Chinese Journal of Tissue Engineering Research ›› 2023, Vol. 27 ›› Issue (15): 2420-2426.doi: 10.12307/2023.383
Previous Articles Next Articles
Mechanism and effect of platelet-rich plasma-derived exosomes for treating knee osteoarthritis
Huang Shuangshuang1, 2, Xiang Xiaona1, 3, 4, 5, Yu Xi1, 3, He Jing1, 3, 4
- 1Department of Rehabilitation Medicine, West China Hospital, Sichuan University, Chengdu 610041, China; 2The Fifth People's Hospital of Sichuan Province, Chengdu 610031, China; 3West China School of Medicine, Sichuan University, Chengdu 610041, China; 4Key Laboratory of Rehabilitation Medicine in Sichuan Province, Chengdu 610041, China; 5Institute of Rehabilitation Medicine, West China Hospital, Sichuan University, Chengdu 610041, China
-
Received:2022-05-05Accepted:2022-07-07Online:2023-05-28Published:2022-10-18 -
Contact:He Jing, MD, Professor, Chief physician, Department of Rehabilitation Medicine, West China Hospital, Sichuan University, Chengdu 610041, Sichuan Province, China; West China School of Medicine, Sichuan University, Chengdu 610041, Sichuan Province, China; Key Laboratory of Rehabilitation Medicine in Sichuan Province, Chengdu 610041, Sichuan Province, China -
About author:Huang Shuangshuang, Master candidate, Physician, Department of Rehabilitation Medicine, West China Hospital, Sichuan University, Chengdu 610041, Sichuan Province, China; The Fifth People's Hospital of Sichuan Province, Chengdu 610031, Sichuan Province, China -
Supported by:Scientific Research Project of Sichuan Provincial Health Commission, No. 20PJ037 (to YX); Natural Science Foundation of Sichuan Province, No. 2022NSFSC1312 (to YX)
CLC Number:
Cite this article
Huang Shuangshuang, Xiang Xiaona, Yu Xi, He Jing. Mechanism and effect of platelet-rich plasma-derived exosomes for treating knee osteoarthritis[J]. Chinese Journal of Tissue Engineering Research, 2023, 27(15): 2420-2426.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
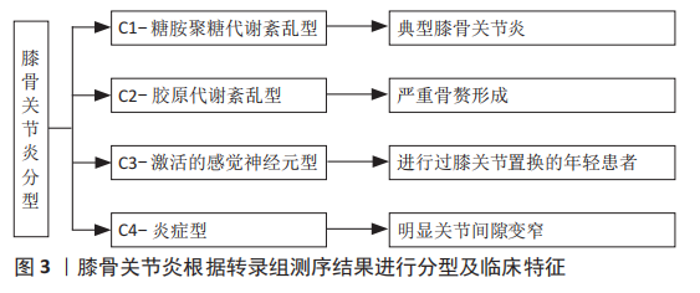
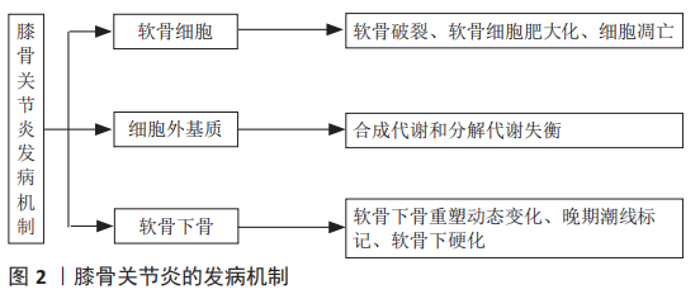
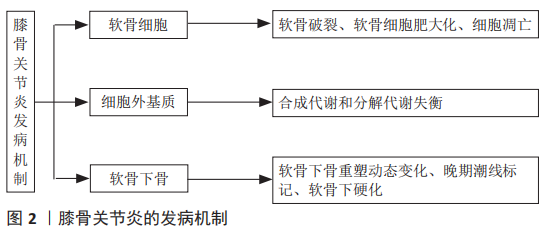
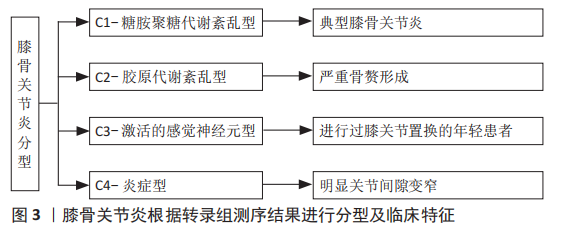
2.1.1 软骨细胞导致的变化 早期膝骨关节炎的治疗措施主要集中在软骨上。在病理结构上观察到软骨破裂,细胞水平上观察到软骨细胞肥大化、发生凋亡等细胞程序性死亡,因此治疗目标常包括重建受损的软骨、防止软骨分解、减少软骨细胞死亡等[28],进一步研究发现,软骨细胞的改变可能是基因错误表达导致的。目前发现2号染色体上编码的卷曲同源相关蛋白、6号染色体上编码的骨形成蛋白5和16号染色体上编码的白细胞介素4受体蛋白参与软骨细胞信号传导和信号转导通路,而这些基因可能引发异常的细胞信号传导或对信号的异常反应[29]。 2.1.2 细胞外基质导致的变化 软骨基质的合成和分解之间的动态平衡受合成代谢和分解代谢影响,合成代谢主要受到胰岛素样生长因子等生长因子的调节,而分解代谢主要受到白细胞介素1β、肿瘤坏死因子、蛋白酶的调节[27]。在一定范围内,软骨细胞受到机械刺激后可以通过改变细胞的代谢活动来消除或补偿导致骨关节炎的有害影响,但是当这些有害影响超出系统的补偿能力时,就会发生基质退化[30]。 2.1.3 软骨下骨导致的变化 软骨下骨异常可能是引起膝骨关节炎的初始原因,在疾病过程中软骨下骨的重塑处于动态变化[31]。软骨下骨指的是皮质骨板和软骨下松质骨,皮质骨板位于钙化软骨正下方,一般厚度为1-3 mm,而松质骨位于远端,比皮质骨板多孔且代谢活跃,并且具有更低的骨体积、密度和刚度[31]。在早期膝骨关节炎中,由于重塑率增加,软骨下皮质骨板变薄,同时,随着松质骨丢失,骨小梁板变得更薄。而在疾病晚期,软骨下皮质骨板增厚,但软骨下松质骨仍为骨质减少,钙化软骨开始向关节软骨推进,随着钙化软骨向前矿化,留下多个潮线标记,因此产生更厚的矿化板,并使得无法自行更新的关节软骨厚度减少,伴随着从关节软骨表面开始的蛋白聚糖丢失和纤维震颤[32]。因此在病理上体现出膝骨关节炎晚期是软骨下硬化(包括软骨下皮质骨板和钙化软骨)和更薄、更细的关节软骨。 2.1.4 膝骨关节炎分型 虽然膝骨关节炎的病理表现相似,但并非所有患者对药物等干预的反应是一致的,说明膝骨关节炎可能存在不同亚型。YUAN等[33]通过转录组测序将骨关节炎分为C1-糖胺聚糖代谢紊乱骨关节炎亚型、C2-胶原代谢紊乱骨关节炎亚型、C3-激活的感觉神经元骨关节炎亚型及C4-炎症骨关节炎亚型,见图3。C1的特征是软骨聚蛋白聚糖表达增加、多功能蛋白聚糖表达下降,反映膝骨关节炎早期;C2的特征是软骨下骨血管丛丰富、5A1与6A1胶原高表达;C3的特征是感觉神经纤维与软骨接触,高表达神经分子,体现出疼痛敏感;C4的特征是T淋巴细胞及单核细胞被激活,促进炎症反应。这项研究也反映出膝骨关节炎的不同分型或许与最初或最主要的发病机制相关。"
