Chinese Journal of Tissue Engineering Research ›› 2023, Vol. 27 ›› Issue (15): 2427-2434.doi: 10.12307/2023.607
Previous Articles Next Articles
Application status and prospect of menstrual blood-derived stem cells and their exosomes
Ma Yujiu1, 2, Zhang Xudong1, 2, Tan Jichun1, 2
- 1Center of Reproductive Medicine, Department of Obstetrics and Gynecology, Shengjing Hospital of China Medical University, Shenyang 110022, Liaoning Province, China; 2Key Laboratory for Reproductive Disorder and Fertility Remodeling of Liaoning Province, Shenyang 110022, Liaoning Province, China
-
Received:2022-06-15Accepted:2022-07-21Online:2023-05-28Published:2022-10-18 -
Contact:Tan Jichun, MD, Doctoral supervisor, Center of Reproductive Medicine, Department of Obstetrics and Gynecology, Shengjing Hospital of China Medical University, Shenyang 110022, Liaoning Province, China; Key Laboratory for Reproductive Disorder and Fertility Remodeling of Liaoning Province, Shenyang 110022, Liaoning Province, China -
About author:Ma Yujiu, Master candidate, Center of Reproductive Medicine, Department of Obstetrics and Gynecology, Shengjing Hospital of China Medical University, Shenyang 110022, Liaoning Province, China; Key Laboratory for Reproductive Disorder and Fertility Remodeling of Liaoning Province, Shenyang 110022, Liaoning Province, China -
Supported by:National Natural Science Foundation of China, No. 82071601 (to TJC); the Liaoning Province Central Leading Local Science and Technology Development Special Project, No. 2020JH6/10500006 (to TJC); the Liaoning Province Key Research and Development Program Project, No. 2018225093 (to TJC); Subject Enhancement Program of China Medical University (to TJC); the Shengjing Free Investigator Fund (to TJC)
CLC Number:
Cite this article
Ma Yujiu, Zhang Xudong, Tan Jichun. Application status and prospect of menstrual blood-derived stem cells and their exosomes[J]. Chinese Journal of Tissue Engineering Research, 2023, 27(15): 2427-2434.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
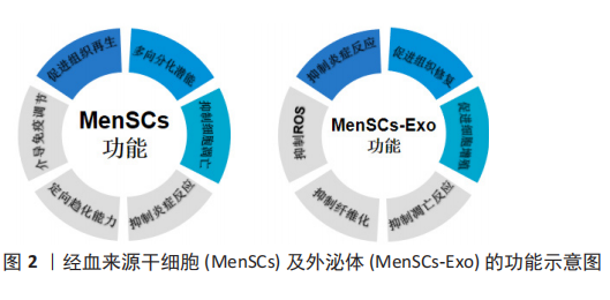
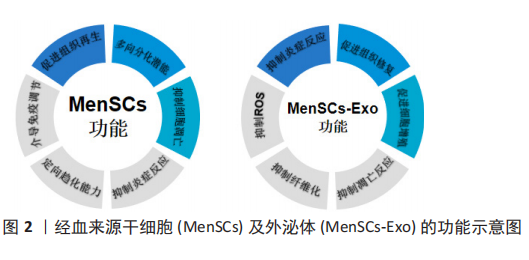
2.1 经血来源干细胞及其外泌体的特征 2.1.1 经血来源干细胞的特征 经血来源干细胞的典型细胞形态与骨髓间充质干细胞相似,多数为梭形,并呈栅栏状或涡旋状排列,细胞的核质色深,较成体细胞大,胞质色略浅,似成纤维细胞形态[5]。表型特征分析显示,经血来源干细胞不表达CD14,CD34,CD38,CD45,CD117,CD133,STRO-1,SSEA-4,HLA-DR,但CD9,CD29,CD59,CD73,CD41a,CD44,CD73,CD90,CD105,CD166,hTERT,OCT-4,HLA-ABC,SOX2,SSEA-4,Nanog等标志物呈阳性表达[2,6-7]。核型分析显示经血来源干细胞在多次传代扩增后仍能保持正常的染色体核型[2]。与脐血间充质干细胞相比,基质金属蛋白酶3、基质金属蛋白酶10、粒细胞-巨噬细胞集落刺激因子、血小板衍生生长因子BB、ANG-2在蛋白质组学分析表达更为显著[2],体现其具有强大的旁分泌功能。 经血来源干细胞可通过密度梯度离心法从月经血中分离,具有极强的增殖能力,在培养液中培养48 h后进入对数生长期,生长迅速,培养96 h后逐渐进入平台期。且经血来源干细胞在体外可稳定增殖22代后仍保持核型稳定[8]。 2.1.2 经血来源干细胞外泌体的特征 通过透射电镜观察外泌体呈典型的圆形杯状囊泡样,有完整的膜结构,NTA粒径分析仪显示粒径大小为30-150 nm,Western blot结果可显示CD63和CD81的表达[9]。经血来源干细胞及其外泌体的功能,见图2。"
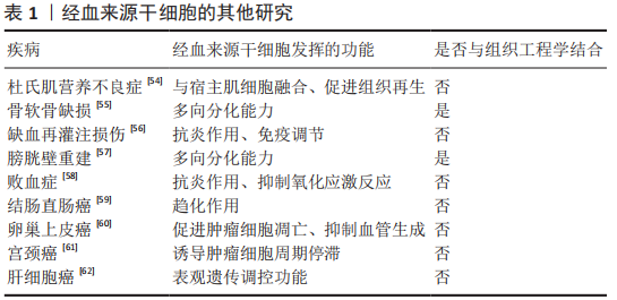
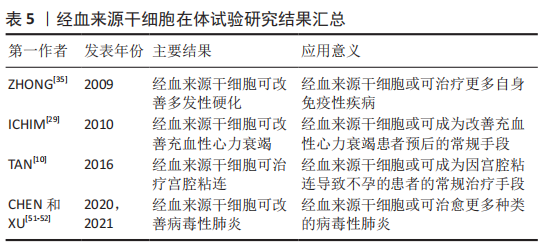
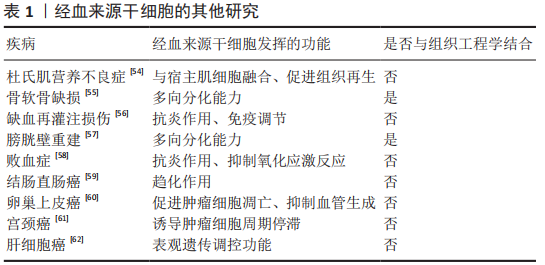
