Chinese Journal of Tissue Engineering Research ›› 2018, Vol. 22 ›› Issue (12): 1950-1955.doi: 10.3969/j.issn.2095-4344.0830
Previous Articles Next Articles
Exercise training for bone and lipid metabolisms: exercise intensity and duration are important factors
Hou Xi-he, Zhang Ling-li, Li Hui, Wu Wei
- Shanghai University of Sport, Shanghai 200438, China
-
Received:2017-11-27Online:2018-04-28Published:2018-04-28 -
Contact:Wu Wei, Master, Intermediate experimentalist, Shanghai University of Sport, Shanghai 200438, China -
About author:Hou Xi-he, Master, Intermediate experimentalist, Shanghai University of Sport, Shanghai 200438, China -
Supported by:the Project of Shanghai Science and Technology Commission, No. 14490503800; the Tackle Key Program for the Sports Science and Technology of Shanghai, No. 17J001; the Project of Shanghai Sports Bureau, No. HJTY-2016-E24
CLC Number:
Cite this article
Hou Xi-he, Zhang Ling-li, Li Hui, Wu Wei. Exercise training for bone and lipid metabolisms: exercise intensity and duration are important factors[J]. Chinese Journal of Tissue Engineering Research, 2018, 22(12): 1950-1955.
share this article
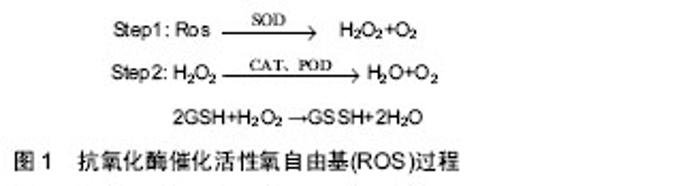
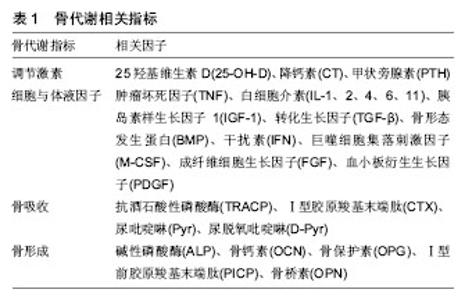
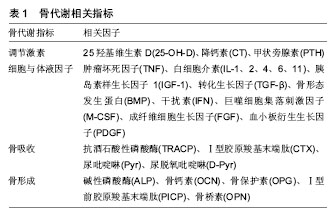
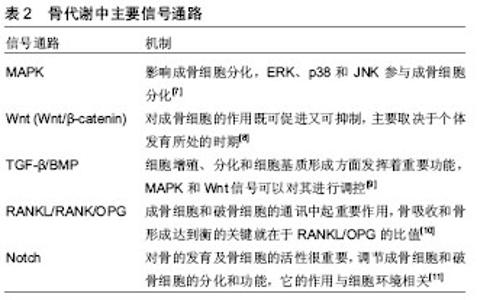
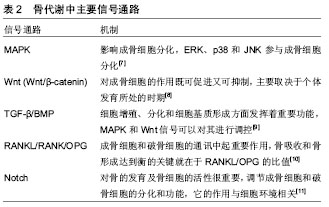
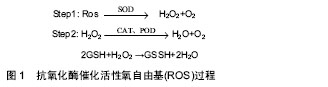
最明显的是通过骨组织受到的应力,细胞对力学刺激信号的影响和传导,将机械信号转变为化学信号,从而发挥作用。机械应力刺激、激活骨代谢指标,进一步调节骨代谢相关信号通路,促进骨形成或骨吸收。如机械刺激成骨细胞系MC3T3,会使碱性磷酸酶、骨形态发生蛋白2、骨桥素及骨钙素水平上升,使Ca2+分泌增多,进一步上调骨形态发生蛋白2、骨形态发生蛋白4促进骨形成[12]。适宜的运动对骨组织产生的应力可以调节骨代谢中的信号通路,如MAPK、Wnt、骨形态发生蛋白、RANKL/RANK/OPG、Notch等,从而促进骨形成。而运动强度和持续时间不同对骨代谢的影响将产生差异,当强度过高、持续时间长时,运动不利于骨形成。Bourrin等[13]研究发现,大强度(80%VO2 max)跑台11周后会引起大鼠成骨细胞活性降低,导致骨丢失,而短期内具有帮助作用。原因是可能造成了细胞的机械磨损,阻碍了相关信号通路(MAPK、Wnt、骨形态发生蛋白等)。也可能是大强度、长时间运动造成了细胞缺氧的应激反应,产生过多的活性氧自由基(reactive oxygen species, ROS)损伤细胞,过多的ROS不能及时被清除,则MAPK信号通路会被抑制[14]。人体内氧化系统与抗氧化系统处于平衡,维持人体正常状态,当某些原因导致体内需氧量增大时,这种平衡会慢慢偏向于氧化系统,而当抗氧化系统无法再平衡氧化系统时,体内则产生过剩的ROS和中间产物过氧化氢(H2O2),造成细胞损伤[15](图1)。运动即是导致体内需氧量增大的原因之一。"
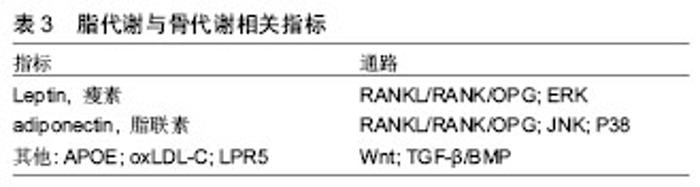
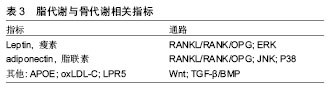
