Chinese Journal of Tissue Engineering Research ›› 2019, Vol. 23 ›› Issue (30): 4868-4874.doi: 10.3969/j.issn.2095-4344.1407
Previous Articles Next Articles
Pulsed electromagnetic fields and platelet rich plasma therapy for treatment of osteoarthritis: theoretical progress and clinical research
- 1 West China School of Medicine, Sichuan University, Chengdu 610041, Sichuan Province, China; 2Rehabilitation Medicine Center, West China Hospital, Sichuan University, Chengdu 610041, Sichuan Province, China
-
Received:2019-05-11Online:2019-10-28Published:2019-10-28 -
Contact:He Hongchen, MD, Rehabilitation Medicine Center, West China Hospital, Sichuan University, Chengdu 610041, Sichuan Province, China -
About author:Xiang Xiaona, Master candidate, West China School of Medicine, Sichuan University, Chengdu 610041, Sichuan Province, China; Rehabilitation Medicine Center, West China Hospital, Sichuan University, Chengdu 610041, Sichuan Province, China -
Supported by:Applied Basic Research Foundation of Sichuan Science and Technology Department, No. 2017JY0342 (to HHC); Sichuan University Young Teachers Scientific Research Startup Fund, No. 20826041A4195 (to HHC)
CLC Number:
Cite this article
Xiang Xiaona, Yu Xi, Liu Yan, Jiang Hongying, He Chengqi, He Hongchen. Pulsed electromagnetic fields and platelet rich plasma therapy for treatment of osteoarthritis: theoretical progress and clinical research[J]. Chinese Journal of Tissue Engineering Research, 2019, 23(30): 4868-4874.
share this article
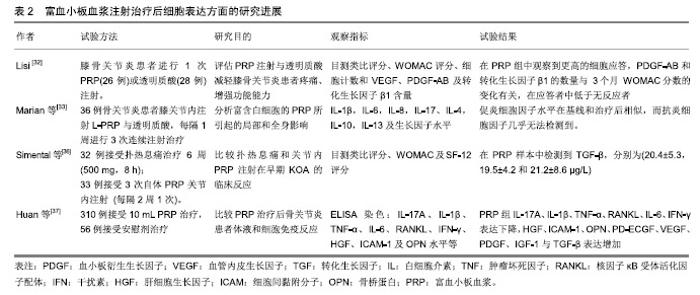
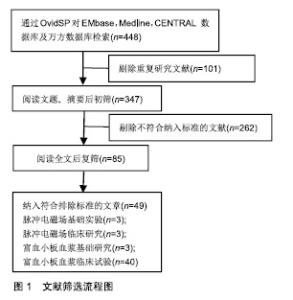
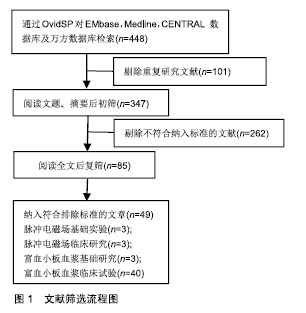
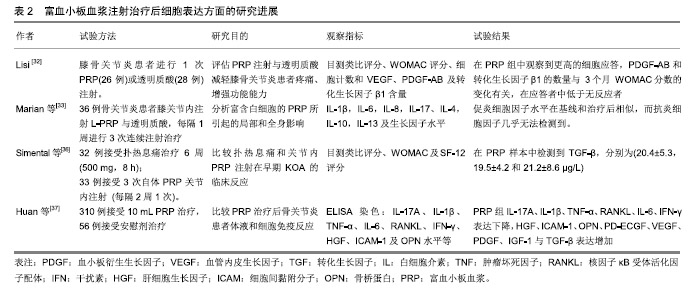
2.2 临床应用研究 目前缺乏脉冲电磁场联合富血小板血浆治疗骨关节炎的临床研究,但两者单独治疗骨关节炎多数研究认为有效。 脉冲电磁场治疗骨关节炎临床研究:脉冲电磁场治疗骨关节炎的临床试验数量较少,火焱等[14]使用30次强度为2.5-3.5 mT及3.5-4.2 mT、8档频率的脉冲电磁场治疗后,观察到骨关节炎患者疼痛缓解、西大略湖与麦克马斯特大学骨关节炎(Western Ontario McMaster Universities,WOMAC)评分较治疗前有明显改善。Bagnato等[15]使用可穿戴脉冲电磁场仪器4周,发现26%患者停止非类固醇抗炎药与镇痛药使用、关节炎症状缓解20%以上。但有研究使用50 Hz,105 μT的脉冲电磁场与经皮电刺激、热疗对比,发现两组认为疼痛改善无差别[16]。作者认为应用脉冲电磁场治疗骨关节炎有一定疗效,但治疗结果与选择的参数有关。 富血小板血浆治疗骨关节炎临床研究:Yu等[17]每隔1周在10例膝骨关节炎女性患膝关节中注射1次6 mL无白细胞富血小板血浆,注射后会出现轻微不良事件,48 h内症状得到缓解。在6个月随访中,80%的目测类比评分下降超过50%。Smith等[18]发现12个月后,少白细胞富血小板血浆组的WOMAC评分比基线提高78%,而安慰剂组为7%。Rahimzadeh等[19]及Jubert等[20]认为富血小板血浆治疗对骨关节炎患者WOMAC评分、目测类比评分、生活质量评价量表(SF-36)均有益处。王一帆等[21]发现富血小板血浆的血小板浓度在(900-1 800)×109 L-1时,临床症状改善程度同血小板浓度呈一定的剂量依赖关系。 与透明质酸、皮质类固醇以及运动疗法相比,富血小板血浆更有优势。Nabi等[22]发现注射治疗6个月后富血小板血浆组疼痛评分仍低于皮质类固醇组。有研究发现富血小板血浆治疗在6,12个月后疗效优于透明质酸注射[23-26]。Lana等[27]认为富血小板血浆联合透明质酸治疗效果更好。 但Di Sante等[28]使用3 mL的富血小板血浆与透明质酸(30 mg/2 mL),发现透明质酸对疼痛缓效果更佳持 久。Akan等[29]发现接受富血小板血浆治疗的患者中,WOMAC疼痛评分减少30% 、身体功能增加24%。