Chinese Journal of Tissue Engineering Research ›› 2023, Vol. 27 ›› Issue (10): 1603-1609.doi: 10.12307/2023.344
Previous Articles Next Articles
Mesenchymal stem cells-derived exosomes in treatment of secondary lymphedema
Wang Xinxin1, Wang Jingxin2
- 1Xinxiang Medical University, Xinxiang 453000, Henan Province, China; 2Zhengzhou Central Hospital, Zhengzhou 450007, Henan Province, China
-
Received:2022-04-26Accepted:2022-06-15Online:2023-04-08Published:2022-09-09 -
Contact:Wang Jingxin, MD, Master’s supervisor, Zhengzhou Central Hospital, Zhengzhou 450007, Henan Province, China -
About author:Wang Xinxin, Master candidate, Xinxiang Medical University, Xinxiang 453000, Henan Province, China -
Supported by:Foundation for the Medical Science and Technology Research Project of Henan Province, No. 2018020778 (to WJX); the Key Research Development and Promotion (Science and Technology) Project in Henan Province, No. 222102310657 (to WJX); Graduate Science and Technology Innovation Support Program of Xinxiang Medical University, No. YJSCX202146Y (to WXX)
CLC Number:
Cite this article
Wang Xinxin, Wang Jingxin. Mesenchymal stem cells-derived exosomes in treatment of secondary lymphedema[J]. Chinese Journal of Tissue Engineering Research, 2023, 27(10): 1603-1609.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
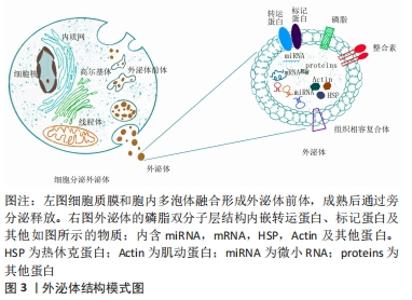
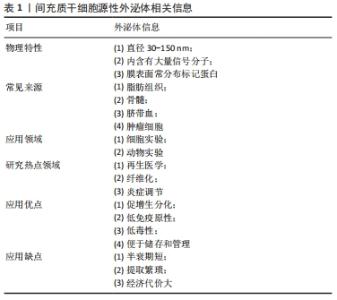
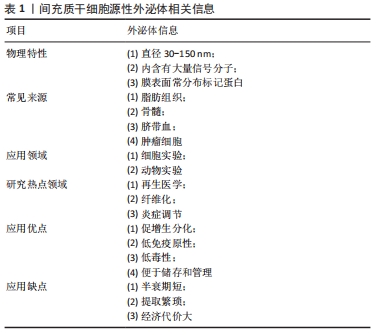
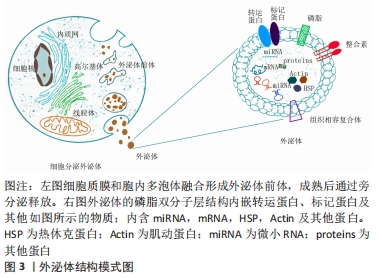
2.1 间充质干细胞源性外泌体在继发性淋巴水肿中研究发展史 1924年,俄罗斯形态学家ALEXANDER等发现间质中存在一种能分化为不同类型血细胞的前体细胞[9];1980年代,JOHNSTONE等[10]从网织红细胞成熟的过程中首次分离并提及“外泌体”概念。20世纪90年代CAPLAN[11]正式规范了间充质干细胞的命名,在21世纪,间充质干细胞源性外泌体成为最常见的选择之一。 细胞生理状态下或对刺激产生应答而分泌释放的物质称为外囊泡,根据直径大小和细胞来源分为外泌体、微囊泡、纳米颗粒、囊泡和凋亡小体,外泌体(Exosomes)是体积最小的外囊泡,由质膜和胞内多泡体融合而成,直径30-150 nm,起初被认为是细胞的“代谢产物”而没有引起重视,随后发现外泌体中含有microRNA、鞘磷脂及神经酰胺等,通过转运到受体细胞介导细胞内和细胞间的通讯,进而影响损伤和感染进程[12]。外泌体来源广泛,包括间充质干细胞、肿瘤细胞、血液、尿液、乳汁和脑脊液等[13-17],其中间充质干细胞源性外泌体应用最广。外泌体表面蛋白组成因来源而异,但Alix,Tsg101和CD9,CD63,CD81跨膜蛋白等是外泌体常见的组成部分,常作为表面标记物[18],外泌体结构模式图见图3。 "
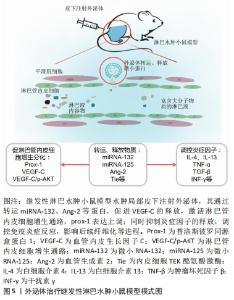
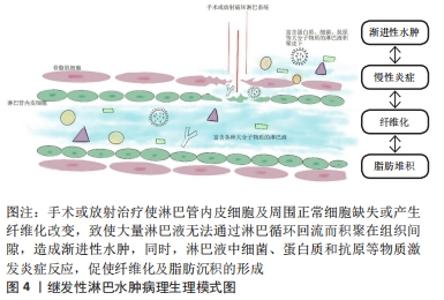
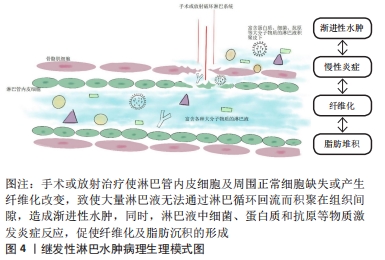
