Chinese Journal of Tissue Engineering Research ›› 2018, Vol. 22 ›› Issue (9): 1470-1476.doi: 10.3969/j.issn.2095-4344.0481
Human umbilical cord-derived mesenchymal stem cells in the treatment of myocardial infarction: existing problems, effects and prospects
Wang Yuan-fei1, Li Ya-xiong1, Zhang Ya-yong1, Jiang Li-hong1, 2
- 1Department of Cardiothoracic Surgery, Yan’an Affiliated Hospital of Kunming Medical University, Institute of Cardiovascular Surgery in Yunnan, Key Laboratory of Cardiovascular Diseases in Yunnan, Yan’an Hospital of Kunming, Kunming 650051, Yunnan Province, China; 2the First People’s Hospital of Yunnan Province, Kunming 650100, Yunnan Province, China
-
Revised:2017-02-10Online:2018-03-28Published:2018-04-03 -
Contact:Jiang Li-hong, M.D., Professor, Doctoral supervisor, Master’s supervisor, Department of Cardiothoracic Surgery, Yan’an Affiliated Hospital of Kunming Medical University, Institute of Cardiovascular Surgery in Yunnan, Key Laboratory of Cardiovascular Diseases in Yunnan, Yan’an Hospital of Kunming, Kunming 650051, Yunnan Province, China; the First People’s Hospital of Yunnan Province, Kunming 650100, Yunnan Province, China;Zhang Ya-yong, Master, Attending physician, Department of Cardiothoracic Surgery, Yan’an Affiliated Hospital of Kunming Medical University, Institute of Cardiovascular Surgery in Yunnan, Key Laboratory of Cardiovascular Diseases in Yunnan, Yan’an Hospital of Kunming, Kunming 650051, Yunnan Province, China -
About author:Wang Yuan-fei, Master candidate, Department of Cardiothoracic Surgery, Yan’an Affiliated Hospital of Kunming Medical University, Institute of Cardiovascular Surgery in Yunnan, Key Laboratory of Cardiovascular Diseases in Yunnan, Yan’an Hospital of Kunming, Kunming 650051, Yunnan Province, China -
Supported by:the Clinical Department Construction Project of China; the Therapeutic Technology Innovation Team for Cardiovascular Surgery in Yunnan Province, No. 2015HC015
CLC Number:
Cite this article
Wang Yuan-fei, Li Ya-xiong, Zhang Ya-yong, Jiang Li-hong. Human umbilical cord-derived mesenchymal stem cells in the treatment of myocardial infarction: existing problems, effects and prospects[J]. Chinese Journal of Tissue Engineering Research, 2018, 22(9): 1470-1476.
share this article