Montanez-Heredia等[30]发现注射富血小板血浆3月后,生活质量高于透明质酸注射。Rayegani等[31]发现富血小板血浆治疗与运动疗法相比,更能改善运动表现。 影像学上可观察到富血小板血浆治疗后关节的好转,也细胞表达中可以得到体现,见表2。Lisi等[32]通过核磁共振发现富血小板血浆治疗后48.3%患者至少有膝骨关节炎Kellgren-Lawrence(K-L)分级1级改善,而对照组(透明质酸)为8%。Hassan等[33]进行6个月、每月1次的5 mL富血小板血浆治疗后滑膜增厚有显著改善。Louis等[34]注射1次富血小板血浆后发现在富血小板血浆组中血小板源性生长因子AB(platelet derived growth factor-AB,PDGF-AB)和转化生长因子β1的数量与WOMAC分数的变化有关,在应答者中低于无反应者。有趣的是,Mariani等[35]对36例骨关节炎患者膝关节内注射富白细胞富血小板血浆与透明质酸,在注射前抽取 1 mL滑液,同时,获得外周血样本,并分离血浆;在第2,6,12个月时再采集外周血样本,炎细胞因子水平在基线和治疗后相似,而抗炎细胞因子几乎无法检测 到。作者认为临床应用富血小板血浆治疗骨关节炎时以少或无白细胞富血小板血浆为主,同时作者发现富血小板血浆治疗后患者炎症因子的表达下降,生长因子表达增加。 2.3 脉冲电磁场、富血小板血浆治疗骨关节炎基础研究 2.3.1 脉冲电磁场治疗骨关节炎基础研究 Parate 等[38]已证实脉冲电磁场具有在软骨中聚集钙转导的作用,Ca2+可能由瞬时受体电位通道(TRPC)包括TRPC1,TRPC6,TRPV1,TRPV4,TRPV6或电压门控钙通道介导。脉冲电磁场增加细胞中Ca2+浓度改变膜电位,可能引起细胞死亡,也可能在骨形成点沉积,加速骨与软骨的形成。 2.3.2 富血小板血浆治疗骨关节炎基础研究 富血小板血浆中的炎症调节因子可以使软骨细胞核因子κB通路激活的炎症反应抑制,从而抑制白细胞介素1介导的无菌性炎症因子的表达和软骨基质的降解,从而同血管内皮生长因子、血小板衍生生长因子、转化生长因子β及类胰岛素生长因子等促进合成代谢的因子一起协同作用[39]。 2.4 脉冲电磁场联合富血小板血浆治疗骨关节炎可能机制 2.4.1 共同抑制白细胞介素1及肿瘤坏死因子α等炎症因子表达 Zou等[40]发现脉冲电磁场对细胞形态、髓核细胞表达白细胞介素1β和肿瘤坏死因子α并无影响、对细胞增殖无明显影响,但分泌到培养基中的白细胞介素1β和肿瘤坏死因子α水平以强度依赖性方式显着降低,低频脉冲电磁场刺激可抑制培养基中的髓核细胞分泌白细胞介素1β和肿瘤坏死因子α。脉冲电磁场对活体的髓核细胞可能具有类似效应,有助于炎症治疗,如骨关节炎。Zhu等[41]进行小鼠实验得出敲除肿瘤坏死因子α与白细胞介素6可增加核因子κB受体活化因子配体(RANKL)基因的表达。Wu等[42]进行动物实验,取膝骨关节炎新西兰兔的软骨细胞,分为对照组、白细胞介素1β、富血小板血浆、重组人Dickkopf相关蛋白1、富血小板血浆+DKK-1治疗组,通过白细胞介素1β治疗24 h,用电子显微镜观察软骨形态,发现富血小板血浆、重组人Dickkopf相关蛋白1、富血小板血浆+重组人Dickkopf相关蛋白1治疗组软骨异常较少,白细胞介素1组mRNA、Wnt1及Wnt/β-连环蛋白表达增高,认为富血小板血浆可通过抑制Wnt/β-连环蛋白通路激活白细胞介素1,从而保护软骨。 临床试验与动物实验均表明脉冲电磁场与富血小板血浆治疗骨关节炎患者之后,血液学指标里的炎症因子水平下降。炎症和骨代谢之间的联系已在不同的骨质疏松症动物模型中得到进一步证实,其中关键炎性细胞因子受体,如白细胞介素1和肿瘤坏死因子α,显示骨质减少;相反,骨折部位需要一定程度的炎症才能使骨折愈合,过量使用抗炎药会抑制骨折的巩固。白细胞介素1通过与靶细胞上的受体结合而发挥作用, 而骨关节炎患者软骨细胞表面的受体数量是正常细胞表面的2倍,使骨关节炎患者的软骨和滑膜细胞对白细胞介素1具有高度敏感性,从而刺激软骨细胞和滑膜产生基质金属蛋白酶等,促进软骨基质的降解;刺激软骨细胞和滑膜产生前列腺素E2参与促进成骨细胞样细胞的增殖和骨吸收,参与骨质增生和软骨下骨囊性变的形成;介导软骨细胞分化,调节软骨细胞表型变成成纤维细胞表型;抑制软骨细胞的增殖。白细胞介素1对骨关节炎的影响是全方位的,从改变软骨细胞正常结构和和促进软骨细胞凋亡发生、降解软骨细胞基质、参与滑膜无菌性炎性病变及影响骨代谢等方面发挥作用。 2.4.2 通过促进Wnt/β-连环蛋白信号通路协同促进血管内皮生长因子分泌 Yang等[43]使用75 Hz、1.6 mT的PEFMs进行小鼠试验发现骨小梁厚度、数量及骨体积分数有增加、促进Wnt3a,Wnt β-连环蛋白与骨桥蛋白的表达。Zhou等[44]使用75 Hz,3.8 mT的脉冲电磁场得出类似结论。Wu等[42]发现Wnt/β-连环蛋白在富血小板血浆治疗时表达增高。Wnt/β-连环蛋白通路主要参与细胞增殖、分化及凋亡等,Wnt/β-连环蛋白信号通路已被证明通过直接影响骨,软骨和滑膜组织在骨和关节病理学中发挥作用。 近年来研究表明,在骨形成方面,抑制间充质干细细胞分化为脂肪细胞,并促进其分化为成骨细胞;促进成骨细胞增殖、成熟;抑制成骨细胞的凋亡。在骨吸收方面,促进成骨细胞分泌骨桥蛋白,同时抑制成骨细胞分泌RANKL,从而减少RANKL/骨桥蛋白的比率,抑制骨吸收[45]。成熟软骨细胞中的Wnt通路同时诱导基质金属蛋白酶13和血管内皮生长因子的表达,调节无菌性炎症反应、避免关节软骨免受炎症侵害的同时,可刺激软骨细胞和干细胞的增殖,促进细胞分泌Ⅱ型胶原和蛋白多糖,抑制细胞凋亡,促进干细胞的迁移和向软骨方向分化,抑制软骨和基质的降解,促进关节软骨在一定程度上进行修复,从而改善骨关节炎症状。新出现的证据表明,Wnts和Wnt相关分子参与人类基因研究和体外研究中的关节炎发展和进展。抑制骨细胞的凋亡,激活下游靶基因,调节骨代谢,但目前骨细胞内Wnt信号通路激活对其他细胞的调控机制有待进一步深入研究。激活的Wnt/β-连环蛋白信号通路可减少骨吸收,同时增加骨形成,Wnt/β-连环蛋白信号传导的介质和下游效应子在骨关节炎以及其他形式的关节炎中增加[46]。