2.2 经血来源干细胞的临床应用及展望 2.2.1 生殖系统 宫腔粘连是造成妇女不孕的主要原因之一,目前主要依靠手术治疗,但中重度宫腔粘连的术后复发率很高。目前已在临床试验中证实移植自体经血来源干细胞可改善薄型子宫内膜症状,使患者能够利用辅助生殖技术受孕[10],且经血来源干细胞与富血小板血浆联合应用可提升治疗效果[11]。也有研究发现经血来源干细胞是通过激活Hippo/TAZ通路来改善宫腔粘连症状的[12]。此外,经血来源干细胞的胞外囊泡(经血来源干细胞-细胞外囊泡)也能安全有效地促进子宫内膜的修复[13]。 宫腔粘连也继发于宫腔内炎症,促炎因子白细胞介素6和干扰素γ增加负反馈地使基质金属蛋白酶活性下降,促进了子宫内膜纤维化的产生[14],经血来源干细胞具有比脐血间充质干细胞更强的促进基质金属蛋白酶表达的能力,可能是治疗宫腔粘连的原因之一。 卵巢功能不全是指由于各种原因导致卵巢功能受损,卵泡提早耗竭,血清性激素水平下降,促性腺激素水平上升,并出现包含月经稀发、不孕等一系列临床症状的疾病。YAMCHI等[15]发现,向患有早发性卵巢功能不全的大鼠体内注射经血来源干细胞可增加早衰卵巢中卵泡的数量,减少卵巢纤维化,改善妊娠结局。经血来源干细胞可通过激活细胞外基质依赖的FAK/Akt信号通路中卵巢转录表达来部分恢复卵巢功能[16]。促性腺激素释放激素与经血来源干细胞联合治疗可上调PI3K-Akt信号通路表达,改善卵巢储备,实现卵巢修复和保护[17]。 一项体外研究提示经血来源干细胞可在20%人类卵泡液的诱导下分化形成卵母细胞样细胞,这种细胞表达生殖细胞标记也含有卵泡刺激激素受体和黄体生成素受体,并可在促性腺激素的调节下产生雌激素和孕激素[18],这也为治疗卵巢功能不全提供了新的研究方向,即是否可通过培养卵巢类似物来替代现有的激素治疗。 盆腔器官脱垂与怀孕和经阴道分娩造成的盆底韧带和肌肉的直接损伤高度相关,研究发现在聚酰胺/明胶复合支架上用外源转化生长因子β、血小板衍生生长因子BB、结缔组织生长因子诱导经血来源干细胞分化为平滑肌细胞和成纤维细胞用于盆腔器官脱垂的修复[19-20],表明经血来源干细胞也具有应用于组织工程的潜力。 近年来,大量的家庭使用体外受精技术助孕,但仍有部分患者未能成功受孕。优化胚胎培养环境和改善胚胎质量是改善体外受精结局的两个主要途径,研究表明经血来源干细胞分泌的血管生成素2和肝细胞生长因子可能在小鼠胚胎与经血来源干细胞共培养中发挥了重要作用[21]。经血来源干细胞-细胞外囊泡也可使胚胎分泌更多血管内皮生长因子和血小板衍生生长因子AA,不仅促进子宫内膜新生血管生成和组织重塑,从而提高子宫内膜的容受性,也可促进胚胎发育,改善体外受精结局[22],表明经血来源干细胞可通过改善胚胎发育的微环境来促进胚胎发育。 子宫内膜异位症的病因尚不完全明确,目前的各种学说都不能解释所有类型的子宫内膜异位症的成因,血管生成和细胞迁移是形成子宫内膜异位病变的重要过程[23],多项研究表明内异症组织中高表达促进血管生成的生长因子[24-25],根据经血来源干细胞的生物学特性,常琦圆等[23]提出假设,子宫内膜异位症可能是由于经血来源干细胞迁移到病灶部位并分泌促进血管生成的因子,从而形成异位内膜组织。因此,阻断经血来源干细胞向其他部位迁移可能是治愈内异症的关键。 目前,全球各地的学者进行了诸多利用经血来源干细胞及其衍生物治疗生殖系统疾病的研究,发现它们主要发挥抑制纤维化和细胞凋亡、促进功能细胞再生和促生长因子表达以及促进自身分化成功能细胞的作用来达到缓解或治愈疾病的效果。 2.2.2 循环系统 心肌梗死是因冠状动脉急性缺血缺氧导致的心肌坏死症状,有较高的致残及致死率。动物研究证实,移植经血来源干细胞可缩小心肌纤维化面积并恢复左室收缩功能,移植的经血来源干细胞也可在梗死区域内分化为心肌细胞[26],但也有研究提示分化成心肌细胞的经血来源干细胞对改善心肌梗死没有明显作用[27]。经血来源干细胞也可通过旁分泌抑制p38信号通路来减轻细胞凋亡,刺激受损心肌细胞重新进入细胞周期,促进梗死部位的心肌再生[27],或通过抑制转化生长因子β/Smad通路来抑制心肌纤维化[28],这为改善梗死后心肌功能指明了方向。 充血性心力衰竭是各种心脏疾病的终末阶段,2010年,ICHIM等[29]发现体内注射经血来源干细胞可改善充血性心力衰竭患者的射血分数,提示患者预后的氨基末端B型利钠肽前体(Pro-BNP)也有所减少,这说明注射经血来源干细胞可作为延长充血性心力衰竭患者生命的治疗方法。 心脏移植是各种心脏疾病终末期的惟一治疗手段,但易出现免疫排斥反应导致供体心脏再次失效,因此找到建立免疫耐受的途径可延长供体心脏的使用周期,移植经血来源干细胞可抑制心脏移植术后B细胞的活化,减少抗体的产生[30],经血来源干细胞产生的基质细胞源性因子1可诱导建立移植免疫耐受[31]。 综上,经血来源干细胞可通过各种通路抑制心肌纤维化,也可通过其抑制炎症反应的作用来诱导建立免疫耐受,为其他器官移植开辟了新思路。 2.2.3 神经系统 脑卒中是由于脑部血管突然破裂或阻塞导致脑部血供不足而引起脑组织损伤的一种急性脑血管疾病,研究表明移植经血来源干细胞可减少脑卒中大鼠行为异常,在体外将经血来源干细胞与脑卒中大鼠的神经细胞共培养可明显减少神经细胞死亡,并提高培养基中神经保护相关生长因子水平[32]。 胶质瘤是一种来自胶质细胞的中枢神经系统肿瘤。因经血来源干细胞具有定向趋化能力,WANG等[33]以经血来源干细胞为载体开发了一种转导系统,在体内和体外都表现出减瘤能力。而经血来源干细胞自身也可减少胶质瘤新生血管生成,抑制肿瘤的生长[34],但这一结果与先前在多种疾病模型中经血来源干细胞可促进新生血管生成的结论不同,因此找到经血来源干细胞在不同情况下对血管生成的作用机制可为多种与血管改变相关的疾病提供方向。 多发性硬化是一种多发于青壮年的自身免疫性疾病,病理上以中枢神经系统的脱髓鞘损伤为特征。2009年,ZHONG等[35]首次对4例多发性硬化症患者进行经血来源干细胞治疗的临床试验,发现移植经血来源干细胞可延缓多发性硬化进展,且移植经血来源干细胞也没有对他们的身体产生不良反应。也有研究表明经血来源干细胞是通过抑制免疫反应,减少CD4+T细胞产生来减缓多发性硬化的发展的[36]。 帕金森病是一种以中脑黑质多巴胺能神经元变性和死亡、纹状体多巴胺含量减少为病理特征的,常见于老年人的神经系统变性疾病。有研究发现经血来源干细胞与帕金森病神经细胞共培养可提高细胞活力,抑制炎症反应和神经细胞凋亡,且经血来源干细胞可通过释放多种神经营养因子治疗帕金森病[37]。WOLFF等[38]证明移植的经血来源干细胞可迁移到帕金森病小鼠的黑质病变部位,并分化成可分泌多巴胺的细胞,增加纹状体多巴胺及其代谢物的浓度。 此外,经血来源干细胞也通过其抗炎、旁分泌及多向分化作用在阿尔茨海默病、脊髓损伤和其他周围神经疾病的动物模型实验中发挥治疗效果[39-41]。 神经系统疾病因其大多起病隐匿和神经细胞再生能力低,一直是其难以攻克的主要原因。经血来源干细胞在治疗神经系统疾病中展现了强大的促进神经细胞再生能力和抗炎作用,并因其可分化成功能性神经细胞,所以给退行性疾病也带来了新的治疗理念。此外,经血来源干细胞也可作为载体,借助其可向体内受损部位定向迁移的能力证明了其与抗肿瘤药物联合治疗的前景。 2.2.4 消化系统 自身免疫性肝炎是一种T细胞介导的对肝脏抗原异常反应的自身免疫疾病。WANG等[42]发现高表达Gal-9的经血来源干细胞可通过抑制Lck/ZAP70/LAT通路调节小鼠T细胞,减弱炎症反应,从而缓解小鼠自身免疫性肝炎症状。白细胞介素37也能增强经血来源干细胞的免疫调节特性和抗炎性,减少了CD4+ T细胞和巨噬细胞的生成[43]。胆汁淤积性肝病是指由各种原因造成的胆汁生成和运输障碍,导致胆汁在肝内淤积并反流入血引起各种消化系统或全身症状的肝胆系统疾病。YANG等[44] 发现向胆汁淤积性肝病模型小鼠移植经血来源干细胞可以改善小鼠肝功能,缓解肝内胆管扩张和胆汁淤积,减少炎症细胞浸润,经血来源干细胞也修复了肝脏紧密连接通路和胆汁转运体损伤,通过上调肝脏β-catenin的表达抑制α1-Ⅰ型胶原、α平滑肌蛋白和转化生长因子β1的表达改善胆汁淤积性肝病的症状。α1-Ⅰ型胶原、α平滑肌蛋白表达增加会导致肝星状细胞激活从而导致肝纤维化,肝纤维化是慢性肝病向肝硬化进展的关键步骤,而移植经血来源干细胞可减少肝硬化模型小鼠体内α平滑肌蛋白和α1-Ⅰ型胶原的表达[44],或许可成为延长慢性肝病患者生存期的疗法。 炎症性肠病为可累及全消化道的一种特发性肠道炎症性疾病,也与自身免疫反应有关。