Chinese Journal of Tissue Engineering Research ›› 2023, Vol. 27 ›› Issue (31): 5078-5084.doi: 10.12307/2023.593
Research and advance in intervertebral disc annulus fibrosus repair
Liu Xin1, 2, Sun Tianze1, 2, Zhang Jing1, 2, Zhang Wentao1, 2, Li Zhonghai1, 2
- 1Department of Orthopedics, First Affiliated Hospital of Dalian Medical University, Dalian 116011, Liaoning Province, China; 2Key Laboratory of Molecular Mechanism for Repair and Remodeling of Orthopedic Diseases, Dalian 116011, Liaoning Province, China
-
Received:2022-09-22Accepted:2022-11-08Online:2023-11-08Published:2023-01-31 -
Contact:Li Zhonghai, MD, Professor, Chief physician, Department of Orthopedics, First Affiliated Hospital of Dalian Medical University, Dalian 116011, Liaoning Province, China; Key Laboratory of Molecular Mechanism for Repair and Remodeling of Orthopedic Diseases, Dalian 116011, Liaoning Province, China -
About author:Liu Xin, Master candidate, Department of Orthopedics, First Affiliated Hospital of Dalian Medical University, Dalian 116011, Liaoning Province, China; Key Laboratory of Molecular Mechanism for Repair and Remodeling of Orthopedic Diseases, Dalian 116011, Liaoning Province, China -
Supported by:Open Research Foundation of Liaoning Provincial Key Laboratory of Micro and Nanotechnology and Systems of Dalian University of Technology, No. 20210101 (to LZH); Natural Science Foundation of Liaoning (General Project), No. 2022-MS-322 (to LZH)
CLC Number:
Cite this article
Liu Xin, Sun Tianze, Zhang Jing, Zhang Wentao, Li Zhonghai. Research and advance in intervertebral disc annulus fibrosus repair[J]. Chinese Journal of Tissue Engineering Research, 2023, 27(31): 5078-5084.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
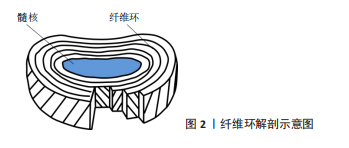
2.1 纤维环的解剖及生理病理特点 椎间盘是位于相邻椎体间重要的解剖结构,由外侧纤维环和内侧髓核以及椎体上下透明软骨板构成,纤维环是由水、Ⅰ型和Ⅱ型胶原纤维、蛋白聚糖和其他蛋白质组成,其内部是由15-25个同心圆的纤维薄片构成,每个薄片的胶原纤维(主要是Ⅰ型胶原纤维)彼此平行,与横轴大约呈30°夹角[8],见图2。纤维薄片层间存在的细胞外基质使纤维环具有非线性、各向异性及黏弹性等特点,同时纤维环层间基质中还存在少量弹性纤维,它可以连接相邻的纤维环薄片以维持椎间盘完整性[9]。纤维环可进一步分为内、外两个部分[10],外部区域主要由Ⅰ型胶原纤维组成,内部则主要由Ⅱ型胶原纤维组成,正因为两种胶原纤维生物学特性不同,使得纤维环可吸收来自髓核的压力,分散脊柱负荷,维持脊柱正常活动[11]。纤维环外部主要通过周围血管丛吸收营养物质,而内部营养物质主要从软骨下骨及软骨终板获得。"
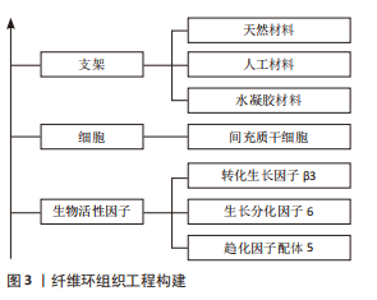
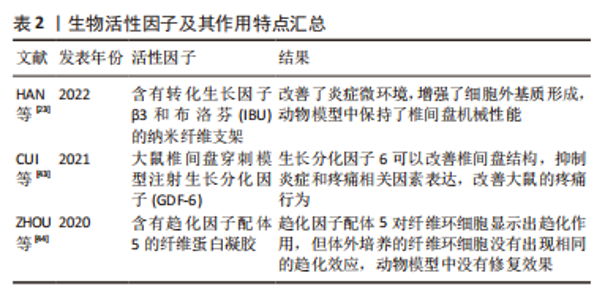
椎间盘退行性改变可导致髓核水合作用减弱、细胞外基质减少以及椎间盘高度下降,使纤维环承受更大的负荷,过度的脊柱负荷在纤维环组织中产生微结构损伤,并随着时间的推移造成纤维环撕裂或断裂。纤维环损伤也会降低椎间盘的整体稳定性,从而导致椎间盘突出,且未经治疗的纤维环损伤有着更高的再遗传率[12-13],纤维环损伤引起的炎症反应还会导致椎间盘内环境紊乱,影响椎间盘的生物力学性能,加速椎间盘退变。 2.2 纤维环组织工程 将细胞和支架植入纤维环缺损部位,通过植入细胞的再生使纤维环得到生物学修复从而恢复其正常的生理功能是组织工程技术的核心。纤维环组织工程的构建是基于支架、细胞和生物活性因子三部分(图3),其中支架可以一定程度上填补纤维环缺损,它为植入细胞的生长、分化以及产生细胞外基质提供了物理基础;各种细胞可以直接或间接修复纤维环;此外,生物活性因子参与纤维环组织工程的构建增加了基因的表达和细胞的增殖,进一步促进纤维环修复再生[14]。DU等[15]研究表明转化生长因子(transforming growth factor,TGF)和胶原-聚氨脂支架能够在体外和体内诱导功能细胞表达修复纤维环缺损,这种结合细胞、生物材料和生物活性因子的组织工程疗法具有巨大的促进纤维环组织修复与再生的潜力。"
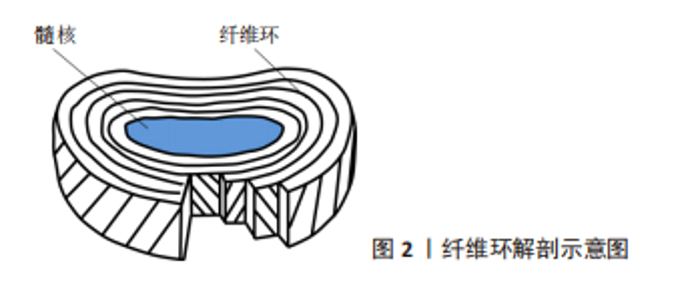