研究表明,巨噬细胞作为淋巴液中数量最多的炎症细胞,常联同其他炎症递质如树突状细胞等通过抗原提呈激活T淋巴细胞,影响淋巴管内皮细胞的生成、纤维化进程和免疫炎症反应[26]。在多种炎症因子共刺激下[27],成纤维细胞合成大量胶原纤维,联合细胞外基质的沉积反之促使集合淋巴管管壁发生纤维化改变,恶化淋巴管泵收缩功能,进一步加重淋巴水肿,淤滞的淋巴液造成更严重的炎症微环境,如此“负反馈”使结局恶化[28]。由此,渐进性水肿、脂肪沉积、皮肤纤维化和慢性免疫炎症反应是继发性淋巴水肿四大病理特征[29],以上病理过程相互作用相互影响,给基础研究和临床治疗带来巨大挑战。 2.3 间充质干细胞源性外泌体对于继发性淋巴水肿的作用 间充质干细胞外泌体在继发性淋巴水肿中已被证实能够通过转运microRNA及调控其他因子,促进淋巴管内皮细胞增生分化,达到重构淋巴管恢复淋巴引流的目的,同时参与调控慢性免疫炎症反应中重要炎症细胞或炎症因子的表达,进而影响脂肪沉积和纤维化进程,其模式图见图5。然而目前结论集中于前者减轻水肿方面,少数文献提及外泌体可使淋巴水肿动物模型皮肤纤维化程度减轻,但具体机制仍不清楚。总之,间充质干细胞外泌体对于缓解继发性淋巴水肿的作用已经十分明确,但部分机制尚不明确,未来值得深入探究。 "
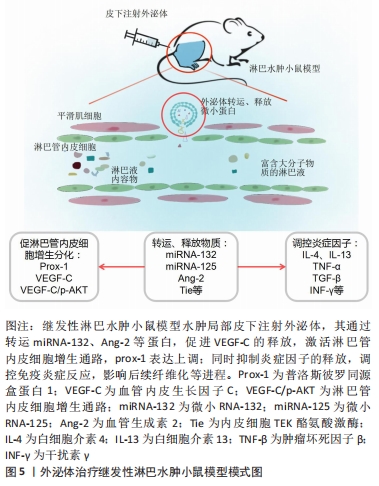
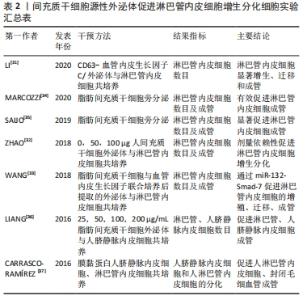
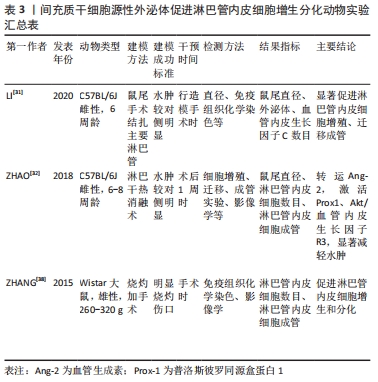
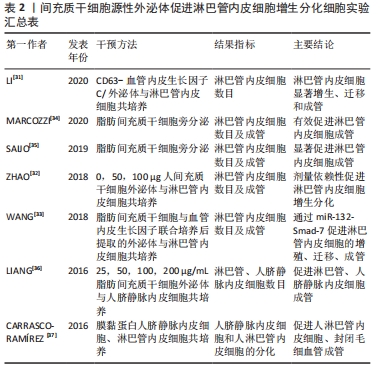
2.3.1 诱导淋巴管内皮细胞增生和分化 淋巴管内皮细胞胞质稀薄,细胞间连接疏松,缺乏紧密连接和黏附连接,相邻内皮细胞连接处为细胞间的重叠结构,又称瓣膜样连接,这种结构特点形成高通透性,允许大分子物质等进入管腔;淋巴管内皮细胞的非管腔面为锚丝,由弹性纤维和胶原纤维组成,两端分别连接内皮细胞与组织间隙中的纤维组织,以固定内皮细胞的空间位置,调节淋巴管与组织内压力变化,稳定淋巴液的正常生理回流[30]。然而手术和放疗破坏局部淋巴管及淋巴结,淋巴管内皮细胞的叠瓦状排列被打乱,致使大量淋巴液不能正常回流,积聚在皮下,引发后续“环式”病理变化。 因此,从结构上恢复受损淋巴管的功能是根治继发性淋巴水肿的首要选择,而间充质干细胞外泌体显然具备修复受损淋巴管的功能。LI等[31]体外构建过表达Exosome-CD63-血管内皮生长因子C融合蛋白(Exos-CD63-VEGFC),显著增加了淋巴管标记物淋巴管内皮透明质酸受体1的表达,促进淋巴管内皮细胞增殖、迁移和成管;在此之前,ZHAO等[32]发现人脐血间充质干细胞来源的外泌体中含有大量血管生成素2和内皮细胞TEK酪氨酸激酶,而血管生成素2是修复淋巴水肿的关键因子,能通过促进淋巴管内皮细胞的增殖、迁移以及小管形成来促进淋巴管再生。TEK酪氨酸激酶经外泌体转运进入细胞后,协同血管生成素2使淋巴管内皮中淋巴管内皮透明质酸受体1表达上调,激活血管内皮生长因子受体3/p-Akt的表达,帮助淋巴管重构。除此之外,WANG等[33]发现脂肪间充质干细胞源性外泌体中的miR-132直接转运到淋巴管内皮细胞,以某种机制扩大血管内皮生长因子C的促淋巴管生成效应,有效缓解了水肿体积。同样,其他相关研究也提示外泌体具有恢复淋巴引流的效应[31-38],见表2,3。 "

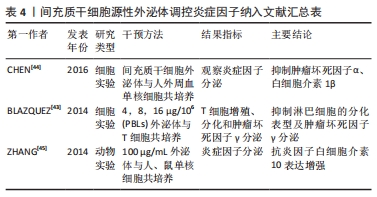
可以肯定的是外泌体的确对于淋巴管内皮细胞有增生作用,但相关机制通常仅明确下游某个蛋白,而不能梳理通顺整个通路;且剂量浓度相关研究为数不多,虽然上述LIANG等[36]设置25,50,100,200 μg/mL质量浓度的脂肪间充质干细胞外泌体探究促进淋巴管内皮细胞增生的最佳浓度,结果显示100 μg/mL效果最为显著,但与ZHAO等[32]的“剂量依赖性”结论相矛盾,原因可能因为实验开展的类型不同所致。因外泌体的低免疫原性,目前尚无外泌体致肿瘤复发的相关报道,后续扩大外泌体的浓度梯度,挖掘外泌体在继发性淋巴水肿中的潜能作用是未来研究重点之一,因此,未来还需开展更多更全面的探索。 2.3.2 潜在调节免疫炎症反应 慢性炎症反应贯穿继发性淋巴水肿始终,早期炎症症状不明显,但镜下观察可见大量炎症细胞及炎症因子弥漫在组织间隙,这为后期脂肪沉积及纤维化的发生提供了“温床”[39]。在新冠肺炎、过敏以及自身免疫疾病的炎症过程中,间充质干细胞外泌体均表现出良好的缓解效果[40-42]。然而缺少对继发性淋巴水肿慢性炎症的直接研究,可能因为慢性炎症反应主要原因起始于淤滞的淋巴液,因其含有大量炎症因子、细菌等物质引发机体保护性炎症反应,造成后续“负反馈”式的恶性结局。因此,大部分研究重点倾向于解决水肿问题。然而在相关领域,BLAZQUEZ等[43]通过体外实验发现人脂肪间充质干细胞来源的外泌体通过细胞-细胞接触直接介导或释放免疫抑制因子间接介导的方式,抑制CD4、CD8、T细胞的分化和增殖,同时抑制肿瘤坏死因子γ的释放,这是否可以说明外泌体在炎症环境中具有“稀释炎症因子”的能力这一问题有待未来继续研究探讨。纳入有关间充质干细胞源性外泌体调控炎症因子的文献归纳[43-45],见表4。 "