XU等[45]发现经血来源干细胞能调节炎症性肠病小鼠体内各种T细胞的比例,抑制B细胞的活化和IgG的产生,也有研究表明具有调节细胞因子谱和淋巴细胞比例功能的程序性死亡受体-配体1(PD-L1)是经血来源干细胞缓解结肠炎症状的必要条件[46],且介导趋化的基质细胞源性因子1/CXCR4轴表达上调可增强经血来源干细胞在缓解小鼠炎症性肠病中的免疫调节作用[47]。 1型糖尿病是由于免疫反应介导胰腺β细胞破坏导致的胰岛素绝对不足。移植经血来源干细胞不仅可减轻小鼠糖尿病症状,增加胰岛面积、可分泌胰岛素细胞数量及血清中的胰岛素水平 [48],也可与材料学相结合,治疗糖尿病导致的皮肤损伤[49]。 经血来源干细胞主要通过抑制炎症反应和纤维化、调节免疫反应来缓解消化系统疾病症状,许多自身免疫性疾病终身难以治愈,只能通过各种内外科手段延缓其进展,结合经血来源干细胞可诱导移植物建立免疫耐受及治疗多发性硬化和自身免疫性肝炎的研究,不难推测经血来源干细胞在治疗自身免疫性疾病具有广阔的前景。 2.2.5 呼吸系统 病毒性肺炎是由呼吸道病毒感染导致的肺部炎症,早期表现为间质性肺炎,严重者肺泡也可受累,甚至会发生肺组织坏死。急性呼吸窘迫综合征、肺衰竭和急性肺炎是H7N9感染患者的主要肺部改变,目前已有研究证实移植经血来源干细胞可缓解小鼠的急性肺损伤[50],有研究对H7N9和COVID-19患者进行了临床试验,发现经血来源干细胞移植可缓解患者的临床症状,降低患者的死亡率[51-52]。 肺纤维化是多种肺部疾病的终末期改变,移植经血来源干细胞可通过调控Bcl2/Bax/Cleaved caspase-3通路减少肺上皮细胞的凋亡,抑制上皮-间质转化及炎症和氧化应激反应[52-53]。 肺部炎症或其他损伤——肺纤维化是多种肺部疾病的病理过程,经血来源干细胞可从抑制炎症到抑制纤维化,促进功能细胞再生等一系列途径来改善肺功能。 2.2.6 其他疾病 文章归纳了经血来源干细胞在他疾病中的临床研究进展[54-62],见表1。"

2.3 经血来源干细胞外泌体的应用 经血来源干细胞外泌体也被证实在早发性卵巢功能不全中发挥治疗作用,不仅可促进模型大鼠颗粒细胞的增殖,抑制早衰卵巢中卵泡凋亡并促进卵泡发育,也能恢复动情周期和血清激素水平,恢复生育能力[64]。 WANG等[65]发现与脂肪源性间质干细胞和骨髓间充质干细胞相比,经血来源干细胞在大鼠心肌梗死的治疗中效果更好,增加了心肌微血管密度,且这一改善是由外泌体介导的,miR-21的表达在经血来源干细胞外泌体中选择性增强,并通过PTEN/Akt途径提高细胞存活率。此外,间充质干细胞具有分泌促进心肌细胞和血管再生、抗炎症反应和细胞凋亡、对抗心肌重塑、改善心脏收缩性和心肌细胞新陈代谢等多种细胞因子的旁分泌能力[66],因此经血来源干细胞外泌体有望成为比经血来源干细胞更佳的治疗心肌梗死的非细胞疗法。 暴发性肝衰竭也被称为急性肝衰竭,是由严重的肝损伤引起的突然出现的肝功能障碍,甚至出现肝性脑病。已有研究证实经血来源干细胞外泌体可抑制小鼠肝细胞凋亡,抑制炎症细胞因子产生,改善肝脏功能,提高暴发性肝衰竭小鼠模型的存活率[67]。提示经血来源干细胞外泌体可能成为乙型肝炎病毒患者长期管理中的常规治疗手段。此外,在一项临床试验中,已经证实了脐带来源间充质干细胞在慢加急性肝衰竭治疗中的有效性和安全性[68],经血来源干细胞外泌体作为一种非细胞治疗手段前景更佳。 前列腺癌是发生在前列腺的上皮性恶性肿瘤,发病率和致死率极高。有研究发现经血来源干细胞外泌体可通过抑制核转录因子κB通路、抑制活性氧途径下调了人前列腺细胞(PC3细胞)中血管内皮生长因子和碱性成纤维细胞生长因子的水平达到减瘤作用[69]。新生血管形成也是肿瘤的病理特征之一,内皮细胞负责血管生成,经血来源干细胞外泌体也具有诱导内皮细胞死亡的细胞毒性作用[70],且外泌体具有低致瘤性,或可与化疗药物一道成为减瘤的常规治疗手段。 经血来源干细胞外泌体也因其具有抑制炎症反应、减轻氧化应激损伤及抑制血管生成的作用,在糖尿病导致的皮肤损伤、肺纤维化等疾病中发挥治疗作用[71-75]。 经血来源干细胞外泌体对各疾病的具体作用机制,见表4。"

| [1] CRISAN M, YAP S, CASTEILLA L, et al. A perivascular origin for mesenchymal stem cells in multiple human organs. Cell Stem Cell. 2008;3(3):301-313. [2] MENG X, ICHIM TE, ZHONG J, et al. Endometrial regenerative cells: a novel stem cell population. J Transl Med. 2007;5:57. [3] BIDARIMATH M, KHALAJ K, KRIDLI RT, et al. Extracellular vesicle mediated intercellular communication at the porcine maternal-fetal interface: a new paradigm for conceptus-endometrial cross-talk. Sci Rep. 2017;7:40476. [4] HUANG-DORAN I, ZHANG CY, VIDAL-PUIG A. Extracellular vesicles: Novel mediators of cell communication in metabolic disease. Trends Endocrinol Metab. 2017;28(1):3-18. [5] 程贤鹦,周锦红.经血来源干细胞临床应用研究进展[J].中华临床医师杂志(电子版),2020,14(5):385-390. [6] LV H, HU Y, CUI Z, et al. Human menstrual blood: a renewable and sustainable source of stem cells for regenerative medicine. Stem Cell Res Ther. 2018;9(1):325. [7] LIU Y, NIU R, YANG F, et al. Biological characteristics of human menstrual blood-derived endometrial stem cells. J Cell Mol Med. 2018; 22(3):1627-1639. [8] 谭季春,李雅璇,王秋实,等.利用月经血建立的经血源性基质干细胞系[J].中国组织工程研究,2015,19(50):8155-8160. [9] 常琦圆.经血源性间充质干细胞来源外泌体治疗大鼠宫腔粘连的研究[D].沈阳:中国医科大学,2020. [10] TAN J, LI P, WANG Q, et al. Autologous menstrual blood-derived stromal cells transplantation for severe asherman’s syndrome. Hum Reprod. 2016;31(12):2723-272910. [11] ZHANG S, LI P, YUAN Z, et al. Platelet-rich plasma improves therapeutic effects of menstrual blood-derived stromal cells in rat model of intrauterine adhesion. Stem Cell Res Ther. 2019;10(1):61. [12] ZHU H, PAN Y, JIANG Y, et al. Activation of the hippo/taz pathway is required for menstrual stem cells to suppress myofibroblast and inhibit transforming growth factor beta signaling in human endometrial stromal cells. Hum Reprod. 