Chinese Journal of Tissue Engineering Research ›› 2022, Vol. 26 ›› Issue (1): 45-51.doi: 10.12307/2022.008
Previous Articles Next Articles
Chondrogenic differentiation of peripheral blood-derived mesenchymal stem cells induced by transforming growth factor beta 3: a dose-effect relationship
Yang Tengyun1, Li Yanlin1, Liu Dejian1, Wang Guoliang1, Zheng Zhujun2
- 1Department of Sports Medicine, 2Department of Rehabilitation, First Affiliated Hospital of Kunming Medical University, Kunming 650032, Yunnan Province, China
-
Received:2020-12-02Revised:2020-12-05Accepted:2021-01-29Online:2022-01-08Published:2021-10-23 -
Contact:Li Yanlin, MD, Professor, Department of Sports Medicine, First Affiliated Hospital of Kunming Medical University, Kunming 650032, Yunnan Province, China -
About author:Yang Tengyun, Master candidate, Department of Sports Medicine, First Affiliated Hospital of Kunming Medical University, Kunming 650032, Yunnan Province, China -
Supported by:the National Natural Science Foundation of China, No. 81960409 (to LYL), No. 81760403 (to LYL); the Expert Workstation Project of Yunnan Province, No. 2018IC102 (to LYL); the Yunnan Province Clinical Center for Bone and Joint Diseases, No. ZX2019-03-04 (to LYL)
CLC Number:
Cite this article
Yang Tengyun, Li Yanlin, Liu Dejian, Wang Guoliang, Zheng Zhujun. Chondrogenic differentiation of peripheral blood-derived mesenchymal stem cells induced by transforming growth factor beta 3: a dose-effect relationship[J]. Chinese Journal of Tissue Engineering Research, 2022, 26(1): 45-51.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
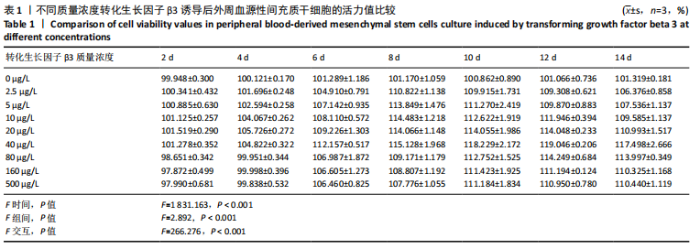
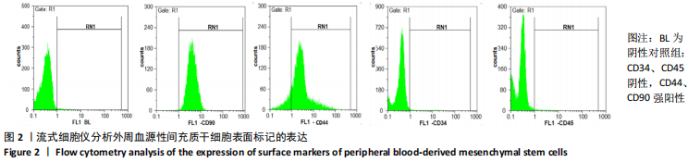
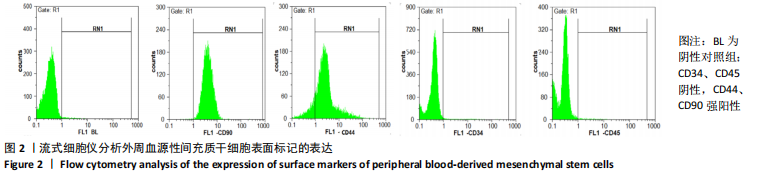
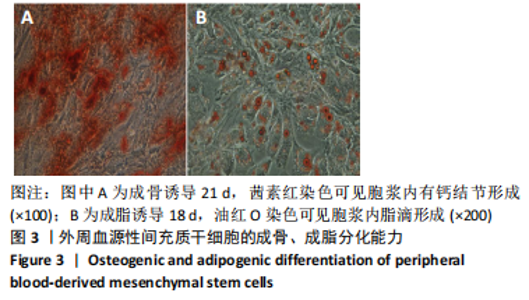
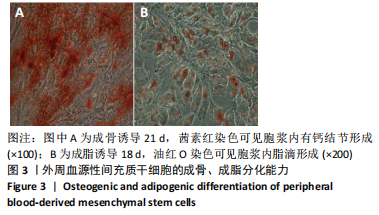
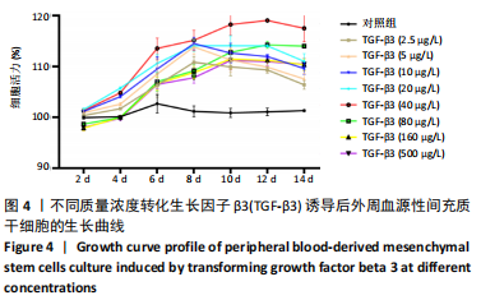
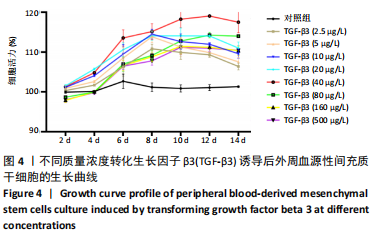
2.2 转化生长因子β3促进PBMSCs成软骨诱导分化的作用 2.2.1 转化生长因子β3诱导后PBMSCs增殖曲线 对照组未加入诱导液培养,其他实验组加入含不同质量浓度转化生长因子β3的诱导液之后,采用统计学两因素重复测量方差分析方法比较各组各时间段细胞活力值,结果如下:球形检验结果显示P < 0.05,证明实验数据不对称,应以“多变量检验结果”为此次研究最终结果。多变量检验结果显示:增殖时间效应显著(F=1 831.163,P < 0.001),表明各实验组随培养天数的递增而出现明显变化;增殖时间*浓度分组交互效应显著(F=2.892,P < 0.001),表明不同质量浓度转化生长因子β3组在不同时间的变化趋势也有显著差异;主体间检验结果显示浓度分组效应显著(F=266.276,P < 0.001),表明细胞增殖随转化生长因子β3质量浓度的变化而出现明显变化。 各质量浓度转化生长因子β3组细胞生长曲线大致相似,与对照组各时间点相比差异均有显著性意义,随着培养时间延长,各组细胞生长曲线基本相似,大致呈“S”形,前期增长不明显,为细胞生长潜伏期,中期进入细胞对数生长期,差异逐渐增大,中后期达生长高峰,而后基本维持水平或下降,表明细胞进入凋亡期。 以上结果显示与对照组(不加入转化生长因子β3)相比,各质量浓度转化生长因子β3均能促进细胞生长,并且随质量浓度增加,细胞生长周期延长,较晚达到平台期;在2.5-40 μg/L时,随质量浓度增加PBMSCs生长增殖能力增强;在80-500 μg/L时,PBMSCs生长增殖能力又出现减弱,见表1,图4。"
