Chinese Journal of Tissue Engineering Research ›› 2024, Vol. 28 ›› Issue (22): 3532-3538.doi: 10.12307/2024.529
Previous Articles Next Articles
Performance of 3D-printed polylactic acid-nano-hydroxyapatite/chitosan/doxycycline antibacterial scaffold
Liu Yan, Zheng Xuexin
- Department of Scientific Research, Fuzhou Second Hospital, Fuzhou 350007, Fujian Province, China
-
Received:2023-09-04Accepted:2023-10-21Online:2024-08-08Published:2024-01-20 -
Contact:Liu Yan, Master, Research assistant, Department of Scientific Research, Fuzhou Second Hospital, Fuzhou 350007, Fujian Province, China -
About author:Liu Yan, Master, Research assistant, Department of Scientific Research, Fuzhou Second Hospital, Fuzhou 350007, Fujian Province, China -
Supported by:Fujian Provincial Health Technology Project, No. 2019-2-27 (to LY); Fujian Provincial Clinical Medical Research Center for First Aid and Rehabilitation in Orthopaedic Trauma, No. 2020Y2014
CLC Number:
Cite this article
Liu Yan, Zheng Xuexin. Performance of 3D-printed polylactic acid-nano-hydroxyapatite/chitosan/doxycycline antibacterial scaffold[J]. Chinese Journal of Tissue Engineering Research, 2024, 28(22): 3532-3538.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
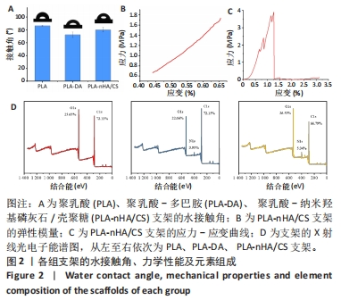
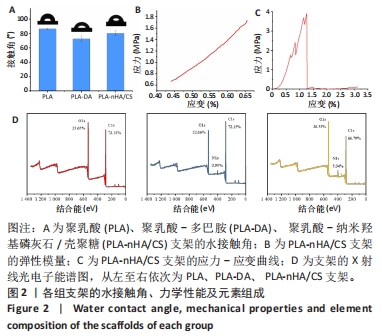
2.2 支架湿润性检测结果 植入生物材料主要通过其表面与生物系统相互作用,而生物材料的表面特性(如润湿性)会影响细胞黏附和蛋白质吸附[24-25],因此为了评估表面湿润性的变化,对所有支架进行静态水接触角测量。图2A显示各组支架的湿润性变。纯PLA支架的疏水性较强,水接触角为(86.50±0.67) °;PLA-DA支架表面的接触角下降到(72.50±4.17) °,材料向亲水型转变,说明多巴胺完全覆盖到支架表面;PLA-nHA/CS支架表面的接触角为(80.30±3.75) °,较PLA-DA支架略有升高,这可能与纳米羟基磷灰石/壳聚糖的涂覆量有关。显然,多巴胺涂层和纳米羟基磷灰石/壳聚糖涂层改善了聚乳酸表面的润湿性,因为多巴胺中邻苯二酚基团和纳米羟基磷灰石中羟基的存在使涂层具有亲水性,这有利于组织愈合和早期骨整合[26]。"
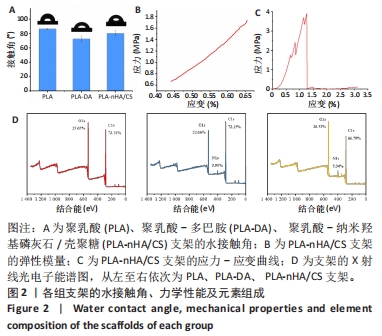

据报道,致密骨的弹性模量为3.9-11.7 GPa、压缩强度为88-164 MPa,松质骨的弹性模量为50-500 MPa、压缩强度为2-12 MPa[28-29]。PLA-nHA/CS支架的弹性模量检测曲线与应力-应变曲线,见图2B、C,显示PLA-nHA/CS为脆性材料,其弹性模量满足松质骨要求,抗压强度介于致密骨和松质骨之间,且明显高于松质骨,因此可以作为松质骨修复的合适候选者。 2.4 支架表面元素组成与结晶结构检测结果 为研究支架的化学结合状态,进行了X射线光电子能谱分析,图2D显示了3种支架的曲线拟合结果。多巴胺修饰后,支架中O1s从25.65%减少到22.66%,同时出现N1s峰,浓度从0%增加到3.95%,表明多巴胺沉积在PLA支架上。涂覆纳米羟基磷灰石和壳聚糖后,支架中C1s从72.15%减少到66.79%,O1s和N1s的浓度均有所增加,分别从22.66%到26.53%、3.95%到5.34%。结果表明多巴胺和纳米羟基磷灰石/壳聚糖成功改性PLA支架。"
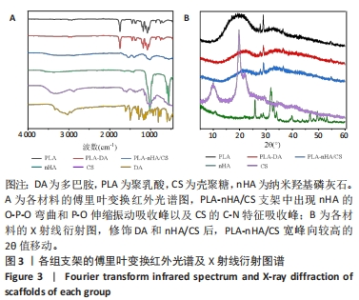
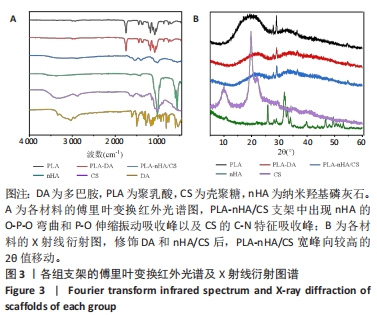
各组材料的傅里叶变换红外光谱分析结果,见图3A。PLA支架在1 748 cm-1处有C-O键伸缩振动的特征吸收峰,在1 180 cm-1和1 453 cm-1处分别有C-O键和甲基C-H键的特征吸收峰[9-30]。在PLA-DA支架中,未出现多巴胺的特征峰。在PLA-nHA/CS支架,556 cm-1和1 017 cm-1处的吸收峰分别对应纳米羟基磷灰石的O-P-O弯曲和P-O伸缩振动[31],1 153 cm-1处对应壳聚糖的C-N特征吸收峰。 各组材料的X射线衍射分析,见图3B。在PLA支架中,以2θ(约19°)为中心的宽衍射峰,显示了聚乳酸结构的浇注结晶特性[20]。当PLA支架经过多巴胺、壳聚糖和纳米羟基磷灰石功能化形成PLA-DA和PLA-nHA/CS支架时,可以观察到宽峰向较高的2θ值(20°-23°)轻微移动。在复合支架的X射线衍射结果中没有出现壳聚糖和纳米羟基磷灰石的衍射峰,尽管它们的结晶良好,这表明PLA-nHA/CS支架表面上壳聚糖和纳米羟基磷灰石的含量低于检测限。由于壳聚糖和纳米羟基磷灰石的涂覆量很低,很难形成均匀的结晶结构。"