有研究表明肿瘤坏死因子α/核转录因子κB信号通路是间充质干细胞抑制T细胞增殖的关键机制[46],而干细胞发挥作用的主要方式依靠其旁分泌功能,从这个意义上说,外泌体是否也通过肿瘤坏死因子α/核转录因子κB信号通路对T细胞产生免疫调节作用尚未可知,明确外泌体对于继发性淋巴水肿慢性炎症的影响或许能为临床药物开发提供新的途径。 2.3.3 缓解皮肤纤维化程度 几乎所有的慢性炎症反应均可致促纤维化细胞因子的积累,并进一步增加促纤维化生长因子白细胞介素4、白细胞介素13以及转化生长因子β的表达,使皮下纤维细胞增多,纤维层加厚,增加胶原沉积,减少胶原分解[47],继发性淋巴水肿的慢性炎症环境也不例外。临床继发性淋巴水肿患者随着纤维化加重,可能诱发水肿肢体麻木、紧胀感甚至疼痛等感觉障碍[48]。目前间充质干细胞外泌体有效减轻心脏、肝脏、肺部疾病的纤维化结论已被证实[49-51],然而对于继发性淋巴水肿皮肤纤维化的研究尚无确切结论。只有1篇研究提及人脐带间充质干细胞来源的外泌体在继发性淋巴水肿小鼠水肿部位皮下注射后,水肿局部真皮层厚度明显变薄,真皮层内结构完整有序,且表皮和真皮层的界限更加完整清晰,而注射磷酸盐缓冲液的对照组真皮层结构紊乱,充满了组织液以及大量增生的纤维组织,皮肤厚度明显变宽[32],这提示人间充质干细胞外泌体具有抑制皮肤纤维化的作用。 转化生长因子β/Smad信号通路是纤维化进程中的经典通路,在继发性淋巴水肿中同样已被证实是纤维化发生的主要机制[52],WANG等[33]发现脂肪间充质干细胞源性外泌体携带的miR-132与Smad-7结合,调控转化生长因子β/Smad信号通路促进淋巴管内皮细胞增生,然而未提及对于纤维化的影响,但提示探索外泌体对继发性淋巴水肿纤维化的作用具有理论可行性。除此之外,其余大部分为间充质干细胞对于纤维化的缓解作用,例如有研究将小鼠脂肪间充质干细胞皮下注射到水肿部位[53],经Masson染色可见蓝染胶原纤维显著减少,Image J软件分析胶原纤维所占面积比下降约20%。综合以上,文章可推断探究间充质干细胞外泌体对于继发性淋巴水肿纤维化存在重要价值,明确间充质干细胞外泌体对于继发性淋巴水肿纤维化的影响机制可能为临床治疗提供新的思路和方法。 2.4 间充质干细胞源性外泌体在组织工程学中的应用 虽然外泌体应用前景广泛,但存在短半衰期及短保留率的问题,鉴于淋巴系统的结构特点和功能多样性,以及继发性淋巴水肿复杂的病理环境,组织工程学方法结合传统疗法带来了新的希望。有研究在体外构建了过表达CD63血管内皮生长因子C融合蛋白的外泌体,并将其嵌入海藻酸钠水凝胶中,通过小鼠模型评估其对淋巴水肿的影响,结果发现经CD63-血管内皮生长因子C融合蛋白外泌体修饰后,血管内皮生长因子C可以成功地传递到淋巴内皮细胞,损伤部位处外泌体序列释放,显著改善淋巴水肿[31]。 还有研究提出通过选择合适的生物材料,体外构建遵循生物力学的三维淋巴管网络,可以重建淋巴系统恢复淋巴引流,同时重建的淋巴网络可以传递信号分子参与调控免疫炎症反应[54]。研究发现透明质酸广泛存在于细胞外基质中,具有良好化学修饰性和低免疫原性的特点,且能影响淋巴管形态,是移植选择或淋巴管体外模型开发的优质选择[55]。ROH等[56]通过对继发性淋巴水肿小鼠局部注射透明质酸,观察到注射区水肿体积明显下降。除了透明质酸外,组织工程中还使用了多种其他生物材料,包括胶原蛋白、纤维蛋白、海藻酸盐和聚乙二醇二丙烯酸酯,都能在体外或体内生成淋巴血管[57]。HADAMITZKY等[58]将纳米纤维胶原蛋白支架植入猪淋巴水肿模型淋巴阻塞区域,并在支架中补充血管内皮生长因子C,植入3个月后观察到支架附近的淋巴集落显著增加,淋巴引流功能部分恢复。 由此可见,经过基因修饰或与特定细胞融合培养联合水凝胶的方法可以最大程度地发挥外泌体的优势,未来还有可能通过人为修改包埋外泌体的表面蛋白,靶向调控、精准高效释放外泌体,解决继发性淋巴水肿相关难题,也为未来组织工程学与外泌体联合应用于解决其他临床问题带来了希望。 "

| [1] BRIX B, SERY O, ONORATO A, et al. Biology of lymphedema. Biology (Basel). 2021;10(4):261. [2] VIEHOFF PB, HIDDING JT, HEERKENS YF, et al. Coding of meaningful concepts in lymphedema-specific questionnaires with the ICF. Disabil Rehabil. 2013;35(25):2105-2112. [3] SCHAVERIEN MV, CORONEOS CJ. Surgical treatment of lymphedema. Plast Reconstr Surg. 2019;144(3):738-758. [4] CHOLLET C, MALLOIZEL-DELAUNAY J, CABARROU B, et al. Liposuction-assisted brachioplasty in breast cancer-related lymphedema:Impact on volume reduction and quality of life. J Plast Reconstr Aesthet Surg. 2021;74(7):1633-1701. [5] BRANDÃO ML, SOARES HPDS, ANDRADE MDA, et