Chinese Journal of Tissue Engineering Research ›› 2018, Vol. 22 ›› Issue (17): 2775-2782.doi: 10.3969/j.issn.2095-4344.0513
Previous Articles Next Articles
Stem cells in orthopedics diseases: existing problems and application prospects
Wu Zhan-yu1, Ye Chuan2, 3
- 1Guizhou Medical University, Guiyang 550004, Guizhou Province, China; 2Department of Orthopedics, the Affiliated Hospital of Guizhou Medical University, Guiyang 550004, Guizhou Province, China; 3Center for Tissue Engineering and Stem Cells, Guizhou Medical University, Guiyang 550004, Guizhou Province, China
-
Revised:2018-03-05Online:2018-06-18Published:2018-06-18 -
Contact:Ye Chuan, M.D., Chief physician, Professor, Department of Orthopedics, the Affiliated Hospital of Guizhou Medical University, Guiyang 550004, Guizhou Province, China; Center for Tissue Engineering and Stem Cells, Guizhou Medical University, Guiyang 550004, Guizhou Province, China -
About author:Wu Zhan-yu, Master candidate, Guizhou Medical University, Guiyang 550004, Guizhou Province, China -
Supported by:the Key Science and Technology Program of the Ministry of Education of China, No. 212156
CLC Number:
Cite this article
Wu Zhan-yu, Ye Chuan. Stem cells in orthopedics diseases: existing problems and application prospects[J]. Chinese Journal of Tissue Engineering Research, 2018, 22(17): 2775-2782.
share this article

2.1 干细胞在骨组织修复中的研究及临床应用 2.1.1 干细胞治疗大段骨缺损 大块骨缺损一般是由于肿瘤、严重的创伤或感染造成的,是骨科一大难题。传统的治疗方案为自体骨移植或异体骨移植,不仅治疗效果有限,同时还存在取骨区域副损伤、移植物排异反应等问题[6]。为解决目前难治性大骨缺损的难题,干细胞治疗应运而生。骨髓血的局部注射就是干细胞疗法的一种较为初始和简单的形式,这种方法早在1990年就出现并尝试性运用于临床[7]。然而,因为细胞没有经过纯化,其中混杂着其他血液成分及细胞,因此还不能称之为严格意义上的干细胞治疗。目前报道的干细胞临床应用,多为自体的、扩增后的及未修饰的人间充质干细胞。虽然这些研究还存在些不足,但也为干细胞治疗大段骨缺损的安全性及潜力提供了依据[8]。 目前的研究表明使用自体间充质干细胞治疗骨缺损是安全的,没有出现感染或过度组织生长的情况。有报道称过长时间培养的间充质干细胞(四五个月),会引发基因组不稳定性,从而转变出恶性标志[9],而相对较短时间的体外扩增(6-8周),目前还没有恶变的报道[10]。 采用局部注射骨髓的方法来治疗骨缺损,有一定效果,但存在细胞流失、不能形成有效的细胞浓度梯度、不具备占位效应等问题。随着组织工程发展,干细胞复合支架材料的应用能有效防止细胞流失,支架材料还能提供细胞黏附及生长的微环境[11],具有占位效应。已有动物实验研究报道获得了很好的效果,刘浩等[12]将成骨诱导后的脂肪干细胞接种到预湿的丝素蛋白/羟基磷灰石材料上,发现细胞能在材料表面正常增殖和分化,说明了丝素蛋白/羟基磷灰石作为人工骨材料具有良好的细胞相容性,能够作为组织工程骨的支架材料。 最近,Dufrane等[13]报道利用3D技术结合自体脂肪干细胞治疗大段骨缺损的案例,其用6例患者的皮下脂肪组织提取干细胞,扩增至第4代,在脱钙骨支架中行成骨诱导,得到立体的骨样结构,然后植入6例大段骨缺损患者,并进行了长达4年的随访,其中3例患者骨愈合良好,没有出现伤口感染、疼痛、炎症反应等早期并发症,并且也没有出现恶变等远期并发症。 虽然有部分大段骨缺损通过干细胞治疗完全再生的报道,但是因为缺乏对照试验,干细胞治疗大段骨缺损的安全有效的结论,尚不能过早得出。 2.1.2 干细胞治疗骨坏死 干细胞治疗骨坏死已较为广泛的用于临床,其中较为多见的是股骨头坏死。股骨头坏死较为传统的治疗方式为股骨头髓芯减压[14],虽然部分患者行髓芯减压术后能够缓解早期股骨头坏死的疼痛症状,然而,术后复查MRI及相关影像学检查显示,单纯的髓芯减压并不能使股骨头坏死区域得到良好的重建修复[15],而干细胞的特性能够弥补传统疗法的不足。研究指出,自体干细胞移植联合髓芯减压较单纯髓芯减压更有效[16]。Rastogi等[17]将40例股骨头坏死患者(ARCO Ⅲ)随机分为两组,一组行骨髓移植,另一组行骨髓单个核细胞移植,术后2年发现两组患者的Harris评分均升高,单个核细胞移植组的坏死面积显著降低,他发现股骨头坏死早期患者治疗效果明显优于中期和晚期患者。Sen等[18]对比了单纯髓芯减压及髓芯减压联合细胞移植的疗效,随访24个月后发现髓芯减压联合细胞移植较单纯髓芯减压效果更优。