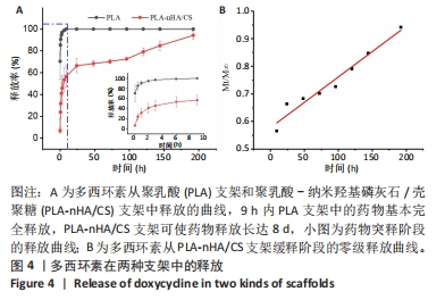
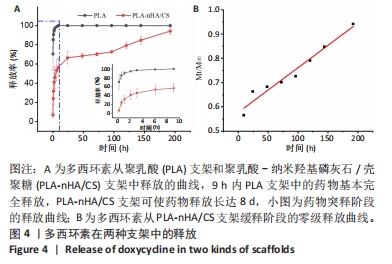
2.5 支架的药物释放 通过计算总药物释放量得到支架的载药量,PLA支架的载药量为(1 870±637) μg,PLA-nHA/CS支架的载药量为(2 207±613) μg。 PLA支架和PLA-nHA/CS支架的药物释放曲线,见图4A。药物在9 h内显示突然释放,大量药物从支架中释放到周围环境,PLA支架中的药物完全释放,而PLA-nHA/CS支架中的药物释放率为(56.55±9.85)%,然后进入缓释阶段,释放时间达到8 d。在缓释阶段,PLA-nHA/CS支架内部的药物迁移到支架表面,由于纳米羟基磷灰石/壳聚糖涂层的覆盖,药物随着该涂层的降解释放到环境中,药物释放速率稳定均匀,呈线性趋势,基本符合零级释放(图4B)。"
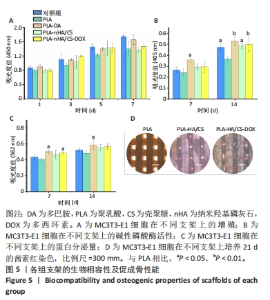
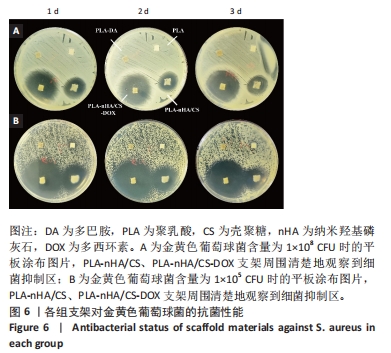
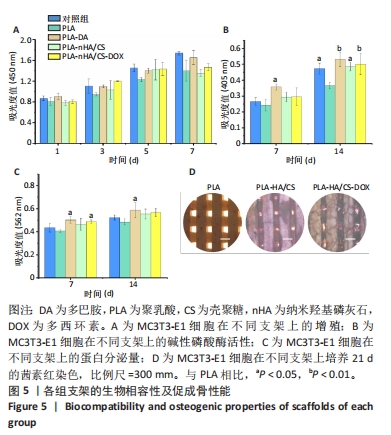
2.6 支架对成骨细胞增殖的影响 CCK-8检测结果显示所有支架均无细胞毒性,见图5A。培养1,3,5和7 d后,所有支架组细胞均明显增殖且与对照组相差不大,说明材料适于细胞生长。细胞的黏附可能与材料表面的亲水性有关[32]。因此得出结论,多巴胺和纳米羟基磷灰石/壳聚糖涂覆后的支架有利于细胞的黏附、迁移和增殖。 2.7 支架对成骨细胞成骨分化的影响 碱性磷酸酶是成骨细胞早期分化的标志酶,其分泌量可以反映成骨细胞的分化状况[33]。培养7,14 d后,在PLA-DA、PLA-nHA/CS和PLA-nHA/CS-DOX支架上培养细胞中的碱性磷酸酶和蛋白分泌量均高于PLA支架,见图5B、C,说明多巴胺和纳米羟基磷灰石/壳聚糖涂层具有显著的成骨作用,且负载多西环素后不影响支架的生物活性。有研究表明,多巴胺和羟基磷灰石涂层材料可促进干细胞的增殖和分化[34-36],因此PLA-nHA/CS支架可在成骨过程中发挥长期作用。 培养21 d后,各组支架上的钙沉积见图5D所示。值得注意的是,该图中只有钙化的无机相被染成红色。与PLA支架相比,PLA-nHA/CS及PLA-nHA/CS-DOX支架上的钙染色更强烈,表现出较高的矿化水平。可以看出,纳米羟基磷灰石/壳聚糖涂层和多西环素药物增强了MC3T3-E1细胞的成骨分化。"
| [1] WUBNEH A, TSEKOURA EK, AYRANCI C, et al. Current state of fabrication technologies and materials for bone tissue engineering. Acta Biomater. 2018;80:1-30. [2] KOONS GL, DIBA M, MIKOS AG. Materials design for bone-tissue engineering. Nat Rev Mater. 2020;5(8):584-603. [3] TETIK H, FENG D, OXANDALE SW, et al. Bioinspired manufacturing of aerogels with precisely manipulated surface microstructure through controlled local temperature gradients. ACS Appl Mater Interfaces. 2021;13(1):924-931. [4] ALDEMIR DIKICI BL, REILLY GC, CLAEYSSENS F. Boosting the osteogenic and angiogenic performance of multiscale porous polycaprolactone scaffolds by in vitro generated extracellular matrix decoration. ACS Appl Mater Interfaces. 2020;12(11):12510-12524. [5] XING H, ZOU B, LIU X, et al. Fabrication strategy of complicated Al2O3-Si3N4 functionally graded materials by stereolithography 3D printing. J Eur Ceram Soc. 2020;40(15):5797-5809. [6] BOULAALA M, ELMESSAOUDI D, BUJ-CORRAL I, et al. Towards design of mechanical part and electronic control of multi-material/multicolor fused deposition modeling 3D printing. Int J Adv Manuf Tech. 2020; 110:45-55. [7] ZHANG W, FENG C, YANG G, et al. 3D-printed scaffolds with synergistic effect of hollow-pipe structure and bioactive ions for vascularized bone regeneration. Biomaterials. 