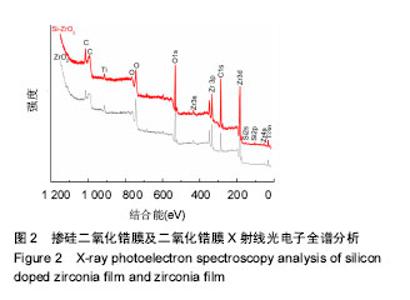
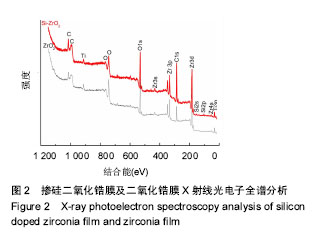
Chinese Journal of Tissue Engineering Research ›› 2018, Vol. 22 ›› Issue (14): 2227-2232.doi: 10.3969/j.issn.2095-4344.0738
Previous Articles Next Articles
Osteoclast activity of MG63 cells on silicon doped zirconia films prepared by cathodic arc deposition
Zhang Shai-lin, Cheng Xiang-yu, Shi Ji-xiang, Zhou Qiang, Shi Wen-jun, Liu Fu-ying, Ji Bin
- Department of Orthopaedics, Putuo Hospital, Shanghai University of Traditional Chinese Medicine, Shanghai 200062, China
-
Received:2017-12-26Online:2018-05-18Published:2018-05-18 -
Contact:Ji Bin, Chief physician, Master’s supervisor, Department of Orthopaedics, Putuo Hospital, Shanghai University of Traditional Chinese Medicine, Shanghai 200062, China -
About author:Zhang Shai-lin, M.D., Attending physician, Department of Orthopaedics, Putuo Hospital, Shanghai University of Traditional Chinese Medicine, Shanghai 200062, China -
Supported by:Independent Innovation Project of Shanghai Putuo District Health System, No. 2013PTKW015; National Natural Science Foundation of China, No. 81673782; Key Clinical Specialist Construction Programs of Putuo District, Shanghai, No. 2016PTZK04
CLC Number:
Cite this article
Zhang Shai-lin, Cheng Xiang-yu, Shi Ji-xiang, Zhou Qiang, Shi Wen-jun, Liu Fu-ying, Ji Bin. Osteoclast activity of MG63 cells on silicon doped zirconia films prepared by cathodic arc deposition[J]. Chinese Journal of Tissue Engineering Research, 2018, 22(14): 2227-2232.
share this article
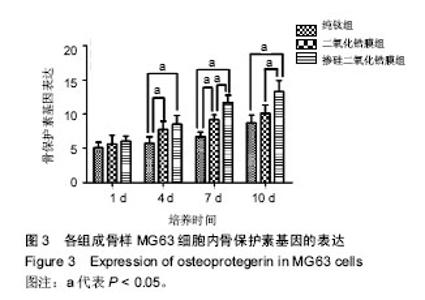
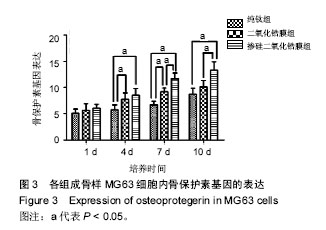
2.3 各组骨保护素基因表达 培养第1天时,各组样品表面的成骨细胞骨保护素基因表达比较差异无显著性意义 (P > 0.05);第4天时,掺硅二氧化锆膜组、二氧化锆膜组骨保护素基因表达显著高于纯钛组(P < 0.05),掺硅二氧化锆膜组、二氧化锆膜组骨保护素基因表达比较差异无显著性意义(P > 0.05);第7天时,掺硅二氧化锆膜组骨保护素基因表达显著高于二氧化锆膜组及纯钛组(P < 0.05),二氧化锆膜组骨保护素基因表达显著高于纯钛组(P < 0.05);第10天时,掺硅二氧化锆膜组骨保护素基因表达显著高于二氧化锆膜组及纯钛组(P < 0.05),二氧化锆膜组和纯钛组骨保护素基因表达比较差异无显著性意义(P > 0.05),见图3。"
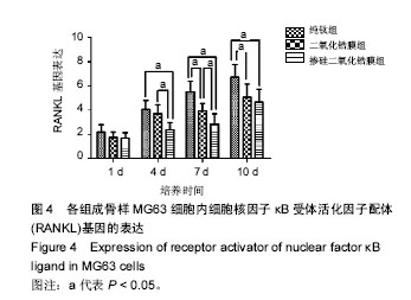
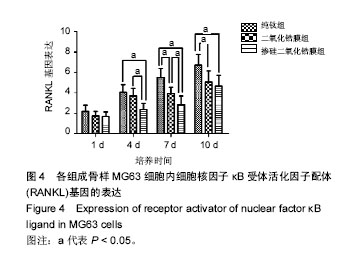
2.4 各组RANKL基因表达 培养第1天时,各组样品表面的成骨细胞RANKL基因表达差异无显著性意义(P > 0.05);第4天时,掺硅二氧化锆膜组RANKL基因表达显著低于二氧化锆膜组及纯钛组(P < 0.05),二氧化锆膜组和纯钛组RANKL基因表达比较差异无显著性意义(P > 0.05);第7天时,掺硅二氧化锆膜组、二氧化锆膜组RANKL基因表达均显著低于纯钛组(P < 0.05),掺硅二氧化锆膜组RANKL基因表达显著低于二氧化锆膜组(P < 0.05);第10天时,掺硅二氧化锆膜组、二氧化锆膜组的RANKL基因表达显著低于纯钛组(P < 0.05),掺硅二氧化锆膜组和二氧化锆膜组的RANKL基因表达比较差异无显著性意义(P > 0.05),见图4。"
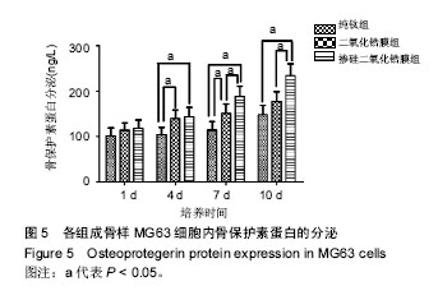
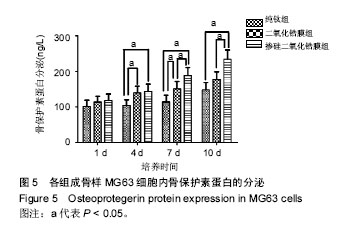
2.5 各组骨保护素蛋白分泌检测 各组样品表面成骨样MG63细胞骨保护素蛋白分泌与骨保护素基因表达具有相同的趋势。培养第1天时,各组骨保护素蛋白分泌无差异 (P > 0.05);第4天时,掺硅二氧化锆膜组、二氧化锆膜组骨保护素蛋白分泌显著高于纯钛组(P < 0.05),掺硅二氧化锆膜组和二氧化锆膜组骨保护素蛋白分泌无差异;第7天时,掺硅二氧化锆膜组骨保护素蛋白分泌显著高于二氧化锆膜组、纯钛组(P < 0.05),二氧化锆膜组骨保护素蛋白分泌显著高于纯钛组(P < 0.05);第10天时,掺硅二氧化锆膜组骨保护素蛋白分泌显著高于二氧化锆膜组、纯钛组 (P < 0.05),二氧化锆膜组和纯钛组骨保护素蛋白分泌无差异(P > 0.05),见图5。"
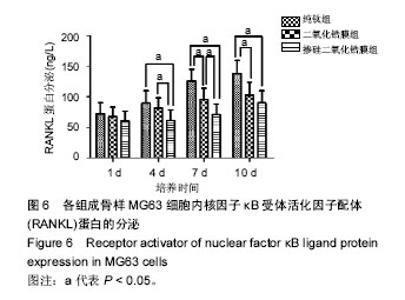
2.6 各组RANKL蛋白分泌检测 培养第1天时,各组样品表面成骨样MG63细胞内RANKL蛋白分泌无差异(P > 0.05);第4天时,掺硅二氧化锆膜组RANKL蛋白分泌显著低于二氧化锆膜组、纯钛组(P < 0.05),二氧化锆膜组和纯钛组RANKL蛋白分泌无差异(P > 0.05);在第7天时,掺硅二氧化锆膜组、二氧化锆膜组的RANKL蛋白分泌均显著低于纯钛组(P < 0.05);掺硅二氧化锆膜组RANKL蛋白分泌均显著低于二氧化锆膜组(P < 0.05);第10天时,掺硅二氧化锆膜组、二氧化锆膜组RANKL蛋白分泌显著低于纯钛组(P < 0.05),掺硅二氧化锆膜组和二氧化锆膜组RANKL蛋白分泌无差异(P > 0.05),见图6。"
