Chinese Journal of Tissue Engineering Research ›› 2022, Vol. 26 ›› Issue (10): 1530-1536.doi: 10.12307/2022.199
Previous Articles Next Articles
Manufacturing of nano-modified polycaprolactone microspheres and its biological effects in dental pulp cells
Li Xuan, Sun Yimin, Li Longbiao, Wang Zhenming, Yang Jing, Wang Chenglin, Ye Ling
- West China School of Stomatology, Sichuan University, Chengdu 610041, Sichuan Province, China
-
Received:2020-10-12Revised:2020-10-14Accepted:2020-11-28Online:2022-04-08Published:2021-10-25 -
Contact:Ye Ling, MD, Professor, West China School of Stomatology, Sichuan University, Chengdu 610041, Sichuan Province, China -
About author:Li Xuan, Doctoral candidate, West China School of Stomatology, Sichuan University, Chengdu 610041, Sichuan Province, China -
Supported by:the Foundation of Basic Research for Application from the Department of Science and Technology, Sichuan Province, No.2019YFS0035 (to YL); the National Science Foundation for Distinguished Young Scholars of China, No.81825005 (to YL)
CLC Number:
Cite this article
Li Xuan, Sun Yimin, Li Longbiao, Wang Zhenming, Yang Jing, Wang Chenglin, Ye Ling. Manufacturing of nano-modified polycaprolactone microspheres and its biological effects in dental pulp cells[J]. Chinese Journal of Tissue Engineering Research, 2022, 26(10): 1530-1536.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
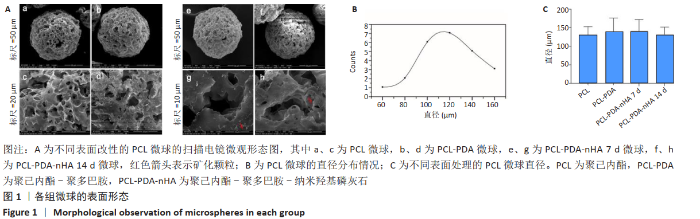
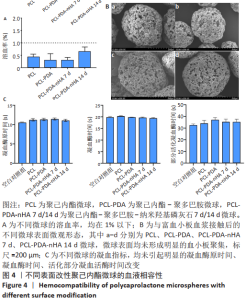
2.1 各组微球的表面形态 如图1A所示,3种微球表面均可见均匀疏松的多孔结构,PCL微球在多巴胺溶液中浸泡 24 h,清洗干燥后可见PDA颗粒覆盖。PCL-PDA-nHA 7 d组和PCL-PDA-nHA 14 d组的尺寸和孔隙无明显差异,但PCL-PDA-nHA 14 d组的微球表面结构更加粗糙,可见更加明显的矿化颗粒形成。由扫描电镜结果计算出PCL组、PCL-PDA组、PCL-PDA-nHA 7 d组和PCL-PDA-nHA 14 d组的直径分布呈正态分布,平均直径分别为(127.82±25.13),(138.93±37.07),(141.50±30.39),(131.50±20.02) μm,4组材料之间的粒径分布无明显差异(P > 0.05),见图1B、C。压汞仪检测得到PCL微球的孔隙率为87.4%,表面孔隙直径在20-50 μm之间。 "
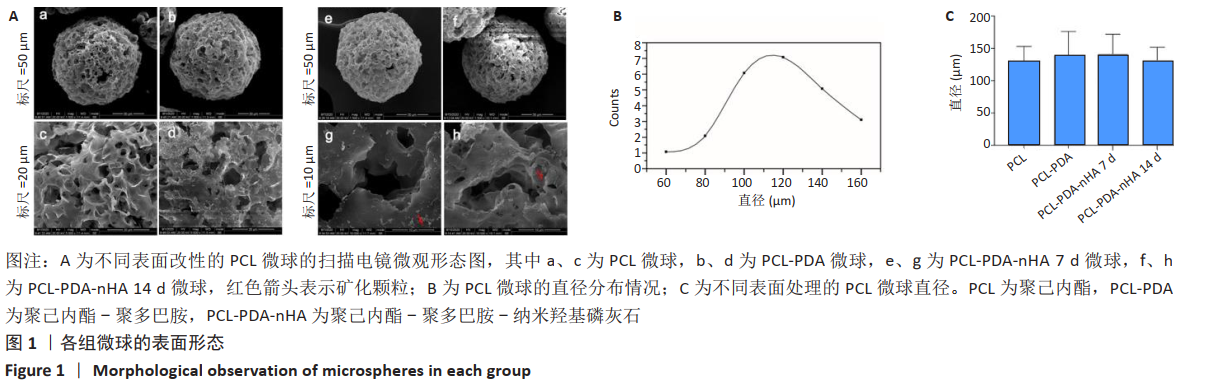
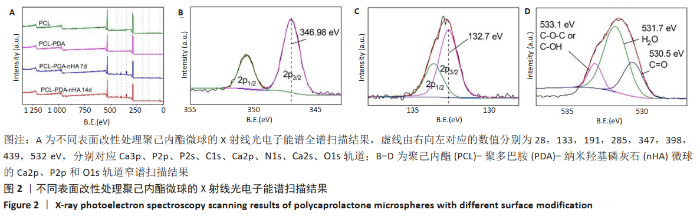
2.2 各组微球X射线光电子能谱分析结果 X射线光电子能谱分析结果如图2A所示,全谱扫描结果显示,PCL组在532,285 eV处有O1s、C1s轨道波峰,这主要来源于PCL的C-O、C=O、C-C等键;PCL-PDA组在398 eV处出现了N1s轨道吸收峰,其来源于PDA的氨基及其形成的化学键;PCL-PDA-nHA组在347 eV处和133 eV处中出现了Ca2p、P2p吸收峰,这可能来源于羟基磷灰石的钙和磷酸根。为了进一步确认化学键的种类,对PCL-PDA-nHA微球的Ca2p、P2p和O1s进行了窄谱扫描。图2B中Ca 2p 3/2轨道的吸收峰在346.98eV处出现,与之前文献报道中HA的吸收峰(347.4,347.2 eV)接 近[17-18],表明钙离子与磷酸基团结合。图2C中P2p吸收峰可以分为2p1/2和2p3/2两个峰,其中2p3/2轨道的吸收峰出现在132.7eV,这与文献报道中羟基磷灰石的P2p3/2轨道的吸收峰(133.4eV)较为相近[18]。在羟基磷灰石中,磷主要与氧形成磷酸根离子。图2D显示O1s的3个吸收峰分别来自O-C、O-H和O=C,这主要来源于PCL自身的C-O-C、C=O化学键和羟基磷灰石中的O-H化学键。结果表明PDA和nHA成功修饰于PCL微球表面。"