2017;135:85-95. [8] LIM SH, CHIA SMY, KANG L, et al. Three-Dimensional Printing of Carbamazepine Sustained-Release Scaffold. J Pharm Sci. 2016;105(7): 2155-2163. [9] YANG Y, QIAO X, HUANG R, et al. E-jet 3D printed drug delivery implants to inhibit growth and metastasis of orthotopic breast cancer. Biomaterials. 2020;230:119618. [10] GAO C, PENG S, FENG P, et al. Bone biomaterials and interactions with stem cells. Bone Res. 2017;5(1):1-33. [11] BAE EB, PARK KH, SHIM JH, et al. Efficacy of rhBMP-2 loaded PCL/β-TCP/bdECM scaffold fabricated by 3D printing technology on bone regeneration. Biomed Res Int. 2018;2018:1-12. [12] TERTULIANO OA, GREER JR. The nanocomposite nature of bone drives its strength and damage resistance. Nat Mater. 2016;15(11):1195-1202. [13] CROSS LM, THAKUR A, JALILI NA, et al. Nanoengineered biomaterials for repair and regeneration of orthopedic tissue interfaces. Acta Biomater. 2016;42:2-17. [14] QUAN H, HE Y, SUN J, et al. Chemical self-assembly of multifunctional hydroxyapatite with a coral-like nanostructure for osteoporotic bone reconstruction. ACS Appl Mater Interfaces. 2018;10(30):25547-25560. [15] REZWAN K, CHEN Q, BLAKER JJ, et al. Biodegradable and bioactive porous polymer/inorganic composite scaffolds for bone tissue engineering. Biomaterials. 2006;27(18):3413-3431. [16] GHORBANI F, ZAMANIAN A, BEHNAMGHADER A, et al. A facile method to synthesize mussel-inspired polydopamine nanospheres as an active template for in situ formation of biomimetic hydroxyapatite. Mat Sci Eng C Mater. 2019;94:729-739. [17] LÓPEZ-IGLESIAS C, BARROS J, ARDAO I, et al. Vancomycin-loaded chitosan aerogel particles for chronic wound applications. Carbohydr Polym. 2019;204:223-231. [18] FERREIRA M, RZHEPISHEVSKA O, GRENHO L, et al. Levofloxacin-loaded bone cement delivery system: Highly effective against intracellular bacteria and Staphylococcus aureus biofilms. Int J Pharm. 2017;532(1):241-248. [19] BAUMANN B, JUNGST T, STICHLER S, et al. Control of nanoparticle release kinetics from 3D printed hydrogel scaffolds. Angew Chem Int Ed. 2017;56(16):4623-4628. [20] MARTIN V, RIBEIRO IA, ALVES MM, et al. Engineering a multifunctional 3D-printed PLA-collagen-minocycline-nanoHydroxyapatite scaffold with combined antimicrobial and osteogenic effects for bone regeneration. Mat Sci Eng C Mater. 2019;101:15-26. [21] HUANG Y, ZHAO X, ZHANG Z, et al. Degradable gelatin-based IPN cryogel hemostat for rapidly stopping deep noncompressible hemorrhage and simultaneously improving wound healing. Chem Mater. 