Chinese Journal of Tissue Engineering Research ›› 2018, Vol. 22 ›› Issue (9): 1450-1456.doi: 10.3969/j.issn.2095-4344.0478
Previous Articles Next Articles
Effects of sustained hypoxia on proliferation of mouse embryonic fibroblasts and preparation of feeder layers
Wei Han-qing1, Pei Yi-jin1, Wang Dan-dan1, Jiang Yang2, Wang Chun1, Li Hong-mei3
- 1Department of Physiology, Institute of Stem Cell and Regenerative Medicine, 2Laboratory of Physiological Science, 3Department of Pathology, Guangdong Medical University, Dongguan 523808, Guangdong Province, China
-
Revised:2018-01-26Online:2018-03-28Published:2018-04-03 -
Contact:Wei Han-qing, Master candidate, Department of Physiology, Institute of Stem Cell and Regenerative Medicine, College of Basic Medicine, Guangdong Medical University, Dongguan 523808, Guangdong Province, China -
About author:Wei Han-qing, Master candidate, Department of Physiology, Institute of Stem Cell and Regenerative Medicine, College of Basic Medicine, Guangdong Medical University, Dongguan 523808, Guangdong Province, China -
Supported by:the National Natural Science Foundation of China, No. 81173136; the Scientific Research Project of Guangdong Province, No. 2013B022000003; the Natural Science Foundation of Guangdong Province, No. 2015A030313524; the International Scientific Cooperation Project of Dongguan in Guangdong Province, No. 2015508102004; the Scientific Research Foundation of Guangdong Medical University, No. M2015019
CLC Number:
Cite this article
Wei Han-qing, Pei Yi-jin, Wang Dan-dan, Jiang Yang, Wang Chun, Li Hong-mei. Effects of sustained hypoxia on proliferation of mouse embryonic fibroblasts and preparation of feeder layers[J]. Chinese Journal of Tissue Engineering Research, 2018, 22(9): 1450-1456.
share this article
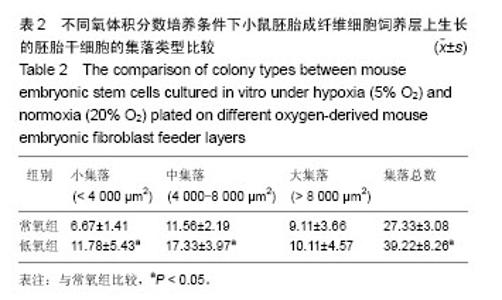
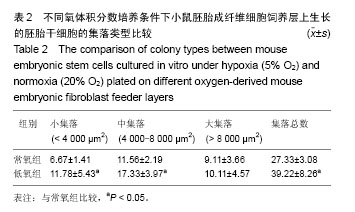
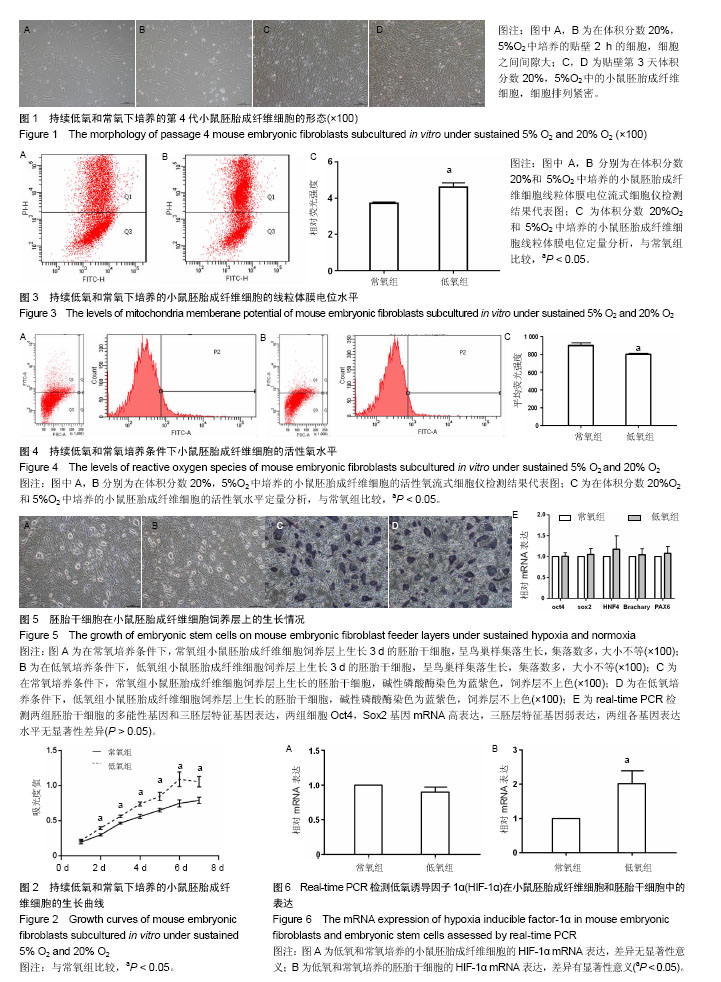
2.1 不同氧体积分数持续培养的小鼠胚胎成纤维细胞形态特征及增殖状况 在体积分数5%O2和体积分数20%O2中分别持续培养小鼠胚胎成纤维细胞,在传代后数小时开始贴壁生长,两组细胞形态一致,均呈典型成纤维细胞形态,梭状或多角状或不规则状,细胞质均匀,细胞核呈卵圆形,可见多个核仁(图1A,B)。细胞贴壁后,细胞增殖加快,第2天进入对数生长期,细胞逐渐排列紧密,细胞与细胞之间形成连接(图1C,D)。低氧组细胞数在第6天达到高峰,进入平台期,而常氧组细胞数在第7天达到高峰。低氧组细胞群体倍增时间为34.87 h,常氧组群体倍增时间38.1 h。 采用MTT法检测两组小鼠胚胎成纤维细胞增殖情况,贴壁第24小时,低氧组细胞数量比常氧组稍多,但差异无显著性意义(P > 0.05)。从第2天至第7天,同一天细胞数量低氧组比常氧组显著增多,差异有显著性意义(P < 0.05,图2),说明体积分数5%O2持续培养的小鼠胚胎成纤维细胞生长增殖更快。 2.2 不同氧体积分数持续培养对小鼠胚胎成纤维细胞线粒体膜电位的影响 经JC-1探针染色后流式细胞仪检测,以红绿荧光平均强度比值代表线粒体膜电位,体积分数5%O2,20%O2下持续培养至对数生长期的小鼠胚胎成纤维细胞线粒体膜电位分别为4.60±0.29,3.72±0.09,结果显示低氧组小鼠胚胎成纤维细胞线粒体膜电位明显上升(P < 0.05,图3)。 2.3 不同氧体积分数持续培养对小鼠胚胎成纤维细胞活性氧水平的影响 经过DCFH-DA染色后流式细胞仪检测,以绿色平均荧光强度代表活性氧水平,体积分数5%O2,20%O2下持续培养至对数生长期的小鼠胚胎成纤维细胞的活性氧水平分别为801.00±11.31,899.50±30.41,结果显示低氧组活性氧显著下降(P < 0.05,图4)。 2.4 不同氧体积分数持续培养的小鼠胚胎成纤维细胞制备的饲养层对胚胎干细胞保持不分化状态的自我更新维持作用 两组胚胎干细胞接种后,呈集落样生长。在第2天即可观察到有小集落出现,集落呈鸟巢状隆起,集落里细胞排列紧密,细胞体积小,核大,核质比高。第3天有大量集落生成,集落增大,各集落大小不一(图5A,B)。第4天即可扩增、传代。第3天两组胚胎干细胞的集落根据大小进行分类计数统计分析,结果显示低氧组胚胎干细胞的集落总数多,其中小集落数及中等大小集落数均比常氧组显著增加(P < 0.05,表2),而大集落数差异无显著性意义(P > 0.05,表2)。 对两组胚胎干细胞的集落用碱性磷酸酶试剂盒染色,均为蓝紫色,碱性磷酸酶表达阳性(图5C,D)。进一步检测两组胚胎干细胞的多能性基因Oct4,Sox2和三胚层标志基因HNF4(内胚层),Brachary(中胚层),PAX6(外胚层)的mRNA表达,结果可见两组Oct4,Sox2的mRNA都高表达,三胚层标志基因弱表达,两组细胞Oct4,Sox2表达差异无显著性意义(P > 0.05,图5E)。结果显示这两组胚胎干细胞都能在各自的培养系统里维持多能性状态。 2.5 小鼠胚胎成纤维细胞和胚胎干细胞中HIF-1α基因表达 体积分数5%O2,20%O2持续培养的小鼠胚胎成纤维细胞均有HIF-1α mRNA表达,两组HIF-1α mRNA表达差异无显著性意义(P > 0.05,图6A)。体积分数5%O2,20%O2持续培养的胚胎干细胞均有HIF-1α mRNA表达,两组比较差异有显著性意义(P < 0.05,图6B)。"