2.1 huc-MSCs概述 脐带由卵黄囊和尿囊所构成,是胚胎或胎儿与胎盘之间的通道。其中,脐带基质中含有的胶状物质叫沃顿胶(Wharton's jelly, WJ),主要由多糖组成。1991年,Mcelreavey等[9]第一次从沃顿胶中分离出成纤维样细胞,这些细胞因为表达CD29、CD44、CD73、CD105,不表达CD34、CD45,并能向脂肪细胞和成骨细胞谱系分化,于2004年被确定为间充质干细胞。目前,己知人脐带中有4种间充质干细胞的来源:沃顿胶、脐血管周、脐带血和脐静脉内皮下膜,其中,沃顿胶是当前脐带源间充质干细胞的主要来源[10]。 沃顿胶间充质干细胞起源于胚胎发育第13天时的中胚层或内胚层,此时,卵黄囊壁的胚外中胚层内由间充质干细胞密集形成血岛,血岛周边细胞分化为内皮细胞,内皮细胞围成内皮管,即原始血管,内皮管不断向外出芽延伸,逐渐形成一个丛状分布的内皮管网,分布于胚体内外的间充质中,被称为“心血管系统发源地”-“干细胞池”[11]。有研究证实,沃顿胶间充质干细胞能被诱导分化为表达α-肌动蛋白、肌钙蛋白T和Connexin-43的细胞,这些细胞为心肌再生所必需[12]。更多证据显示,沃顿胶间充质干细胞可诱导分化为内皮细胞、心肌细胞,它们能够融入到血管和缺血的心肌组织中,从而改善心功能[13-14]。因此,有研究者认为沃顿胶间充质干细胞才是真正意义的原始干细胞[15]。 Garzon等[16]研究沃顿胶间充质干细胞传代培养的细胞活性,发现可能存在着复杂的促凋亡基因和抗凋亡基因平衡的问题,尤其是促凋亡基因FASTKD2、BNIP3L和抗凋亡基因TNFAIP8、BCL2L2;并且指出,沃顿胶间充质干细胞的存活率最高的时期是第5代和第6代。因此,用于细胞治疗和组织工程的首选方案是第5代和第6代沃顿胶间充质干细胞。 2.2 huc-MSCs移植对动物心肌梗死模型心功能修复效果的研究 由于huc-MSCs具有较低的免疫原性和较高的血管生成率使其成为移植治疗心肌梗死的重要替代细胞,得到了广泛的应用,并获得了显著效果,见表1[5,13,17-19]。"

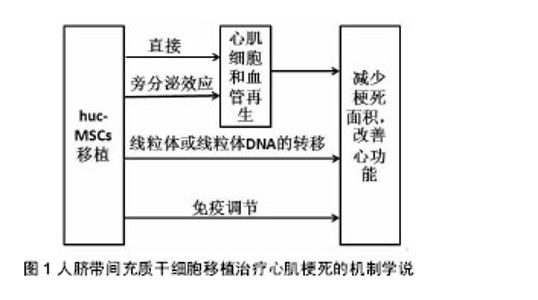
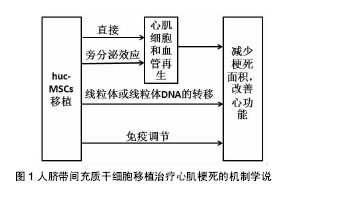
2.3 huc-MSCs移植治疗心肌梗死的临床研究 尽管huc-MSCs治疗心肌梗死的确切机制仍不清楚,但由于临床前研究的丰富成果显示出huc-MSCs移植治疗的可行性、有效性和安全性,目前正逐步开展人心肌细胞再生的临床试验。 2.3.1 移植的时间与剂量 移植的时间:关于细胞移植时间的选择,目前尚没有一个系统的关于移植时机的研究。但对于急性心脏病发作后,存在一个最佳的治疗窗口,在此期间干细胞疗法可以发挥最佳的治疗作用[20]。然而,由于自体骨髓间充质干细胞难以在14 d内进行完整的分离、鉴定、纯化及获得所需的剂量,因此错过了细胞治疗的最佳时间窗口[21]。而huc-MSCs的出现为临床细胞治疗的探索提供了新的路线,因为它有经过验证的基础设施和趋于完善的操作程序,可以在最有利的时间窗口给符合条件的患者使用。此外,有研究指出huc-MSCs对陈旧性心肌梗死患者同样可以显著降低患者心肌梗死面积和提高左室射血分数,但具体机制仍不清楚[22-23]。 移植剂量:与其他药物一样,干细胞的注射剂量与治疗效果存在一定的相关性,剂量过低起不到治疗效果;剂量过高,对移植区增加负担,细胞存活率低,同样达不到治疗作用。目前关于干细胞的移植数量仍没有统一的说法,可能与培养的干细胞质量和(或)活性有关。Li等[24]分别采用3×106、4×106和5×106数量级的huc-MSCs通过冠状动脉内注射治疗老年人冠状动脉慢性完全闭塞,结果发现,3种剂量的干细胞均能显著降低患者心肌梗死面积和提高左室射血分数,尤以3×106数量级最明显,但3者没有差异,说明3×106数量级的huc-MSCs治疗效果更好。但Gao等[25]采用6×106数量级的沃顿胶间充质干细胞治疗118例ST-T段抬高的急性心肌梗死患者,结果同样表明,患者的心肌存活率、梗死区再灌注和左室射血分数均显著高于对照组(P < 0.05)。然而,上述结果没有统一的最佳移植剂量,这也是限制临床应用细胞治疗疾病的因素之一。 2.3.2 移植的效果与安全性 移植的效果:Gao等[25]进行18个月的随访结果显示沃顿胶间充质干细胞治疗是有效的。Li等[24]研究在2年的随访时间内发现,患者的心功能显著提高。田洪榛等[26]通过经冠状动脉移植沃顿胶间充质干细胞治疗缺血性心力衰竭指出,18个月后患者左室功能得到提高,左室容量减少,左心室重构速度减慢,甚至2年后患者的活动耐量及生活质量仍得到了提高。此外,梁晓辉等[27]指出,沃顿胶间充质干细胞移植治疗心肌梗死后心力衰竭,可以显著降低患者的NT-proBNP水平。另外,移植的效果还与心肌梗死人群、局部心肌微环境、是否与组织工程技术结合、移植后疗效评价体系均有关系。 移植的安全性:Gao等[25]、Li等[24]和田洪榛等[26]研究证明了huc-MSCs经冠状动脉内注射是安全的。Musialek等[28]评价了10例使用沃顿胶间充质干细胞治疗心肌梗死的效果,随访1年后,无心脏不良事件的发生。田洪榛等[26]也发现,患者在治疗的过程中并没有出现血流减慢、肌钙蛋白异常升高等急性并发症,表明移植沃顿胶间充质干细胞时不会出现微血管及冠状动脉的梗死。此外,由于沃顿胶间充质干细胞能够低水平表达多潜能胚胎干细胞标记,如POUF1,NANOG,SOX2和LIN28等,能够明显上调多种细胞因子如IL-12A等,从而说明了其为何不引起畸胎瘤的形成和解释了具有抗癌的特性[29]。Gauthaman等[30]比较了沃顿胶间充质干细胞和胚胎干细胞在免疫缺陷小鼠中的肿瘤形成能力,结果发现所有接受人胚胎干细胞的动物均出现了免疫应答并发生了肿瘤,而接受沃顿胶间充质干细胞的动物却并没有出现免疫应答和肿瘤的发生,甚至到20周时依然没有肿瘤发生。作者认为,沃顿胶间充质干细胞在移植治疗心肌梗死时既不会引起微血管及冠状动脉的梗死也不会诱发肿瘤,在细胞治疗上是安全的。 2.4 huc-MSCs移植治疗心肌梗死的机制 目前对于huc-MSCs移植治疗心肌梗死改善心功能的机制尚不明确,认为可能的机制有以下几种学说(图1)。"