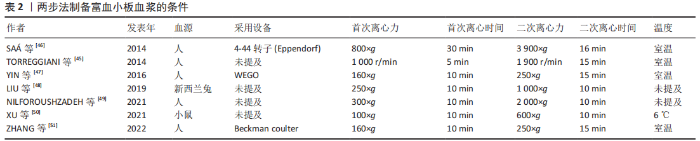
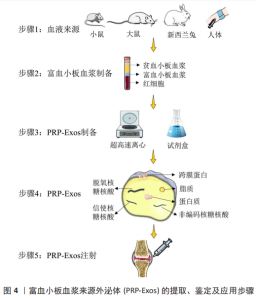
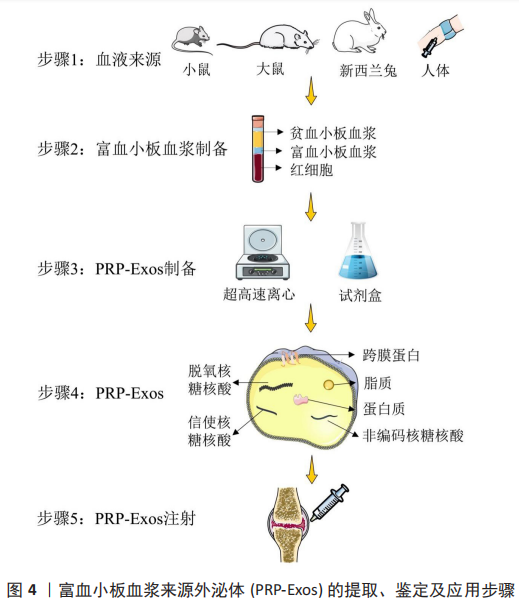
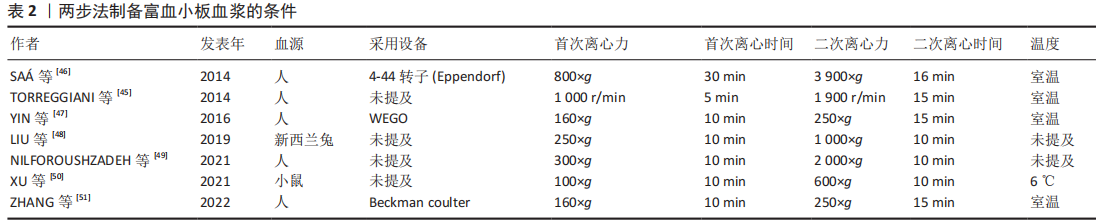
2.2 PRP-Exos的特征及应用 PRP-Exos是一种特定直径的细胞外囊泡,密度为1.1-1.2 g/mL[34],可完成细胞间的具有生物活性的脂质、核酸(DNA、mRNAs及非编码RNAs)及蛋白质的运输[35],促进受体细胞的基因表达以及细胞增殖、凋亡的调节与免疫调节等[36-38]。外泌体来源于内体,细胞质膜内陷可以形成早期内体,内体向腔内出芽形成腔内囊泡,成熟多囊泡内体与细胞膜融合释放腔内囊泡到细胞外[39]。 2.2.1 PRP-Exos的提取 PRP-Exos的提取可分为2步:首先需完成富血小板血浆的制备,多数研究选择2次离心。将血液加入带抗凝剂的采血管中,首次离心将血小板从血浆中的红细胞和白细胞中分离,然后将分离的含有血小板的血浆转移到新的离心管中,并以较高离心力再次离心,弃去大部分上清血浆,然后将血小板沉淀重新悬浮在残留血浆中以获得富血小板血浆[40]。IYER等[41]采用一步法离心,150 r/min离心5 min制备大鼠富血小板血浆,而ZHANG等[42]在4 ℃下采用1 000×g离心20 min制备兔源富血小板血浆。第二步需完成外泌体的提取,采用的方法包括超高速离心法及试剂盒法。CERVENAKOVA等[43]采用ExoQuickTM等试剂盒可较为便捷地完成外泌体的提取,4 ℃过夜孵育后1 500×g离心30 min。超高速离心法步骤较为复杂[44-45],如非新鲜富血小板血浆需重新离心悬浮,血小板颗粒被激活,然后通过低速离心以丢弃细胞碎片,取得的上清液通过0.22 μm过滤器完成大直径囊泡的去除,其次以 10 000-20 000×g连续离心30-60 min,将外泌体与微囊泡分离。以100 000-1 300 000×g的离心力完成70-90 min的2次超高速离心以获取PRP-Exos,以少量PBS重悬后可保存于-80 ℃。也有部分研究直接从血浆或者血小板裂解物中收集外泌体,见表2[45-51]。"
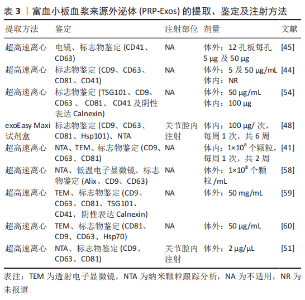
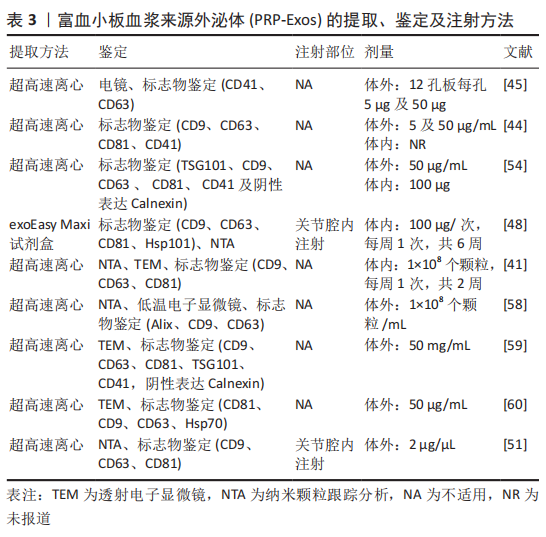
2.2.2 PRP-Exos的鉴定 PRP-Exos的鉴定与其他外泌体一致,包括通过透射电子显微镜表征囊泡形态[42]、纳米粒子跟踪分析显示粒径和浓度[41],以及外泌体带有的标志物验证,见表3。PRP-Exos的大小和表面标志物与其他细胞来源的外泌体相似,但外泌体的含量随着组织类型的不同而不同。目前研究报道了异源三聚体G蛋白、Hsp70[46]、Hsp101[48]、血小板表面糖蛋白CD41[44-45]、CD61[42,52],以及内体来源的四跨膜蛋白家族成员[53-54],如 CD9、CD63 和 CD81,其余还包括肿瘤易感基因 101(tumor suppressor gene 101,TSG101)和Alix[54-55]。TSG101是微囊泡体形成特异性转运相关蛋白所需的内体分选复合物,在外泌体中并不特异性表达[56]。尽管最近的一项研究表明,与其他和非CD63相比,CD63是一种更具特异性的微囊泡体表面标志物[57]。 2.2.3 PRP-Exos的治疗 目前PRP-Exos暂未进行临床应用,多数研究为细胞层面的研究,体外使用剂量包含1×108 个外泌体颗粒/mL、5 μg/mL、50 μg/mL及50 mg/mL[45,58-60]。动物体内注射采取原位关节腔内注射,100 μg/次,每周1次,共进行6周注射治疗[48],而结合生物材料后采取的剂量明显减低[51]。PRP-Exos应用流程中涉及的提取、鉴定及注射方式见表3及图4。"
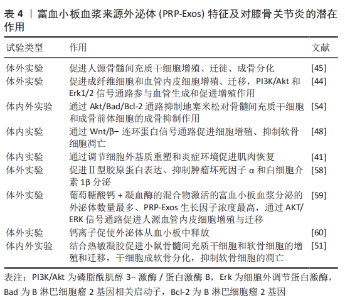
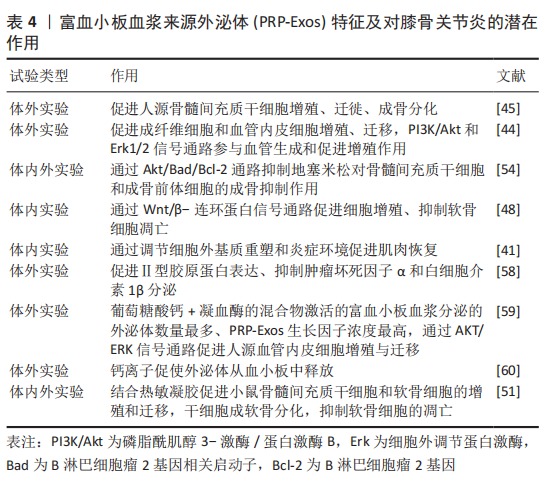
2.3 PRP-Exos的效应原理 2.3.1 PRP-Exos与富血小板血浆的异同 一般根据其白细胞与纤维蛋白的含量比可以将富血小板血浆分为4类:①富含白细胞的富血小板血浆;②贫白细胞的富血小板血浆;③富含白细胞的血小板纤维蛋白;④纯富血小板纤维蛋白[16]。富血小板纤维蛋白是三维结构的纤维蛋白基质,因此富血小板纤维蛋白在给药或移植后不会迅速溶解,且其制备无需使用抗凝剂或活化剂[61]。但无论是哪种富血小板血浆其发挥作用的主要机制在于活化的血小板分泌大量的生长因子、趋化因子和细胞因子,促进血管生成,调节细胞外基质,加速细胞募集、增殖和分化,并抑制软骨细胞的凋亡[54]。活化后的富血小板血浆可上调碱性成纤维生长因子(basic fibroblast growth factor,bFGF)、血小板衍生生长因子(platelet- derived growth factor,PDGF)AB/BB、转化生长因子β(transforming growth factor-β,TGF-β)和血管内皮生长因子(vascular endothelial growth factor,VEGF)的表达[45]。虽然富血小板血浆通常在37 ℃或室温下制备,但ETULAIN等[62]发现在4 ℃条件下预孵育富血小板血浆可最大限度地释放血小板衍生的促血管生成分子。 PRP-Exos以往被认为是从活化的血小板中释放的促凝物质[63],富血小板血浆起效的各类因子一旦从血小板中释放出来将失去磷脂膜的保护,可能会被细胞外环境中的裂解酶破坏并迅速失去其生物活性[64],因此外泌体这样的天然囊泡或许是一种延效工具。