YU等[17]制备了一种含有岩藻衣聚糖的聚氨酯支架,研究表明该支架能缓解脂多糖引起的炎症和氧化应激反应,阻止纤维环细胞外基质降解,并在体内促进沉积以保持椎间盘的高度、含水量和机械性能。BOREM等[18-19]将糖胺聚糖加入到胶原蛋白角层状多层贴片支架中,评估其承受模拟冲击负荷和抵抗天然椎间盘组织突出的能力,结果显示此支架能够在施加超生理负荷压力时防止椎间盘组织疝出,该结论表明此法可能有促进纤维缺损修复的效果。SAGHARI等[20]用可吸收聚乙醇酸和透明质酸制备的支架植入绵羊椎间盘纤维环缺损处,将术后3个月的组织学切片与未处理组对比,结果显示支架植入组的纤维环组织修复显著增强,并伴有蛋白聚糖形成,表明此支架在绵羊腰椎间盘缺陷模型中修复了纤维环组织。CHRISTIANI等[21]使用3D打印制造的聚己内酯支柱多层支架复制了天然纤维环组织的层间排列角度,该支架轴向压缩和周向拉伸性能与天然纤维环组织相似,体外研究表明该人工支架对牛纤维环细胞代谢活性、形态和蛋白的表达产生了影响;共聚焦显微镜图像表明细胞能够在聚己内酯支架上附着和扩散,其纹理能够诱导细胞排列和增殖;免疫荧光分析显示Ⅰ型胶原、蛋白聚糖和纤维环特异性蛋白标记物在支架上一致沉积,显示了此支架作为纤维环再生模版的潜力。DE PIERI等[22]研究表明静电纺丝技术能够制备特定的3D支架,并能模拟细胞外基质微米至纳米级的纤维结构。此外,HAN等[23]用静电纺丝技术构建的纳米纤维支架也被证实具有高度仿生纤维环的结构。ZHU等[24]通过3D打印和静电纺丝技术构建的仿生人工椎间盘支架,在形态学和力学试验中表明,纤维环结构中纤维束呈同心定向排列,并且可以承受髓核变性过程中产生的张力。MA等[25]用静电纺丝技术开发了纳米亚尼支架(aligned nanoyarn scaffold,AYS)和纳米合成支架(aligned nanofibrous scaffold,AFS)以及三维多孔纳米纤维杂化支架(nanoyarn/three-dimensional porous nanofibrous hybrid scaffold,HS),将间充质干细胞接种于支架上测定细胞增殖率并评估细胞浸润和分化程度,形态学结果表明纳米亚尼支架具有更大的孔径、更高的孔隙率和更好的纤维排列;机械测试结果表明纳米合成支架和纳米亚尼支架的拉伸性能与天然纤维环组织相似;接种间充质干细胞7 d后发现三维多孔纳米纤维杂化支架和纳米亚尼支架上培养的细胞数量显著高于纳米合成支架。LIU等[26]使用静电纺丝技术合成了脱细胞环纤维基质-聚醚碳酸酯氨基甲酸酯混合支架,该支架弹性模量接近天然纤维环,在支架上培养的纤维环干细胞增加了Ⅰ型胶原纤维的基因表达,核磁共振成像、组织学染色及免疫组化评价表明脱细胞环纤维基质-聚醚碳酸酯氨基甲酸酯混合支架可以有效地修复纤维环缺损。尽管人工材料和天然材料制备支架各具优势,但各种材料的选择目前尚无最佳定论,在临床应用前还需要更多研究探索出最适合纤维环修复的材料。 作为支架的凝胶材料因其具有高强度的粘连性常用于封堵纤维环的缺损,但其力学强度有限,单独应用难以取得满意效果,目前多将凝胶材料与其他复合材料交联后使用。京尼平因其细胞毒性较低且具有较好的生物学稳定性,现被广泛用于关节软骨及椎间盘组织工程的研究。京尼平交联纤维蛋白作为针对纤维环修复的粘合剂,与纤维环有较好的匹配特性,可以输送细胞。PANEBIANCO等[27]在京尼平交联纤维蛋白水凝胶与氧化海藻酸盐微珠复合材料中递送纤维环细胞,结果显示纤维环细胞在材料中保持了高活力,并在释放时显示出纤维环细胞表型和基因表达;此外,富含细胞的此种材料具有生物学稳定性,在体外长期培养中促进了细胞外基质合成,并且将此种材料植入牛尾椎间盘缺损中可降低椎间盘疝出的风险。CRUZ等[28]将牛纤维环细胞包封在京尼平交联纤维蛋白凝胶中,发现纤维环细胞在此凝胶中可以保持高活力;此外,京尼平交联纤维蛋白凝胶能够匹配纤维环的剪切和压缩特性,并显著提高纤维环强度,增加细胞活力和糖胺聚糖的产生。FARUCHIGER等[29]将京尼平纤维蛋白水凝胶与工程丝支架相结合,在无负载、静态负载和复杂负载的条件下培养14 d后评估修复效果,结果显示3种负载条件下均未检测到椎间盘组织疝出,细胞外基质含量与未处理对照组椎间盘相当,表明此水凝胶与支架结合的方法对纤维环修复具有可行性。LONG等[30]将京尼平纤维蛋白水凝胶与聚碳酸三亚甲基酯支架结合来修复纤维环缺损,12个月的长期随访显示纤维环已有愈合,生物材料再吸收,并且没有明显的水凝胶相关并发症发生。PENG等[31]研究发现京尼平交联脱细胞纤维环水凝胶可诱导骨髓间充质干细胞特异性分化和椎间盘再生,同时此凝胶还有较好的生物相容性、良好的生物活性和较高的机械强度。DISTEFANO等[32]用纤维蛋白和聚乙二醇二丙烯酸酯组成的水凝胶与氧化甲基丙烯酸酯糖胺聚糖两种修复策略对比,结果显示水凝胶组对纤维环组织的黏附性更大,在牛体外模型中证明了水凝胶的机械性能能够降低疝出风险,表明水凝胶与椎间盘细胞外基质蛋白的结合可用于密封纤维环缺损。DISTEFANO等[33]研究表明利用水凝胶作为纤维环密封剂的组织工程仍处于动物模型阶段,因为这些生物材料经常受到纤维环组织整合不良的困扰,导致修复结果具有巨大差异,在评估了18种水凝胶制剂后发现纤维环组织共价结合的水凝胶在改善缺损的椎间盘的生物力学性质方面最有效,但没有一种能够恢复到完整的天然状态。 综上,尽管凝胶技术在实验室试验取得了成功,但目前凝胶交联剂种类繁多,缺乏最佳选择,且凝胶修复纤维环难以在日常活动中抵抗高应力[34],所以未来还需要在众多的材料中进行筛选并进行更多的动物研究和临床试验以验证其有效性和最佳的材料配比。 2.2.2 细胞 目前常用于椎间盘纤维环修复与再生治疗的细胞为人体间充质干细胞,包括骨髓间充质干细胞、脂肪来源的间充质干细胞、脐带间充质干细胞等[35],这些细胞含量丰富、易于获取,且具有出色的增殖、分化能力。LI等[36]在人体间充质干细胞衍生的外泌体(囊泡)减弱白细胞介素诱导的纤维环细胞损伤实验中表明,人体间充质干细胞可通过分泌外泌体抑制白细胞介素1β诱导的纤维环细胞炎症和凋亡。DISTEFANO等[37]用有氧和缺氧预处理人体间充质干细胞衍生外泌体,研究它们对纤维环细胞增殖、迁移和基因表达的影响,并用水凝胶包埋的聚乳酸-共乙醇酸微球持续外泌体的递送,结果显示缺氧组的纤维环细胞增殖和迁移反应更强,并且在体外肿瘤坏死因子α激发下维持了健康的纤维环细胞表型并减弱了分解代谢反应,该结论表明缺氧处理人体间充质干细胞外泌体可以通过刺激纤维环细胞增殖,促进细胞迁移和保护细胞免受退行性促炎微环境的侵害。人体椎间盘即为厌氧环境,故而厌氧条件下细胞培养后移植可能是未来纤维环修复再生的一大方向!XIAO等[38]用抗氧化剂超氧化物歧化酶2和过氧化氢酶对人脂肪来源间充质干细胞进行修饰,通过核磁共振、组织学及免疫组化等分析检测细胞增殖及基因表达水平,结果显示注射修饰后间充质干细胞的小鼠椎间盘高度指数、MRI指数和平均T2强度增加,蛋白聚糖和Ⅱ型胶原纤维表达增加,炎症反应减少,该结果表明椎间盘组织的病理状况已改善。THORPE等[39]直接将人体间充质干细胞移植到变性纤维环后,细胞外基质含量明显增多,该结论证明人体间充质干细胞在细胞外基质重塑中发挥着促进纤维环修复再生的作用。然而,SMITH等[40]、SAKAI等[41]临床前研究表明,由于纤维环结构完整性的破坏,在间充质干细胞注射后,物理维持适当间充质干细胞生存的生理环境对预防细胞凋亡很重要,单纯的细胞治疗不太可能在高度退化的椎间盘环境中再生纤维环细胞,因此中度椎间盘变性和支架植入患者可能从这类治疗中获益最多。 2.2.3 生物活性因子 组织工程常用的生物活性因子有TGFβ-1、TGF-β3、骨形态发生蛋白(bone morphogenetic protein,BMP)2、BMP-7、表皮生长因子、胰岛素样生长因子Ⅰ、碱性成纤维细胞生长因子和血小板衍生生长因子等[42]。生物活性因子及其作用特点汇总,见表2。"
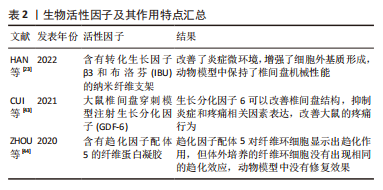
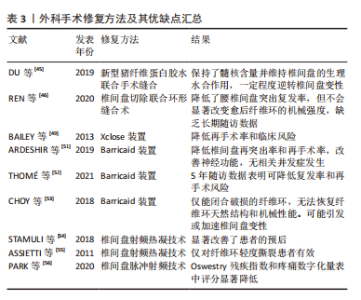
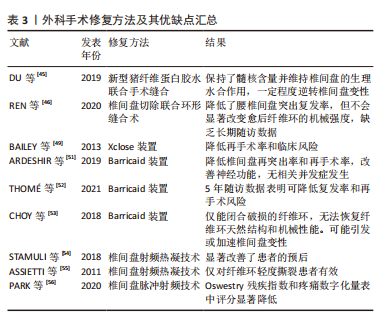