| [1] Evans MJ, Kaufman MH. Establishment in culture of pluripotential cells from mouse embryos. Nature. 1981;292(5819):154-156.[2] Martin GR. Isolation of a pluripotent cell line from early mouse embryos cultured in medium conditioned by teratocarcinoma stem cells. Proc Natl Acad Sci U S A. 1981;78(12):7634-7638.[3] Thomson JA, Itskovitz-Eldor J, Shapiro SS, et al. Embryonic stem cell lines derived from human blastocysts. Science. 1998;282(5391):1145-1147.[4] Yue W, Li Y, Zhang T, et al. ESC-Derived Basal Forebrain Cholinergic Neurons Ameliorate the Cognitive Symptoms Associated with Alzheimer's Disease in Mouse Models. Stem Cell Reports. 2015;5(5):776-790.[5] Du V, Luciani N, Richard S, et al. A 3D magnetic tissue stretcher for remote mechanical control of embryonic stem cell differentiation. Nat Commun. 2017;8(1):400.[6] 贾源君,裴轶劲.干细胞在发育毒性体外模型建立中的应用研究进展[J].中国医药导报,2015,12(2): 152-155.[7] Colaianna M, Ilmjärv S, Peterson H, et al. Fingerprinting of neurotoxic compounds using a mouse embryonic stem cell dual luminescence reporter assay. Arch Toxicol. 2017;91(1):365-391.[8] Jain K, Verma PJ, Liu J. Isolation and handling of mouse embryonic fibroblasts. Methods Mol Biol. 2014;1194:247-252. [9] Fleischmann G, Müller T, Blasczyk R, et al. Growth characteristics of the nonhuman primate embryonic stem cell line cjes001 depending on feeder cell treatment. Cloning Stem Cells. 2009;11(2):225-233. [10] Dowell KG, Simons AK, Bai H, et al. Novel insights into embryonic stem cell self-renewal revealed through comparative human and mouse systems biology networks. Stem Cells. 2014;32(5):1161-1172.[11] Takahashi K, Tanabe K, Ohnuki M, et al. Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell. 2007;131(5):861-872.[12] Yu J, Vodyanik MA, Smuga-Otto K, et al. Induced pluripotent stem cell lines derived from human somatic cells. Science. 2007;318(5858):1917-1920.[13] 胡三强,王妍妍,马永宾,等.小鼠胚胎成纤维细胞的分离培养及饲养层制备[J].中国组织工程研究, 2014,18(45): 7306-7311.[14] Oh HR, Kim J, Kim J. Critical roles of Cyclin D1 in mouse embryonic fibroblast cell reprogramming. FEBS J. 2016;283(24):4549-4568.[15] Zhang CL, Liu X, He QJ, et al. miR?342?5p promotes Zmpste24?deficient mouse embryonic fibroblasts proliferation by suppressing GAS2. Mol Med Rep. 2017;16(6):8944-8952. [16] Lim JW, Bodnar A. Proteome analysis of conditioned medium from mouse embryonic fibroblast feeder layers which support the growth of human embryonic stem cells. Proteomics. 2002;2(9):1187-1203.[17] Mamada H, Sato T, Ota M, et al. Cell competition in mouse NIH3T3 embryonic fibroblasts is controlled by the activity of Tead family proteins and Myc. J Cell Sci. 2015;128(4):790-803.