al. Efficacy of complex decongestive therapy for lymphedema of the lower limbs: a systematic review. J Vasc Bras. 2020;19:e20190074. [6] JØRGENSEN MG, TOYSERKANI NM, JENSEN CH, et al. Adipose-derived regenerative cells and lipotransfer in alleviating breast cancer-related lymphedema: an open-label phase I trial with 4 years of follow-up. Stem Cells Transl Med. 2021;10(6):844-854. [7] SHUKLA L, YUAN Y, SHAYAN R, et al. Fat therapeutics:the clinical capacity of adipose-derived stem cells and exosomes for human disease and tissue regeneration. Front Pharmacol. 2020;11:158. [8] LI ZJ, YANG E, LI YZ, et al. Application and prospect of adipose stem cell transplantation in treating lymphedema. World J Stem Cells. 2020; 12(7):676-687. [9] DURAND. Characterization of the Hematopoietic Stem Cell Niche: Cellular and Molecular Analysis, in Stem Cells Handbook, Editor. Springer NY. 2013:211-221. [10] JOHNSTONE RM, ADAM M, HAMMOND JR, et al. Vesicle formation during reticulocyte maturation. Association of plasma membrane activities with released vesicles (exosomes). J Biol Chem. 1987;262(19): 9412-9420. [11] CAPLAN. Mesenchymal stem cells. J Orthop Res. 1991;9(5):641-650. [12] PEGTEL DM, GOULD SJ. Exosomes. Annu Rev Biochem. 2019;88:487-514. [13] ZHOU CF, MA J, HUANG L, et al. Cervical squamous cell carcinoma-secreted exosomal miR-221-3p promotes lymphangiogenesis and lymphatic metastasis by targeting VASH1. Oncogene. 2019;38(8):1256-1268. [14] BARANYAI T, HERCZEG K, ONÓDI Z, et al. Isolation of exosomes from blood plasma: qualitative and quantitative comparison of ultracentrifugation and size exclusion chromatography methods. PLoS One. 2015;10(12):e0145686. [15] ROYO F, ZUÑIGA-GARCIA P, SANCHEZ-MOSQUERA P, et al. Different EV enrichment methods suitable for clinical settings yield different subpopulations of urinary extracellular vesicles from human samples. J Extracell Vesicles. 2016;5:29497. [16] ZONNEVELD MI, BRISSON AR, VAN HERWIJNEN MJ, et al. Recovery of extracellular vesicles from human breast milk is influenced by sample collection and vesicle isolation procedures. J Extracell Vesicles. 2014. doi: 10.3402/jev.v3.24215. [17] HADE MD, SUIRE CN, SUO Z. Mesenchymal stem cell-derived exosomes: applications in regenerative medicine. Cells. 