通过转基因小鼠的临床前研究以及转化和人体研究,在人类遗传学研究中,已经提出与Wnt/β-连环蛋白信号传导相关的基因的改变作为骨关节炎发展的易感因子。sFRP3是Wnt信号传导的拮抗剂,由sFrp3基因编码。sFrp3中的单核苷酸多态性导致在羧基末端的Arg324Gly取代,并且与关节负重区域中骨关节炎的风险增加相关[47]。 因此作者认为白细胞介素1及肿瘤坏死因子α等炎症因子表达的抑制、Wnt/β-连环蛋白表达的促进在脉冲电磁场与富血小板血浆的有效治疗中发挥了重要作用,但两者是否存在相互作用目前尚不清楚。 2.4.3 Toll样受体 是一类具有模式识别受体功能的大家族,引发炎症反应是其最突出的生物学功能。Toll样受体活化后,可通过一系列的生化反应,诱导许多促炎症因子的合成与释放,如肿瘤坏死因子α、白细胞介素1,6,8、基质金属蛋白酶1,3,9,13等。脉冲电磁场与富血小板血浆治疗能抑制炎症因子释放或许是抑制了Toll样受体活化。同时,Toll样受体4在骨髓单核细胞的细胞分化中起关键作用。在第一阶段,单独的Toll样受体4活化促进这些前体细胞转化为巨噬细胞,并阻断破骨细胞生成。然而,成骨细胞可以将这种途径转变为破骨细胞的形成。成骨细胞的Toll样受体4活化诱导核因子κB配体和巨噬细胞集落刺激因子的受体活化剂的产生。在聚集阶段存在这些因子可防止巨噬细胞成熟并驱使破骨细胞形成。相反,在发展阶段,Toll样受体4激活会触发细胞活化。因此,由Toll样受体4介导的破骨细胞形成取决于在聚集阶段期间成骨细胞衍生的RANKL的存在或不存在[48]。由此可见由Toll样受体介导骨髓细胞分化能影响骨代谢,与Wnt/β-连环蛋白的表达可能相关。但有研究表明,膝关节骨关节炎中Toll样受体4的蛋白质水平显着高于掌指骨关节炎,ELISA测定证实滑液中存在可溶形式的Toll样受体2,4[49]。 "
| [1]Bruyère O, Cooper C, Arden N, et al. Can we identify patients with high risk of osteoarthritis progression who will respond to treatment? A focus on epidemiology and phenotype of osteoarthritis. Drugs Aging. 2015;32(3):179-187. [2]中华医学会骨科学分会关节外科学组.骨关节炎诊疗指南(2018年版)[J].中华骨科杂志,2018,38(12):705-715. [3]薛庆云,王坤正,裴福兴,等.中国40岁以上人群原发性骨关节炎患病状况调查[J].中华骨科杂志,2015,35(12):1206-1212. [4]Allen KD, Golightly YM. State of the evidence. Curr Opin Rheumatol. 2015;27(3):276-83.[5]Turkiewicz A, Gerhardsson de Verdier M, Engström G, et al. Prevalence of knee pain and knee OA in southern Sweden and the proportion that seeks medical care. Rheum. 2015; 54(5):827-835. [6]Driban JB, Eaton CB, Amin M, et al. Glucose homeostasis influences the risk of incident knee osteoarthritis: Data from the osteoarthritis initiative. J Orthop Res. 2017;35(10): 2282-2287.[7]Chen A, Gupte C, Akhtar K, et al. The global economic cost of osteoarthritis: how the UK compares. Arthritis. 2012;2012: 698709.[8]Kotlarz H, Gunnarsson CL, Fang H, et al. Insurer and out-of-pocket costs of osteoarthritis in the US: evidence from national survey data. Arthritis Rheum. 2009;6(12):3546-53. doi: 10.1002/art.24984.[9]Rosen AD, Chastney EE. Effect of long term exposure to 0.5 T static magnetic fields on growth and size of GH3 cells. Bioelectromagnetics. 2009;30(2):114-119.[10]Sullivan K, Balin AK, Allen RG. Effects of static magnetic fields on the growth of various types of human cells. Bioelectromagnetics. 2011;32(2):140-147.[11]Galli C, Pedrazzi G, Mattioli-Belmonte M, Guizzardi S. The use of pulsed electromagnetic fields to promote bone responses to biomaterials in vitro and in vivo. Int J Biomater. 