| [1] van den Borne SW, Diez J, Blankesteijn WM, et al. Myocardial remodeling after infarction: the role of myofibroblasts. Nat Rev Cardiol. 2010;7(1):30-37.[2] Coulombe KL, Bajpai VK, Andreadis ST, et al. Heart regeneration with engineered myocardial tissue. Annu Rev Biomed Eng. 2014;16:1-28.[3] Winters AA, Bou-Ghannam S, Thorp H, et al. Evaluation of Multiple Biological Therapies for Ischemic Cardiac Disease. Cell Transplant. 2016;25(9):1591-1607.[4] Ma T, Sun J, Zhao Z, et al. A brief review: adipose-derived stem cells and their therapeutic potential in cardiovascular diseases. Stem Cell Res Ther. 2017;8(1):124.[5] Santos Nascimento D, Mosqueira D, Sousa LM, et al. Human umbilical cord tissue-derived mesenchymal stromal cells attenuate remodeling after myocardial infarction by proangiogenic, antiapoptotic, and endogenous cell-activation mechanisms. Stem Cell Res Ther. 2014;5(1):5.[6] Li L, Jaiswal PK, Makhoul G, et al. Hypoxia modulates cell migration and proliferation in placenta-derived mesenchymal stem cells. J Thorac Cardiovasc Surg. 2017;154(2):543-552.[7] Perea-Gil I, Monguió-Tortajada M, Gálvez-Montón C, et al. Preclinical evaluation of the immunomodulatory properties of cardiac adipose tissue progenitor cells using umbilical cord blood mesenchymal stem cells: a direct comparative study. Biomed Res Int. 2015;2015:439808.[8] 王永伟,张晋,徐曼,等. 人脐带和骨髓来源间充质干细胞移植治疗小鼠心肌梗死的比较研究[J]. 中国循证心血管医学杂志, 2013,5(2):196-199.[9] McElreavey KD, Irvine AI, Ennis KT, et al. Isolation, culture and characterisation of fibroblast-like cells derived from the Wharton's jelly portion of human umbilical cord. Biochem Soc Trans. 1991;19(1):29S.[10] 张嵬,刘晓程. 沃顿胶体间充质干细胞生物学特性及在心血管领域的应用[J]. 中国组织工程研究,2013,17(23):4320-4327.[11] 高连如. “生物药”——Wharton's jelly源间充质干细胞[J]. 转化医学杂志, 2016, 5(4): 193-197.[12] Gao LR, Zhang NK, Ding QA, et al. Common expression of stemness molecular markers and early cardiac transcription factors in human Wharton's jelly-derived mesenchymal stem cells and embryonic stem cells. Cell Transplant. 2013;22(10): 1883-900.[13] Zhang W, Liu XC, Yang L, et al. Wharton's jelly-derived mesenchymal stem cells promote myocardial regeneration and cardiac repair after miniswine acute myocardial infarction. Coron Artery Dis. 2013;24(7):549-558.[14] López Y, Lutjemeier B, Seshareddy K, et al. Wharton's jelly or bone marrow mesenchymal stromal cells improve cardiac function following myocardial infarction for more than 32 weeks in a rat model: a preliminary report. Curr Stem Cell Res Ther. 2013;8(1):46-59.[15] Kim DW, Staples M, Shinozuka K, et al. Wharton's jelly-derived mesenchymal stem cells: phenotypic characterization and optimizing their therapeutic potential for clinical applications. Int J Mol Sci. 2013;14(6):11692-11712.[16] Garzón I, Pérez-Köhler B, Garrido-Gómez J, et al. Evaluation of the cell viability of human Wharton's jelly stem cells for use in cell therapy. Tissue Eng Part C Methods. 2012;18(6): 408-419.[17] 裴宇,刘晓程,张嵬,等. 同种、异种脐带间充质干细胞移植治疗猪急性心肌梗死模型疗效对比[J]. 中华全科医学, 2015, 13(12): 1935-1937.