2019;34(4):635-645. [13] ZHANG S, CHANG Q, LI P, et al. Concentrated small extracellular vesicles from menstrual blood-derived stromal cells improve intrauterine adhesion, a pre-clinical study in a rat model. Nanoscale. 2021;13(15): 7334-7347. [14] CHEN JM, HUANG QY, ZHAO YX, et al. The latest developments in immunomodulation of mesenchymal stem cells in the treatment of intrauterine adhesions, both allogeneic and autologous. Front Immunol. 2021;12:785717. [15] YAMCHI NN, RAHBARGHAZI R, BEDATE AM, et al. Menstrual blood cd146(+) mesenchymal stem cells reduced fibrosis rate in the rat model of premature ovarian failure. Cell Biochem Funct. 2021;39(8):998-1008. [16] FENG P, LI P, TAN J. Human menstrual blood-derived stromal cells promote recovery of premature ovarian insufficiency via regulating the ecm-dependent fak/akt signaling. Stem Cell Rev Rep. 2019;15(2): 241-255. [17] SUN F, WEI W. Effect of gnrh agonist combined with menstrual blood-derived stem cell transplantation on ovarian function in mice. Nan Fang Yi Ke Da Xue Xue Bao. 2021;41(12):1850-1856. [18] LAI D, GUO Y, ZHANG Q, et al. Differentiation of human menstrual blood-derived endometrial mesenchymal stem cells into oocyte-like cells. Acta Biochim Biophys Sin (Shanghai). 2016;48(11):998-1005. [19] SU K, EDWARDS SL, TAN KS, et al. Induction of endometrial mesenchymal stem cells into tissue-forming cells suitable for fascial repair. Acta Biomater. 2014;10(12):5012-5020. [20] EMMERSON SJ, GARGETT CE. Endometrial mesenchymal stem cells as a cell based therapy for pelvic organ prolapse. World J Stem Cells. 2016;8(5):202-215. [21] GONçALVES MF, ASENSI KD, NASCIMENTO ALL, et al. Human menstrual blood-derived mesenchymal cells improve mouse embryonic development. Tissue Eng Part A. 2020;26(13-14):769-779. [22] BLAZQUEZ R, SANCHEZ-MARGALLO FM, ALVAREZ V, et al. Murine embryos exposed to human endometrial mscs-derived extracellular vesicles exhibit higher vegf/pdgf aa release, increased blastomere count and hatching rates. PLoS One. 2018;13(4):e0196080. [23] 常琦圆,谭季春.经血源性间充质干细胞生物学特性及在组织修复重建中的临床应用[J].中国组织工程研究,2019,23(21):3432-3438 [24] PROESTLING K, BIRNER P, BALENDRAN S, et al. Enhanced expression of the stemness-related factors oct4, sox15 and twist1 in ectopic endometrium of endometriosis patients. Reprod Biol Endocrinol. 2016; 14(1):81. [25] YERLIKAYA G, BALENDRAN S, PROSTLING K, et al. Comprehensive study of angiogenic factors in women with endometriosis compared to women without endometriosis. Eur J Obstet Gynecol Reprod Biol. 2016;204:88-98. [26] HIDA N, NISHIYAMA N, MIYOSHI S, et al. Novel cardiac precursor-like cells from human menstrual blood-derived mesenchymal cells. Stem Cells. 2008;26(7):1695-1704. [27] JIANG Z, HU X, YU H, et al. Human endometrial stem cells confer enhanced myocardial salvage and regeneration by paracrine mechanisms. J Cell Mol Med. 2013;17(10):1247-1260. [28] ZHANG Z, WANG JA, XU Y, et al. Menstrual blood derived mesenchymal cells ameliorate cardiac fibrosis via inhibition of endothelial to mesenchymal transition in myocardial infarction. Int J Cardiol. 