2021;10(8):1959. [18] LIN J, LI J, HUANG B, et al. Exosomes: novel biomarkers for clinical diagnosis. ScientificWorldJournal. 2015;2015:657086. [19] KALLURI R, LEBLEU VS. The biology, function, and biomedical applications of exosomes. Science. 2020;367(6478):eaau6977. [20] ZHAO G, LIU F, LIU Z, et al. MSC-derived exosomes attenuate cell death through suppressing AIF nucleus translocation and enhance cutaneous wound healing. Stem Cell Res Ther. 2020;11(1):174. [21] HA DH, KIM HK, LEE J, et al. Mesenchymal Stem/Stromal Cell-Derived Exosomes for Immunomodulatory Therapeutics and Skin Regeneration. Cells. 2020;9(5):1157. [22] WEI F, LI Z, CRAWFORD R, et al. Immunoregulatory role of exosomes derived from differentiating mesenchymal stromal cells on inflammation and osteogenesis. J Tissue Eng Regen Med. 2019;13(11): 1978-1991. [23] BALBI C, VASSALLI G. Exosomes: beyond stem cells for cardiac protection and repair. Stem Cells. 2020;38(11):1387-1399. [24] LOU G, CHEN Z, ZHENG M, et al. Mesenchymal stem cell-derived exosomes as a new therapeutic strategy for liver diseases. Exp Mol Med. 2017;49(6):e346. [25] GAO F, ZUO B, WANG Y, et al. Protective function of exosomes from adipose tissue-derived mesenchymal stem cells in acute kidney injury through SIRT1 pathway. Life Sci. 2020;255:117719. [26] GHANTA S, CUZZONE DA, TORRISI JS, et al. Regulation of inflammation and fibrosis by macrophages in lymphedema. Am J Physiol Heart Circ Physiol. 2015;308(9):H1065-H1077. [27] LY CL, KATARU RP, MEHRARA BJ. Inflammatory Manifestations of Lymphedema. Int J Mol Sci. 2017;18(1):171. [28] 罗雅馨,毕浩然,陈晓旭,等.间充质干细胞来源外泌体与再生医学:无细胞疗法临床应用的未来[J].中国组织工程研究,2020,24(19): 3055-3062. [29] SCHAVERIEN MV, ALDRICH MB. New and Emerging Treatments for Lymphedema. Semin Plast Surg. 2018;32(1):48-52. [30] BECKER J, SCHWOCH S, ZELENT C, et al. Transcriptome analysis of hypoxic lymphatic endothelial cells indicates their potential to contribute to extracellular matrix rearrangement. Cells. 2021;10(5): 1008. [31] LI B, YANG J, WANG R, et al. Delivery of vascular endothelial growth factor (VEGFC) via engineered exosomes improves lymphedema. Ann Transl Med. 2020;8(22):1498. [32] ZHAO M. Mesenchymal stem cell derived exosomes enhance lymphangiogenesis via exosomal transfer of Ang-2/Tie2. BioRxiv. 