[18] Latifpour M, Nematollahi-Mahani SN, Deilamy M, et al. Improvement in cardiac function following transplantation of human umbilical cord matrix-derived mesenchymal cells. Cardiology. 2011;120(1):9-18.[19] Liu CB, Huang H, Sun P, et al. Human Umbilical Cord-Derived Mesenchymal Stromal Cells Improve Left Ventricular Function, Perfusion, and Remodeling in a Porcine Model of Chronic Myocardial Ischemia. Stem Cells Transl Med. 2016;5(8): 1004-1013.[20] Fisher SA, Zhang H, Doree C, et al. Stem cell treatment for acute myocardial infarction. Cochrane Database Syst Rev. 2015;(9):CD006536.[21] Gao LR, Pei XT, Ding QA, et al. A critical challenge: dosage-related efficacy and acute complication intracoronary injection of autologous bone marrow mesenchymal stem cells in acute myocardial infarction. Int J Cardiol. 2013;168(4): 3191-3199.[22] 徐培敬,胡有东,张枫林,等. 人脐带间充质干细胞治疗老年心肌梗死的CD34+细胞、Toll样受体2和Toll样受体4的表达水平分析[J]. 中华损伤与修复杂志:电子版, 2013, 8(5):487-491.[23] 李侠,郭燕,胡有东,等. 脐带间充质干细胞治疗老年人陈旧性心肌梗死对血小板糖蛋白和内皮细胞黏附分子的影响[J]. 中华老年医学杂志, 2013, 32(6):582-585.[24] Li X, Hu YD, Guo Y, et al. Safety and efficacy of intracoronary human umbilical cord-derived mesenchymal stem cell treatment for very old patients with coronary chronic total occlusion. Curr Pharm Des. 2015;21(11):1426-1432.[25] Gao LR, Chen Y, Zhang NK, et al. Intracoronary infusion of Wharton's jelly-derived mesenchymal stem cells in acute myocardial infarction: double-blind, randomized controlled trial. BMC Med. 2015;13:162.[26] 田洪榛,陈江,张晓霞,等. 脐带华通胶间充质干细胞治疗缺血性心力衰竭的2年随访观察[J]. 解放军医学院学报, 2016, 37(5): 407-412.[27] 梁晓辉,赵子粼,罗建春,等. WJ-MHCs对心肌梗死后心力衰竭患者NT-proBNP水平的影响[J]. 西南国防医药, 2014, 24(5): 483-486.[28] Musialek P, Mazurek A, Jarocha D, et al. Myocardial regeneration strategy using Wharton's jelly mesenchymal stem cells as an off-the-shelf 'unlimited' therapeutic agent: results from the Acute Myocardial Infarction First-in-Man Study. Postepy Kardiol Interwencyjnej. 2015;11(2):100-107.[29] Fong CY, Chak LL, Biswas A, et al. Human Wharton's jelly stem cells have unique transcriptome profiles compared to human embryonic stem cells and other mesenchymal stem cells. Stem Cell Rev. 2011;7(1):1-16.[30] Gauthaman K, Fong CY, Suganya CA, et al. Extra-embryonic human Wharton's jelly stem cells do not induce tumorigenesis, unlike human embryonic stem cells. Reprod Biomed Online. 2012;24(2):235-246.[31] Di Siena S, Gimmelli R, Nori SL, et al. Activated c-Kit receptor in the heart promotes cardiac repair and regeneration after injury. Cell Death Dis. 2016;7(7):e2317.[32] Sullivan KE, Quinn KP, Tang KM, et al. Extracellular matrix remodeling following myocardial infarction influences the therapeutic potential of mesenchymal stem cells. Stem Cell Res Ther. 2014;5(1):14.