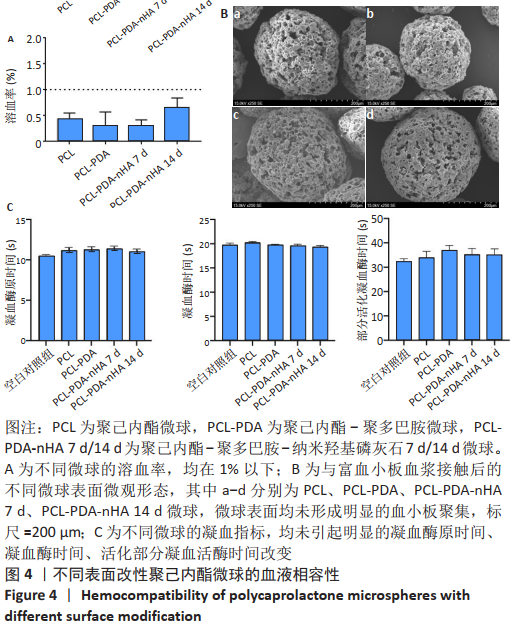
2.3 各组微球的蛋白吸附能力 如图3显示,与未修饰的PCL组相比,PCL-PDA组、PCL-PDA-nHA 7 d组和PCL-PDA-nHA 14 d组的蛋白吸附能力明显增强(P < 0.01);PCL-PDA组与PCL-PDA-nHA 7 d组的蛋白吸附能力相似(P > 0.05),PCL-PDA-nHA 14 d组的蛋白吸附能力高于PCL-PDA-nHA 7 d组(P < 0.001)。 2.4 各组微球血液相容性检测结果 如图4A所示,各组微球的溶血率均在1%以下,符合ISO 10993-4:2017《医疗器械生物学评价第4部分:与血液相互作用试验选择》中标准溶血试验的相关要求(<5%),结果显示修饰了PDA和nHA的PCL微球不会引起明显的溶血。nHA修饰的PCL微球植入体内后不会造成红细胞破裂,避免了因为血红蛋白释放而带来的细胞毒性。"

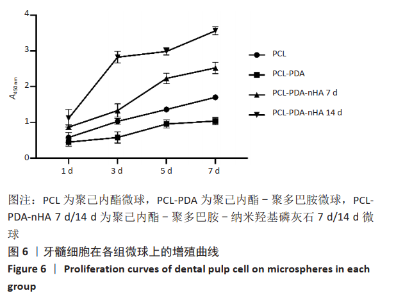
2.6 与微球三维共培养后牙髓细胞的增殖 CCK-8法评价微球的细胞毒性结果如图6所示。细胞与微球共培养1 d后,PCL-PDA组/PCL-PDA-nHA 7 d组的细胞数量与PCL组相比差异无显著性意义(P > 0.05),PCL-PDA-nHA 14 d组的细胞数量多于PCL组(P < 0.05)。随着培养时间的增加,各组微球中的细胞数量均呈上升趋势,PCL-PDA组、PCL-PDA-nHA 7 d组培养5,7 d的细胞数量少于PCL组(P < 0.05),PCL-PDA-nHA 14 d组培养3,5,7 d的细胞数量多于PCL组(P < 0.05),PCL-PDA-nHA 7 d组培养3,5,7 d的细胞数量少于PCL-PDA-nHA 14 d组(P < 0.05)。结果可以看出,经羟基磷灰石修饰后材料的生物相容性明显增强,且羟基磷灰石修饰量增加后,材料的细胞相容性得到了进一步提高。"
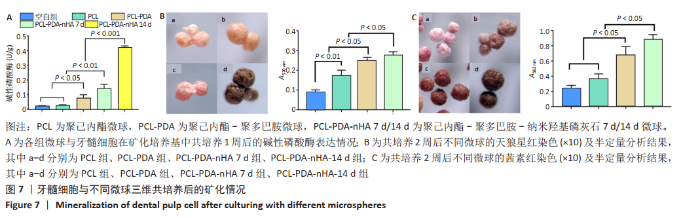
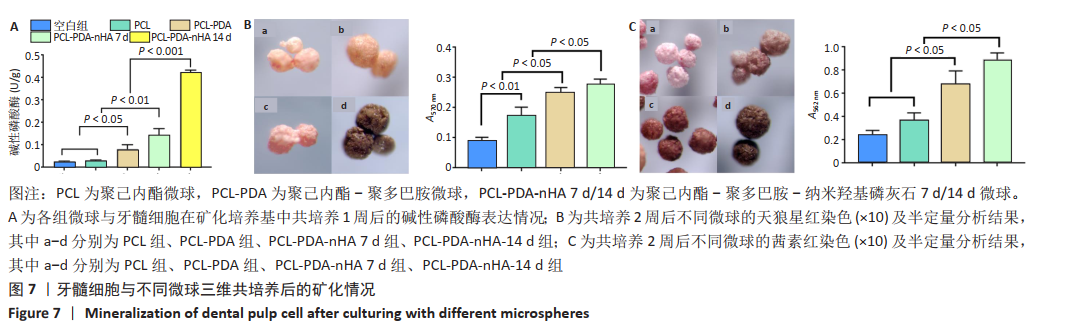
2.7 不同微球对牙髓细胞的矿化诱导作用 碱性磷酸酶检测结果显示,PCL组与空白组的碱性磷酸酶表达水平比较差异无显著性意义(P > 0.05),PCL-PDA组高于空白组、PCL组 (P < 0.05),PCL-PDA-nHA 7 d组高于空白组、PCL微球组、PCL-PDA组(P < 0.01),PCL-PDA-nHA 14 d组高于空白组、PCL微球组、PCL-PDA组、PCL-PDA-nHA 7 d组(P < 0.001),见图7A。 采用天狼星红染色及半定量分析检测胶原分泌水平,见图7B。半定量分析结果显示,PCL-PDA组的细胞胶原分泌水平高于PCL组(P < 0.01),PCL-PDA-nHA 7 d组高于PCL微球组、PCL-PDA组(P < 0.05),PCL-PDA-nHA 14 d组高于PCL组、PCL-PDA组、PCL-PDA-nHA 7 d组(P < 0.05)。 采用茜素红染色及半定量分析检测细胞外基质的矿化水平,见图7C。半定量分析结果显示,PCL-PDA组与PCL组矿化水平比较差异无显著性意义(P > 0.05),PCL-PDA-nHA 7 d组高于PCL-PDA组、PCL组(P < 0.05),PCL-PDA-nHA 14 d组高于PCL-PDA-nHA 7 d组、PCL-PDA组、PCL组(P < 0.05),说明PDA和nHA在PCL表面的共同修饰促进了骨样表型的建立。"
| [1] 樊明文.牙体牙髓病学[M]. 4版.人民卫生出版社,2012. [2] SPIELMAN H, SCHAFFER SB, COHEN MG, et al. Restorative outcomes for endodontically treated teeth in the Practitioners Engaged in Applied Research and Learning Network. J Am Dent Assoc. 2012;143(7):746-755. [3] PETRINO JA, BODA KK, SHAMBARGER S, et al. Challenges in regenerative endodontics: a case series. J Endod. 