[18] Desai N, Ludgin J, Goldberg J, et al. Development of a xeno-free non-contact co-culture system for derivation and maintenance of embryonic stem cells using a novel human endometrial cell line. J Assist Reprod Genet. 2013;30(5):609-615.[19] Trimarchi JR, Liu L, Porterfield DM, et al. A non-invasive method for measuring preimplantation embryo physiology. Zygote. 2000;8(1):15-24.[20] Caroti CM, Ahn H, Salazar HF, et al. A Novel Technique for Accelerated Culture of Murine Mesenchymal Stem Cells that Allows for Sustained Multipotency. Sci Rep. 2017;7(1):13334.[21] Chen R, Liu Y, Su Q, et al. Hypoxia stimulates proliferation of rat neural stem/progenitor cells by regulating mir-21: an in vitro study. Neurosci Lett. 2017;661:71-76.[22] Choi W, Kwon SJ, Jin HJ, et al. Optimization of culture conditions for rapid clinical-scale expansion of human umbilical cord blood-derived mesenchymal stem cells. Clin Transl Med. 2017;6(1):38-50.[23] Prasad SM, Czepiel M, Cetinkaya C, et al. Continuous hypoxic culturing maintains activation of Notch and allows long-term propagation of human embryonic stem cells without spontaneous differentiation. Cell Prolif. 2009;42(1):63-74.[24] Betts DH, Perrault SD, King WA. Low oxygen delays fibroblast senescence despite shorter telomeres. Biogerontology. 2008;9(1):19-31.[25] 王春,魏含清,裴轶劲.低氧培养对小鼠胚胎成纤维细胞生长和线粒体分布及功能的影响[J].重庆医学,2017,46(19):2599-2603.[26] Ezashi T, Das P, Roberts RM. Low O2 tensions and the prevention of differentiation of hES cells. Proc Natl Acad Sci U S A. 2005;102(13): 4783-4788.[27] Prado-Lopez S, Conesa A, Armiñán A, et al. Hypoxia promotes efficient differentiation of human embryonic stem cells to functional endothelium. Stem Cells. 2010;28(3):407-418.[28] Binó L, Ku?era J, Štefková K, et al. The stabilization of hypoxia inducible factor modulates differentiation status and inhibits the proliferation of mouse embryonic stem cells. Chem Biol Interact. 2016;244:204-214.[29] 罗阳,彭芳,杜红姿, 等.不同浓度氧气对人未成熟卵子体外成熟和早期胚胎发育的影响[J].实用医学杂志,2017,33(20):3394-3397.[30] Harvey AJ. The role of oxygen in ruminant preimplantation embryo development and metabolism. Anim Reprod Sci. 2007;98(1-2):113-128.[31] Karja NW, Wongsrikeao P, Murakami M, et al. Effects of oxygen tension on the development and quality of porcine in vitro fertilized embryos. Theriogenology. 2004;62(9):1585-1595.[32] Peng H, Shi W, Zhang W, et al. Better Quality and More Usable Embryos Obtained on Day 3 Cultured in 5% Than 20% Oxygen: A Controlled and Randomized Study Using the Sibling Oocytes. Reprod Sci. 2016;23(3):372-378.[33] Li B, Li C, Zhu M, et al. Hypoxia-Induced Mesenchymal Stromal Cells Exhibit an Enhanced Therapeutic Effect on Radiation-Induced Lung Injury in Mice due to an Increased Proliferation Potential and Enhanced Antioxidant Ability. Cell Physiol Biochem. 2017;44(4):1295-1310.[34] Weli SC, Fink T, Cetinkaya C, et al. Notch and hedgehog signaling cooperate to maintain self-renewal of human embryonic stem cells exposed to low oxygen concentration. Int J Stem Cells. 