2013; 168(2):1711-1714. [29] ICHIM TE, SOLANO F, LARA F, et al. Combination stem cell therapy for heart failure. Int Arch Med. 2010;3(1):5. [30] XU X, LI X, GU X, et al. Prolongation of cardiac allograft survival by endometrial regenerative cells:Focusing on b-cell responses. Stem Cells Transl Med. 2017;6(3):778-78710. [31] LAN X, WANG G, XU X, et al. Stromal cell-derived factor-1 mediates cardiac allograft tolerance induced by human endometrial regenerative cell-based therapy. Stem Cells Transl Med. 2017;6(11):1997-2008. [32] BORLONGAN CV, KANEKO Y, MAKI M, et al. Menstrual blood cells display stem cell-like phenotypic markers and exert neuroprotection following transplantation in experimental stroke. Stem Cells Dev. 2010; 19(4):439-4521. [33] WANG XJ, XIANG BY, DING YH, et al. Human menstrual blood-derived mesenchymal stem cells as a cellular vehicle for malignant glioma gene therapy. Oncotarget. 2017;8(35):58309-58321. [34] HAN X, MENG X, YIN Z, et al. Inhibition of intracranial glioma growth by endometrial regenerative cells. Cell Cycle. 2009;8(4):606-610. [35] ZHONG Z, PATEL AN, ICHIM TE, et al. Feasibility investigation of allogeneic endometrial regenerative cells. J Transl Med. 2009;7:15. [36] 高海瑶.经血源性子宫内膜干细胞对实验性自身免疫性脑脊髓炎的治疗效果及作用机制研究[D].新乡:新乡医学院,2019. [37] LI H, YAHAYA BH, NG WH, et al. Conditioned medium of human menstrual blood-derived endometrial stem cells protects against mpp(+)-induced cytotoxicity in vitro. Front Mol Neurosci. 2019;12:80. [38] WOLFF EF, GAO XB, YAO KV, et al. Endometrial stem cell transplantation restores dopamine production in a parkinson’s disease model. J Cell Mol Med. 2011;15(4):747-755. [39] ZHAO Y, CHEN X, WU Y, et al. Transplantation of human menstrual blood-derived mesenchymal stem cells alleviates alzheimer’s disease-like pathology in app/ps1 transgenic mice. Front Mol Neurosci. 2018; 11:140. [40] WU Q, WANG Q, LI Z, et al. Human menstrual blood-derived stem cells promote functional recovery in a rat spinal cord hemisection model. Cell Death Dis. 2018;9(9):882. [41] FARZAMFAR S, NASERI-NOSAR M, GHANAVATINEJAD A, et al. Sciatic nerve regeneration by transplantation of menstrual blood-derived stem cells. Mol Biol Rep. 2017;44(5):407-41210. [42] WANG H, ZHAO Y, REN B, et al. Endometrial regenerative cells with galectin-9 high-expression attenuate experimental autoimmune hepatitis. Stem Cell Res Ther. 2021;12(1):541. [43] LI G, KONG D, QIN Y, et al. Il-37 overexpression enhances the therapeutic effect of endometrial regenerative cells in concanavalin a-induced hepatitis. Cytotherapy. 2021;23(7):617-626. [44] YANG Y, CHEN Y, ZHAO Y, et al. Human menstrual blood-derived stem cell transplantation suppresses liver injury in ddc-induced chronic cholestasis. Stem Cell Res Ther. 2022;13(1):57. [45] XU X, WANG Y, ZHANG B, et al. Treatment of experimental colitis by endometrial regenerative cells through regulation of b lymphocytes in mice. Stem Cell Res Ther. 2018;9(1):146. [46] SHI G, WANG G, LU S, et al. Pd-l1 is required for human endometrial regenerative cells-associated attenuation of experimental colitis in mice. Am J Transl Res. 2019;11(8):4696-4712. [47] LI X, LAN X, ZHAO Y, et al. Sdf-1/cxcr4 axis enhances the immunomodulation of human endometrial regenerative cells in alleviating experimental colitis. Stem Cell Res Ther. 2019;10(1):204. [48] SUN YL, SHANG LR, LIU RH, et al. Therapeutic effects of menstrual blood-derived endometrial stem cells on mouse models of streptozotocin-induced type 1 diabetes. World J Stem Cells. 