2018;2018:8935750. [12]Salamanna F, Veronesi F, Maglio M, et al. New and emerging strategies in platelet-rich plasma application in musculoskeletal regenerative procedures: general overview on still open questions and outlook. Biomed Res Int. 2015; 2015:846045.[13]Brossi PM, Moreira JJ, Machado TS, et al. Platelet-rich plasma in orthopedic therapy: a comparative systematic review of clinical and experimental data in equine and human musculoskeletal lesions. BMC Vet Res. 2015;11:98. [14]火焱,陈德志,王昭玲,等.低频脉冲电磁场治疗骨关节炎的有效性及安全性观察[J].中国医药科学,2013,3(13):9-11.[15]Bagnato GL, Miceli G, Marino N, et al. Pulsed electromagnetic fields in knee osteoarthritis: A double blind, placebo-controlled, randomized clinical trial. Rheumatology (United Kingdom). 2016;55(4):755-762.[16]Ay S, Evcik D. The effects of pulsed electromagnetic fields in the treatment of knee osteoarthritis: a randomized, placebo-controlled trial. Rheumatol Int. 2009;29(6):663-666.[17]Taniguchi Y, Yoshioka T, Kanamori A, et al. Intra-articular platelet-rich plasma (PRP) injections for treating knee pain associated with osteoarthritis of the knee in the Japanese population: a phase I and IIa clinical trial. Nagoya J Med Sci. 2018;80(1):39-51.[18]Smith PA. Intra-articular autologous conditioned plasma injections provide safe and efficacious treatment for knee osteoarthritis: an FDA-sanctioned, randomized, double-blind, placebo-controlled clinical trial. Am J Sports Med. 2016; 44(4):884-891.[19]Rahimzadeh P, Imani F, Faiz SHR, et al. The effects of injecting intra-articular platelet-rich plasma or prolotherapy on pain score and function in knee osteoarthritis. Clin Interv Aging. 2018;13:73-79.[20]Jubert NJ, Rodriguez L, Reverte-Vinaixa MM, et al. Platelet-rich plasma injections for advanced knee osteoarthritis: a prospective, randomized, double-blinded clinical trial. Orthop J Sports Med. 2017;5(2): 2325967116689386. [21]王一帆,李小峰,罗道明,等.不同浓度富血小板血浆治疗膝关节骨性关节炎临床疗效研究[J].陕西医学杂志,2018,47(9): 1113-1118.[22]Nabi BN, Sedighinejad A, Mardani-Kivi M, et al. Comparing the effectiveness of intra-articular platelet-rich plasma and corticosteroid injection under ultrasound guidance on pain control of knee osteoarthritis. Iranian Red Crescent Med J. 2018;20(3):e62157.[23]Raeissadat SA, Rayegani SM, Hassanabadi H, et al. Knee osteoarthritis injection choices: platelet-rich plasma (PRP) versus hyaluronic acid (A one-year randomized clinical trial). Clin Med Insights Arthritis Musculoskelet Disord. 2015;8: 1-8.[24]吕帅洁,厉驹,何斌,等.富血小板血浆关节内注射治疗膝骨关节炎的前瞻性随机对照研究[J].中华创伤杂志,2016,32(7):626-631.[25]翟文亮,周亮,刘晖,等.富血小板血浆与透明质酸钠治疗膝关节骨关节炎的临床观察[J].中华关节外科杂志,2017,11(1):96-100.[26]Ahmad HS, Farrag SE, Okasha AE, et al. Clinical outcomes are associated with changes in ultrasonographic structural appearance after platelet-rich plasma treatment for knee osteoarthritis. Int J Rheum Dis. 2018;21(5):960-966.