[33] Yannarelli G, Dayan V, Pacienza N, et al. Human umbilical cord perivascular cells exhibit enhanced cardiomyocyte reprogramming and cardiac function after experimental acute myocardial infarction. Cell Transplant. 2013;22(9):1651-1666.[34] Xu H, Dong H, Zhao M. Effects of human umbilical cord mesenchymal stem cells on vascular endothelial growth factor and IL-6 expression in tissue of AMI rats. Zhonghua Wei Zhong Bing Ji Jiu Yi Xue. 2017;29(6):511-514.[35] Zhao Y, Sun X, Cao W, et al. Exosomes Derived from Human Umbilical Cord Mesenchymal Stem Cells Relieve Acute Myocardial Ischemic Injury. Stem Cells Int. 2015;2015: 761643.[36] Wu SZ, Li YL, Huang W, et al. Paracrine effect of CXCR4-overexpressing mesenchymal stem cells on ischemic heart injury. Cell Biochem Funct. 2017;35(2):113-123.[37] Marsano A, Maidhof R, Luo J, et al. The effect of controlled expression of VEGF by transduced myoblasts in a cardiac patch on vascularization in a mouse model of myocardial infarction. Biomaterials. 2013;34(2):393-401.[38] Cho HM, Kim PH, Chang HK, et al. Targeted Genome Engineering to Control VEGF Expression in Human Umbilical Cord Blood-Derived Mesenchymal Stem Cells: Potential Implications for the Treatment of Myocardial Infarction. Stem Cells Transl Med. 2017;6(3):1040-1051.[39] Ma J, Zhao Y, Sun L, et al. Exosomes Derived from Akt-Modified Human Umbilical Cord Mesenchymal Stem Cells Improve Cardiac Regeneration and Promote Angiogenesis via Activating Platelet-Derived Growth Factor D. Stem Cells Transl Med. 2017;6(1):51-59.[40] Zhao L, Liu X, Zhang Y, et al. Enhanced cell survival and paracrine effects of mesenchymal stem cells overexpressing hepatocyte growth factor promote cardioprotection in myocardial infarction. Exp Cell Res. 2016;344(1):30-39.[41] Arechederra M, Carmona R, González-Nuñez M, et al. Met signaling in cardiomyocytes is required for normal cardiac function in adult mice. Biochim Biophys Acta. 2013;1832(12): 2204-2215.[42] Kim BR, Seo SH, Park MS, et al. sMEK1 inhibits endothelial cell proliferation by attenuating VEGFR-2-dependent-Akt/eNOS/HIF-1α signaling pathways. Oncotarget. 2015;6(31):31830-31843.[43] 曹文明,孙丽,赵媛媛,等. 脐带MSC来源外体上调Smad7表达修复大鼠急性心肌损伤[J]. 临床检验杂志, 2015, 33(7): 527-531.[44] 李佳,辛毅,崔巍,等.人脐带间充质干细胞外泌小体保护缺氧复氧损伤的心肌细胞[J]. 中国病理生理杂志, 2016,32(4): 577-583.[45] Sabapathy V, Sundaram B, V M S, et al. Human Wharton's Jelly Mesenchymal Stem Cells plasticity augments scar-free skin wound healing with hair growth. PLoS One. 2014;9(4): e93726.[46] Schernthaner C, Paar V, Wernly B, et al. Elevated plasma levels of interleukin-16 in patients with acute myocardial infarction. Medicine (Baltimore). 