2022;14(1): 104-116. [49] MIRZADEGAN E, GOLSHAHI H, SAFFARIAN Z, et al. The remarkable effect of menstrual blood stem cells seeded on bilayer scaffold composed of amniotic membrane and silk fibroin aiming to promote wound healing in diabetic mice. Int Immunopharmacol. 2022;102:108404. [50] XIANG B, CHEN L, WANG X, et al. Transplantation of menstrual blood-derived mesenchymal stem cells promotes the repair of lps-induced acute lung injury. Int J Mol Sci. 2017;18(4):689. [51] XU X, JIANG W, CHEN L, et al. Evaluation of the safety and efficacy of using human menstrual blood-derived mesenchymal stromal cells in treating severe and critically ill covid-19 patients: an exploratory clinical trial. Clin Transl Med. 2021;11(2):e297. [52] CHEN J, HU C, CHEN L, et al. Clinical study of mesenchymal stem cell treatment for acute respiratory distress syndrome induced by epidemic influenza a (h7n9) infection: a hint for covid-19 treatment. Engineering (Beijing, China). 2020;6(10):1153-116110. [53] CHEN X, WU Y, WANG Y, et al. Human menstrual blood-derived stem cells mitigate bleomycin-induced pulmonary fibrosis through anti-apoptosis and anti-inflammatory effects. Stem Cell Res Ther. 2020; 11(1):477. [54] ZHAO Y, LAN X, WANG Y, et al. Human endometrial regenerative cells attenuate bleomycin-induced pulmonary fibrosis in mice. Stem Cells Int. 2018;2018:3475137. [55] CUI CH, UYAMA T, MIYADO K, et al. Menstrual blood-derived cells confer human dystrophin expression in the murine model of duchenne muscular dystrophy via cell fusion and myogenic transdifferentiation. Mol Biol Cell. 2007;18(5):1586-1594. [56] KHANMOHAMMADI M, GOLSHAHI H, SAFFARIAN Z, et al. Repair of osteochondral defects in rabbit knee using menstrual blood stem cells encapsulated in fibrin glue: a good stem cell candidate for the treatment of osteochondral defects. Tissue Eng Regen Med. 2019; 16(3):311-324. [57] SUN P, LIU J, LI W, et al. Human endometrial regenerative cells attenuate renal ischemia reperfusion injury in mice. J Transl Med. 2016; 14:28. [58] SHOAE-HASSANI A, MORTAZAVI-TABATABAEI SA, SHARIF S, et al. Differentiation of human endometrial stem cells into urothelial cells on a three-dimensional nanofibrous silk-collagen scaffold: an autologous cell resource for reconstruction of the urinary bladder wall. J Tissue Eng Regen Med. 2015;9(11):1268-1276. [59] JIN W, ZHAO Y, HU Y, et al. Stromal cell-derived factor-1 enhances the therapeutic effects of human endometrial regenerative cells in a mouse sepsis model. Stem Cells Int. 2020;2020:4820543. [60] GUO Y, ZHANG Z, XU X, et al. Menstrual blood-derived stem cells as delivery vehicles for oncolytic adenovirus virotherapy for colorectal cancer. Stem Cells Dev. 2019;28(13):882-896. [61] BU S, WANG Q, ZHANG Q, et al. Human endometrial mesenchymal stem cells exhibit intrinsic anti-tumor properties on human epithelial ovarian cancer cells. Sci Rep. 2016;6:37019. [62] LIU QY, RUAN F, LI JY, et al. Human menstrual blood-derived stem cells inhibit the proliferation of hela cells via