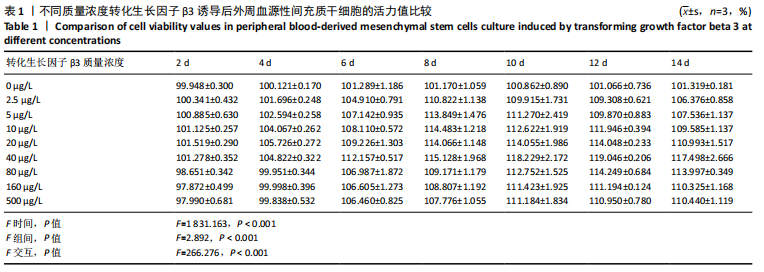
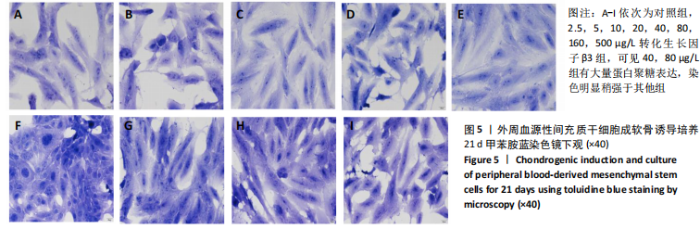
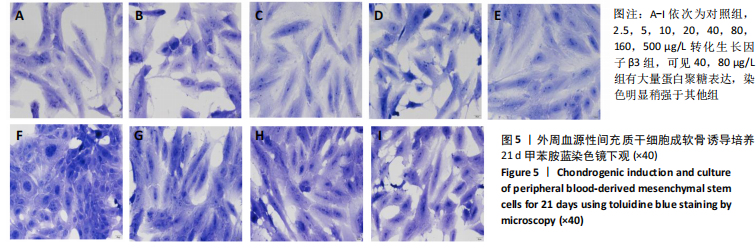
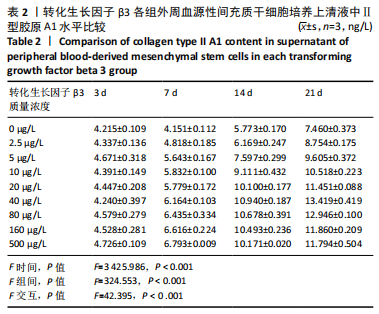
2.2.2 酶联免疫吸附法检测Ⅱ型胶原A1水平 对照组未加入诱导液培养,其他实验组加入含不同质量浓度转化生长因子β3的诱导液之后,采用统计学两因素重复测量方差分析方法比较各组各时间段细胞培养上清中Ⅱ型胶原A1水平,结果如下:球形检验结果显示P > 0.05,证明此次研究数据对称,以“主体内效应检验结果”为此次研究最终结果。主体内效应检验结果显示:时间效应显著(F=3 425.986,P < 0.001),表明各质量浓度转化生长因子β3组Ⅱ型胶原A1水平随培养天数的递增而出现明显变化;时间*浓度分组交互效应显著(F=42.395,P < 0.001),表明不同质量浓度转化生长因子β3组在不同培养天数Ⅱ型胶原A1水平的变化趋势也有显著差异;主体间效应检验结果显示浓度分组效应显著(F=324.553,P < 0.001),表明Ⅱ型胶原A1水平随转化生长因子β3质量浓度的变化而出现明显变化。 各组PBMSCs分化曲线基本相似,第一时点细胞处于增殖期,未见明显分化,与对照组之间差异无显著性意义(P > 0.05),从第二时间点开始,各组间差异逐渐显著,各组Ⅱ型胶原A1水平随培养时间的增长逐步递增。随着转化生长因子β3质量浓度增加,在7-14 d时Ⅱ型胶原A1水平增长较明显,在第14天后增长减缓,第21天时达到分化高峰,此时40,80 μg/L转化生长因子β3组Ⅱ型胶原A1水平差异无显著性意义(P > 0.05),但明显优于其他质量浓度组。 以上结果表明,高于2.5 μg/L时转化生长因子β3均能促进PBMSCs分化,并且在5-40 μg/L范围有一定时间浓度依赖,随着转化生长因子β3质量浓度增高和培养时间延长,分化产生Ⅱ型胶原A1更多;80 μg/L转化生长因子β3促进 PBMSCs分化能力与40 μg/L相当,高于80 μg/L时促进PBMSCs 分化能力又减弱。各组相比较之下转化生长因子β3质量浓度在40,80 μg/L时更容易促进PBMSCs成软骨分化,见表2。"
| [1] RAGNI E, PERUCCA ORFEI C, DE LUCA P, et al. Secreted Factors and EV-miRNAs Orchestrate the Healing Capacity of Adipose Mesenchymal Stem Cells for the Treatment of Knee Osteoarthritis. Int J Mol Sci. 2020; 21(5):1582. [2] ARSHI A, PETRIGLIANO FA, WILLIAMS RJ, et al. Stem Cell Treatment for Knee Articular Cartilage Defects and Osteoarthritis. Curr Rev Musculoskelet Med. 2020;13(1):20-27. [3] JAFRI MA, KALAMEGAM G, ABBAS M, et al. Deciphering the Association of Cytokines, Chemokines, and Growth Factors in Chondrogenic Differentiation of Human Bone Marrow Mesenchymal Stem Cells Using an ex vivo Osteochondral Culture System. Front Cell Dev Biol. 2020;7:380. [4] 杨春水,杨志刚,李建英,等.成人外周血间充质干细胞诱导为神经元样细胞的体外研究[J].交通医学,2010,24(6):602-604. [5] LOTFY A, EL-SHERBINY YM, CUTHBERT R, et al. Comparative study of biological characteristics of mesenchymal stem cells isolated from mouse bone marrow and peripheral blood. Biomed Rep. 2019; 11(4):165-170. [6] FU WL, ZHOU CY, YU JK. A new source of mesenchymal stem cells for articular cartilage repair: MSCs derived from mobilized peripheral blood share similar biological characteristics in vitro and chondrogenesis in vivo as MSCs from bone marrow in a rabbit model. Am J Sports Med. 2014;42(3):592-601. [7] WANG SJ, YIN MH, JIANG D, et al. The Chondrogenic Potential of Progenitor Cells Derived from Peripheral Blood: A Systematic