| [1] Martin JY,Schwartz Z,Hummert TW,et al.Effect of titanium surface roughness on proliferation, differentiation, and protein synthesis of human osteoblast-like cells(MG63).J Biomed Mater Res.1995; 29(3): 389-401.[2] Fini M,Cigada A,Rondelli G,et al.In vitro and in vivo behaviour of Ca- and P-enriched anodized titanium.Biomaterials.1999;20(17):1587-1594.[3] Sugino A Ohtsuki C,Tsuru K,et al.Effect of spatial design and thermal oxidation on apatite formation on Ti-15Zr-4Ta-4Nb alloy.Acta Biomater. 2009;5(1):298-304.[4] Kasuga T,Nogami M,Niinomi M,et al.Bioactive calcium phosphate invert glass-ceramic coating on beta-type Ti-29Nb-13Ta-4.6Zr alloy. Biomaterials.2003;24(2):283-290.[5] Yan YY,Han Y.Structure and bioactivity of micro-arc oxidized zirconia films.Surf Coat Techno.2007;201(9-11):5692-5695.[6] Wang G,Meng F,Ding C,et al.Microstructure, bioactivity and osteoblast behavior of monoclinic zirconia coating with nanostructured surface. Acta Biomater.2010;6(3):990-1000.[7] Sollazzo V,Pezzetti F,Scarano A,et al.Zirconium oxide coating improves implant osseointegration in vivo.Dent Mater.2008;24(3):357-361.[8] Zhang S,Sun J,Xu Y,et al.Biological behavior of osteoblast-like cells on titania and zirconia films deposited by cathodic arc deposition. Biointerphases.2012;7(1-4):60.[9] Zhang S,Sun J,Xu Y,et al.Adhesion, proliferation and differentiation of osteoblasts on zirconia films prepared by cathodic arc deposition. Biomed Mater Eng.2013;23(5):373-385.[10] Thian ES,Huang J,Best SM,et al.Magnetron co-sputtered silicon-containing hydroxyapatite thin films--an in vitro study. Biomaterials. 2005;26(16):2947- 2956.[11] Thian ES,Huang J,Best SM,et al.Silicon-substituted hydroxyapatite thin films: effect of annealing temperature on coating stability and bioactivity.J Biomed Mater Res A.2006;78(1):121-128.[12] Thian ES,Huang J,Best SM,et al.A new way of incorporating silicon in hydroxyapatite (Si-HA) as thin films.J Mater Sci Mater Med. 2005; 16(5):411-415.[13] Liu XY,Huang AP,Ding CX,et al.Bioactivity and cytocompatibility of zirconia (ZrO2) films fabricated by cathodic arc deposition.Biomaterials. 2006;27(21):3904-3911.[14] Hench LL.Bioactive materials: the potential for tissue regeneration.J Biomed Mater Res.1998;41(4):511-518.[15] Buser D,Nydegger T,Oxland T,et al.Interface shear strength of titanium implants with a sandblasted and acid-etched surface: a biomechanical study in the maxilla of miniature pigs.J Biomed Mater Res. 1999;45(2): 75-83.[16] Jing W,Zhang M,Jin L,et al.Assessment of osteoinduction using a porous hydroxyapatite coating prepared by micro-arc oxidation on a new titanium alloy.Int J Surg.2015;24(Pt A):51-56.[17] Lynn AK,Duquesnay DL.Hydroxyapatite-coated Ti-6Al-4V Part 2: the effects of post-deposition heat treatment at low temperatures. Biomaterials.2002;23(9):1947-1953.