另外PRP-Exos中生长因子的含量高于富血小板血浆[45],但目前暂无富血小板血浆与PRP-Exos的内容成分、机制及疗效的对比研究,PRP-Exos能否替代富血小板血浆仍需大量研究。 2.3.2 不同激活剂对PRP-Exos的影响 有多种方法可以激活富血小板血浆,不同的活化剂可能会影响生物活性分子的释放和纤维蛋白原的裂解。RUI等[59]设置0.1 U/mL 凝血酶,2 mmol/L葡萄糖酸钙溶液,等体积的 2 mmol/L葡萄糖酸钙溶液+ 0.1 U/mL凝血酶的混合物及等体积的生理盐水为活化剂探究不同激活方式对富血小板血浆及PRP-Exos的影响,研究发现用凝血酶+葡萄糖酸钙激活的富血小板血浆收获了最高浓度的外泌体,其次为葡萄糖酸钙、凝血酶。同时凝血酶+葡萄糖酸钙激活后产生的PRP-Exos中,细胞因子(血管内皮生长因子、血小板衍生生长因子BB、碱性成纤维生长因子、转化生长因子β)浓度最高。此外,SAUMELL-ESNAOLA等[60]发现CaCl2活化的血小板比未活化的血小板释放更多的外泌体,透射电子显微镜下形态典型,并且CaCl2活化后的PRP-Exos更加符合外泌体的分子特征。包括生长因子和细胞因子在内的105种分析物的阵列分析表明,CaCl2活化后的PRP-Exos中大多数分析物水平较低,但外泌体标志蛋白水平相对较高。因此,认为钙离子促使外泌体从血小板中释放。 2.4 PRP-Exos的作用 2.4.1 PRP-Exos对膝骨关节炎的作用 PRP-Exos对膝骨关节炎的治疗作用主要体现在促进软骨细胞增殖、迁徙,减少软骨细胞凋亡及肥大化,抑制炎症因子的释放,促进细胞外基质分泌,见表4。LIU等[48]在软骨细胞体外实验中添加白细胞介素1β模拟炎症环境,研究发现PRP-Exos仍具有促进软骨细胞增殖与迁徙能力,同时促进软骨基质分泌、减少软骨细胞凋亡。OTAHAL等[58]通过实时定量聚合酶链式反应对软骨细胞的基因表达分析显示,PRP-Exos加入激活后的M1型巨噬细胞共培养的骨关节炎来源软骨细胞后Ⅱ型胶原蛋白表达增加,而通过酶联免疫吸附法检测显示肿瘤坏死因子α 和白细胞介素1β 水平较低,说明PRP-Exos对炎症因子有抑制作用,并促进细胞外基质的分泌。 除此之外,PRP-Exos发挥作用可能与剂量相关。TORREGGIANI等[45]比较了0.6,5,50 μg这3种浓度的血小板裂解物来源外泌体对人源骨髓间充质干细胞的影响,5 μg 及50 μg的外泌体分别可促进10.3倍及27.1倍的细胞增殖,同时促进细胞迁徙,5 μg的外泌体还可促进骨髓间充质干细胞成骨分化。GUO等[44]发现5 μg/mL PRP-Exos和50 μg/mL PRP-Exos对成纤维细胞及血管内皮细胞的生长显示出相似的增殖率。 2.4.2 PRP-Exos对其他骨骼肌肉系统疾病的作用 目前PRP-Exos对膝骨关节炎的研究较少,但PRP-Exos在其他骨骼肌肉系统疾病中的研究也有一定参考意义。如TAO等[54]发现PRP-Exos 可以促进骨组织的维持和再生,并促进细胞增殖。体外实验结果表明PRP-Exos 可以在添加地塞米松的激素环境下促进血管生成,用PRP-Exos处理的骨髓间充质干细胞和成骨前体细胞的凋亡细胞比例更少。PRP-Exos 可以抑制体外激素环境下对骨髓间充质干细胞和成骨前体细胞的成骨抑制作用。另外,IYER 等[41]认为PRP-Exos及间充质干细胞来源外泌体均可以加速损伤肌肉的恢复。通过大鼠构建的肌肉损伤模型发现两者可以通过调节细胞外基质和抑制炎症,从而促进肌肉恢复,但涉及机制不完全相同,PRP-Exos治疗后肌细胞生成素增加,而间充质干细胞来源外泌体治疗后炎症因子表达明显下降。PRP-Exos与组织材料的结合近年也在积极探索中。ZHANG等[51]将PRP-Exos与热敏水凝胶(PRP-Exo-Gel)结合后移植于距下关节,发现PRP-Exo-Gel可连续释放外泌体长达28 d,并促进小鼠骨髓间充质干细胞和软骨细胞的增殖、迁移,同时增强干细胞的成软骨分化,抑制炎症诱导的软骨细胞变性。通过体内实验证实,PRP-Exo-Gel增加了外泌体的释放时间,抑制了软骨细胞的凋亡和肥大,促进其增殖,并可能在干细胞募集中发挥作用,延缓骨关节炎的进展。PRP-Exos在不同的骨骼肌肉系统疾病模型上均表现出对细胞生长、细胞外基质及炎症的调节作用,与生物材料的结合也是膝骨关节炎治疗的未来方向。 2.5 PRP-Exos作用机制 目前关于PRP-Exos的机制探究主要在分子层面。PRP-Exos在促血管生成方面的机制涉及PI3K/Akt 和 Erk1/2信号通路[44,59]。此外,PRP-Exos 通过 Akt/Bad/Bcl-2 信号通路抑制激素环境下细胞凋亡[54],并通过抑制白细胞介素1β刺激的软骨细胞中的 Wnt/β-连环蛋白信号通路来促进细胞增殖和软骨基质分泌[48]。就PRP-Exos包含的非编码RNA而言,目前仅XU等[65]研究进行了miR-141-3p对髓核细胞的保护作用探究。PRP-Exos通过下调 Keap1,导致 Nrf2 从 Keap1-Nrf2 复合物中释放,进一步从细胞质转移到细胞核以实现其抗氧化生物学功能。XU等[65]进一步研究发现PRP-Exos对Nrf2 缺陷的髓核细胞没有生物学作用。此外,PRP-Exos中富含miR-141-3p,miR-141-3p可以靶向Keap1 mRNA的3’非翻译区降解,导致Nrf2易位。"
| [1] VOS T, ABAJOBIR AA, ABATE KH, et al. Global, regional, and national incidence, prevalence, and years lived with disability for 328 diseases and injuries for 195 countries, 1990–2016: a systematic analysis for the Global Burden of Disease Study 2016. Lancet. 2017;390(10100):1211-1259. [2] HUNTER DJ, Bierma-Zeinstra S. Osteoarthritis. Lancet. 2019;393(10182): 1745-1759. [3] SAFIRI S, KOLAHI AA, SMITH E, et al. Global, regional and national burden of osteoarthritis 1990-2017: a systematic analysis of the Global Burden of Disease Study 2017. Ann Rheum Dis. 2020;79(6):819-828. [4] KOLASINSKI SL, NEOGI T, HOCHBERG MC, et al. 2019 American College of Rheumatology/Arthritis Foundation Guideline for the Management of Osteoarthritis of the Hand, Hip, and Knee. Arthritis Care Res (Hoboken). 