[27]Lana JFSD, Weglein A, Sampson SE, et al. Randomized controlled trial comparing hyaluronic acid, platelet-rich plasma and the combination of both in the treatment of mild and moderate osteoarthritis of the knee. Int J Rheum Dis. 2016; 12(2):69-78.[28]Di Sante L, Villani C, Santilli V, et al. Intra-articular hyaluronic acid vs platelet-rich plasma in the treatment of hip osteoarthritis. Med Ultrason. 2016;18(4):463-468.[29]Akan O, Sarikaya NO, Kocyigit H. Efficacy of platelet-rich plasma administration in patients with severe knee osteoarthritis: Can platelet-rich plasma administration delay arthroplasty in this patient population? Int J Clin Exp Med. 2018;11(9):9473-9483.[30]Montanez-Heredia E, Irizar S, Huertas PJ, et al. Intra-articular injections of platelet-rich plasma versus hyaluronic acid in the treatment of osteoarthritic knee pain: a randomized clinical trial in the context of the spanish national health care system. Int J Mol Sci. 2016;17(7):E1064.[31]Rayegani SM, Raeissadat SA, Taheri MS, et al. Does intra articular platelet rich plasma injection improve function, pain and quality of life in patients with osteoarthritis of the knee? A randomized clinical trial. Orthopedic Reviews. 2014;6(3): 112-117.[32]Lisi C, Perotti C, Scudeller L, et al. Treatment of knee osteoarthritis: platelet-derived growth factors vs. hyaluronic acid. A randomized controlled trial. Clin Rehabil. 2018;32(3): 330-339.[33]Hassan AS, El-Shafey AM, Ahmed HS, et al. Effectiveness of the intra-articular injection of platelet rich plasma in the treatment of patients with primary knee osteoarthritis. Egyptian Rheumatologist. 2015;37(3):119-124.[34]Louis ML, Magalon J, Jouve E, et al. Growth factors levels determine efficacy of platelets rich plasma injection in knee osteoarthritis: a randomized double blind noninferiority trial compared with viscosupplementation. Arthroscopy. 2018; 34(5):1530-1540.[35]Mariani E, Canella V, Cattini L, et al. Leukocyte-rich Platelet-rich Plasma injections do not up-modulate intra- articular pro-inflammatory cytokines in the osteoarthritic knee. PLoS One. 2016;11(6):e0156137.[36]Simental-Mendia M, Vilchez-Cavazos JF, Pena-Martinez VM, et al. Leukocyte-poor platelet-rich plasma is more effective than the conventional therapy with acetaminophen for the treatment of early knee osteoarthritis. Arch Orthop Trauma Surg. 2016;136(12):1723-1732.[37]Huang G, Hua S, Yang T, et al. Platelet-rich plasma shows beneficial effects for patients with knee osteoarthritis by suppressing inflammatory factors. Exp Ther Med. 2018;15(3): 3096-3102.