HAN等[23]采用静电纺丝技术构建含有TGFβ-3和布洛芬的纳米纤维支架,结果证实布洛芬的快速释放改善了炎症微环境,并且TGF-β3持续释放增强了新生细胞外基质形成,在两种动物模型中发现具有良好抗炎能力的仿生支架可增强细胞外基质形成并保持椎间盘的机械性能。CUI等[43]在构建大鼠椎间盘穿刺模型术后一两周通过椎间盘注射生长分化因子(growth differentiation factor,GDF-6)或磷酸盐溶液,核磁共振成像显示GDF-6抑制了纤维环的变性信号;组织学染色表明术后8周GDF-6对椎间盘中肿瘤坏死因子α和白细胞介素1β的表达有抑制作用;机械和热痛觉过敏试验表明GDF-6可显著改善大鼠椎间盘机械性能和热刺激性疼痛行为;综上,GDF-6可以改善椎间盘结构,抑制炎症和疼痛相关因素表达,改善大鼠的疼痛行为,但GDF-6的再生特性仍需进一步临床前研究和临床试验获取全面的数据以验证。ZHOU等[44]使用含有趋化因子配体5的纤维蛋白凝胶处理动态负载条件下培养的牛尾椎间盘并在绵羊颈椎间盘进行动物研究,14 d后收集牛尾椎间盘样本行组织学检查,14周后处死绵羊并在病理学检查前用核磁共振扫描椎间盘,结果显示趋化因子配体5以剂量依赖性的方式对纤维环细胞显示出趋化作用,但体外培养的纤维环细胞没有出现相同的趋化效应,体外释放研究表明趋化因子配体5从纤维蛋白凝胶到培养基中持续释放,然而其在绵羊颈椎间盘中并没有刺激纤维环细胞向缺损部位聚集,试点动物研究没有显示趋化因子配体5的任何修复效果,表明趋化因子配体5在体外对纤维环细胞具有趋化作用,但在离体或体内纤维蛋白凝胶内递送时没有再生作用。生物活性因子的种类繁多,不同的生物活性因子在组织工程应用的效果也不同,未来还需要更多的研究找寻对纤维环修复作用更强的生物活性因子。 组织工程是近年来兴起的对纤维环修复再生有较好应用前景的研究,在支架、细胞及生物活性因子方面的研究也取得了一定进展,但具体修复机制的阐明以及修复材料的选择在未来仍需探索,以更好地修复纤维环,提高脊柱手术的疗效。 2.3 外科手术修复 2.3.1 纤维环缝合术 将破损的纤维环直接缝合是最简单直接的修复方法,腰椎间盘突出摘除髓核后进行纤维环缝合已在临床上应用。DU等[45]研究表明新型猪纤维蛋白胶水可有效提升手术缝合线对纤维环缺损的闭合效果,保持髓核含量并维持椎间盘的生理水合作用,同时一定程度逆转受损椎间盘的变性。REN等[46]对135例行椎间盘切除联合环形缝合术或单纯椎间盘切除术患者进行观察,分别在术后3 d及3,6,12,24和36个月对患者进行评估,结果表明腰椎间盘切除联合环形缝合术与椎间盘高度恢复有更强的相关性,同时该法还降低了腰椎间盘突出复发率,但使用腰椎间盘切除联合环形缝合术并不会显著改变愈后纤维环的机械强度。然而现有的研究病例几乎都是短期随访,没有长期随访的数据[47],缝合手术本身的人为操作也可能引起纤维环的进一步损伤,加速椎间盘退变的进程[48]。由此可见,纤维环缝合技术虽能短期缓解患者的不适,但并未恢复纤维环结构的完整性,且长期效果未知,因此单纯的缝合技术已不再是当前研究的热点与重点。 2.3.2 纤维环成形器 早期的纤维环成形器是由Anulex Technologies Inc(美国)生产的Xclose和Incolse装置。Xclose是利用编织的聚脂带在纤维环缺损处作X形缝合,Inclose是一种网状结构,包裹纤维环破裂部分并防止再突出。BAILEY等[49]将750例腰椎间盘突出患者以2∶1的比例随机分配到Xclose装置系统修复组,术后2周、6个月、1年和2年的随访结果表明两组中患者的症状均得到了缓解,且差异不大,但Xclose组再次手术概率较低,且术后3,6个月的出血风险显著降低,临床相关风险降低45%,尽管没有统计学意义,但减少了再次手术的需要。然而,该成形器存在一些缺点,例如高昂的成本和复杂的技术,无法承受纤维环所承受的高强度机械负荷,无法有效地提高愈合率等,所以该成形器未能在临床中得到广泛应用[50]。另一种成形器Barricaid装置能够更好地修复破损的纤维环,防止髓核突出。ARDESHIR等[51]对50例患者行椎间盘切除术或Barricaid装置治疗,并进行了6,12,26和52周随访,结果表明Barricaid装置治疗使椎间盘再突出率和再手术率降低,患者的神经功能得到改善,并且没有严重的装置相关并发症。THOMé等[52]对554例患者行Barricaid装置治疗或单纯腰椎间盘切除术,术后5年随访结果表明Barricaid装置闭合纤维环缺损可降低复发率和再手术风险。但CHOY等[53]发现Barricaid装置仅能闭合破损的纤维环,无法恢复纤维环天然结构和机械性能,由于此装置锚定于相邻椎骨,易造成骨赘形成,甚至引发或加速椎间盘变性。纤维环成形器在防止髓核突出和恢复破损纤维环的拉伸强度等功能主要在体外实验被证实,缺乏人体试验及随访数据,所以纤维环成形器的应用并未得到广泛的认可。 2.3.3 射频热凝技术 椎间盘射频热凝技术(电热疗法)是一种微创或局限创伤的纤维环修复方法,它可使纤维环局部胶原纤维及组织发生变性,从而修复缺损,达到闭合纤维环的效果。STAMULI等[54]对85例接受椎间盘电热疗法的患者进行24个月的随访,评估了Oswestry残疾指数、视觉模拟量表、生活质量等,结果表明电热疗法显著改善了患者的预后。但ASSIETTI等[55]对50例行射频热凝治疗的患者进行2年随访,结果表明该治疗方法仅对纤维环轻度撕裂患者的症状实现了较长期的改善,对纤维环完全撕裂患者的治疗效果不佳。改进后的椎间盘脉冲射频技术操作更为便捷,创伤更小。PARK等[56]对45例接受7 min或15 min椎间盘脉冲射频的患者在治疗后2周和6个月随访评估,结果显示两组患者在Oswestry残疾指数和疼痛数字化量表中评分显著降低,且差异不大,结果表明无论椎间盘内脉冲射频应用的持续时间如何,其对治疗腰椎间盘源性腰痛均有效。目前,椎间盘射频脉冲的作用机制尚不确切,需要长期随访研究阐明其作用机制并确认其安全性和有效性[57-58]。 外科手术修复方法及其优缺点汇总,见表3。"

2.4 生物疗法 生物疗法的主要策略包括生物分子治疗、核酸疗法以及细胞内信号通路靶向治疗[59]。生物分子疗法即组织工程的生物活性因子的应用;核酸疗法即使用短DNA片段将基因递送到患者纤维环细胞中,操纵编码蛋白的合成和分泌以增强纤维环细胞的稳定性并抑制椎间盘退变;靶向治疗即使用信号分子改变细胞内信号转导通路以延缓或改变纤维环细胞变性的结局。REN等[60]观察重组腺病毒介导成骨蛋白1(adeno-associated virus (AAV)-mediated osteogenic protein-1,OP-1)和软骨细胞关键转录因子9(SRY-box transcription factor 9,SOX-9)双基因共转染在兔变性椎间盘体内的生物学效应,结果表明双基因转染显著改善了退行性椎间盘的高度,同时显著促进了蛋白聚糖和Ⅱ型胶原蛋白的表达。SUGIMOTO等[61]研究发现压电型机械敏感离子通道蛋白1可以通过调节BMP-2的表达来调节间充质干细胞对纤维环细胞增殖、分化和凋亡的影响。由于生物疗法长期疗效较差且稳定性不足,它们的临床应用还有待进一步研究。 2.5 其他修复办法——透明质酸的应用 SLOAN等[62]注射透明质酸以增强髓核联合光交联胶原蛋白贴片修复纤维环,这种方法可修复纤维缺损,恢复髓核水合作用,并且在损伤后6周可保持椎间盘天然的扭转能力和压缩特性。FULLER等[63]使用透明质酸寡糖刺激绵羊椎间盘细胞,通过免疫定位表明透明质酸寡糖能在椎间盘病变纤维环外侧区域进行主动重塑,但无法达到修复内部缺损的效果。RUSSO等[64]评估了透明质酸和血小板血浆组成的水凝胶作为人体间充质干细胞载体用于椎间盘再生的效果,组织学分析显示凝胶周围的椎间盘组织活力保存较好,水凝胶保留了组织的正常代谢。以上结果表明透明质酸可多样性地应用于纤维环的修复,关于透明质酸如何作用于纤维环修复再生的研究还需进一步探索,阐明透明质酸的作用机制以确定其修复效果。"