[18] Suchanek W,Yoshimura M.Processing and properties of hydroxyapatite-based biomaterials for use as hard tissue replacement implants.J Mater Res.1998;13(1):94-117.[19] Yang YC,Chang E,Lee SY.Mechanical properties and Young's modulus of plasma-sprayed hydroxyapatite coating on Ti substrate in simulated body fluid.J Biomed Mater Res A.2003;67(3):886-899.[20] 章筛林,成翔宇,纪斌.硅在生物材料领域的应用:增加材料生物活性不影响其机械性能[J].中国组织工程研究,2017,21(2):296-301.[21] Carlisle EM.Silicon: a possible factor in bone calcification.Science. 1970;167(916):279-280.[22] Schwarz K,Milne DB.Growth-promoting effects of silicon in rats.Nature. 1972;239(5371):333-334.[23] Xynos ID,Edgar AJ,Buttery LD,et al.Ionic products of bioactive glass dissolution increase proliferation of human osteoblasts and induce insulin-like growth factor II mRNA expression and protein synthesis. Biochem Biophys Res Commun.2000;276(2):461-465.[24] Darley WM,Volcani BE.Role of silicon in diatom metabolism. A silicon requirement for deoxyribonucleic acid synthesis in the diatom Cylindrotheca fusiformis Reimann and Lewin.Exp Cell Res. 1969;58(2): 334-342.[25] Keeting PE,Oursler MJ,Wiegand KE,et al.Zeolite A increases proliferation, differentiation, and transforming growth factor beta production in normal adult human osteoblast-like cells in vitro. J Bone Miner Res.1992;7(11):1281-1289.[26] Reffitt DM,Ogston N,Jugdaohsingh R,et al.Orthosilicic acid stimulates collagen type 1 synthesis and osteoblastic differentiation in human osteoblast-like cells in vitro.Bone.2003;32(2):127-135.[27] Arumugam MQ,Ireland DC,Brooks RA,et al.The effect orthosilicic acid on collagen type I, alkaline phosphatase and osteocalcin mRNA expression in human bone-derived osteoblasts in vitro.Key Eng Mater. 2006;309-311:121-124.[28] Shepherd JH,Shepherd DV,Best SM.Substituted hydroxyapatites for bone repair.J Mater Sci Mater Med.2012;23(10):2335-2347.[29] Hu HJ,Liu XY,Ding CX.Preparation and cytocompatibility of Si-incorporated nanostructured TiO2 coating.Surf Coat Technol. 2010;204(20):3265-3271.[30] Zhang Z,Sun J,Hu H,et al.Osteoblast-like cell adhesion on porous silicon-incorporated TiO2 coating prepared by micro-arc oxidation.J Biomed Mater Res B Appl Biomater.2011;97(2):224-234.[31] Matsuo K,Irie N.Osteoclast-osteoblast communication.Arch Biochem Biophys.2008;473(2):201-209.[32] Boyle WJ,Lacey DL,Timms E,et al.Osteoprotegerin ligand is a cytokine that regulates osteoclast differentiation and activation.Cell.1998;93(2):165-176.[33] Roodman GD.Advances in bone biology: the osteoclast.Endocr Rev. 1996;17(4):308-332.[34] Suda T,Nakamura I,Jimi E,et al.Regulation of osteoclast function.J Bone Miner Res.1997;12(6):869-879.[35] Suda T,Udagawa N,Nakamura I,et al.Modulation of osteoclast differentiation by local factors. Bone.1995;17(2 Suppl):87S-91S.[36] Hofbauer LC,Heufelder AE.Clinical review 114: hot topic. The role of receptor activator of nuclear factor-kappaB ligand and osteoprotegerin in the pathogenesis and treatment of metabolic bone diseases.J Clin Endocrinol Metab.2000;85(7):2355-2363.