此外,应用型研究发现,不同运动项目对骨代谢影响也存在差异。长期运动训练运动员的骨形态结构、骨量、骨密度不同于普通人。运动员之间,也由于其专项和训练强度不同,其骨形态结构、骨量、骨密度也会出现差异。Ari等[16]对女子定向越野、自行车、滑雪和举重运动员的研究得出,举重运动员的骨密度高于其他项目运动员。Jürim等[17]比较女子优秀足球运动员和女子优秀游泳运动员骨密度之间的差异,在身高和体质量无差异的情况下,得出女子优秀足球运动员的骨密度显著高于游泳运动员。Min等[18]比较了跑步、摔跤和高尔夫女子运动员,得出摔跤运动员所测定骨密度要高于跑步和高尔夫运动员。Morel等[19]研究发现,划艇、游泳运动员的全身骨密度和下肢骨密度较低,而橄榄球、足球、拳击运动爱好者的全身骨密度以及下肢骨密度较高;足球、田径运动员的下肢骨密度占全身百分比较高,从事健美、拳击、攀岩和游泳的则是上肢骨密度比率大,橄榄球运动员的脊柱骨密度比率较大。 不同运动项目对骨代谢影响有所不同,对骨组织产生相对较大应力的项目(举重、摔跤等)更有利于骨生长及骨量的累积。需注意的是,在运动训练中,大强度的运动不宜进行过长时间,并且间隔不宜太过频繁,以免造成细胞机械磨损和氧化应激损伤而不利于骨生长和骨量的累积。 2.2 运动训练对脂代谢的影响 2.2.1 脂代谢相关检测指标 脂代谢过程中,常用的指标有三酰甘油、血清总胆固醇、高密度脂蛋白胆固醇(HDL-C)和低密度脂蛋白胆固醇(LDL-C)等。此外,瘦素、脂联素、载脂蛋白E(APOE)、氧化型低密度脂蛋白胆固醇(oxLDL-C)和低密度脂蛋白受体相关蛋白5(LPR5)等既可作为脂代谢指标,又可影响骨代谢[20-24]。其中瘦素和脂联素较为重要。瘦素和脂联素是脂肪细胞分泌的两个重要因子,共同参与维持脂肪代谢的平衡,对骨代谢也产生一定影响(表3)。"
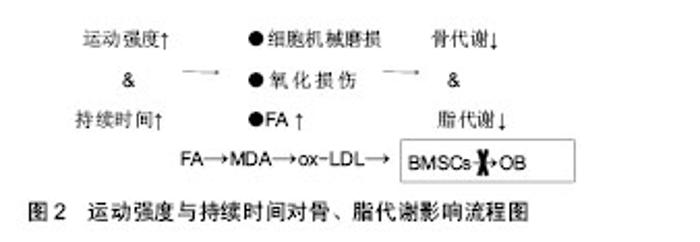
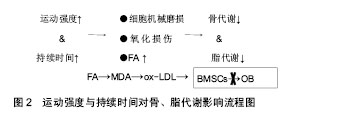
瘦素主要由脂肪细胞分泌的多功能蛋白类激素,可抑制食欲减少能量摄入、抑制脂肪合成和增加能量的消耗。肥胖程度越高,瘦素水平也越高,且常伴有瘦素抵抗[25]。瘦素对骨代谢的调节作用主要有中枢调控和外周调控作用,中枢调控主要通过下丘脑交感神经系统,其过程较为复杂,既可激活相应的交感神经(β2)引起骨流失,也可激活相应交感神经(β1)引起骨形成[26];外周调控中瘦素通过OPG/RANKL信号通路可作用于骨髓间充质干细胞使骨保护素分泌增多,RANKL分泌减少,抑制破骨细胞分化,促进成骨细胞的增殖分化[26]。同时,瘦素可抑制骨髓间充质干细胞向脂肪细胞分化,促进骨髓间充质干细胞向成骨细胞分化[27]。而近期研究中还发现,瘦素还可通过ERK信号通路(MAPK家族,可诱导碱性磷酸酶表达,使基质矿化增强、向成骨方向分化速率加快[28])对软骨细胞上的雌激素受体的表达调控骨代谢[29]。瘦素分泌浓度对骨代谢产生不同的影响,少量或适量分泌可促进骨形成,当瘦素分泌过量(30 nmol/L)时,可使骨髓间充质干细胞凋亡,促进骨吸收,导致了骨量流失[30]。总之,瘦素对骨代谢的影响是一个十分复杂的过程,通过各个信号通路的综合作用,作用性质可能取决于分泌浓度。 脂联素是脂肪组织表达、分泌的拮抗炎症因子,主要通过靶细胞膜上的脂联素受体结合而参与糖脂代谢的调节,可促进胰岛素增敏[31]。脂联素对骨代谢的调节作用主要3方面:①骨骼局部产生的脂联素促进骨形成,②循环中的脂联素抑制骨形成,③脂联素通过影响胰岛素(insulin)信号通路促进骨形成[32]。其中主要涉及的信号通路包括JNK信号通路(MAPK家族,在转导胞外信号至核转录因子时起着重要的作用,可以提高转录的能力[33])、P38信号(MAPK家族,可促进骨髓间充质干细胞向成骨细胞分化,抑制骨髓间充质干细胞向脂肪细胞分化[34])通路及RANKL/RANK/OPG信号通路。脂联素可通过JNK及P38促进成骨细胞的增殖分化;诱导NF-κB受体活化素配体(receptor activator of NF-kappaβ ligand,RANKL)表达,阻断骨保护素表达,促进骨吸收[35-36]。此外,脂联素可促进胰岛素分泌及增加其活性,胰岛素可促进成骨细胞及骨钙素含量,促进骨形成[37-38]。脂联素对骨代谢的影响也是一个复杂的过程,通过不同的信号通路产生不同的作用,其关键因素可能与脂联素的浓度、持续作用时间及变化量有关,作用机制有待进一步研究。 瘦素与脂联素对骨代谢的影响是一个复杂的过程,与其浓度、作用时间等关系密切。 2.2.2 运动训练对脂代谢的影响 运动训练对脂代谢常规指标具有改善作用。Saremi等[39]研究中发现,12周有氧运动(每周5 d)可以显著降低低密度脂蛋白胆固醇的水平,改善腰臀比(WHR)。在动物实验中,John等[40]研究发现,中等强度运动可降低胰岛素抵抗小鼠的血清三酰甘油和总胆固醇浓度,并适当提高高密度脂蛋白胆固醇水平。运动对三酰甘油、血清总胆固醇、高密度脂蛋白胆固醇和低密度脂蛋白胆固醇具有一定改善作用,长期有规律的运动习惯可以改善身体形态,消耗多余的脂肪。运动强度和运动训练持续时间是影响脂肪代谢的两个重要因素。一般而言,中低强度的运动可以使脂肪分解加快、加速脂肪氧化,大强度运动则会减少脂肪氧化。脂肪氧化分解产生能量需要经过一定时间段的持续运动累积,一般而言,运动后30 min脂肪氧化分解产能的效率将显著提高。 运动训练对瘦素和脂联素的影响由诸多因素决定,除了运动强度及持续时间外,体质量、体脂和体质水平都会对其影响,而且是综合多方面的共同影响。适宜的运动可以降低血瘦素水平,改善瘦素抵抗,并增加血液脂联素水平。一次性大强度运动后,体内的瘦素和脂联素水平变化不明显或易恢复到原有水平。Jurmae等[41]研究发现,优秀男性划船运动员在一次性极限运动后血清脂联素并未出现明显变化。Zaccaria等[42]研究发现,短时间一次性大强度运动后(60 min),体内的瘦素水平没有显著改变;而长期运动对瘦素和脂联素的水平变化还需取决于其体质量和体脂是否在长期运动的过程有所改变,对于体质量较大、体脂含量较高的人,长期运动对其瘦素及脂联素将产生积极影响,有利于过剩脂肪消耗及骨形成、骨量累积。Hulver等[43]研究发现,6个月的中等强度运动可使减重者脂联素显著上升,而体质量没有变化者其脂联素没有明显变化。