| [1] URBAN JPG, FAIRBANK JCT. Current perspectives on the role of biomechanical loading and genetics in development of disc degeneration and low back pain; a narrative review. J Biomech. 2020;102:109573. [2] KUPKA J, KOHLER A, EL BAGDADI K, et al. Adrenoceptor Expression during Intervertebral Disc Degeneration. Int J Mol Sci. 2020;21(6):2085. [3] RUIZ-FERNÁNDEZ C, FRANCISCO V, PINO J, et al. Molecular Relationships among Obesity, Inflammation and Intervertebral Disc Degeneration: Are Adipokines the Common Link? Int J Mol Sci. 2019; 20(8):2030. [4] AHMED AS, BERG S, ALKASS K, et al. NF-κB-Associated Pain-Related Neuropeptide Expression in Patients with Degenerative Disc Disease. Int J Mol Sci. 2019;20(3):658 [5] LONG RG, TORRE OM, HOM WW, et al. Design Requirements for Annulus Fibrosus Repair: Review of Forces, Displacements, and Material Properties of the Intervertebral Disk and a Summary of Candidate Hydrogels for Repair. J Biomech Eng. 2016;138(2):021007. [6] RAJPUROHIT R, RISBUD MV, DUCHEYNE P, et al. Phenotypic characteristics of the nucleus pulposus: expression of hypoxia inducing factor-1, glucose transporter-1 and MMP-2. Cell Tissue Res. 2002;308(3):401-407 [7] 王宇鹏,银和平,吴一民,等.缝合和粘合两种方法修复山羊腰椎间盘纤维环缺损[J].中国组织工程研究,2018,22(26):4156-4161. [8] MICHALEK AJ. A growth-based model for the prediction of fiber angle distribution in the intervertebral disc annulus fibrosus. Biomech Model Mechanobiol. 2019;18(5):1363-1369. [9] TAVAKOLI J, COSTI JJ. Ultrastructural organization of elastic fibres in the partition boundaries of the annulus fibrosus within the intervertebral disc. Acta Biomater. 2018;68:67-77. [10] GHEZELBASH F, SHIRAZI-ADL A, BAGHANI M, et al. On the modeling of human intervertebral disc annulus fibrosus: Elastic, permanent deformation and failure responses. J Biomech. 2020;102:109463. [11] OICHI T, TANIGUCHI Y, OSHIMA Y, et al. Pathomechanism of intervertebral disc degeneration. JOR Spine. 2020;3(1):e1076. [12] STRENGE KB, DIPAOLA CP, MILLER LE, et al. Multicenter study of lumbar discectomy with Barricaid annular closure device for prevention of lumbar disc reherniation in US patients: A historically controlled post-market study protocol. Medicine (Baltimore). 2019;98(35):e16953. [13] MILLER LE, MCGIRT MJ, GARFIN SR, et al. Association of Annular Defect Width After Lumbar Discectomy With Risk of Symptom Recurrence and Reoperation: Systematic Review and Meta-analysis of Comparative Studies. Spine (Phila Pa 1976). 2018;43(5):E308-E315. [14] 张维昊,徐宝山,马信龙,等.仿生可降解组织工程纤维环支架的制备与评估[J].中国组织工程研究,2020,24(4):524-531. [15] DU J, LONG RG, NAKAI T, et al. Functional cell phenotype induction with TGF-β1 and collagen-polyurethane scaffold for annulus fibrosus rupture repair. Eur Cell Mater. 2020;39:1-17. [16] 何冠宇,徐宝山,杜立龙,等.天然丝素蛋白构建仿生取向微通道纤维环支架[J].中国组织工程研究,2022,26(4):560-566. [17] YU Q, HAN F, YUAN Z, et al. Fucoidan-loaded nanofibrous scaffolds promote annulus fibrosus repair by ameliorating the inflammatory and oxidative microenvironments in degenerative intervertebral discs. Acta Biomater. 2022;148:73-89. [18] BOREM R, MADELINE A, Vela R Jr, et al. Multi-laminate annulus fibrosus repair scaffold with an interlamellar matrix enhances impact resistance, prevents herniation and assists in restoringspinal kinematics. J Mech Behav Biomed Mater. 