Ma等[19]做了前瞻性的双盲随机对照研究,实验组为髓芯减压+自体骨移植+骨髓血单个核细胞移植,对照组为髓芯减压+自体骨移植。随访24个月,实验组的疗效优于对照组,并且治疗组中坏死程度为Ⅰ期或Ⅱ期的,没有一例病程进展。Martin等[20]用骨髓细胞悬液和富血小板血浆治疗Ⅰ、Ⅱ期股骨头坏死,术后患者有明显的疼痛缓解。 这些研究均显示,骨髓干细胞移植治疗股骨头坏死早期有超越传统疗法的疗效,但也有文献报道,两者之间治疗效果并没有明显区别[21],并且从单个患者疗效上看,部分患者行自体干细胞移植联合髓芯减压的效果仍然不及预期,其中较近的一次研究是Lim等[22]完成的,他的研究收录了128例患者(190髋),分为髓芯减压联合骨移植及髓芯减压联合干细胞移植两组,评估术后两组患者的临床症状恢复情况及影像学变化,两组患者间没有明显差别。 干细胞治疗也应用于一些特殊类型的骨坏死,如镰状细胞病。Hernigou等[23]用同种异体干细胞静脉输注治疗镰状细胞病继发肱骨头坏死(Ⅱ期)患者,通过随访4年后发现治疗效果良好,坏死区域完全修复。 干细胞治疗骨坏死在临床上已被较多的团队研究并开展,取得了一些可喜的结果,但大多数报道的研究没有设置对照组,大多数研究的方向主要针对股骨头缺血坏死,而其中最常使用的手段就是联合髓芯减压将骨髓细胞悬液植入坏死区域,少部分研究是将干细胞体外扩增后植入,部分研究使用了复合支架材料,主要包括纤维蛋白胶、富血小板血浆、β-磷酸三钙、自体骨和钽棒等。不同研究团队干细胞治疗骨坏死的疗效评价结果不同,但是从理论上讲,干细胞治疗骨坏死是极具前景的。 2.1.3 干细胞促进骨折愈合 大部分的骨折在正确的治疗后都能顺利愈合,尽管如此,仍然希望能缩短愈合时间,促进肢体功能的快速康复,郑艳峰等[24]使用骨髓血移植治疗严重下肢粉碎性骨折22例,结果显示患者均在术后4-10周出现典型骨痂,术后4-6个月出现连续骨痂,提示骨折临床愈合。贾世双等[25]认为细胞移植结合内固定是治疗股骨颈骨折的一种较好的方法,其操作简单、固定可靠、疗效满意,为股骨颈骨折的治疗提供了一种能降低股骨头早期坏死发生率的新思路。Rodriguez-Collazo等[26]报道了骨髓干细胞+富血小板血浆联合Ilizarov外固定架治疗双踝关节骨折,10例患者使用骨髓干细胞为治疗组,10例不用骨髓干细胞为对照组,研究发现骨髓干细胞治疗组患者的骨折完全愈合时间明显短于对照组。自体骨髓浓缩干细胞悬液局部注射能促进骨折愈合。 骨折延迟愈合与骨不连也是骨折后常见并发症,对于骨不连患者开放手术加自体骨移植是目前治疗的金标准。但有手术创伤大、供区副损伤等缺点,尤其局部软组织条件差时则更加棘手。而局部注射骨髓间充质干细胞进行治疗,可以避免开放手术的缺点,现在临床上已有部分应用。Desai等[27]运用骨髓干细胞局部注射治疗49例胫骨骨不连患者,取得了不错的疗效。Murena等[28]将骨髓干细胞与同种异体骨及骨形态发生蛋白7复合,治疗2例肱骨干骨折不愈合,分别在术后4个月及8个月达到了愈合标准。Qu等[29]用细胞疗法治疗骨不连患者,3例用自体骨髓干细胞,6例用脐带间充质干细胞,对比两组患者均达到了临床愈合标准。 骨髓的注射应该包括骨不连区域和近远端骨断端周围,但目前还不清楚当断端有纤维组织时,干细胞注入后是否会改变该组织的性质,并促进成骨,还是只在该纤维组织周围形成骨桥,包绕纤维组织,连接两个断端。但无论如何,细胞疗法在治疗骨不连的传统疗法上迈出了一大步,具有很大的优势,但也需要进一步的研究。 干细胞在骨组织修复中的临床应用见表1。"

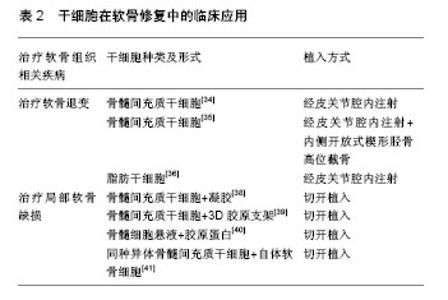
2.2 干细胞在软骨修复中的研究与临床应用 随着社会老龄化的加重,骨性关节炎发病率上升,并且随着膝关节镜的应用及人们健康意识及生活质量要求的提高,越来越多的各种原因引起的软骨缺损患者需要得到更有效、更微创、更个性化的治疗。软骨再生在科研及临床中居于越来越重要的地位。 在科研领域,Johnson等[30]对间充质干细胞进行化学筛检并发现一个叫做kartogenin的小分子,它可诱使间充质干细胞转变成为软骨细胞而促进软骨产生,并且发现给小鼠局部注射kartogenin,可触发其体内的软骨发育。韩志军和他的团队[31]将猪骨髓间充质干细胞作为种子细胞,转化生长因子β1作为诱导因子,以聚羟基乙酸为支架,植入自体猪皮下经过8-10周的体内培养,支架材料基本降解,成功构建了具有一定弹性的组织工程软骨,并通过组织学证实。杨萌等[32]将滑膜间充质干细胞混合软骨细胞培养于壳聚糖/Ⅰ型胶原复合支架材料中,移植于SD大鼠皮下,4,8周时取材进行苏木精-伊红染色、甲苯胺蓝染色和免疫组织化学检测。结果表明两种细胞混合培养于壳聚糖/Ⅰ型胶原复合支架材料中,在体内环境下能够形成软骨样组织。 目前临床上已经开展的干细胞软骨修复主要有两种方式,一是干细胞关节腔注射治疗软骨退变,二是可吸收支架材料负载干细胞治疗局部软骨缺损。 2.2.1 干细胞关节腔注射治疗软骨退变 2008年,Centeno等[33]报道了2例软骨退变患者行干细胞治疗后,疼痛缓解,并且MRI检查可见软骨增加。Davatchi等[34]也进行了类似的报道,他将(8.0-9.0)×106个骨髓间充质干细胞分别注入3例膝关节骨性关节炎患者的膝关节腔中,6个月以内各项参数都有所改善,之后便开始下降,但5年后仍然高于其治疗前水平。Wong和他的团队[35]观察关节腔内注射自体骨髓间充质干细胞联合内侧开放式楔形胫骨高位截骨治疗膝关节骨性关节炎的疗效,共纳入56膝,全为单间室膝关节骨性关节炎合并膝内翻患者,随机分为细胞治疗组及对照组,细胞治疗组患者在术后3周行微创膝关节腔注射扩增的骨髓间充质干细胞+透明质酸,而对照组只注射透明质酸。在排除年龄、基础评分等因素后,接受细胞治疗的患者表现出更好的疗效。 目前临床上用于软骨修复的除骨髓间充质干细胞外,还有脂肪间充质干细胞,Pak[36]提取患者自体脂肪组织干细胞,治疗了2例膝关节骨性关节炎患者和2例髋关节骨性关节炎患者,都达到了较满意的疗效,治疗12周后,2例膝关节骨性关节炎患者MRI上显示半月板厚度有所增加。