同样,持续有氧运动4-10个月,肥胖者的瘦素水平下降,Hermoso等[44]推测体脂变化时导致瘦素变化的关键因素。长期坚持运动人群其体内瘦素和脂联素水平变化不会太大,脂代谢和骨代谢处于良好的动态平衡中。 运动强度过大时,脂质易受氧化应激影响,过剩的ROS与多不饱和脂肪酸生成次级氧化产物丙二醛与低密度脂蛋白结合生成氧化低密度脂蛋白[45],过多的氧化低密度脂蛋白不仅可以造成脂质异变,而且也可抑制骨髓间充质干细胞向成骨细胞分化,不利于骨形成[46]。这个与上述骨代谢中ROS恶性效果相似,因此,无论是脂代谢还是骨代谢,当运动强度过大或大强度运动持续时间较长,都将不利于代谢,产生不利影响。 2.3 运动训练对骨代谢和脂代谢影响中的思考 运动强度和持续时间是影响骨代谢及脂代谢的两个重要因素,前文中表明,当运动强度过大时,会造成细胞的机械磨损及氧化损伤,甚至出现氧化损伤标志物,如丙二醛、H2O2、共轭二烯(conjugated diolefins,CD)等。当细胞损伤得不到修复或者氧化损伤标志物在体内的含量达到一定值时,不仅不利于骨代谢及脂代谢,乃至对身体健康状况造成伤害[47]。而运动持续时间对骨代谢的影响,目前较少,Ghorbanzadeh等[48]认为,在剧烈运动过程中,初期对骨代谢的影响最为明显,随着运动持续时间的增加而有所下降;运动时体内脂肪大量动员,血浆游离脂肪酸浓度上升,为肌肉组织和其他器官提供了氧化代谢的底物,随着脂肪酸的浓度增高,对肌肉组织、器官又是一种不良作用[49]。如果运动强度过大、持续时间较长,有氧氧化较弱,体内的脂肪酸含量高而无法被很有效地分解产生能量,则导致血液脂肪酸含量越来越高,给机体带来巨大负担,引起机能下降。此外,血浆游离脂肪酸含量增高也易诱导丙二醛的生成,最终结合成氧化低密度脂蛋白,抑制了骨髓间充质干细胞向成骨细胞分化[50]。 因此,从分子机制角度来讲,运动训练时,运动强度过大则只能持续短暂时间,运动强度较大时,也不能持续太长时间。以免造成细胞机械磨损、氧化损伤及过 量血浆脂肪酸堆积,不利于骨代谢和脂代谢(图2)。 除了运动强度和持续时间这两个重要因素外,运动"
| [1] Ghodsi M, Iarijani B, Keshtkar AA,et al. Mechanisms involved in altered bone metabolism in diabetes: a narrative review. J Diabetes MetabDisord.2016;15:52.[2] Betancor MB, Ortega A, Gandara F, et al. Lipid metabolism- related gene expression pattern of Atlantic Bluefin tuna (Thunnusthynnus L.) larvae fed on live prey. Fish Physiol Biochem. 2017;43(2):493-516.[3] Klein-Nulend J, Bacabac RG, Bakker AD. Mechanical loading and how it affects bone cells: the role of the osteocyte cytoskeleton in maintaining our skeleton. Eur Cell Mater 2012;24(12): 278-291.[4] Lee S, Suzuki T, Izawa H, et al. The influence of the type of continuous exercise stress applied during growth periods on bone metabolism and osteogenesis. J Bone Metab. 2016;23(3):157-164.[5] Chanda M, Srikuea R, Cherdchutam W, et al. Modulating effects of exercise training regimen on skeletal muscle properities in female polo ponies. BMC Vet Res. 2016;12:245.[6] Styner M, Pagnotti GM, Mcgrath C,et al. Exercise decreases marrow adipose tissue through B-Oxidation in obese running mice. J Bone Miner Res. 2017;32(8):1692-1702.[7] Zhang YL, Smolen P, Baxter DA,et al. Biphasic regulation of p38 MAPK by serotonin contributes to the efficacy of stimulus protocols that induce long-term synaptic facilitation. ENEURO. 2017; 4(1): 0373-0316.[8] Katoh M.Networking of WNT, FGF, Notch, BMP, and hedgehog signaling pathways during carcinogenesis.Sterm Cell Rev 2007; 3(1): 30-38.