[37] Hofbauer LC,Khosla S,Dunstan CR,et al.The roles of osteoprotegerin and osteoprotegerin ligand in the paracrine regulation of bone resorption.J Bone Miner Res.2000;15(1):2-12.[38] Hsu H,Lacey DL,Dunstan CR,et al.Tumor necrosis factor receptor family member RANK mediates osteoclast differentiation and activation induced by osteoprotegerin ligand.Proc Natl Acad Sci U S A.1999;96(7):3540-3545.[39] Kulkarni RN,Bakker AD,Everts V,et al.Inhibition of osteoclastogenesis by mechanically loaded osteocytes: involvement of MEPE.Calcif Tissue Int.2010;87(5):461-468.[40] 黎彦龙,明何,陈秉雄,等.OPG-RANKL-RANK信号系统是调节破骨细胞及骨质疏松症的重要途径[J].中国组织工程研究,2015,19(24):3894-3898. |
| [1] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [2] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [3] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [4] | Li Xingping, Xiao Dongqin, Zhao Qiao, Chen Shuo, Bai Yiguang, Liu Kang, Feng Gang, Duan Ke. Preparation and properties of copper-loaded antibacterial functional film on titanium surface [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 553-557. |
| [5] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [6] | Shi Xiaoxiu, Mao Shilong, Liu Yang, Ma Xingshuang, Luo Yanfeng. Comparison of tantalum and titanium (alloy) as orthopedic materials: physical and chemical indexes, antibacterial and osteogenic ability [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 593-599. |
| [7] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| [8] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [9] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| [10] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| [11] | Chen Jiana, Qiu Yanling, Nie Minhai, Liu Xuqian. Tissue engineering scaffolds in repairing oral and maxillofacial soft tissue defects [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 644-650. |
| [12] | Xing Hao, Zhang Yonghong, Wang Dong. Advantages and disadvantages of repairing large-segment bone defect [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 426-430. |
| [13] | Chen Siqi, Xian Debin, Xu Rongsheng, Qin Zhongjie, Zhang Lei, Xia Delin. Effects of bone marrow mesenchymal stem cells and human umbilical vein endothelial cells combined with hydroxyapatite-tricalcium phosphate scaffolds on early angiogenesis in skull defect repair in rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3458-3465. |
| [14] | Wang Hao, Chen Mingxue, Li Junkang, Luo Xujiang, Peng Liqing, Li Huo, Huang Bo, Tian Guangzhao, Liu Shuyun, Sui Xiang, Huang Jingxiang, Guo Quanyi, Lu Xiaobo. Decellularized porcine skin matrix for tissue-engineered meniscus scaffold [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3473-3478. |
| [15] | Mo Jianling, He Shaoru, Feng Bowen, Jian Minqiao, Zhang Xiaohui, Liu Caisheng, Liang Yijing, Liu Yumei, Chen Liang, Zhou Haiyu, Liu Yanhui. Forming prevascularized cell sheets and the expression of angiogenesis-related factors [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3479-3486. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||