2010;36(3):536-541. [4] DOYON GE, DUMSHA T, FRAUNHOFER JAV. Fracture Resistance of Human Root Dentin Exposed to Intracanal Calcium Hydroxide. J Endod. 2005;31(12):895-897. [5] Yao Q, Wei B, Liu N, et al. Chondrogenic regeneration using bone marrow clots and a porous polycaprolactone-hydroxyapatite scaffold by three-dimensional printing. Tissue Eng Part A. 2015;21(7-8):1388-1397. [6] 朱惠光,计剑,高长有,等.聚乳酸组织工程支架材料[J].功能高分子学报,2001,14(4):488-92. [7] 戴炜枫,茹敏良,何月英,等.新型官能团化聚己内酯的研究进展[J].高分子通报,2008(9):10-19. [8] LEE H, LEE BP, MESSERSMITH PB. A reversible wet/dry adhesive inspired by mussels and geckos. Nature. 2007;448(7151):338-341. [9] HO CC, DING SJ. Structure, properties and applications of mussel-inspired polydopamine. J Biomed Nanotechnol. 2014;10(10):3063-3084. [10] SONG Y, JIANG H, WANG B, et al. Silver-Incorporated Mussel-Inspired Polydopamine Coatings on Mesoporous Silica as an Efficient Nanocatalyst and Antimicrobial Agent. Acs Appl Mater Interfaces. 2018;10(2):1792-1801. [11] QIAN Y, ZHOU X, ZHANG F, et al. Triple PLGA/PCL Scaffold Modification Including Silver Impregnation, Collagen Coating, and Electrospinning Significantly Improve Biocompatibility, Antimicrobial, and Osteogenic Properties for Orofacial Tissue Regeneration. ACS Appl Mater Interfaces. 2019;11(41):37381-37396. [12] PAN H, ZHENG Q, YANG S, et al. Effects of functionalization of PLGA-[Asp-PEG]n copolymer surfaces with Arg-Gly-Asp peptides, hydroxyapatite nanoparticles, and BMP-2-derived peptides on cell behavior in vitro. J Biomed Mater Res A. 2014;102(12):4526-4535. [13] 谭帼馨,欧阳孔友,周蕾,等.聚多巴胺螯合钙离子对钛表面的修饰及修饰后的细胞相容性[J].无机材料学报,2015(10):1075-1080. [14] GAO X, SONG J, JI P, et al. Polydopamine-Templated Hydroxyapatite Reinforced Polycaprolactone Composite Nanofibers with Enhanced Cytocompatibility and Osteogenesis for Bone Tissue Engineering. ACS Appl Mater Interfaces. 2016;8(5): 3499-3515. [15] LIN B, ZHONG M, ZHENG C, et al. Preparation and characterization of dopamine-induced biomimetic hydroxyapatite coatings on the AZ31 magnesium alloy. Surf Coat Technol. 2015;281:82-88. [16] RYU J, KU SH, LEE H, et al. Mussel‐Inspired Polydopamine Coating as a Universal Route to Hydroxyapatite Crystallization. Adv Funct Mater. 