Jo的团队[37]开展了一项前瞻性对照研究,纳入了18例膝关节骨性关节炎患者,依据注入细胞数量的不同(1.0×107,5.0×107和1.0×108)分3组,注射6个月后患者的膝关节相关功能评分均有提高,并认为膝关节腔注射1.0×108脂肪干细胞,能够有效缓解膝关节骨性关节炎的疼痛症状并改善膝关节功能。 干细胞已被单独或与其他外科手术方式相结合治疗骨性关节炎,一些团队将其和透明质酸、激素或血小板血浆联合进行一些临床相关研究。有少数实验团队报道了干细胞注射治疗后不仅膝关节功能明显改善,而且软骨及半月板也得到了修复。干细胞治疗骨性关节炎的前景是乐观的,但还需要更严谨、样本量更大及更深入的研究。 2.2.2 可吸收支架材料负载干细胞治疗局部软骨缺损 Kuroda等[38]于2007年报道了用自体骨髓间充质干细胞修复软骨缺损,将扩增后的间充质干细胞加入胶原溶液中,移植于软骨缺损部位,以滑膜覆盖。术后7个月行关节镜检发现,软骨缺损部位被修复,1年后,关节功能有明显改善,能够满足于日常生活,并且没有疼痛及并发症的发生。Kasemkijwattana和他的团队[39]用骨髓干细胞移植治疗2例面积较大的创伤性软骨缺损,细胞与3D胶原支架复合覆盖缺损区域,术后31个月,关节镜检发现缺损区域修复良好,并且与周围软骨整合,患者功能也大有改善。Gobbi等[40]用骨髓干细胞修复Ⅳ级软骨损伤,6例均以微创手术植入细胞胶原复合物,术后随访发现患者的各项功能评分有明显升高,软骨损伤面积较小的或只有单处损伤的患者疗效更加优越,MRI检查显示缺损区域被软骨样组织覆盖。最近的研究是De Windt等[41]于2016年报道的,他用同种异体间充质干细胞混合自体软骨细胞治疗软骨缺损,12个月后患者的症状明显缓解,功能明显改善,缺损区域完全修复,并且修复区域的组织只含有患者自己的DNA,他认为植入的同种异体干细胞只起了旁分泌的效应。 大部分研究报道的都是令人高兴的结果,软骨缺损区域在干细胞治疗后都得到了很好的修复,但是覆盖坏死区域的组织并没被证实为软骨组织,同样也缺乏对照组来证实干细胞治疗的真正有效性,仍需要样本量更大、证据指数更高的研究。诱导性多能干细胞在软骨组织修复领域有着可深入挖掘的潜力。 干细胞在软骨修复中的临床应用见表2。"

| [1] Sampson S, Botto-van Bemden A, Aufiero D. Stem cell therapies for treatment of cartilage and bone disorders: osteoarthritis, avascular necrosis, and non-union fractures. PM R. 2015; 7(4 Suppl):S26-32.[2] Zhao DW, Yu XB. Core decompression treatment of early-stage osteonecrosis of femoral head resulted from venous stasis or artery blood supply insufficiency. J Surg Res. 2015;194(2): 614-621.[3] Jarvis NE, Halliday L, Sinnott M, et al. Surgery for the fractured clavicle: factors predicting nonunion. J Shoulder Elbow Surg. 2017 Dec 19. doi: 10.1016/j.jse.2017.10.010. [Epub ahead of print] [4] Im GI. Regeneration of articular cartilage using adipose stem cells. J Biomed Mater Res A. 2016;104(7):1830-1844. [5] Ellera Gomes JL, da Silva RC, Silla LM, et al. Conventional rotator cuff repair complemented by the aid of mononuclear autologous stem cells. Knee Surg Sports Traumatol Arthrosc. 2012;20(2): 373-377.[6] Im GI. Nonviral gene transfer strategies to promote bone regeneration. J Biomed Mater Res A. 2013;101(10):3009-3018. [7] Healey JH, Zimmerman PA, McDonnell JM, et al. Percutaneous bone marrow grafting of delayed union and nonunion in cancer patients. Clin Orthop Relat Res. 1990;(256):280-285.[8] Imam MA, Holton J, Ernstbrunner L, et al. A systematic review of the clinical applications and complications of bone marrow aspirate concentrate in management of bone defects and nonunions. Int Orthop. 2017;41(11):2213-2220.[9] Røsland GV, Svendsen A, Torsvik A, et al. Long-term cultures of bone marrow-derived human mesenchymal stem cells frequently undergo spontaneous malignant transformation. Cancer Res. 2009;69(13):5331-5339.[10] Im GI. Stem cells for reutilization in bone regeneration. J Cell Biochem. 2015;116(4):487-493.