2019;95:41-52. [19] BOREM R, MADELINE A, Theos C, et al. Angle-ply scaffold supports annulus fibrosus matrix expression and remodeling by mesenchymal stromal and annulus fibrosus cells. J Biomed Mater Res B Appl Biomater. 2022;110(5):1056-1068. [20] SAGHARI FARD MR, KRUEGER JP, STICH S, et al. A Biodegradable Polymeric Matrix for the Repair of Annulus Fibrosus Defects in Intervertebral Discs. Tissue Eng Regen Med. 2022 Jul 11. doi: 10.1007/s13770-022-00466-0. [21] CHRISTIANI TR, BARONCINI E, STANZIONE J, et al. In vitro evaluation of 3D printed polycaprolactone scaffolds with angle-ply architecture for annulus fibrosus tissue engineering. Regen Biomater. 2019;6(3):175-184. [22] DE PIERI A, BYERLEY AM, MUSUMECI CR, et al. Electrospinning and 3D bioprinting for intervertebral disc tissue engineering. JOR Spine. 2020;3(4):e1117. [23] HAN F, YU Q, CHU G, et al. Multifunctional Nanofibrous Scaffolds with Angle-Ply Microstructure and Co-Delivery Capacity Promote Partial Repair and Total Replacement of Intervertebral Disc. Adv Healthc Mater. 2022;11(19):e2200895. [24] ZHU M, TAN J, LIU L, et al. Construction of biomimetic artificial intervertebral disc scaffold via 3D printing and electrospinning. Mater Sci Eng C Mater Biol Appl. 2021;128:112310. [25] MA J, HE Y, LIU X, et al. A novel electrospun-aligned nanoyarn/three-dimensional porous nanofibrous hybrid scaffold for annulus fibrosus tissue engineering. Int J Nanomedicine. 2018;13:1553-1567. [26] LIU C, LI Y, ZHANG Y, et al. The experimental study of regeneration of annulus fibrosus using decellularized annulus fibrosus matrix/poly(ether carbonate urethane)urea-blended fibrous scaffolds with varying elastic moduli. J Biomed Mater Res A. 2022;110(5):991-1003. [27] PANEBIANCO CJ, RAO S, HOM WW, et al. Genipin-crosslinked fibrin seeded with oxidized alginate microbeads as a novel composite biomaterial strategy for intervertebral disc cell therapy. Biomaterials. 2022;287:121641. [28] CRUZ MA, HOM WW, DISTEFANO TJ, et al. Cell-Seeded Adhesive Biomaterial for Repair of Annulus Fibrosus Defects in Intervertebral Discs. Tissue Eng Part A. 2018;24(3-4):187-198. [29] FRAUCHIGER DA, MAY RD, BAKIRCI E, et al. Genipin-Enhanced Fibrin Hydrogel and Novel Silk for Intervertebral Disc Repair in a Loaded Bovine Organ Culture Model. J Funct Biomater. 2018;9(3):40. [30] LONG RG, FERGUSON SJ, BENNEKER LM, et al. Morphological and biomechanical effects of annulus fibrosus injury and repair in an ovine cervical model. JOR Spine. 2019;3(1):e1074. [31] PENG Y, HUANG D, LI J, et al. Genipin-crosslinked decellularized annulus fibrosus hydrogels induces tissue-specific differentiation of bone mesenchymal stem cells and intervertebral disc regeneration. J Tissue Eng Regen Med. 2020;14(3):497-509. [32] DISTEFANO TJ, SHMUKLER JO, DANIAS G, et al. Development of a two-part biomaterial adhesive strategy for annulus fibrosus repair and ex vivo evaluation of implant herniation risk. Biomaterials. 