2018; 466987. [33] WANG X, WANG H, CAO J, et al. Exosomes from Adipose-Derived Stem Cells Promotes VEGF-C-Dependent Lymphangiogenesis by Regulating miRNA-132/TGF-beta Pathway. Cell Physiol Biochem. 2018;49(1):160-171. [34] MARCOZZI C, FRATTINI A, BORGESE M, et al. Paracrine effect of human adipose-derived stem cells on lymphatic endothelial cells. Regen Med. 2020;15(9):2085-2098. [35] SAIJO H, SUZUKI K, YOSHIMOTO H, et al. Paracrine Effects of Adipose-Derived Stem Cells Promote Lymphangiogenesis in Irradiated Lymphatic Endothelial Cells. Plast Reconstr Surg. 2019;143(6):1189e-1200e. [36] LIANG X, ZHANG L, WANG S, et al. Exosomes secreted by mesenchymal stem cells promote endothelial cell angiogenesis by transferring miR-125a. J Cell Sci. 2016;129(11):2182-2189. [37] CARRASCO-RAMÍREZ P, GREENING DW, ANDRÉS G, et al. Podoplanin is a component of extracellular vesicles that reprograms cell-derived exosomal proteins and modulates lymphatic vessel formation. Oncotarget. 2016;7(13):16070-16089. [38] ZHANG B, WU X, ZHANG X, et al. Human umbilical cord mesenchymal stem cell exosomes enhance angiogenesis through the Wnt4/beta-catenin pathway. Stem Cells Transl Med. 2015;4(5):513-222. [39] KATARU RP, BAIK JE, PARK HJ, et al. Regulation of Immune Function by the Lymphatic System in Lymphedema. Front Immunol. 2019;10:470. [40] PINKY, GUPTA S, KRISHNAKUMAR V, et al. Mesenchymal stem cell derived exosomes:a nano platform for therapeutics and drug delivery in combating COVID-19. Stem Cell Rev Rep. 2021;17(1):33-43. [41] REN J, LIU Y, YAO Y, et al. Intranasal delivery of MSC-derived exosomes attenuates allergic asthma via expanding IL-10 producing lung interstitial macrophages in mice. Int Immunopharmacol. 2021;91: 107288. [42] RIAZIFAR M, MOHAMMADI MR, PONE EJ, et al. Stem cell-derived exosomes as nanotherapeutics for autoimmune and neurodegenerative disorders. ACS Nano. 2019;13(6):6670-6688. [43] BLAZQUEZ R, SANCHEZ-MARGALLO FM, DE LA ROSA O, et al. Immunomodulatory potential of human adipose mesenchymal stem cells derived exosomes on in vitro stimulated T cells. Front Immunol. 