[11] Hoshino M, Egi T, Terai H, et al. Regenerative repair of long intercalated rib defects using porous cylinders of beta-tricalcium phosphate: an experimental study in a canine model. Plast Reconstr Surg. 2007;119(5):1431-1439.[12] 刘浩,褚亚伟,丁涛,等. 丝素蛋白/羟基磷灰石支架对成骨诱导脂肪干细胞活性及成骨性能的影响[J]. 中国组织工程研究, 2015,19(45): 7224-7229. [13] Dufrane D, Docquier PL, Delloye C, et al. Scaffold-free Three-dimensional Graft From Autologous Adipose-derived Stem Cells for Large Bone Defect Reconstruction: Clinical Proof of Concept. Medicine (Baltimore). 2015;94(50):e2220.[14] Steinberg ME, Steinberg DR. Classification systems for osteonecrosis: an overview. Orthop Clin North Am. 2004;35(3):273-283.[15] Plenk H Jr, Hofmann S, Breitenseher M, et al. Pathomorphological aspects and repair mechanisms of femur head necrosis. Orthopade. 2000;29(5):389-402.[16] Tabatabaee RM, Saberi S, Parvizi J, et al. Combining Concentrated Autologous Bone Marrow Stem Cells Injection With Core Decompression Improves Outcome for Patients with Early-Stage Osteonecrosis of the Femoral Head: A Comparative Study. J Arthroplasty. 2015;30(9 Suppl):11-15.[17] Rastogi S, Sankineani SR, Nag HL, et al. Intralesional autologous mesenchymal stem cells in management of osteonecrosis of femur: a preliminary study. Musculoskelet Surg. 2013;97(3):223-228.[18] Sen RK, Tripathy SK, Aggarwal S, et al. Early results of core decompression and autologous bone marrow mononuclear cells instillation in femoral head osteonecrosis: a randomized control study. J Arthroplasty. 2012;27(5):679-686. [19] Ma Y, Wang T, Liao J, et al. Efficacy of autologous bone marrow buffy coat grafting combined with core decompression in patients with avascular necrosis of femoral head: a prospective, double-blinded, randomized, controlled study. Stem Cell Res Ther. 2014;5(5):115.[20] Martin JR, Houdek MT, Sierra RJ. Use of concentrated bone marrow aspirate and platelet rich plasma during minimally invasive decompression of the femoral head in the treatment of osteonecrosis. Croat Med J. 2013;54(3):219-224.[21] Pepke W, Kasten P, Beckmann NA, et al. Core Decompression and Autologous Bone Marrow Concentrate for Treatment of Femoral Head Osteonecrosis: A Randomized Prospective Study. Orthop Rev (Pavia). 2016;8(1):6162.[22] Lim YW, Kim YS, Lee JW, et al. Stem cell implantation for osteonecrosis of the femoral head. Exp Mol Med. 2013;45:e61.[23] Hernigou P, Bernaudin F, Reinert P, et al. Bone-marrow transplantation in sickle-cell disease. Effect on osteonecrosis: a case report with a four-year follow-up. J Bone Joint Surg Am. 1997;79(11): 1726-1730.[24] 郑艳峰,朱家骏,徐志文,等. 研究经皮自体红骨髓移植治疗严重粉碎性骨折[J]. 中国骨与关节损伤杂志, 2008,23(8):693-694.[25] 贾世双,张堃,李双,等.股骨颈并股骨干骨折的修复:骨移植与组织及细胞移植效果评价[J]. 