2020;72(2):149-162. [5] MUSUMECI G, AIELLO FC, SZYCHLINSKA MA, et al. Osteoarthritis in the XXIst century: risk factors and behaviours that influence disease onset and progression. Int J Mol Sci. 2015;16(3):6093-6112. [6] 向小娜,余曦,刘岩,等.富血小板血浆与脉冲电磁场联合应用治疗骨关节炎:理论与临床研究的进展[J].中国组织工程研究,2019, 23(30): 4868-4874. [7] DOBSON GP, LETSON HL, GRANT A, et al. Defining the osteoarthritis patient: back to the future. Osteoarthritis Cartilage. 2018;26(8):1003-1007. [8] MURPHY C, WITHROW J, HUNTER M, et al. Emerging role of extracellular vesicles in musculoskeletal diseases. Mol Aspects Med. 2018;60:123-128. [9] LI J, PEI M. Cell senescence: a challenge in cartilage engineering and regeneration. Tissue Eng Part B Rev. 2012;18(4):270-287. [10] LI Y, WU Y, JIANG K, et al. Mangiferin Prevents TBHP-Induced Apoptosis and ECM Degradation in Mouse Osteoarthritic Chondrocytes via Restoring Autophagy and Ameliorates Murine Osteoarthritis. Oxid Med Cell Longev. 2019;2019:8783197. [11] GUILAK F, NIMS RJ, DICKS A, et al. Osteoarthritis as a disease of the cartilage pericellular matrix. Matrix Biol. 2018;71-72:40-50. [12] TU J, HUANG W, ZHANG W, et al. TWIST1-MicroRNA-10a-MAP3K7 Axis Ameliorates Synovitis of Osteoarthritis in Fibroblast-like Synoviocytes. Mol Ther Nucleic Acids. 2020;22:1107-1120. [13] RAHMATI M, NALESSO G, MOBASHERI A, et al. Aging and osteoarthritis: Central role of the extracellular matrix. Ageing Res Rev. 2017;40:20-30. [14] HUNZIKER EB, QUINN TM, HÄUSELMANN HJ. Quantitative structural organization of normal adult human articular cartilage. Osteoarthritis Cartilage. 2002;10(7):564-572. [15] GLYN-JONES S, PALMER AJ, AGRICOLA R, et al. Osteoarthritis. Lancet. 2015;386(9991):376-387. [16] DE VOS RJ, WEIR A, VAN SCHIE HT, et al. Platelet-rich plasma injection for chronic Achilles tendinopathy: a randomized controlled trial. JAMA. 2010;303(2):144-149. [17] FOSTER TE, PUSKAS BL, MANDELBAUM BR, et al. Platelet-rich plasma: from basic science to clinical applications. Am J Sports Med. 2009; 37(11):2259-2272. [18] DOHAN EHRENFEST DM, RASMUSSON L, ALBREKTSSON T. Classification of platelet concentrates: from pure platelet-rich plasma (P-PRP) to leucocyte- and platelet-rich fibrin (L-PRF). Trends Biotechnol. 2009;27(3):158-167. [19] LOUIS ML, MAGALON J, JOUVE E, et al. Growth Factors Levels Determine Efficacy of Platelets Rich Plasma Injection in Knee Osteoarthritis: A Randomized Double Blind Noninferiority Trial Compared With Viscosupplementation. Arthroscopy. 2018;34(5):1530-1540.e2. [20] KON E, BUDA R, FILARDO G, et al. Platelet-rich plasma: intra-articular knee injections produced favorable results on degenerative cartilage lesions. Knee Surg Sports Traumatol Arthrosc. 