tgf-beta1-mediated jnk/p21 signaling pathways. Stem Cells Int. 2019;2019:9280298. [63] WU Y, CHEN X, ZHAO Y, et al. Genome-wide DNA methylation and hydroxymethylation analysis reveal human menstrual blood-derived stem cells inhibit hepatocellular carcinoma growth through oncogenic pathway suppression via regulating 5-hmc in enhancer elements. Stem Cell Res Ther. 2019;10(1):151. [64] ZHANG S, HUANG B, SU P, et al. Concentrated exosomes from menstrual blood-derived stromal cells improves ovarian activity in a rat model of premature ovarian insufficiency. Stem Cell Res Ther. 2021;12(1):178. [65] WANG K, JIANG Z, WEBSTER KA, et al. Enhanced cardioprotection by human endometrium mesenchymal stem cells driven by exosomal microrna-21. Stem Cells Transl Med. 2017;6(1):209-222. [66] SHAFEI AE, ALI MA, GHANEM HG, et al. Mesenchymal stem cell therapy: a promising cell-based therapy for treatment of myocardial infarction. J Gene Med. 2017;19(12):2995. [67] CHEN L, XIANG B, WANG X, et al. Exosomes derived from human menstrual blood-derived stem cells alleviate fulminant hepatic failure. Stem Cell Res Ther. 2017;8(1):9. [68] SHI M, ZHANG Z, XU R, et al. Human mesenchymal stem cell transfusion is safe and improves liver function in acute-on-chronic liver failure patients. Stem Cells Transl Med. 2012;1(10):725-731. [69] ALCAYAGA-MIRANDA F, GONZALEZ PL, LOPEZ-VERRILLI A, et al. Prostate tumor-induced angiogenesis is blocked by exosomes derived from menstrual stem cells through the inhibition of reactive oxygen species. Oncotarget. 2016;7(28):44462-44477. [70] ROSENBERGER L, EZQUER M, LILLO-VERA F, et al. Stem cell exosomes inhibit angiogenesis and tumor growth of oral squamous cell carcinoma. Sci Rep. 2019;9(1):663. [71] DALIRFARDOUEI R, JAMIALAHMADI K, JAFARIAN AH, et al. Promising effects of exosomes isolated from menstrual blood-derived mesenchymal stem cell on wound-healing process in diabetic mouse model. J Tissue Eng Regen Med. 2019;13(4):555-568. [72] SUN L, ZHU M, FENG W, et al. Exosomal mirna let-7 from menstrual blood-derived endometrial stem cells alleviates pulmonary fibrosis through regulating mitochondrial DNA damage. Oxid Med Cell Longev. 2019;2019:4506303. [73] FARAMARZI H, MEHRABANI D, FARD M, et al. The potential of menstrual blood-derived stem cells in differentiation to epidermal lineage: a preliminary report. World journal of plastic surgery. 2016; 5(1):26-31. [74] AKHAVAN-TAVAKOLI M, FARD M, KHANJANI S, et al. In vitro differentiation of menstrual blood stem cells into keratinocytes: a potential approach for management of wound healing. Biologicals. 2017;48:66-73. [75] CHANG QY, ZHANG SW, LI PP, et al. Safety of menstrual blood-derived stromal cell transplantation in treatment of intrauterine adhesion. World J Stem Cells. 2020;12(5):368-380. |
| [1] | Fang Xingyan, Tian Zhenli, Zhao Zheyi, Wen Ping, Xie Tingting. Effects of sodium arsenite on human umbilical vein endothelial cell injury and sphingosine kinases 1/sphingosine 1-phosphate signaling axis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(在线): 1-7. |
| [2] | Nong Fuxiang, Jiang Zhixiong, Li Yinghao, Xu Wencong, Shi Zhilan, Luo Hui, Zhang Qinglang, Zhong Shuang, Tang Meiwen. Bone cement augmented proximal femoral nail antirotation for type A3.3 intertrochanteric femoral fracturalysis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(在线): 1-10. |
