] SUN J, LIANG Q, HAN Q, et al. One-step synthesis of magnetic graphene oxide nanocomposite and its application in magnetic solid phase extraction of heavy metal ions from biological samples. Talanta. 2015;132:557-563. [17] FU C, PAN S, MA Y, et al. Effect of electrical stimulation combined with graphene-oxide-based membranes on neural stem cell proliferation and differentiation. Artif Cells Nanomed Biotechnol. 2019;47(1):1867-1876. [18] CHOI YJ, GURUNATHAN S, KIM JH. Graphene Oxide-Silver Nanocomposite Enhances Cytotoxic and Apoptotic Potential of Salinomycin in Human Ovarian Cancer Stem Cells (OvCSCs): A Novel Approach for Cancer Therapy. Int J Mol Sci. 2018;19(3):710. [19] GARCIA-ALEGRIA E, ILIUT M, STEFANSKA M, et al. Graphene Oxide promotes embryonic stem cell differentiation to haematopoietic lineage. Sci Rep. 2016;6:25917. [20] YANG D, LI T, XU M, et al. Graphene oxide promotes the differentiation of mouse embryonic stem cells to dopamine neurons. Nanomedicine (Lond). 2014;9(16):2445-2455. [21] TAKAHASHI K, YAMANAKA S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell. 2006;126(4):663-676. [22] SABURI E, ISLAMI M, HOSSEINZADEH S, et al. In vitro osteogenic differentiation potential of the human induced pluripotent stem cells augments when grown on Graphene oxide-modified nanofibers. Gene. 2019;696:72-79. [23] PITTENGER MF, MACKAY AM, BECK SC, et al. Multilineage potential of adult human mesenchymal stem cells. Science. 1999 ;284(5411):143-147. [24] LI N, HUA J. Interactions between mesenchymal stem cells and the immune system. Cell Mol Life Sci. 2017;74(13):2345-2360. [25] ZHOU M, LOZANO N, WYCHOWANIEC JK, et al. Graphene oxide: A growth factor delivery carrier to enhance chondrogenic differentiation of human mesenchymal stem cells in 3D hydrogels. Acta Biomater. 2019;96:271-280. [26] HE Y, LI Y, CHEN G, et al. Concentration-dependent cellular behavior and osteogenic differentiation effect induced in bone marrow mesenchymal stem cells treated with magnetic graphene oxide. J Biomed Mater Res A. 2020;108(1):50-60. [27] DELIORMANLI AM. Direct Write Assembly of Graphene/Poly(ε-Caprolactone) Composite Scaffolds and Evaluation of Their Biological Performance Using Mouse Bone Marrow Mesenchymal Stem Cells. Appl Biochem Biotechnol. 2019;188(4):1117-1133. [28] VERRE AF, FARONI A, ILIUT M, et al. Improving the glial differentiation of human Schwann-like adipose-derived stem cells with graphene oxide substrates. Interface Focus. 2018;8(3):20180002. [29] SEONWOO H, JANG KJ, LEE D, et al. Neurogenic Differentiation of Human Dental Pulp Stem Cells on Graphene-Polycaprolactone Hybrid Nanofibers. Nanomaterials (Basel). 2018;8(7):554. [30] BÁTIZ LF, CASTRO MA, BURGOS PV, et al. Exosomes as Novel Regulators of Adult Neurogenic Niches. Front Cell Neurosci. 2016;9:501. [31] CHANG KA, KIM JW, KIM JA, et al. Biphasic electrical currents stimulation promotes both proliferation and differentiation of fetal neural stem cells. PLoS One. 2011;6(4):e18738. [32] ZHAO H, STEIGER A, NOHNER M, et al. Specific Intensity Direct Current (DC) Electric Field Improves Neural Stem Cell Migration and Enhances Differentiation towards βIII-Tubulin+ Neurons. PLoS One. 2015;10(6):e0129625. [33] KIM J, YANG K, LEE JS, et al. Enhanced Self-Renewal and Accelerated Differentiation of Human Fetal Neural Stem Cells Using Graphene Oxide Nanoparticles. Macromol Biosci. 2017;17(8). doi: 10.1002/mabi.201600540. [34] TANG M, LI J, HE L, et al. Transcriptomic profiling of neural stem cell differentiation on graphene substrates. Colloids Surf B Biointerfaces. 2019;182:110324. [35] TROTT KR. Tumour stem cells: the biological concept and its application in cancer treatment. Radiother Oncol. 