2010;18(4):472-479. [21] SAMPSON S, REED M, SILVERS H, et al. Injection of platelet-rich plasma in patients with primary and secondary knee osteoarthritis: a pilot study. Am J Phys Med Rehabil. 2010;89(12):961-969. [22] THÉRY C, WITWER KW, AIKAWA E, et al. Minimal information for studies of extracellular vesicles 2018 (MISEV2018): a position statement of the International Society for Extracellular Vesicles and update of the MISEV2014 guidelines. J Extracell Vesicles. 2018;7(1):1535750. [23] KALLURI R, LEBLEU VS. The biology, function, and biomedical applications of exosomes. Science. 2020;367(6478):eaau6977. [24] CHENG N, DU D, WANG X, et al. Recent Advances in Biosensors for Detecting Cancer-Derived Exosomes. Trends Biotechnol. 2019;37(11): 1236-1254. [25] LI D, WANG Y, JIN X, et al. NK cell-derived exosomes carry miR-207 and alleviate depression-like symptoms in mice. J Neuroinflammation. 2020;17(1):126. [26] CHEW JRJ, CHUAH SJ, TEO KYW, et al. Mesenchymal stem cell exosomes enhance periodontal ligament cell functions and promote periodontal regeneration. Acta Biomater. 2019;89:252-264. [27] MICHAEL JW, SCHLÜTER-BRUST KU, EYSEL P. The epidemiology, etiology, diagnosis, and treatment of osteoarthritis of the knee. Dtsch Arztebl Int. 2010;107(9):152-162. [28] ECKSTEIN F, LE GRAVERAND MP, CHARLES HC, et al. Clinical, radiographic, molecular and MRI-based predictors of cartilage loss in knee osteoarthritis. Ann Rheum Dis. 2011;70(7):1223-1230. [29] WILSON JF. To stop osteoarthritis, fixing cartilage may not be enough. Ann Intern Med. 2007;147(6):437-439. [30] ANDRIACCHI TP. Osteoarthritis: Probing knee OA as a system responding to a stimulus. Nat Rev Rheumatol. 2012;8(7):371-372. [31] BURR DB, GALLANT MA. Bone remodelling in osteoarthritis. Nat Rev Rheumatol. 2012;8(11):665-673. [32] ASO K, SHAHTAHERI SM, HILL R, et al. Associations of Symptomatic Knee Osteoarthritis With Histopathologic Features in Subchondral Bone. Arthritis Rheumatol. 2019;71(6):916-924. [33] YUAN C, PAN Z, ZHAO K, et al. Classification of four distinct osteoarthritis subtypes with a knee joint tissue transcriptome atlas. Bone Res. 2020;8(1):38. [34] DOYLE LM, WANG MZ. Overview of Extracellular Vesicles, Their Origin, Composition, Purpose, and Methods for Exosome Isolation and Analysis. Cells. 2019;8(7):727. [35] CAO Q, GUO Z, YAN Y, et al. Exosomal long noncoding RNAs in aging and age-related diseases. IUBMB Life. 2019;71(12):1846-1856. [36] WHITESIDE TL. Exosome and mesenchymal stem cell cross-talk in the tumor microenvironment. Semin Immunol. 2018;35:69-79. [37] LIU Y, LIN L, ZOU R, et al. MSC-derived exosomes promote proliferation and inhibit apoptosis of chondrocytes via lncRNA-KLF3-AS1/miR-206/GIT1 axis in osteoarthritis. Cell Cycle. 2018;17(21-22):2411-2422. [38] HA DH, KIM HK, LEE J, et al. Mesenchymal Stem/Stromal Cell-Derived Exosomes for Immunomodulatory Therapeutics and Skin Regeneration. Cells. 