2014;5:556. [44] CHEN W, HUANG Y, HAN J, et al. Immunomodulatory effects of mesenchymal stromal cells-derived exosome. Immunol Res. 2016;64(4): 831-840. [45] ZHANG B, YIN Y, LAI RC, et al. Mesenchymal stem cells secrete immunologically active exosomes. Stem Cells Dev. 2014;23(11):1233-1244. [46] DORRONSORO A, FERRIN I, SALCEDO JM, et al. Human mesenchymal stromal cells modulate T-cell responses through TNF-alpha-mediated activation of NF-kappaB. Eur J Immunol. 2014;44(2):480-488. [47] AZHAR SH, LIM HY, TAN BK, et al. The Unresolved Pathophysiology of Lymphedema. Front Physiol. 2020;11:137. [48] KATARU RP, WISER I, BAIK JE, et al. Fibrosis and secondary lymphedema:chicken or egg? Transl Res. 2019;209:68-76. [49] LI Q, JIN Y, YE X, et al. Bone marrow mesenchymal stem cell-derived exosomal microRNA-133a restrains myocardial fibrosis and epithelial-mesenchymal transition in viral myocarditis rats through suppressing MAML1. Nanoscale Res Lett. 2021;16(1):111. [50] 李洪超,王皙,李莉,等.人脂肪干细胞来源外泌体对四氯化碳诱导肝纤维化模型大鼠的治疗作用[J].中国组织工程研究,2020, 24(13):1996. [51] DINH PC, PAUDEL D, BROCHU H, et al. Inhalation of lung spheroid cell secretome and exosomes promotes lung repair in pulmonary fibrosis. Nat Commun. 2020;11(1):1064. [52] YOON SH, KIM KY, WANG Z, et al. EW-7197, a transforming growth factor-beta type I receptor kinase inhibitor, ameliorates acquired lymphedema in a mouse tail model. Lymphat Res Biol. 2020;18(5):433-438. [53] 周晨笑,苏万春,李娜,等.脂肪干细胞局部注射改善淋巴水肿的实验研究[J].首都医科大学学报,2020,41(6):869-875. [54] CHEN H, WANG L, ZENG X, et al. Exosomes, a New Star for Targeted Delivery. Front Cell Dev Biol. 2021;9:751079. [55] CAMPBELL KT, HADLEY DJ, KUKIS DL, et al. Alginate hydrogels allow for bioactive and sustained release of VEGF-C and VEGF-D for lymphangiogenic therapeutic applications. PLoS One. 2017;12(7): e0181484. [56] ROH K, CHO S, PARK JH, et al. Therapeutic effects of hyaluronidase on acquired lymphedema using a newly developed mouse limb model. Exp Biol Med (Maywood). 2017;242(6):584-592. [57] ALDERFER L, HALL E, HANJAYA-PUTRA D. Harnessing biomaterials for lymphatic system modulation. Acta Biomater. 2021;133:34-45. [58] HADAMITZKY C, ZAITSEVA TS, BAZALOVA-CARTER M, et al. Aligned nanofibrillar collagen scaffolds-Guiding lymphangiogenesis for treatment of acquired lymphedema. Biomaterials. 2016;102:259-267. |