中国组织工程研究, 2015,19(21): 3424-3428.[26] Rodriguez-Collazo ER, Urso ML. Combined use of the Ilizarov method, concentrated bone marrow aspirate (cBMA), and platelet-rich plasma (PRP) to expedite healing of bimalleolar fractures. Strategies Trauma Limb Reconstr. 2015;10(3):161-166.[27] Desai P, Hasan SM, Zambrana L, et al. Bone Mesenchymal Stem Cells with Growth Factors Successfully Treat Nonunions and Delayed Unions. HSS J. 2015;11(2):104-111.[28] Murena L, Canton G, Vulcano E, et al. Treatment of humeral shaft aseptic nonunions in elderly patients with opposite structural allograft, BMP-7, and mesenchymal stem cells. Orthopedics. 2014;37(2):e201-206.[29] Qu Z, Fang G, Cui Z, et al. Cell therapy for bone nonunion: a retrospective study. Minerva Med. 2015;106(6):315-321.[30] Johnson K, Zhu S, Tremblay MS, et al. A stem cell-based approach to cartilage repair. Science. 2012;336(6082):717-721.[31] 韩志军,刘晓峥,任华. 猪骨髓间充质干细胞体外诱导构建组织工程化软骨[J]. 中国组织工程研究与临床康复, 2008,12(2):209-212. [32] 杨萌,邵博,龚忠诚,等.三维环境下软骨细胞诱导滑膜间充质干细胞向软骨样细胞的分化[J]. 中国组织工程研究, 2016,20(11):1544-1550. [33] Centeno CJ, Busse D, Kisiday J, et al. Regeneration of meniscus cartilage in a knee treated with percutaneously implanted autologous mesenchymal stem cells. Med Hypotheses. 2008; 71(6):900-908. [34] Davatchi F, Sadeghi Abdollahi B, Mohyeddin M, et al. Mesenchymal stem cell therapy for knee osteoarthritis: 5 years follow-up of three patients. Int J Rheum Dis. 2016;19(3):219-225.[35] Wong KL, Lee KB, Tai BC, et al. Injectable cultured bone marrow-derived mesenchymal stem cells in varus knees with cartilage defects undergoing high tibial osteotomy: a prospective, randomized controlled clinical trial with 2 years' follow-up. Arthroscopy. 2013;29(12):2020-2028.[36] Pak J. Regeneration of human bones in hip osteonecrosis and human cartilage in knee osteoarthritis with autologous adipose-tissue-derived stem cells: a case series. J Med Case Rep. 2011;5:296.[37] Jo CH, Lee YG, Shin WH, et al. Intra-articular injection of mesenchymal stem cells for the treatment of osteoarthritis of the knee: a proof-of-concept clinical trial. Stem Cells. 2014;32(5): 1254-1266. [38] Kuroda R, Ishida K, Matsumoto T, et al. Treatment of a full-thickness articular cartilage defect in the femoral condyle of an athlete with autologous bone-marrow stromal cells. Osteoarthritis Cartilage. 2007;15(2):226-231. [39] Kasemkijwattana C, Hongeng S, Kesprayura S, et al. Autologous bone marrow mesenchymal stem cells implantation for cartilage defects: two cases report. J Med Assoc Thai. 2011;94(3):395-400. [40] Gobbi A, Karnatzikos G, Scotti C, et al. One-Step Cartilage Repair with Bone Marrow Aspirate Concentrated Cells and Collagen Matrix in Full-Thickness Knee Cartilage Lesions: Results at 2-Year Follow-up. Cartilage. 