2010; 20(13):2132-2139. [17] NEGRILA CC, PREDOI MV, ICONARU SL, et al. Development of Zinc-Doped Hydroxyapatite by Sol-Gel Method for Medical Applications. Molecules. 2018; 23(11):2986. [18] STOICA TF, MOROSANU C, SLAV A, et al. Hydroxyapatite films obtained by sol–gel and sputtering. Thin Solid Films. 2008;516(22):8112-8116. [19] NAKAO S, TSUKAMOTO T, UEYAMA T, et al. STAT3 for Cardiac Regenerative Medicine: Involvement in Stem Cell Biology, Pathophysiology, and Bioengineering. Int J Mol Sci. 2020;21(6):1937. [20] RAHAMAN MN, MAO JJ. Stem cell-based composite tissue constructs for regenerative medicine. Biotechnol Bioeng. 2010;91(3):261-284. [21] WILLIAMS JM, ADEWUNMI A, SCHEK RM, et al. Bone tissue engineering using polycaprolactone scaffolds fabricated via selective laser sintering. Biomaterials. 2005;26(23):4817-4827. [22] YANG X, YANG F, WALBOOMERS XF, et al. The performance of dental pulp stem cells on nanofibrous PCL/gelatin/nHA scaffolds. J Biomed Mater Res A. 2010; 93(1):247-257. [23] LOUVRIER A, EUVRARD E, NICOD L, et al. Odontoblastic differentiation of dental pulp stem cells from healthy and carious teeth on an original PCL-based 3D scaffold. Int Endod J. 2018;51 Suppl 4:e252-e263. [24] 张一.聚己内酯的表面改性及其对细胞行为的影响[D].上海:华东理工大学, 2012. [25] Yoon H, Kim GH, Koh YH. A micro-scale surface-structured PCL scaffold fabricated by a 3D plotter and a chemical blowing agent. J Biomater Sci Polym Ed. 2010;21(2):159-70. [26] 李俊达,陈美霖,韦晓英,等.覆盖富血小板血浆3D打印聚己内酯支架对牙髓细胞体外生物学行为的影响[J].中华口腔医学研究杂志(电子版),2017, 11(3):149-156. [27] 刘宗光,屈树新,翁杰.聚多巴胺在生物材料表面改性中的应用[J].化学进展, 2015,27(2):212-219. [28] 张嘉敏,汪涛,汤春波.颅骨修复钛网表面PDA/CPP涂层的制备及性能研究[J].材料科学,2017,7(1):134-142. [29] ZHAO H, WAITE JH. Linking adhesive and structural proteins in the attachment plaque of Mytilus californianus. J Biol Chem. 2006;281(36): 26150-26158. [30] 叶玲.再生性牙髓治疗方法的前景[J].口腔医学,2016,36(11):961-967. [31] 侯丽,乔春霞,赵增琳.解读ISO 10993-4:2017《医疗器械生物学评价第4部分:与血液相互作用试验选择》[J].中国医疗设备,2018,33(11):1-6. [32] NAKASHIMA M, IOHARA K, MURAKAMI M, et al. Pulp regeneration by transplantation of dental pulp stem cells in pulpitis: a pilot clinical study. Stem Cell Res Ther. 2017;8(1):61. [33] GRONTHOS S, MANKANI M, BRAHIM J, et al. Postnatal human dental pulp stem cells (DPSCs) in vitro and invivo. Proc Natl Acad Sci U S A. 2000;97(25): 13625-13630. [34] MIURA