[38]Parate D, Franco-Obregón A, Fröhlich J, et al. Enhancement of mesenchymal stem cell chondrogenesis with short-term low intensity pulsed electromagnetic fields. Sci Rep. 2017; 7(1):9421. [39]郭燕庆.富血小板血浆(PRP)治疗膝骨关节炎的临床疗效评价[D].济南:山东大学,2017.[40]Zou J, Chen Y, Qian J, et al. Effect of a low-frequency pulsed electromagnetic field on expression and secretion of IL-1β and TNF-α in nucleus pulposus cells. J Int Med Res. 2017; 45(2):462-470.[41]Zhu SZ, He H, Wang Q, et al. Abnormal bone and cartilage metabolism could be antagonised by pulsed electromagnetic fields (PEMFS) and TNF-alpha and IL-6 gene knockouts in a similar mechanism. Annals of the Rheumatic Diseases. 2018;77:897-898.[42]Wu J, Huang JF, Qin XX, et al. Platelet-rich plasma inhibits Wnt/beta-catenin signaling in rabbit cartilage cells activated by IL-1beta. Int Immunopharmacol. 2018;55:282-289.[43]Yang X, He H, Gao Q, et al. Pulsed electromagnetic field improves subchondral bone microstructure in knee osteoarthritis rats through a Wnt/beta-catenin signaling- associated mechanism. Bioelectromagnetics. 2018;39(2): 89-97.[44]周予靖,王朴,陈红英,等.脉冲电磁场对大鼠骨髓间充质干细胞增殖?成骨分化和Wnt/β-catenin信号通路的影响[J].四川大学学报(医学版),2015,46(3):347-353.[45]王谦,王朴,刘小菁,等.脉冲电磁场调控干细胞增殖与分化[J].中国康复医学杂志,2014,29(2):180-183.[46]陶可,熊养,曾晖.Wnt信号通路与骨关节炎关系的研究进展[J].中华风湿病学杂志,2011,15(8):564-568.[47]Zhou Y, Wang T, Hamilton JL, et al. Wnt/β-catenin signaling in osteoarthritis and in other forms of arthritis. Curr Rheumatol Rep. 2017;19(9):53.[48]Alonso-Pérez A, Franco-Trepat E, Guillán-Fresco M, et al. Role of toll-like receptor 4 on osteoblast metabolism and function. Front Physiol. 2018;9:504. [49]Barreto G, Sandelin J, Salem A, et al. Toll-like receptors and their soluble forms differ in the knee and thumb basal osteoarthritic joints. Acta Orthop. 2017;88(3):326-333. |
| [1] | Pu Rui, Chen Ziyang, Yuan Lingyan. Characteristics and effects of exosomes from different cell sources in cardioprotection [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-. |
| [2] | Huang Dengcheng, Wang Zhike, Cao Xuewei. Comparison of the short-term efficacy of extracorporeal shock wave therapy for middle-aged and elderly knee osteoarthritis: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1471-1476. |
| [3] | Peng Zhihao, Feng Zongquan, Zou Yonggen, Niu Guoqing, Wu Feng. Relationship of lower limb force line and the progression of lateral compartment arthritis after unicompartmental knee arthroplasty with mobile bearing [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1368-1374. |