2020;9(5):1157. [39] LIU J, REN L, LI S, et al. The biology, function, and applications of exosomes in cancer. Acta Pharm Sin B. 2021;11(9):2783-2797. [40] DOHAN EHRENFEST DM, RASMUSSON L, ALBREKTSSON T. Classification of platelet concentrates: from pure platelet-rich plasma (P-PRP) to leucocyte- and platelet-rich fibrin (L-PRF). Trends Biotechnol. 2009; 27(3):158-167. [41] IYER SR, SCHEIBER AL, YAROWSKY P, et al. Exosomes Isolated From Platelet-Rich Plasma and Mesenchymal Stem Cells Promote Recovery of Function After Muscle Injury. Am J Sports Med. 2020;48(9):2277-2286. [42] ZHANG W, DONG X, WANG T, et al. Exosomes derived from platelet-rich plasma mediate hyperglycemia-induced retinal endothelial injury via targeting the TLR4 signaling pathway. Exp Eye Res. 2019;189:107813. [43] CERVENAKOVA L, SAÁ P, YAKOVLEVA O, et al. Are prions transported by plasma exosomes? Transfus Apher Sci. 2016;55(1):70-83. [44] GUO SC, TAO SC, YIN WJ, et al. Exosomes derived from platelet-rich plasma promote the re-epithelization of chronic cutaneous wounds via activation of YAP in a diabetic rat model. Theranostics. 2017;7(1):81-96. [45] TORREGGIANI E, PERUT F, RONCUZZI L, et al. Exosomes: novel effectors of human platelet lysate activity. Eur Cell Mater. 2014;28:137-151. [46] SAÁ P, YAKOVLEVA O, DE CASTRO J, et al. First demonstration of transmissible spongiform encephalopathy-associated prion protein (PrPTSE) in extracellular vesicles from plasma of mice infected with mouse-adapted variant Creutzfeldt-Jakob disease by in vitro amplification. J Biol Chem. 2014;289(42):29247-29260. [47] YIN W, QI X, ZHANG Y, et al. Advantages of pure platelet-rich plasma compared with leukocyte- and platelet-rich plasma in promoting repair of bone defects. J Transl Med. 2016;14:73. [48] LIU X, WANG L, MA C, et al. Exosomes derived from platelet-rich plasma present a novel potential in alleviating knee osteoarthritis by promoting proliferation and inhibiting apoptosis of chondrocyte via Wnt/β-catenin signaling pathway. J Orthop Surg Res. 2019;14(1):470. [49] NILFOROUSHZADEH MA, AGHDAMI N, TAGHIABADI E. Effects of Adipose-Derived Stem Cells and Platelet-Rich Plasma Exosomes on The Inductivity of Hair Dermal Papilla Cells. Cell J. 2021;23(5):576-583. [50] XU Y, LIN Z, HE L, et al. Platelet-Rich Plasma-Derived Exosomal USP15 Promotes Cutaneous Wound Healing via Deubiquitinating EIF4A1. Oxid Med Cell Longev. 2021;2021:9674809. [51] ZHANG Y, WANG X, CHEN J, et al. Exosomes derived from platelet-rich plasma administration in site mediate cartilage protection in subtalar osteoarthritis. J Nanobiotechnology. 