2011;2(3):286-299.[41] De Windt TS, Vonk LA, Slaper-Cortenbach IC, et al. Allogeneic Mesenchymal Stem Cells Stimulate Cartilage Regeneration and Are Safe for Single-Stage Cartilage Repair in Humans upon Mixture with Recycled Autologous Chondrons. Stem Cells. 2017;35(1):256-264.[42] Wu D, Lee S, Luo J, et al. Intraneural Injection of ATP Stimulates Regeneration of Primary Sensory Axons in the Spinal Cord. J Neurosci. 2018;38(6):1351-1365.[43] Karamouzian S, Nematollahi-Mahani SN, Nakhaee N, et al. Clinical safety and primary efficacy of bone marrow mesenchymal cell transplantation in subacute spinal cord injured patients. Clin Neurol Neurosurg. 2012;114(7):935-939.[44] Park JH, Kim DY, Sung IY, et al. Long-term results of spinal cord injury therapy using mesenchymal stem cells derived from bone marrow in humans. Neurosurgery. 2012;70(5):1238-1247. [45] Dai G, Liu X, Zhang Z, et al. Transplantation of autologous bone marrow mesenchymal stem cells in the treatment of complete and chronic cervical spinal cord injury. Brain Res. 2013;1533:73-79.[46] Cheng H, Liu X, Hua R, et al. Clinical observation of umbilical cord mesenchymal stem cell transplantation in treatment for sequelae of thoracolumbar spinal cord injury. J Transl Med. 2014;12:253.[47] Mendonça MV, Larocca TF, de Freitas Souza BS, et al. Safety and neurological assessments after autologous transplantation of bone marrow mesenchymal stem cells in subjects with chronic spinal cord injury. Stem Cell Res Ther. 2014;5(6):126.[48] Oe K, Kushida T, Okamoto N, et al. New strategies for anterior cruciate ligament partial rupture using bone marrow transplantation in rats. Stem Cells Dev. 2011;20(4):671-679.[49] Lacitignola L, Crovace A, Rossi G, et al. Cell therapy for tendinitis, experimental and clinical report. Vet Res Commun. 2008;32 Suppl 1:S33-38.[50] Lee SY, Kim W, Lim C, et al. Treatment of Lateral Epicondylosis by Using Allogeneic Adipose-Derived Mesenchymal Stem Cells: A Pilot Study. Stem Cells. 2015;33(10):2995-3005.[51] Pascual-Garrido C, Rolón A, Makino A. Treatment of chronic patellar tendinopathy with autologous bone marrow stem cells: a 5-year-followup. Stem Cells Int. 2012;2012:953510.[52] Sahoo S, Toh SL, Goh JC. A bFGF-releasing silk/PLGA-based biohybrid scaffold for ligament/tendon tissue engineering using mesenchymal progenitor cells. Biomaterials. 2010;31(11): 2990-2998. [53] Chen X, Zou XH, Yin GL, et al. Tendon tissue engineering with mesenchymal stem cells and biografts: an option for large tendon defects. Front Biosci (Schol Ed). 2009;1:23-32.