2020;32(15):6595-6610. [22] XIE C, LI P, LIU Y, et al. Preparation of TiO2 nanotubes/mesoporous calcium silicate composites with controllable drug release. Mat Sci Eng C-Mater. 2016;67:433-439. [23] GOUDARZI A, SADRNEZHAAD SK, JOHARI N. The prominent role of fully-controlled surface co-modification procedure using titanium nanotubes and silk fibroin nanofibers in the performance enhancement of Ti6Al4V implants - ScienceDirect. Surf Coat Technol. 2021;412:127001. [24] MATSUMOTO T, TASHIRO Y, KOMASA S, et al. Effects of surface modification on adsorption behavior of cell and protein on titanium surface by using quartz crystal microbalance system. Materials. 2020; 14(1):97. [25] JIA Z, SHI Y, XIONG P, et al. From solution to biointerface: graphene self-assemblies of varying lateral sizes and surface properties for biofilm control and osteodifferentiation. ACS Appl Mater Interfaces. 2016;8(27):17151-17165. [26] GITTENS RA, SCHEIDELER L, RUPP F, et al. A review on the wettability of dental implant surfaces II: Biological and clinical aspects. Acta Biomater. 2014;10(7):2907-2918. [27] GOMES FILHO MS, OLIVEIRA FA, BARBOSA MAA. A statistical mechanical model for drug release: Investigations on size and porosity dependence. Physica A. 2016;460:29-37. [28] WANG Y, SHI X, REN L, et al. Poly (lactide‐co‐glycolide)/titania composite microsphere‐sintered scaffolds for bone tissue engineering applications. J Biomed Mater Res B. 2010;93(1):84-92. [29] SUN H, ZHANG C, ZHANG B, et al. 3D printed calcium phosphate scaffolds with controlled release of osteogenic drugs for bone regeneration. Chem Eng J. 2022;427:130961. [30] OLIVEIRA JE, MATTOSO LH, ORTS WJ, et al. Structural and morphological characterization of micro and nanofibers produced by electrospinning and solution blow spinning: a comparative study. Adv Mater Sci Eng. 2013;2013:1-14. [31] NAZEER MA, YILGOR E, YAGCI MB, et al. Effect of reaction solvent on hydroxyapatite synthesis in sol–gel process. R Soc Open Sci. 2017; 4(12):171098. [32] WU C, ZHANG Y, ZHOU Y, et al. A comparative study of mesoporous glass/silk and non-mesoporous glass/silk scaffolds: physiochemistry and in vivo osteogenesis. Acta Biomater. 2011;7(5):2229-2236. [33] VIMALRAJ S. Alkaline phosphatase: Structure, expression and its function in bone mineralization. Gene. 2020;754:144855. [34] SHI X, LI L, OSTROVIDOV S, et al. Stretchable and micropatterned membrane for osteogenic differentation of stem cells. ACS Appl Mater Interfaces. 2014;6(15):11915-11923. [35] YU Y, LI X, LI J, et al. Dopamine-assisted co-deposition of hydroxyapatite-functionalised nanoparticles of polydopamine on implant surfaces to promote osteogenesis in environments with high ROS levels. Mat Sci Eng C Mater. 