2022;20(1):56. [52] ZHANG W, JIANG H, KONG Y. Exosomes derived from platelet-rich plasma activate YAP and promote the fibrogenic activity of Müller cells via the PI3K/Akt pathway. Exp Eye Res. 2020;193:107973. [53] FLAMANT S, TAMARAT R. Extracellular Vesicles and Vascular Injury: New Insights for Radiation Exposure. Radiat Res. 2016;186(2):203-218. [54] TAO SC, YUAN T, RUI BY, et al. Exosomes derived from human platelet-rich plasma prevent apoptosis induced by glucocorticoid-associated endoplasmic reticulum stress in rat osteonecrosis of the femoral head via the Akt/Bad/Bcl-2 signal pathway. Theranostics. 2017;7(3):733-750. [55] LEBLANC P, ARELLANO-ANAYA ZE, BERNARD E, et al. Isolation of Exosomes and Microvesicles from Cell Culture Systems to Study Prion Transmission. Methods Mol Biol. 2017;1545:153-176. [56] XU X, LIANG Y, LI X, et al. Exosome-mediated delivery of kartogenin for chondrogenesis of synovial fluid-derived mesenchymal stem cells and cartilage regeneration. Biomaterials. 2021;269:120539. [57] MATHIEU M, NÉVO N, JOUVE M, et al. Specificities of exosome versus small ectosome secretion revealed by live intracellular tracking of CD63 and CD9. Nat Commun. 2021;12(1):4389. [58] OTAHAL A, KRAMER K, KUTEN-PELLA O, et al. Effects of Extracellular Vesicles from Blood-Derived Products on Osteoarthritic Chondrocytes within an Inflammation Model. Int J Mol Sci. 2021;22(13):7224. [59] RUI S, YUAN Y, DU C, et al. Comparison and Investigation of Exosomes Derived from Platelet-Rich Plasma Activated by Different Agonists. Cell Transplant. 2021;30:9636897211017833. [60] SAUMELL-ESNAOLA M, DELGADO D, GARCÍA DEL CAÑO G, et al. Isolation of Platelet-Derived Exosomes from Human Platelet-Rich Plasma: Biochemical and Morphological Characterization. Int J Mol Sci. 2022;23(5):2861. [61] SHAO S, PAN R, CHEN Y. Autologous Platelet-Rich Plasma for Diabetic Foot Ulcer. Trends Endocrinol Metab. 2020;31(12):885-890. [62] ETULAIN J, MENA HA, MEISS RP, et al. An optimised protocol for platelet-rich plasma preparation to improve its angiogenic and regenerative properties. Sci Rep. 2018;8(1):1513. [63] MELKI I, TESSANDIER N, ZUFFEREY A, et al. Platelet microvesicles in health and disease. Platelets. 2017;28(3):214-221. [64] WU J, PIAO Y, LIU Q, et al. Platelet-rich plasma-derived extracellular vesicles: A superior alternative in regenerative medicine? Cell Prolif. 2021;54(12):e13123. [65] XU J, XIE G, YANG W, et al. Platelet-rich plasma attenuates intervertebral disc degeneration via delivering miR-141-3p-containing exosomes. Cell Cycle. 2021;20(15):1487-1499. |
| [1] | Nong Fuxiang, Jiang Zhixiong, Li Yinghao, Xu Wencong, Shi Zhilan, Luo Hui, Zhang Qinglang, Zhong Shuang, Tang Meiwen. Bone cement augmented proximal femoral nail antirotation for type A3.3 intertrochanteric femoral fracturalysis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(在线): 1-10. |
| [2] | Li Xiaomin, Tian Xiangdong, Tan Yetong, Zhu Guangyu, Wang Rongtian, Wang Jian, Xue Zhipeng, Ma Sheng, Hu Yuanyi, Huang Ye, Ding Tiansong. Changes of lower limb force line and knee function after high tibial osteotomy in osteoporotic medial ventricular knee osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1325-1329. |