2020; 258:120309. [33] DISTEFANO TJ, SHMUKLER JO, DANIAS G, et al. The Functional Role of Interface Tissue Engineering in Annulus Fibrosus Repair: Bridging Mechanisms of Hydrogel Integration with Regenerative Outcomes. ACS Biomater Sci Eng. 2020;6(12):6556-6586. [34] MCGUIRE R, BOREM R, MERCURI J. The fabrication and characterization of a multi-laminate, angle-ply collagen patch for annulus fibrosus repair. J Tissue Eng Regen Med. 2017;11(12):3488-3493. [35] ZHANG W, SUN T, LI Y, et al. Application of stem cells in the repair of intervertebral disc degeneration. Stem Cell Res Ther. 2022;13(1):70. [36] LI ZQ, KONG L, LIU C, et al. Human Bone Marrow Mesenchymal Stem Cell-derived Exosomes Attenuate IL-1β-induced Annulus Fibrosus Cell Damage. Am J Med Sci. 2020;360(6):693-700. [37] DISTEFANO TJ, VASO K, PANEBIANCO CJ, et al. Hydrogel-Embedded Poly(Lactic-co-Glycolic Acid) Microspheres for the Delivery of hMSC-Derived Exosomes to Promote Bioactive Annulus Fibrosus Repair. Cartilage. 2022;13(3):19476035221113959. [38] XIAO L, XU SJ, LIU C, et al. Sod2 and catalase improve pathological conditions of intervertebral disc degeneration by modifying human adipose-derived mesenchymal stem cells. Life Sci. 2021;267:118929. [39] THORPE AA, BACH FC, TRYFONIDOU MA, et al. Leaping the hurdles in developing regenerative treatments for the intervertebral disc from preclinical to clinical. JOR Spine. 2018;1(3):e1027. [40] SMITH LJ, SILVERMAN L, SAKAI D, et al. Advancing cell therapies for intervertebral disc regeneration from the lab to the clinic: Recommendations of the ORS spine section. JOR Spine. 2018;1(4): e1036. [41] SAKAI D, SCHOL J. Cell therapy for intervertebral disc repair: Clinical perspective. J Orthop Translat. 2017;9:8-18. [42] CHU G, SHI C, WANG H, et al. Strategies for Annulus Fibrosus Regeneration: From Biological Therapies to Tissue Engineering. Front Bioeng Biotechnol. 2018,6:90. [43] CUI H, ZHANG J, LI Z, et al. Growth differentiation factor-6 attenuates inflammatory and pain-related factors and degenerated disc-induced pain behaviors in rat model. J Orthop Res. 2021; 39(5):959-970. [44] ZHOU Z, ZEITER S, SCHMID T, et al. Effect of the CCL5-Releasing Fibrin Gel for Intervertebral Disc Regeneration. Cartilage. 2020;11(2):169-180. [45] DU ZC, ZHU LX. A Heterologous Fibrin Glue Enhances the Closure Effect of Surgical Suture on the Repair of Annulus Fibrous Defect in a Sheep Model. Curr Med Sci. 2019;39(4):597-603. [46] REN C, QIN R, LI Y, et al. Microendoscopic Discectomy Combined with Annular Suture Versus Percutaneous Transforaminal Endoscopic Discectomy for Lumbar Disc Herniation: A Prospective Observational Study. Pain Physician. 2020;23(6):E713-E721. [47] TAVAKOLI J, DIWAN AD, TIPPER JL. Advanced Strategies for the Regeneration of Lumbar Disc Annulus Fibrosus. Int J Mol Sci. 2020; 21(14):4889. [48] PEREDO AP, GULLBRAND SE, SMITH HE, et al. Putting the Pieces in Place: Mobilizing Cellular Players to Improve Annulus Fibrosus Repair. Tissue Eng Part B Rev. 2021;27(4):295-312. [49] BAILEY A, ARAGHI A, BLUMENTHAL S, et al. Anular Repair Clinical Study Group. Prospective, multicenter, randomized, controlled study of anular repair in lumbar discectomy: two-year follow-up. Spine (Phila Pa 1976). 2013;38(14):1161-1169. [50] BOWLES RD, SETTON LA. Biomaterials for intervertebral disc regeneration and r epair. Biomaterials. 