M, GRONTHOS S, ZHAO M, et al. SHED: stem cells from human exfoliated deciduous teeth . Proc Natl Acad Sci U S A. 2003;100(10):5807-5812. [35] D’ANTÒ V, RAUCCI MG, GUARINO V, et al. Behaviour of human mesenchymal stem cells on chemically synthesized HA-PCL scaffolds for hard tissue regeneration. J Tissue Eng Regen Med. 2016;10(2):E147-154. [36] GUO T, CAO G, LI Y, et al. Signals in Stem Cell Differentiation on Fluorapatite-Modified Scaffolds. J Dent Res. 2018;97(12):1331-1338. [37] ALIPOUR M, AGHAZADEH M, AKBARZADEH A, et al. Towards osteogenic differentiation of human dental pulp stem cells on PCL-PEG-PCL/zeolite nanofibrous scaffolds. Artif Cells Nanomed Biotechnol. 2019;47(1):3431-3437. [38] 梁倩.PCOS/bFGF调控人牙槽骨成骨细胞增殖矿化的作用研究[D].广州:暨南大学,2018. |
| [1] | Dai Xianglin, Zhang Wenfeng, Yao Xijun, Shang Jiaqi, Huang Qiujin, Ren Yifan, Deng Jiupeng. Barium titanate/polylactic acid piezoelectric composite film affects adhesion, proliferation, and osteogenic differentiation of MC3T3-E1 cells [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(3): 367-373. |
| [2] | Liu Huan, Li Han, Ma Yunhao, Zhong Weijian, Ma Guowu. Osteogenic capacity of partially demineralized dentin particles in the maxillary sinus lift [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(3): 354-359. |
| [3] | Wu Lihao, Shao Anliang, Xu Lin, Ren Kang, Wang Hongjian, Chen Liang, Xu Ling. Evaluation of immunotoxicity of the absorbable macroporous polysaccharides composite hemostatic material [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(3): 329-334. |
| [4] | Bi Jintong, Hu Xin, Liu Jinshu. Wear properties of dental ceramics [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(3): 406-412. |
| [5] | Jiang Haifang, Liu Rong, Hu Peng, Chen Wei, Wei Zairong, Yang Chenglan, Nie Kaiyu. Application of 3D printing technology in the precise and personalized treatment of cleft lip and palate [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(3): 413-419. |
| [6] | Zhang Jinglin, Leng Min, Zhu Boheng, Wang Hong. Mechanism and application of stem cell-derived exosomes in promoting diabetic wound healing [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1113-1118. |