| [4] | Zhang Chao, Lü Xin. Heterotopic ossification after acetabular fracture fixation: risk factors, prevention and treatment progress [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1434-1439. |
| [5] | Zhou Jihui, Li Xinzhi, Zhou You, Huang Wei, Chen Wenyao. Multiple problems in the selection of implants for patellar fracture [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1440-1445. |
| [6] | Wang Debin, Bi Zhenggang. Related problems in anatomy mechanics, injury characteristics, fixed repair and three-dimensional technology application for olecranon fracture-dislocations [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1446-1451. |
| [7] | Ji Zhixiang, Lan Changgong. Polymorphism of urate transporter in gout and its correlation with gout treatment [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1290-1298. |
| [8] | Yuan Mei, Zhang Xinxin, Guo Yisha, Bi Xia. Diagnostic potential of circulating microRNA in vascular cognitive impairment [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1299-1304. |
| [9] | Chen Jiming, Wu Xiaojing, Liu Tianfeng, Chen Haicong, Huang Chengshuo. Effects of silymarin on liver injury and bone metabolism induced by carbon tetrachloride in mice [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1224-1228. |
| [10] | Liu Xiangxiang, Huang Yunmei, Chen Wenlie, Lin Ruhui, Lu Xiaodong, Li Zuanfang, Xu Yaye, Huang Meiya, Li Xihai. Ultrastructural changes of the white zone cells of the meniscus in a rat model of early osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1237-1242. |
| [11] | Wang Xianyao, Guan Yalin, Liu Zhongshan. Strategies for improving the therapeutic efficacy of mesenchymal stem cells in the treatment of nonhealing wounds [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1081-1087. |
| [12] | Wan Ran, Shi Xu, Liu Jingsong, Wang Yansong. Research progress in the treatment of spinal cord injury with mesenchymal stem cell secretome [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1088-1095. |
| [13] | Liao Chengcheng, An Jiaxing, Tan Zhangxue, Wang Qian, Liu Jianguo. Therapeutic target and application prospects of oral squamous cell carcinoma stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1096-1103. |
| [14] | Zhao Min, Feng Liuxiang, Chen Yao, Gu Xia, Wang Pingyi, Li Yimei, Li Wenhua. Exosomes as a disease marker under hypoxic conditions [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1104-1108. |
| [15] | Xie Wenjia, Xia Tianjiao, Zhou Qingyun, Liu Yujia, Gu Xiaoping. Role of microglia-mediated neuronal injury in neurodegenerative diseases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1109-1115. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||