[9] Sebald. The BMP signaling family is a diverse subset of the transforming growth factor-β (TGF-β) superfamily. BiolChem 2004; 385: 697-710.[10] Oh JH,Lee NK. Up-regulation of RANK expression via ERK1/2 by insulin contributes to the enhancement of osteoclast differentiation. Mol Cells. 2017;40(5):371-377.[11] Bruegge K,Jelkmann W,Metzen E.Hydroxylation of hypoxia-inducible transcription factors and chemical compounds targeting the HIF-alpha hydroxylases. Curr Med Chem. 2007;14(17):1853-1862.[12] Guo Y, Zhang CQ, Zeng QC,et al. Mechanical strain promotesosteoblast ECM formation and improves its osteoinductivepotential. Biomed Eng Online.2012;11:80.[13] Bourrin S, Genty C, Palle S,et al.Adverse effects of strenuous exercise: a densitometric and histomorphometric study in the rat.J Appl Physiol. 1994;76(5):1999-2005.[14] Fatokun AA,Stone TW,Smith RA. Hydrogen peroxide-induce doxidative stress in MC 3T3-E 1 cells: the effects of glutamate and protection by purines. Bone.2006;39(3):542-551.[15] Miyamoto H,Doita M,Nishida K.Effects of cyclic mechanical stress on the production of inflammatory agents by nucleus pulposus and anulusfibrosus derived cells invitro. Spine.2006;31(1): 4-9.[16] Ari H.Bone mineral density of female athletes in different sports. Bone and Mineral.1993;23(1):1-14.[17] Jürim EJ,Rmson R,Mestu J.Interactions between adipose, bone, and muscle tissue markers during acute negative energy balance in male rowers. J Sports Med Phys Fitness. 2011;51(2):347-354.[18] Min SK, Lim ST, Kim CS. Association of ACTN3 polymorphisms with BMD, and physical fitness of elderly women.J Phys Ther Sci. 2016; 28(10): 2731-2736.[19] Morel J, Combe B, Francisco J.Bone mineral density of 704 amateur sports men involved in different physical activities. OsteoporosInt 2011;12(2):152-159.[20] Peter I, Crosier MD, Yoshida M,et al. Associations of APOE gene polymorphisms with bone mineral density and fracture risk: a meta-analysis. Osteoporosis International.2011;22(4): 1199-1209.[21] Kartz GA,Holme RL,Nicholson K,et al.SR-BI/CD36 chimeric receptors define extracellular subdomains of SR-BI critical for cholesterol transport. Biochemistry.2014;53(39): 6173-6182.[22] Lin CH,Ji T,Chen CF,et al.Wnt signaling in osteosarcoma. Adv Exp Med Biol.2014; 804: 33-34.[23] Karthic R, Gokul K, Anishetty S.173 role of free cysteines in leptin receptor. Journal of Biomolecular Structure and Dynamics. 2013; 31(Supl):112-117.