2010;3(2):129-137.[35] Christensen DR, Calder PC, Houghton FD. GLUT3 and PKM2 regulate OCT4 expression and support the hypoxic culture of human embryonic stem cells. Sci Rep. 2015;5:17500.[36] Närvä E, Pursiheimo JP, Laiho A, et al. Continuous hypoxic culturing of human embryonic stem cells enhances SSEA-3 and MYC levels. PLoS One. 2013;8(11):e78847.[37] Liu S, Song N, He J, et al. Effect of Hypoxia on the Differentiation and the Self-Renewal of Metanephrogenic Mesenchymal Stem Cells. Stem Cells Int. 2017;2017:7168687.[38] Kawasaki T, Sumita Y, Egashira K, et al. Transient Exposure to Hypoxic and Anoxic Oxygen Concentrations Promotes Either Osteogenic or Ligamentogenic Characteristics of PDL Cells. Biores Open Access. 2015;4(1):175-187.[39] Forristal CE, Christensen DR, Chinnery FE, et al. Environmental oxygen tension regulates the energy metabolism and self-renewal of human embryonic stem cells. PLoS One. 2013;8(5):e62507.[40] 马文军,储加强,陶立,等.持续低氧预处理促进兔尿源性干细胞增殖和减少凋亡[J].基础医学与临床,2017, 37(1):13-18.[41] 赵惠卿,朱玲玲,赵彤,等.低氧对胚胎干细胞增殖的影响[J].中国应用生理学杂志,2004,20(3): 3209-3213.[42] Fischer B, Künzel W, Kleinstein J, et al. Oxygen tension in follicular fluid falls with follicle maturation. Eur J Obstet Gynecol Reprod Biol. 1992;43(1):39-43.[43] Weli SC, Fink T, Cetinkaya C, et al. Notch and hedgehog signaling cooperate to maintain self-renewal of human embryonic stem cells exposed to low oxygen concentration. Int J Stem Cells. 2010;3(2):129-137.[44] Chiu SC, Lin YJ, Huang SY, et al. The Role of Intermittent Hypoxia on the Proliferative Inhibition of Rat Cerebellar Astrocytes. PLoS One. 2015;10(7):e0132263.[45] Bellio MA, Rodrigues CO, Landin AM, et al. Physiological and hypoxic oxygen concentration differentially regulates human c-Kit+ cardiac stem cell proliferation and migration. Am J Physiol Heart Circ Physiol. 2016;311(6):H1509-H1519.[46] Lages YM, Nascimento JM, Lemos GA, et al. Low oxygen alters mitochondrial function and response to oxidative stress in human neural progenitor cells. PeerJ. 2015;3:e1486.[47] Ying QL, Wray J, Nichols J, et al. The ground state of embryonic stem cell self-renewal. Nature. 2008;453(7194):519-523.[48] Ateghang B, Wartenberg M, Gassmann M, et al. Regulation of cardiotrophin-1 expression in mouse embryonic stem cells by HIF-1alpha and intracellular reactive oxygen species. J Cell Sci. 2006; 119(Pt 6):1043-1052.[49] Garreta E, Melo E, Navajas D, et al. Low oxygen tension enhances the generation of lung progenitor cells from mouse embryonic and induced pluripotent stem cells. Physiol Rep. 2014;2(7): e12075.[50] Lee SW, Jeong HK, Lee JY, et al. Hypoxic priming of mESCs accelerates vascular-lineage differentiation through HIF1-mediated inverse regulation of Oct4 and VEGF. EMBO Mol Med. 2012;4(9):924-938.[51] Ramírez MÁ, Pericuesta E, Yáñez-Mó M, et al. Effect of long-term culture of mouse embryonic stem cells under low oxygen concentration as well as on glycosaminoglycan hyaluronan on cell proliferation and differentiation. Cell Prolif. 