2017;96(44):e8396.[47] Banerjee I, Fuseler JW, Intwala AR, et al. IL-6 loss causes ventricular dysfunction, fibrosis, reduced capillary density, and dramatically alters the cell populations of the developing and adult heart. Am J Physiol Heart Circ Physiol. 2009;296(5): H1694-1704.[48] Prockop DJ, Oh JY. Medical therapies with adult stem/progenitor cells (MSCs): a backward journey from dramatic results in vivo to the cellular and molecular explanations. J Cell Biochem. 2012;113(5):1460-1469.[49] Arutyunyan I, Elchaninov A, Makarov A, et al. Umbilical Cord as Prospective Source for Mesenchymal Stem Cell-Based Therapy. Stem Cells Int. 2016;2016:6901286.[50] Lin HY, Liou CW, Chen SD, et al. Mitochondrial transfer from Wharton's jelly-derived mesenchymal stem cells to mitochondria-defective cells recaptures impaired mitochondrial function. Mitochondrion. 2015;22:31-44.[51] Weiss ML, Anderson C, Medicetty S, et al. Immune properties of human umbilical cord Wharton's jelly-derived cells. Stem Cells. 2008;26(11):2865-2874.[52] 武丽萍,蔡力,陶剑虹,等. 不同月龄人脐带间充质干细胞移植梗死心肌的血运重建[J]. 中国组织工程研究, 2013, 17(49): 8520-8526.[53] 王巍,李肖甫,李中健. 不同孕周人脐带间充质干细胞移植改善心肌梗死模型心脏功能的比较[J]. 中国组织工程研究, 2016, 20(6):79-806.[54] 赵璐洋,孙瑛,李连冲. 心室内注射人脐带间充质干细胞改善心肌梗死大鼠心功能及作用机制[J]. 中国组织工程研究, 2017, 21(25):4026-4031. |
| [1] | Li Xuan, Sun Yimin, Li Longbiao, Wang Zhenming, Yang Jing, Wang Chenglin, Ye Ling. Manufacturing of nano-modified polycaprolactone microspheres and its biological effects in dental pulp cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(10): 1602-1608. |
| [2] | Liu Xiaogang, Li Tian, Zhang Duo. Effect and mechanism of the effective components of Chinese medicine on promoting the differentiation of bone marrow mesenchymal stem cells into chondrocytes [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(1): 121-126. |
| [3] | Shuai Zhiqin, Chen Jiameng, Liu Taotao, Hu Anling, Li Lisheng, Yu Limei, Xu Shangfu. Research status and problems of stem cells and their derived exosomes for prevention and treatment of vascular restenosis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(1): 138-144. |
| [4] | Li Zhongkang, Zheng Jiahua, Tian Yanpeng, Huang Xianghua. Latest progress and mechanisms of mesenchymal stem cells on premature ovarian failure [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(1): 145-152. |
| [5] | Wang Shuyun, Xie Junhui, Yu Xuefeng. Effect and mechanism of mesenchymal stem cells in the treatment of diabetic nephropathy [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(1): 153-158. |
| [6] | Lin Miaoyuan, Li Yuwan, Liu Yi, Chen Bei, Zhang Li. Research hotspots and application value of tissue-engineered skin [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(1): 159-166. |