[24] Mendez-Sanchez N,Chavez-Tapia NC,Zamora-Valdes D,et al. Adiponectin, structure, function and pathophy-siological implications in non-alcoholic fatty liver disease. Mini Rev Med Chem. 2006;6(6): 651-656.[25] Wabitsch M, Pridzun L, Ranke M, et al.Measurement of immunofunctional leptin to detect and monitor patients with functional leptin deficiency. Eur J Endocrinol.2016;176(3): 315-322.[26] Hamrick MW, Ferrari SL.Leptin and the sympathetic connection of fat to bone. OsteoporosInt.2008;19(7):905-912.[27] Hamrick MW. Role of the cytokine-like hormone leptin in muscle-bone crosstalk with aging.J Bone Metab. 2017;24(1):1-8.[28] Fathi E, Farahzadi R. Enhancement of osteogenic differentiation of rat adipose tissue-derived mesenchymal stem cells by zinc sulphate under electromagnetic field via tha PKA, ERK1/2 and Wnt/β-catenin signaling pathways.Plo S One. 2017; 12(3): e0173877.[29] Wang SJ, Li XF, Jiang LS. Leptin regulates estrogen receptor gene expression in ATDC5 cells through the extracellular signal regulated kinase signaling pathway. J Cell Biochem.2012;113(4):1323-1332.[30] Cirmanova V, Bayer M, Starka L,et al.The effect of leptin on bone: an evolving concept of action. Physiol Res.2008; 57 (Suppl 1): S143-51.[31] Ahima RS. Meta solic actions of adipocyte hormones focus on adiponectin. Obesity(Silver Spring). 2006;14(1):9S-15S.[32] Shinoda Y,Yamaguchi M,Ogata N,et al.Regulation of bone formation by adiponectin through autocrine/paracrine and endocrine pathways. Cell Biochem.2006;99(1):196-208.[33] Lee JS,Huh KW,Bhargava A,et al.Comprehensive analysis of protein-protein interactions between Arabidopsis MAPKs and MAPK kinases helps define potential MAPK signalling modules. Plant Signal Behav.2008;3(12):1037-1041.[34] Yi C, Liu D, Fong CC,et al.Gold Nanoparticles Promote Osteogenic Differentiation of Mesenchymal Stem Cells through p38 MAPK Pathway. ACS Nano.2010;4(11):116439-116448.[35] Luo XH,Guo LJ,Yuan LQ,et al.Adiponectin stimulates human osteoblasts proliferation and differentiation via the MAPK signaling pathway. Exp Cell Res.2005;309(1): 99-109.[36] Luo XH, Guo LJ, Xie H,et al.Adiponectin stimulates RANKL and inhibits OPG expression in human osteoblasts through the MAPKsignaling pathway. Bone Miner Res.2006;21(10): 1648-1656.[37] Tong HV,Luu NK,Son HA,et al.Adiponectin and pro-inflammatory cytokines are modulated in Vietnamese patients with type 2 diabetes mellitus. J Diabetes Investig. 2017;8(3):295-305.