| [7] | Shen Song, Xu Bin. Diffuse distribution of bone cement in percutaneous vertebroplasty reduces the incidence of refracture of adjacent vertebral bodies [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 499-503. |
| [8] | Hou Wanxing, Li Hongwei, Zheng Xin, Zhu Xianren. Correlation between preoperative magnetic resonance imaging findings and bone cement leakage after percutaneous vertebral augmentation [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 504-509. |
| [9] | Yang Sidi, Wang Qian, Xu Nuo, Wang Ronghan, Jin Chuanqi, Lu Ying, Dong Ming. Biodentine enhances the proliferation and differentiation of osteoblasts through upregulating bone morphogenetic protein-2 [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 516-520. |
| [10] | Le Guoping, Zhang Ming, Xi Licheng, Luo Hanwen. Preparation and in vitro evaluation of vancomycin hydrochloride@polylactic acid-glycolic acid copolymer-chitosan-hyaluronic acid composite sustained-release microspheres [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 528-534. |
| [11] | Liu Tongbin, Lin Peng, Zhang Xiaoming, Dong Xiling, Cao Fei, Wang Le, Guo Xinxing. Optimization of preparation method of atorvastatin calcium sustained-release microspheres [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 535-539. |
| [12] | Qiu Peng, Fu Qilin, Liu Min, Lan Yuyan, Wang Pin. Comparison of oral micro-adhesion on polyetheretherketone, zirconium dioxide, and pure titanium abutment [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 540-545. |
| [13] | He Guanyu, Xu Baoshan, Du Lilong, Zhang Tongxing, Huo Zhenxin, Shen Li. Biomimetic orientated microchannel annulus fibrosus scaffold constructed by silk fibroin [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 560-566. |
| [14] | Wang Can, Gu Weiping, Jiang Yubin, Zhu Lin, Chen Gang. Finite element analysis of the influence of different implant designs on the stress of mandibular edentulous jaw [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 573-578. |
| [15] | Yang Feng, Zhao Qian, Zhang Shixuan, Zhao Tienan, Feng Bo. Effectiveness and safety of rapamycin combined with CD133 antibody stent in preventing vascular restenosis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 579-584. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||