2021;131:112473. [36] FITZPATRICK V, MARTíN-MOLDES Z, DECK A, et al. Functionalized 3D-printed silk-hydroxyapatite scaffolds for enhanced bone regeneration with innervation and vascularization. Biomaterials. 2021;276:120995. [37] CAMPANA V, MILANO G, PAGANO E, et al. Bone substitutes in orthopaedic surgery: from basic science to clinical practice. J Mater Sci Mater Med. 2014;25:2445-2461. [38] BAUER T, MUSCHLER G. Bone graft materials. An overview of the basic science. Clin Oryhop Relat R. 2000;371:10-27. [39] INZANA JA, OLVERA D, FULLER SM, et al. 3D printing of composite calcium phosphate and collagen scaffolds for bone regeneration. Biomaterials. 2014;35(13):4026-4034. [40] GAO C, PENG S, FENG P, et al. Bone biomaterials and interactions with stem cells. Bone Res. 2017;5(4):17059. [41] KRASOWSKA A, SIGLER K. How microorganisms use hydrophobicity and what does this mean for human needs? Front Cell Infect Microbiol. 2014;4:112. [42] QUAN H, HE Y, SUN J, et al. Chemical Self-Assembly of Multifunctional Hydroxyapatite with a Coral-like Nanostructure for Osteoporotic Bone Reconstruction. ACS Appl Mater Interfaces. 2018;10(30):25547-25560. [43] ZHANG B, WANG L, SONG P, et al. 3D printed bone tissue regenerative PLA/HA scaffolds with comprehensive performance optimizations. Mater Des. 2021;201:109490. [44] CAMPOCCIA D, MONTANARO L, ARCIOLA CR. The significance of infection related to orthopedic devices and issues of antibiotic resistance. Biomaterials. 2006;27(11):2331-2339. [45] VISSCHER L, DANG HP, KNACKSTEDT M, et al. 3D printed Polycaprolactone scaffolds with dual macro-microporosity for applications in local delivery of antibiotics. Mat Sci Eng C Mater. 2018; 87:78-89. |
| [1] | Cheng Weilu, Wang Zehua, Zhang Yidan, Liu Yinghui. Application and regulatory challenges of organoid technology in medical field [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(1): 202-210. |
| [2] | Yang Yufang, Yang Zhishan, Duan Mianmian, Liu Yiheng, Tang Zhenglong, Wang Yu. Application and prospects of erythropoietin in bone tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(9): 1443-1449. |
| [3] | Wang Menghan, Qi Han, Zhang Yuan, Chen Yanzhi. Three kinds of 3D printed models assisted in treatment of Robinson type II B2 clavicle fracture [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(9): 1403-1408. |
| [4] | Ning Tianliang, Wang Kun, Wang Lingbiao, Han Pengfei. Finite element analysis on correction effect of varus foot orthosis based on the three-point force principle [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(6): 891-899. |