2017;129:54-67. [51] ARDESHIRI A, MILLER LE, SYNOWITZ M, et al. Surgical Experience and Complications in 50 Patients Treated with an Anular Closure Device Following Lumbar Discectomy. Orthop Surg. 2019;11(3):431-437. [52] THOMÉ C, KURŠUMOVIC A, KLASSEN PD, et al. Annular Closure RCT Study Group. Effectiveness of an Annular Closure Device to Prevent Recurrent Lumbar Disc Herniation: A Secondary Analysis With 5 Years of Follow-up. JAMA Netw Open. 2021;4(12):e2136809. [53] CHOY WJ, PHAN K, DIWAN AD, et al. Annular closure device for disc herniation: meta-analysis of clinical outcome and complications. BMC Musculoskelet Disord. 2018;19(1):290. [54] STAMULI E, KESORNSAK W, GREVITT MP, et al. A Cost-Effectiveness Analysis of Intradiscal Electrothermal Therapy Compared with Circumferential Lumbar Fusion. Pain Pract. 2018;18(4):515-522. [55] ASSIETTI R, MOROSI M, MIGLIACCIO G, et al. Treatment of discogenic low back pain with Intradiscal Electrothermal Therapy (IDET): 24 months follow-up in 50 consecutive patients. Acta Neurochir Suppl. 2011;108:103-105. [56] PARK CH, LEE SH, LEE PB. Intradiscal Pulsed Radiofrequency Application Duration Effect on Lumbar Discogenic Low Back Pain. Pain Physician. 2020;23(5):E535-E540. [57] FUKUI S, NITTA K, IWASHITA N, et al. Intradiscal pulsed radiofrequency for chronic lumbar discogenic low back pain: a one year prospective outcome study using discoblock for diagnosis. Pain Physician. 2013; 16(4):E435-442. [58] KIM HS, WU PH, JANG IT. Lumbar Degenerative Disease Part 1: Anatomy and Pathophysiology of Intervertebral Discogenic Pain and Radiofrequency Ablation of Basivertebral and Sinuvertebral Nerve Treatment for Chronic Discogenic Back Pain: A Prospective Case Series and Review of Literature. Int J Mol Sci. 2020;21(4):1483. [59] CHU G, SHI C, WANG H, et al. Strategies for Annulus Fibrosus Regeneration: From Biological Therapies to Tissue Engineering. Front Bioeng Biotechnol. 2018;6:90. [60] REN S, LIU Y, MA J, et al. Treatment of rabbit intervertebral disc degeneration with co-transfection by adeno-associated virus-mediated SOX9 and osteogenic protein-1 double genes in vivo. Int J Mol Med. 2013;32(5):1063-1068. [61] SUGIMOTO A, MIYAZAKI A, KAWARABAYASHI K, et al. Piezo type mechanosensitive ion channel component 1 functions as a regulator of the cell fate determination of mesenchymal stem cells. Sci Rep. 2017;7(1):17696. [62] SLOAN SR JR, WIPPLINGER C, KIRNAZ S, et al. Combined nucleus pulposus augmentation and annulus fibrosus repair prevents acute intervertebral disc degeneration after discectomy. Sci Transl Med. 2020;12(534):eaay2380. [63] FULLER ES, SHU C, SMITH MM, et al. Hyaluronan oligosaccharides stimulate matrix metalloproteinase and anabolic gene expression in vitro by intervertebral disc cells and annular repair in vivo. J Tissue Eng Regen Med. 2018;12(1):e216-e226. [64] RUSSO F, AMBROSIO L, PEROGLIO M, et al. A Hyaluronan and Platelet-Rich Plasma Hydrogel for Mesenchymal Stem Cell Delivery in the Intervertebral Disc: An Organ Culture Study. Int J Mol Sci. 2021;22(6): 2963. |
| [1] | Sun Kexin, Zeng Jinshi, Li Jia, Jiang Haiyue, Liu Xia. Mechanical stimulation enhances matrix formation of three-dimensional bioprinted cartilage constructs [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(在线): 1-7. |