1994;30(1):1-5. [36] PRIETO-VILA M, TAKAHASHI R, USUBA W, et al. Drug Resistance Driven by Cancer Stem Cells and Their Niche. Int J Mol Sci. 2017;18(12):2574. [37] FIORILLO M, VERRE AF, ILIUT M, et al. Graphene oxide selectively targets cancer stem cells, across multiple tumor types: implications for non-toxic cancer treatment, via “differentiation-based nano-therapy”. Oncotarget. 2015;6(6):3553-3562. [38] WEI C, LIU Z, JIANG F, et al. Cellular behaviours of bone marrow-derived mesenchymal stem cells towards pristine graphene oxide nanosheets. Cell Prolif. 2017;50(5):e12367. [39] SHUAI Y, MAO C, YANG M. Protein Nanofibril Assemblies Templated by Graphene Oxide Nanosheets Accelerate Early Cell Adhesion and Induce Osteogenic Differentiation of Human Mesenchymal Stem Cells. ACS Appl Mater Interfaces. 2018;10(38):31988-31997. [40] NEZAKATI T, COUSINS BG, SEIFALIAN AM. Toxicology of chemically modified graphene-based materials for medical application. Arch Toxicol. 2014;88(11):1987-2012. [41] LALWANI G, D’AGATI M, KHAN AM, et al. Toxicology of graphene-based nanomaterials. Adv Drug Deliv Rev. 2016;105(Pt B):109-144. [42] OU L, SONG B, LIANG H, et al. Toxicity of graphene-family nanoparticles: a general review of the origins and mechanisms. Part Fibre Toxicol. 2016;13(1):57. [43] LIAO C, LI Y, TJONG SC. Graphene Nanomaterials: Synthesis, Biocompatibility, and Cytotoxicity. Int J Mol Sci. 2018;19(11):3564. [44] LI Y, LIU Y, FU Y, et al. The triggering of apoptosis in macrophages by pristine graphene through the MAPK and TGF-beta signaling pathways. Biomaterials. 2012;33(2):402-411. [45] CHEN G, YANG H, LU C, et al. Simultaneous induction of autophagy and toll-like receptor signaling pathways by graphene oxide. Biomaterials. 2012;33(27):6559-6569. [46] PATTAMMATTEL A, PANDE P, KUTTAPPAN D, et al. Controlling the Graphene-Bio Interface: Dispersions in Animal Sera for Enhanced Stability and Reduced Toxicity. Langmuir. 2017;33(49):14184-14194. [47] TALUKDAR Y, RASHKOW J, LALWANI G, et al. The effects of graphene nanostructures on mesenchymal stem cells. Biomaterials. 2014;35(18): 4863-4877. [48] LI Y, YUAN H, VON DEM BUSSCHE A, et al. Graphene microsheets enter cells through spontaneous membrane penetration at edge asperities and corner sites. Proc Natl Acad Sci U S A. 2013;110(30):12295-12300. [49] TU Y, LV M, XIU P, et al. Destructive extraction of phospholipids from Escherichia coli membranes by graphene nanosheets. Nat Nanotechnol. 2013;8(8):594-601. [50] CHANG Y, YANG S, LIU J, et al. In vitro toxicity evaluation of graphene oxide on A549 cells. Toxicol Lett. 2011;200(3):201-210. [51] YUE H, WEI W, YUE Z, et al. The role of the lateral dimension of graphene oxide in the regulation of cellular responses. Biomaterials. 2012;33(16):4013-4021. [52] ZHANG Y, ALI SF, DERVISHI E, et al. Cytotoxicity effects of graphene and single-wall carbon nanotubes in neural phaeochromocytoma-derived PC12 cells. ACS Nano. 2010;4(6):3181-3186. [53] LV M, ZHANG Y, LIANG L, et al. Effect of graphene oxide on undifferentiated and retinoic acid-differentiated SH-SY5Y cells line. Nanoscale. 2012;4(13):3861-3866. [54] SaSIDHARAN A, PANCHAKARLA LS, SADANANDAN AR, et al. Hemocompatibility and macrophage response of pristine and functionalized graphene. Small. 2012;8(8):1251-1263. [55] SASIDHARAN A, PANCHAKARLA LS, CHANDRAN P, et al. Differential nano-bio interactions and toxicity effects of pristine versus functionalized graphene. Nanoscale. 2011;3(6):2461-2464. [56] WOJTONISZAK M, CHEN X, KALENCZUK RJ, et al. Synthesis, dispersion, and cytocompatibility of graphene oxide and reduced graphene oxide. Colloids Surf B Biointerfaces. 2012;89:79-85. [57] HU W, PENG C, LV M, et al. Protein corona-mediated mitigation of cytotoxicity of graphene oxide. ACS Nano. 2011;5(5):3693-3700. |