Review. Stem Cells Dev. 2016;25(16):1195-1207. [8] BRANLY T, BERTONI L, CONTENTIN R, et al. Characterization and use of Equine Bone Marrow Mesenchymal Stem Cells in Equine Cartilage Engineering. Study of their Hyaline Cartilage Forming Potential when Cultured under Hypoxia within a Biomaterial in the Presence of BMP-2 and TGF-ß1. Stem Cell Rev Rep. 2017;13(5):611-630. [9] FAN L, CHEN J, TAO Y, et al. Enhancement of the chondrogenic differentiation of mesenchymal stem cells and cartilage repair by ghrelin. J Orthop Res. 2019;37(6):1387-1397. [10] OCCHETTA P, PIGEOT S, RASPONI M, et al. Developmentally inspired programming of adult human mesenchymal stromal cells toward stable chondrogenesis. Proc Natl Acad Sci U S A. 2018;115(18):4625-4630. [11] BIANCHI VJ, WEBER JF, WALDMAN SD, et al. Formation of Hyaline Cartilage Tissue by Passaged Human Osteoarthritic Chondrocytes. Tissue Eng Part A. 2017;23(3-4):156-165. [12] WANG T, NIMKINGRATANA P, SMITH CA, et al. Enhanced chondrogenesis from human embryonic stem cells. Stem Cell Res. 2019;39:101497. [13] CHIJIMATSU R, KOBAYASHI M, EBINA K, et al. Impact of dexamethasone concentration on cartilage tissue formation from human synovial derived stem cells in vitro. Cytotechnology. 2018;70(2):819-829. [14] MAHBOUDI H, SOLEIMANI M, ENDERAMI SE, et al. Enhanced chondrogenesis differentiation of human induced pluripotent stem cells by MicroRNA-140 and transforming growth factor beta 3 (TGFβ3). Biologicals. 2018;52:30-36. [15] ZHAO D, LI Y, ZHOU X, et al. Peripheral Blood Mesenchymal Stem Cells Combined with Modified Demineralized Bone Matrix Promote Pig Cartilage Defect Repair. Cells Tissues Organs. 2018;206(1-2):26-34. [16] LI J, DONG S. The Signaling Pathways Involved in Chondrocyte Differentiation and Hypertrophic Differentiation. Stem Cells Int. 2016; 2016:2470351. [17] KELLER B, YANG T, CHEN Y, et al. Interaction of TGFbeta and BMP signaling pathways during chondrogenesis. PLoS One. 2011;6(1): e16421. [18] 赵道洪. BMP-2、TGF-β3协同调控滇南小耳猪PBMSCs成软骨分化研究[D].昆明:昆明医科大学,2018. [19] LIU Y, MA Y, ZHANG J, et al. Exosomes: A Novel Therapeutic Agent for Cartilage and Bone Tissue Regeneration. Dose Response. 