| [7] | Song Huifang, Tan Jiayin, Kang Yi, Li Bin, Bi Zhifei, Long Nü, Xia Zhongnian, Guo Rui. Hypoxic pretreatment enhances the protective effect of aged human bone marrow mesenchymal stem cells conditioned medium against H9C2 oxidative stress damage [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(1): 1-6. |
| [8] | Xie Xingqi, Hu Wei, Tu Guanjun. Bone marrow mesenchymal stem cells-derived exosomes combined with chondroitinase ABC for treating spinal cord injury in rats [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(1): 20-26. |
| [9] | Zhang Xuelei, Luo Gan, Yu Shenghui, Gu Zuchao, Peng Xu, He Xueling, Liu Yan, Zhang Xiaomei. Acellular nerve scaffold combined with bone marrow mesenchymal stem cells and platelet-rich gel for femoral nerve injury [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(1): 27-32. |
| [10] | Zhao Laihe, Xia Bing, Ma Teng, Gao Jianbo, Li Shengyou, Gao Xue, Zheng Yi, Hu Guangwen, Luo Zhuojing, Huang Jinghui. Extracellular matrix of Schwann-like cells induced by bone marrow mesenchymal stem cells promotes axonal regeneration after peripheral nerve injury [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(1): 33-39. |
| [11] | Mai Liping, He Guodong, Chen Shaoxian, Zhu Jiening, Hou Xinghua, Zhang Mengzhen, Li Xiaohong. Expression of aldehyde dehydrogenase 3B1 during human bone marrow mesenchymal stem cells senescence [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(1): 40-44. |
| [12] | Yang Tengyun, Li Yanlin, Liu Dejian, Wang Guoliang, Zheng Zhujun. Chondrogenic differentiation of peripheral blood-derived mesenchymal stem cells induced by transforming growth factor beta 3: a dose-effect relationship [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(1): 45-51. |
| [13] | Huang Tao, Cheng Zhijian, Jia Zhiqiang, Zhao Xiaoguang, Wang Lei, Zhai Wenjing, Zhou Yongxin. Mechanism by which miR-146a regulates osteogenic differentiation of adipose derived mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(1): 70-75. |
| [14] | Jing Jin, Zhao Shandi, Chen Long, Peng Shuanglin, Tang Hui, Guo Daijin, Zeng Xinyi, Xiao Jingang. Repair of calvarial defects in osteoporotic mice by adipose-derived stem cells combined with biphasic calcium phosphate ceramic scaffold [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(1): 90-95. |
| [15] | Cui Shuaishuai, Yang Xiaohong. Effects of miRNA in self-renewal, multidirectional differentiation, fate and function regulation of bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(1): 101-106. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||