[54] Ju YJ, Muneta T, Yoshimura H, et al. Synovial mesenchymal stem cells accelerate early remodeling of tendon-bone healing. Cell Tissue Res. 2008;332(3):469-478.[55] Ellera Gomes JL, da Silva RC, Silla LM, et al. Conventional rotator cuff repair complemented by the aid of mononuclear autologous stem cells. Knee Surg Sports Traumatol Arthrosc. 2012;20(2): 373-377. [56] Hernigou P, Flouzat Lachaniette CH, et al. Biologic augmentation of rotator cuff repair with mesenchymal stem cells during arthroscopy improves healing and prevents further tears: a case-controlled study. Int Orthop. 2014;38(9):1811-1818.[57] Le Maitre CL, Freemont AJ, Hoyland JA. Accelerated cellular senescence in degenerate intervertebral discs: a possible role in the pathogenesis of intervertebral disc degeneration. Arthritis Res Ther. 2007;9(3):R45.[58] Zhang Y, Drapeau S, Howard SA, et al. Transplantation of goat bone marrow stromal cells to the degenerating intervertebral disc in a goat disc injury model. Spine (Phila Pa 1976). 2011;36(5): 372-377. [59] Sakai D, Mochida J, Iwashina T, et al. Regenerative effects of transplanting mesenchymal stem cells embedded in atelocollagen to the degenerated intervertebral disc. Biomaterials. 2006;27(3): 335-345. [60] Sobajima S, Vadala G, Shimer A, et al. Feasibility of a stem cell therapy for intervertebral disc degeneration. Spine J. 2008;8(6): 888-896.[61] Orozco L, Soler R, Morera C, et al. Intervertebral disc repair by autologous mesenchymal bone marrow cells: a pilot study. Transplantation. 2011;92(7):822-828.[62] Yoshikawa T, Ueda Y, Miyazaki K, et al. Disc regeneration therapy using marrow mesenchymal cell transplantation: a report of two case studies. Spine (Phila Pa 1976). 2010;35(11):E475-480.[63] Pettine K, Suzuki R, Sand T, et al. Treatment of discogenic back pain with autologous bone marrow concentrate injection with minimum two year follow-up. Int Orthop. 2016;40(1):135-140. |
| [1] | Pu Rui, Chen Ziyang, Yuan Lingyan. Characteristics and effects of exosomes from different cell sources in cardioprotection [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-. |
| [2] | Lin Qingfan, Xie Yixin, Chen Wanqing, Ye Zhenzhong, Chen Youfang. Human placenta-derived mesenchymal stem cell conditioned medium can upregulate BeWo cell viability and zonula occludens expression under hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 4970-4975. |
| [3] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [4] | Zhang Chao, Lü Xin. Heterotopic ossification after acetabular fracture fixation: risk factors, prevention and treatment progress [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1434-1439. |