2019; 17(4):1559325819892702. [20] IIJIMA H, ISHO T, KUROKI H, et al. Effectiveness of mesenchymal stem cells for treating patients with knee osteoarthritis: a meta-analysis toward the establishment of effective regenerative rehabilitation. NPJ Regen Med. 2018;3:15. [21] GARZA JR, CAMPBELL RE, TJOUMAKARIS FP, et al. Clinical Efficacy of Intra-articular Mesenchymal Stromal Cells for the Treatment of Knee Osteoarthritis: A Double-Blinded Prospective Randomized Controlled Clinical Trial. Am J Sports Med. 2020;48(3):588-598. [22] BASTOS R, MATHIAS M, ANDRADE R, et al. Intra-articular injections of expanded mesenchymal stem cells with and without addition of platelet-rich plasma are safe and effective for knee osteoarthritis. Knee Surg Sports Traumatol Arthrosc. 2018;26(11):3342-3350. [23] NICKEL J, MUELLER TD. Specification of BMP Signaling. Cells. 2019; 8(12):1579. [24] THIELEN NGM, VAN DER KRAAN PM, VAN CAAM APM. TGFbeta/BMP Signaling Pathway in Cartilage Homeostasis. Cells. 2019;8(9):969. [25] RIM YA, NAM Y, PARK N, et al. Chondrogenic Differentiation from Induced Pluripotent Stem Cells Using Non-Viral Minicircle Vectors. Cells. 2020;9(3):582. [26] 郭宇鹏,唐康来,张吉强,等.不同浓度TGF-β3对体外肌腱干细胞分化的影响研究[J].中国修复重建外科杂志,2015,29(5):620-625. [27] RUHL T, BEIER JP. Quantification of chondrogenic differentiation in monolayer cultures of mesenchymal stromal cells. Anal Biochem. 2019;582:113356. [28] KRÜGER JP, MACHENS I, LAHNER M, et al. Initial boost release of transforming growth factor-β3 and chondrogenesis by freeze-dried bioactive polymer scaffolds. Ann Biomed Eng. 2014;42(12):2562-2576. |
| [1] | Wang Jing, Xiong Shan, Cao Jin, Feng Linwei, Wang Xin. Role and mechanism of interleukin-3 in bone metabolism [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1316-1322. |
| [2] | Xiao Hao, Liu Jing, Zhou Jun. Research progress of pulsed electromagnetic field in the treatment of postmenopausal osteoporosis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1323-1329. |
| [3] | An Weizheng, He Xiao, Ren Shuai, Liu Jianyu. Potential of muscle-derived stem cells in peripheral nerve regeneration [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1183-1190. |
| [4] | Fan Yiming, Liu Fangyu, Zhang Hongyu, Li Shuai, Wang Yansong. Serial questions about endogenous neural stem cell response in the ependymal zone after spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1191-1197. |
| [5] | Tian Chuan, Zhu Xiangqing, Yang Zailing, Yan Donghai, Li Ye, Wang Yanying, Yang Yukun, He Jie, Lü Guanke, Cai Xuemin, Shu Liping, He Zhixu, Pan Xinghua. Bone marrow mesenchymal stem cells regulate ovarian aging in macaques [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1033-1039. |