| [3] | Pan Zhongjie, Qin Zhihong, Zheng Tiejun, Ding Xiaofei, Liao Shijie. Targeting of non-coding RNAs in the pathogenesis of the osteonecrosis of the femoral head [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1441-1447. |
| [4] | Dang Yi, Du Chengyan, Yao Honglin, Yuan Nenghua, Cao Jin, Xiong Shan, Zhang Dingmei, Wang Xin. Hormonal osteonecrosis and oxidative stress [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1469-1476. |
| [5] | Nie Chenchen, Su Kaiqi, Gao Jing, Fan Yongfu, Ruan Xiaodi, Yuan Jie, Duan Zhaoyuan, Feng Xiaodong. The regulatory role of circular RNAs in cerebral ischemia-reperfusion injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1286-1291. |
| [6] | Lian Shilin, Zhang Yan, Jiang Qiang, Zhang Hanshuo, Li Tusheng, Ding Yu. Interventional effects of whole blood and platelet-rich plasma with different preparation methods on nucleus pulposus cells [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1199-1204. |
| [7] | Xu Cong, Zhao He, Sun Yan. Regeneration of facial nerve injury repaired by biomaterial nerve conduits [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1089-1095. |
| [8] | Xue Ting, Zhang Xinri, Kong Xiaomei. Mesenchymal stem cell therapy for pneumoconiosis using nanomaterials combined with multi-modal molecular imaging [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1133-1140. |
| [9] | Li Xinyue, Li Xiheng, Mao Tianjiao, Tang Liang, Li Jiang. Three-dimensional culture affects morphology, activity and osteogenic differentiation of human periodontal ligament stem cells [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 846-852. |
| [10] | Li Qicheng, Deng Jin, Fu Xiaoyang, Han Na. Effects of bone marrow mesenchymal stem cells-derived exosomes on hypoxia-treated myoblasts [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 853-859. |
| [11] | Wang Min, Yin Xiushan, Wang Yingxi, Zhang Yan, Zhao Long, Xia Shuyue. Inhalation of bone marrow mesenchymal stem cells-derived exosomes alleviates inflammatory injury in chronic obstructive pulmonary disease [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 827-834. |
| [12] | Zhang Houjun, Deng Bowen, Jiang Shengyuan, Zhao Yi, Ren Jingpei, Xu Lin, Mu Xiaohong. Proteomic analysis of cerebrospinal fluid exosomes derived from cerebral palsy children [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 903-908. |
| [13] | Gao Ting, Ma Xiaohong, Li Xiaorong. Extraction and identification of exosomes from three different sources of ovarian granulosa cells [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 860-865. |
| [14] | Yuan Wei, Liu Jingdong, Xu Guanghui, Kang Jian, Li Fuping, Wang Yingjie, Zhi Zhongzheng, Li Guanwu. Osteogenic differentiation of human perivascular stem cells and its regulation based on Wnt/beta-catenin signaling pathway [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 866-871. |
| [15] | Hao Liufang, Duan Hongmei, Wang Zijue, Hao Fei, Hao Peng, Zhao Wen, Gao Yudan, Yang Zhaoyang, Li Xiaoguang. Spatiotemporal dynamic changes of ependymal cells after spinal cord injury in transgenic mice [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 883-889. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||