2011;44(1):75-85.[52] Szablowska-Gadomska I, Zayat V, Buzanska L. Influence of low oxygen tensions on expression of pluripotency genes in stem cells. Acta Neurobiol Exp (Wars). 2011;71(1):86-93.[53] Forristal CE, Wright KL, Hanley NA, et al. Hypoxia inducible factors regulate pluripotency and proliferation in human embryonic stem cells cultured at reduced oxygen tensions. Reproduction. 2010;139(1): 85-97.[54] Kudová J, Procházková J, Vaši?ek O, et al. HIF-1alpha Deficiency Attenuates the Cardiomyogenesis of Mouse Embryonic Stem Cells. PLoS One. 2016;11(6):e0158358.[55] 赵丹,唐嘉茵,王艳艳,等.低氧微环境对胎鼠成纤维细胞HIF-1α和PTEN表达及细胞周期的影响[J].解剖科学进展,2012,18(4):305-309. |
| [1] | Li Xuan, Sun Yimin, Li Longbiao, Wang Zhenming, Yang Jing, Wang Chenglin, Ye Ling. Manufacturing of nano-modified polycaprolactone microspheres and its biological effects in dental pulp cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(10): 1602-1608. |
| [2] | Liu Xiaogang, Li Tian, Zhang Duo. Effect and mechanism of the effective components of Chinese medicine on promoting the differentiation of bone marrow mesenchymal stem cells into chondrocytes [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(1): 121-126. |
| [3] | Lin Miaoyuan, Li Yuwan, Liu Yi, Chen Bei, Zhang Li. Research hotspots and application value of tissue-engineered skin [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(1): 159-166. |
| [4] | Xie Xingqi, Hu Wei, Tu Guanjun. Bone marrow mesenchymal stem cells-derived exosomes combined with chondroitinase ABC for treating spinal cord injury in rats [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(1): 20-26. |
| [5] | Yang Tengyun, Li Yanlin, Liu Dejian, Wang Guoliang, Zheng Zhujun. Chondrogenic differentiation of peripheral blood-derived mesenchymal stem cells induced by transforming growth factor beta 3: a dose-effect relationship [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(1): 45-51. |
| [6] | Jing Jin, Zhao Shandi, Chen Long, Peng Shuanglin, Tang Hui, Guo Daijin, Zeng Xinyi, Xiao Jingang. Repair of calvarial defects in osteoporotic mice by adipose-derived stem cells combined with biphasic calcium phosphate ceramic scaffold [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(1): 90-95. |
| [7] | Kang Yue, Liu Jie. Biological function of circular RNAs in osteogenic differentiation of mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(1): 107-112. |
| [8] | Pu Rui, Chen Ziyang, Yuan Lingyan. Characteristics and effects of exosomes from different cell sources in cardioprotection [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-. |
| [9] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [10] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [11] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [12] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [13] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| [14] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [15] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||