| [2] | Cao Sheng, Kong Lingwei, Xu Kun, Sun Zhijie. Correlation of cervical sagittal force line parameters with degenerative segment and Pfirrmann classification in patients with cervical intervertebral disc degeneration [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1319-1324. |
| [3] | You Zhengqiu, Zhang Zhongzu, Wang Qunbo. Early symptomatic intervertebral disc pseudocysts after discectomy detected on MRI [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1403-1409. |
| [4] | Lian Shilin, Zhang Yan, Jiang Qiang, Zhang Hanshuo, Li Tusheng, Ding Yu. Interventional effects of whole blood and platelet-rich plasma with different preparation methods on nucleus pulposus cells [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1199-1204. |
| [5] | Xu Xingxing, Wen Chaoju, Meng Maohua, Wang Qinying, Chen Jingqiao, Dong Qiang. Carbon nanomaterials in oral implant [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1062-1070. |
| [6] | Yang Yitian, Wang Lu, Yao Wei, Zhao Bin. Application of the interaction between biological scaffolds and macrophages in bone regeneration [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1071-1079. |
| [7] | Li Cheng, Zheng Guoshuang, Kuai Xiandong, Yu Weiting. Alginate scaffold in articular cartilage repair [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1080-1088. |
| [8] | Shi Yehong, Wang Cheng, Chen Shijiu. Early thrombosis and prevention of small-diameter blood vessel prosthesis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1110-1116. |
| [9] | Tang Haotian, Liao Rongdong, Tian Jing. Application and design of piezoelectric materials for bone defect repair [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1117-1125. |
| [10] | Xu Yan, Li Ping, Lai Chunhua, Zhu Peijun, Yang Shuo, Xu Shulan. Piezoelectric materials for vascularized bone regeneration [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1126-1132. |
| [11] | Li Xinyue, Li Xiheng, Mao Tianjiao, Tang Liang, Li Jiang. Three-dimensional culture affects morphology, activity and osteogenic differentiation of human periodontal ligament stem cells [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 846-852. |
| [12] | Li Xiaoyin, Yang Xiaoqing, Chen Shulian, Li Zhengchao, Wang Ziqi, Song Zhen, Zhu Daren, Chen Xuyi. Collagen/silk fibroin scaffold combined with neural stem cells in the treatment of traumatic spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 890-896. |
| [13] | Yuan Bo, Xie Lide, Fu Xiumei. Schwann cell-derived exosomes promote the repair and regeneration of injured peripheral nerves [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 935-940. |
| [14] | Qin Yuxing, Ren Qiangui, Li Zilong, Quan Jiaxing, Shen Peifeng, Sun Tao, Wang Haoyu. Action mechanism and prospect of bone microvascular endothelial cells for treating femoral head necrosis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 955-961. |
| [15] | Xiong Bohan, Yu Yang, Lu Xiaojun, Wang Xu, Yang Tengyun, Zhang Yaozhang, Liao Xinyu, Zhou Xiaoxiang, He Lu, Li Yanlin. Research progress in promoting tendon to bone healing during anterior cruciate ligament reconstruction [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(5): 779-786. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||