| [1] | Yao Xiaoling, Peng Jiancheng, Xu Yuerong, Yang Zhidong, Zhang Shuncong. Variable-angle zero-notch anterior interbody fusion system in the treatment of cervical spondylotic myelopathy: 30-month follow-up [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1377-1382. |
| [2] | Kong Yamin, Yan Juntao, Ma Bingxiang, Li Huawei. Massage vibration intervenes with MyoD expression and proliferation and differentiation of muscle satellite cells in rats with sciatic nerve injury [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1160-1166. |
| [3] | Wu Cong, Jia Quanzhong, Liu Lun. Relationship between transforming growth factor beta1 expression and chondrocyte migration in adult articular cartilage after fragmentation [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1167-1172. |
| [4] | Wang Baojuan, Zheng Shuguang, Zhang Qi, Li Tianyang. Miao medicine fumigation can delay extracellular matrix destruction in a rabbit model of knee osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1180-1186. |
| [5] | Wang Qin, Shen Cheng, Liao Jing, Yang Ye. Dapagliflozin improves renal injury in diabetic nephropathy rats [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1216-1222. |
| [6] | Xiao Yang, Gong Liqiong, Fei Jing, Li Leiji. Effect of electroacupuncture on nerve growth factor and its receptor expression in facial nerve nucleus after facial nerve injury in rabbits [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1253-1259. |
| [7] | Wang Jing, Xiong Shan, Cao Jin, Feng Linwei, Wang Xin. Role and mechanism of interleukin-3 in bone metabolism [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1260-1265. |
| [8] | Xiao Hao, Liu Jing, Zhou Jun. Research progress of pulsed electromagnetic field in the treatment of postmenopausal osteoporosis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1266-1271. |
| [9] | An Weizheng, He Xiao, Ren Shuai, Liu Jianyu. Potential of muscle-derived stem cells in peripheral nerve regeneration [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1130-1136. |
| [10] | Fan Yiming, Liu Fangyu, Zhang Hongyu, Li Shuai, Wang Yansong. Serial questions about endogenous neural stem cell response in the ependymal zone after spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1137-1142. |
| [11] | Liang Xuezhen, Yang Xi, Li Jiacheng, Luo Di, Xu Bo, Li Gang. Bushen Huoxue capsule regulates osteogenic and adipogenic differentiation of rat bone marrow mesenchymal stem cells via Hedgehog signaling pathway [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1020-1026. |
| [12] | Wang Jifang, Bao Zhen, Qiao Yahong. miR-206 regulates EVI1 gene expression and cell biological behavior in stem cells of small cell lung cancer [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1027-1031. |
| [13] | Liu Feng, Peng Yuhuan, Luo Liangping, Wu Benqing. Plant-derived basic fibroblast growth factor maintains the growth and differentiation of human embryonic stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1032-1037. |
| [14] | Wen Dandan, Li Qiang, Shen Caiqi, Ji Zhe, Jin Peisheng. Nocardia rubra cell wall skeleton for extemal use improves the viability of adipogenic mesenchymal stem cells and promotes diabetes wound repair [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1038-1044. |
| [15] | Zhu Bingbing, Deng Jianghua, Chen Jingjing, Mu Xiaoling. Interleukin-8 receptor enhances the migration and adhesion of umbilical cord mesenchymal stem cells to injured endothelium [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1045-1050. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||