| [6] | Hou Jingying, Guo Tianzhu, Yu Menglei, Long Huibao, Wu Hao. Hypoxia preconditioning targets and downregulates miR-195 and promotes bone marrow mesenchymal stem cell survival and pro-angiogenic potential by activating MALAT1 [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1053-1059. |
| [7] | Zhou Ying, Zhang Huan, Liao Song, Hu Fanqi, Yi Jing, Liu Yubin, Jin Jide. Immunomodulatory effects of deferoxamine and interferon gamma on human dental pulp stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1060-1067. |
| [8] | Liang Xuezhen, Yang Xi, Li Jiacheng, Luo Di, Xu Bo, Li Gang. Bushen Huoxue capsule regulates osteogenic and adipogenic differentiation of rat bone marrow mesenchymal stem cells via Hedgehog signaling pathway [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1068-1074. |
| [9] | Wang Jifang, Bao Zhen, Qiao Yahong. miR-206 regulates EVI1 gene expression and cell biological behavior in stem cells of small cell lung cancer [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1075-1079. |
| [10] | Liu Feng, Peng Yuhuan, Luo Liangping, Wu Benqing. Plant-derived basic fibroblast growth factor maintains the growth and differentiation of human embryonic stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1080-1085. |
| [11] | Wen Dandan, Li Qiang, Shen Caiqi, Ji Zhe, Jin Peisheng. Nocardia rubra cell wall skeleton for extemal use improves the viability of adipogenic mesenchymal stem cells and promotes diabetes wound repair [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1086-1092. |
| [12] | Zhu Bingbing, Deng Jianghua, Chen Jingjing, Mu Xiaoling. Interleukin-8 receptor enhances the migration and adhesion of umbilical cord mesenchymal stem cells to injured endothelium [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1093-1098. |
| [13] | Luo Xiaoling, Zhang Li, Yang Maohua, Xu Jie, Xu Xiaomei. Effect of naringenin on osteogenic differentiation of human periodontal ligament stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1099-1104. |
| [14] | Wang Xinmin, Liu Fei, Xu Jie, Bai Yuxi, Lü Jian. Core decompression combined with dental pulp stem cells in the treatment of steroid-associated femoral head necrosis in rabbits [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1122-1127. |
| [15] | Fang Xiaolei, Leng Jun, Zhang Chen, Liu Huimin, Guo Wen. Systematic evaluation of different therapeutic effects of mesenchymal stem cell transplantation in the treatment of ischemic stroke [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1133-1140. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||