| [5] | Yin Tong, Yang Jilei, Li Yourui, Liu Zhuoran, Jiang Ming. Application of core-shell structured nanofibers in oral tissue regeneration [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 766-770. |
| [6] | Wang Jiani, Chen Junyu. Angiogenesis mechanism of metal ions and their application in bone tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 804-812. |
| [7] | Wang Wu, Fan Xiaolei, Xie Jie, Hu Yihe, Zeng Min. Hydroxyapatite-polyvinyl alcohol/collagen-chitosan-gelatin composite hydrogel for repairing rabbit osteochondral defect [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 682-689. |
| [8] | Shen Ziqing, Xia Tian, Shan Yibo, Zhu Ruijun, Wan Haoxin, Ding Hao, Pan Shu, Zhao Jun. Vascularized tracheal substitutes constructed by exosome-load hydrogel-modified 3D printed scaffolds [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 697-705. |
| [9] | Zhu Liwei, Wang Jiangyue, Bai Ding. Application value of nanocomposite gelatin methacryloyl hydrogels in different bone defect environments [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 753-758. |
| [10] | Yang Yuqing, Chen Zhiyu. Role and application of early transient presence of M1 macrophages in bone tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(4): 594-601. |
| [11] | Liu Penghui, Wu Fan, Wang Zejie, Wu Gaoyi, Zhou Libo. Constructing an animal model of temporomandibular joint osteoarthritis in Sprague-Dawley rats by digital technology [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(35): 5642-5648. |
| [12] | Li Yulin, Yu Haipeng, Tang Huajing, Zhang Zitong, Lin Xingnan. The mechanism, safety and application of berberine in promoting bone regeneration [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(35): 5702-5708. |
| [13] | Zhang Aili, Huang Jiazheng, Fan Wen, Li Yihuan, Li Shuang, Gan Xuewen, Xiong Ying. Establishing equivalent model to verify the precision of personalized bone model rapidly [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(30): 4795-4799. |
| [14] | Kong Xiangyu, Wang Xing, Pei Zhiwei, Chang Jiale, Li Siqin, Hao Ting, He Wanxiong, Zhang Baoxin, Jia Yanfei. Biological scaffold materials and printing technology for repairing bone defects [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 479-485. |
| [15] | Xu Jing, Lyu Huixin, Bao Xin, Zhang Yi, Wang Yihan, Zhou Yanmin. Application of near infrared responsive hydrogels in tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 486-492. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||