| [3] | Zheng Hongrui, Zhang Wenjie, Wang Yunhua, He Bin, Shen Yajun, Fan Lei. Femoral neck system combined with platelet-rich plasma in the treatment of femoral neck fracture [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1390-1395. |
| [4] | Pan Zhongjie, Qin Zhihong, Zheng Tiejun, Ding Xiaofei, Liao Shijie. Targeting of non-coding RNAs in the pathogenesis of the osteonecrosis of the femoral head [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1441-1447. |
| [5] | Cai Zhihao, Xie Zhaoyong. Femoral neck anteversion measurement assessment: how to establish a unified method and standard [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1448-1454. |
| [6] | Dang Yi, Du Chengyan, Yao Honglin, Yuan Nenghua, Cao Jin, Xiong Shan, Zhang Dingmei, Wang Xin. Hormonal osteonecrosis and oxidative stress [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1469-1476. |
| [7] | Wang Ji, Zhang Min, Yang Zhongya, Zhang Long. A review of physical activity intervention in type 2 diabetes mellitus with sarcopenia [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1272-1277. |
| [8] | Nie Chenchen, Su Kaiqi, Gao Jing, Fan Yongfu, Ruan Xiaodi, Yuan Jie, Duan Zhaoyuan, Feng Xiaodong. The regulatory role of circular RNAs in cerebral ischemia-reperfusion injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1286-1291. |
| [9] | Gao Yu, Han Jiahui, Ge Xin. Immunoinflammatory microenvironment after spinal cord ischemia-reperfusion injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1300-1305. |
| [10] | Huang Linke, Wei Linhua, Jiang Jie, Liu Qian, Chen Weiwei. Effects of estrogen combined with treadmill exercise on bone mass and articular cartilage in ovariectomized mice [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1166-1171. |
| [11] | Lian Shilin, Zhang Yan, Jiang Qiang, Zhang Hanshuo, Li Tusheng, Ding Yu. Interventional effects of whole blood and platelet-rich plasma with different preparation methods on nucleus pulposus cells [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1199-1204. |
| [12] | Xu Xingxing, Wen Chaoju, Meng Maohua, Wang Qinying, Chen Jingqiao, Dong Qiang. Carbon nanomaterials in oral implant [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1062-1070. |
| [13] | Li Cheng, Zheng Guoshuang, Kuai Xiandong, Yu Weiting. Alginate scaffold in articular cartilage repair [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1080-1088. |
| [14] | Chen Shisong, Liu Xiaohong, Xu Zhiyun. Current status and prospects of bioprosthetic heart valves [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1096-1102. |
| [15] | Lu Di, Zhang Cheng, Duan Rongquan, Liu Zongxiang. Osteoinductive properties of calcium phosphate ceramic bone repair materials [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1103-1109. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||