[38] Movahed A,Larijani B,Nabipour I,et al.Reduced serum osteocalcin concentrations are associated with type 2 diabetes mellitus and the metabolic syndrome components in p ostmenopausal women: the cross talk between bone and energy metabolism. J Bone Miner Metab. 2012;30(6): 683-691.[39] Saremi AA, sghari M, Ghorbani A.Effects of aerobic training on serum omentin-1 and cardiometabolic risk factors in overweight and obese men.J Sports Sci.2010; 28(9): 993-998.[40] John R, Jenny M, Hedjazifar S,et al.Overexpressing the novel autocrine/endocrine WISP2 induces hyperplasia of the heart, white and brown adipose tissues and prevents insulin resistance. Sci Rep.2017;7: 43515.[41] Jurimae J, Purse P, Jurimae T.Adiponectin is altered after maximal exercise in highly trained male rowers. Eur J Appl Physiol.2005;93(4): 502-505.[42] Zaccaria M,Ermolao A, Brugin E,et al.Plasma leptin and energy expenditure during prolonged, moderate intensity, treadmill exercise. J Endocrinol Invest. 2013;36(6):396-401.[43] Hulver MW,Zheng D,Tanner CJ,et al.Adiponectin is not altered with exercise training despite enhanced insulin action. Am J Physiol Endocrinol Metab.2002;283(4):E861-865.[44] Hermoso AG, Ceballos RJM, Poblete CE,et al. Exercise, adipokines and pediatric obesity: a meta-analysis of randomized controlled trails.Int J Obes.2017;41(4): 475-482.[45] Zhang M, Jiang L. Oxidized low-density lipoprotein decreases VEGFR2 experssion in HUVECs and impairs angiogenesis. EXP Ther Med.2016;12(6):3742-3748.[46] Montagani A, Gonnelli S, Alessandri M, et al. Osteoporosis and risk of fracture in patients with diabetes: an update. Aging ClinExp Res.2011; 23(2):84-90.[47] Meira LB, Bugni JM, Green SL,et al. DNA damage induced by chronic inflammation contributes to colon carcinogenesis in mice. J Clin Invest. 2008;118(7):2516-2525.[48] Ghorbanzadeh V, Mohammadi M, Dariushnejad H,et al. Cardioprotective effect of crocin combined with voluntary exercise in rat: Role of Mir-126 and Mir-210 in heart angiogenesis. Arq Bras Cardio.2017;109(1): 54-62.[49] Bezaire V,Bruce C,Heigenhauser G,et al.Identification of fatty acid translocase on human skeletal muscle mitochondrial membranes: essential role in fatty acid oxidation. Am J Physiol Endocrinol Metab. 2006;290(3):E509-515.[50] Sun JZ, Wang DH, Wang C, et al. Effects of treadmill exercise at different intensity on the serum total antioxidant capacity, activity of superoxide dismutase and content of malondialdehyde in rats. Zhongguo Linchuang Kangfu. 