| [5] | Zhou Jihui, Li Xinzhi, Zhou You, Huang Wei, Chen Wenyao. Multiple problems in the selection of implants for patellar fracture [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1440-1445. |
| [6] | Wang Debin, Bi Zhenggang. Related problems in anatomy mechanics, injury characteristics, fixed repair and three-dimensional technology application for olecranon fracture-dislocations [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1446-1451. |
| [7] | Zhang Xiumei, Zhai Yunkai, Zhao Jie, Zhao Meng. Research hotspots of organoid models in recent 10 years: a search in domestic and foreign databases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1249-1255. |
| [8] | Ji Zhixiang, Lan Changgong. Polymorphism of urate transporter in gout and its correlation with gout treatment [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1290-1298. |
| [9] | Yuan Mei, Zhang Xinxin, Guo Yisha, Bi Xia. Diagnostic potential of circulating microRNA in vascular cognitive impairment [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1299-1304. |
| [10] | Hou Jingying, Yu Menglei, Guo Tianzhu, Long Huibao, Wu Hao. Hypoxia preconditioning promotes bone marrow mesenchymal stem cells survival and vascularization through the activation of HIF-1α/MALAT1/VEGFA pathway [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 985-990. |
| [11] | Shi Yangyang, Qin Yingfei, Wu Fuling, He Xiao, Zhang Xuejing. Pretreatment of placental mesenchymal stem cells to prevent bronchiolitis in mice [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 991-995. |
| [12] | Liang Xueqi, Guo Lijiao, Chen Hejie, Wu Jie, Sun Yaqi, Xing Zhikun, Zou Hailiang, Chen Xueling, Wu Xiangwei. Alveolar echinococcosis protoscolices inhibits the differentiation of bone marrow mesenchymal stem cells into fibroblasts [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 996-1001. |
| [13] | Fan Quanbao, Luo Huina, Wang Bingyun, Chen Shengfeng, Cui Lianxu, Jiang Wenkang, Zhao Mingming, Wang Jingjing, Luo Dongzhang, Chen Zhisheng, Bai Yinshan, Liu Canying, Zhang Hui. Biological characteristics of canine adipose-derived mesenchymal stem cells cultured in hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1002-1007. |
| [14] | Geng Yao, Yin Zhiliang, Li Xingping, Xiao Dongqin, Hou Weiguang. Role of hsa-miRNA-223-3p in regulating osteogenic differentiation of human bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1008-1013. |
| [15] | Lun Zhigang, Jin Jing, Wang Tianyan, Li Aimin. Effect of peroxiredoxin 6 on proliferation and differentiation of bone marrow mesenchymal stem cells into neural lineage in vitro [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1014-1018. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||