2006;10(48):68-71.[51] Goldberg A, Mitchell K, Soans J,et al. The use of mesenchymal stem cells for cartilage repair and regeneration: a systematic review. J Orthop Surg Res.2017;12:39.[52] Hunter RL,Agnew AM.Intraskeletal variation in human cortical osteocyte lacunar density: Implications for bone quality assessment. Rep.2016;5:252-261. |
| [1] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [2] | Chen Jiming, Wu Xiaojing, Liu Tianfeng, Chen Haicong, Huang Chengshuo. Effects of silymarin on liver injury and bone metabolism induced by carbon tetrachloride in mice [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1224-1228. |
| [3] | Geng Qiudong, Ge Haiya, Wang Heming, Li Nan. Role and mechanism of Guilu Erxianjiao in treatment of osteoarthritis based on network pharmacology [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1229-1236. |
| [4] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [5] | Li Shibin, Lai Yu, Zhou Yi, Liao Jianzhao, Zhang Xiaoyun, Zhang Xuan. Pathogenesis of hormonal osteonecrosis of the femoral head and the target effect of related signaling pathways [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(6): 935-941. |
| [6] | Zheng Xiaolong, He Xiaoming, Gong Shuidi, Pang Fengxiang, Yang Fan, He Wei, Liu Shaojun, Wei Qiushi. Bone turnover characteristics in patients with alcohol-induced osteonecrosis of the femoral head [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 657-661. |
| [7] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [8] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [9] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| [10] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [11] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| [12] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| [13] | Chen Jiana, Qiu Yanling, Nie Minhai, Liu Xuqian. Tissue engineering scaffolds in repairing oral and maxillofacial soft tissue defects [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 644-650. |
| [14] | Xing Hao, Zhang Yonghong, Wang Dong. Advantages and disadvantages of repairing large-segment bone defect [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 426-430. |
| [15] | Zhu Rui, Zeng Qing, Huang Guozhi. Ferroptosis and stroke [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(23): 3734-3739. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||