| [1] | Sun Kexin, Zeng Jinshi, Li Jia, Jiang Haiyue, Liu Xia. Mechanical stimulation enhances matrix formation of three-dimensional bioprinted cartilage constructs [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(在线): 1-7. |
| [2] | Nong Fuxiang, Jiang Zhixiong, Li Yinghao, Xu Wencong, Shi Zhilan, Luo Hui, Zhang Qinglang, Zhong Shuang, Tang Meiwen. Bone cement augmented proximal femoral nail antirotation for type A3.3 intertrochanteric femoral fracturalysis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(在线): 1-10. |
| [3] | Pan Zhongjie, Qin Zhihong, Zheng Tiejun, Ding Xiaofei, Liao Shijie. Targeting of non-coding RNAs in the pathogenesis of the osteonecrosis of the femoral head [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1441-1447. |
| [4] | Cai Zhihao, Xie Zhaoyong. Femoral neck anteversion measurement assessment: how to establish a unified method and standard [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1448-1454. |
| [5] | Dang Yi, Du Chengyan, Yao Honglin, Yuan Nenghua, Cao Jin, Xiong Shan, Zhang Dingmei, Wang Xin. Hormonal osteonecrosis and oxidative stress [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1469-1476. |
| [6] | Wang Ji, Zhang Min, Yang Zhongya, Zhang Long. A review of physical activity intervention in type 2 diabetes mellitus with sarcopenia [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1272-1277. |
| [7] | Nie Chenchen, Su Kaiqi, Gao Jing, Fan Yongfu, Ruan Xiaodi, Yuan Jie, Duan Zhaoyuan, Feng Xiaodong. The regulatory role of circular RNAs in cerebral ischemia-reperfusion injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1286-1291. |
| [8] | Gao Yu, Han Jiahui, Ge Xin. Immunoinflammatory microenvironment after spinal cord ischemia-reperfusion injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1300-1305. |
| [9] | Liu Xiaolin, Mu Xinyue, Ma Ziyu, Liu Shutai, Wang Wenlong, Han Xiaoqian, Dong Zhiheng. Effect of hydrogel-loaded simvastatin microspheres on osteoblast proliferation and differentiation [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 998-1003. |
| [10] | Xu Xingxing, Wen Chaoju, Meng Maohua, Wang Qinying, Chen Jingqiao, Dong Qiang. Carbon nanomaterials in oral implant [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1062-1070. |
| [11] | Yang Yitian, Wang Lu, Yao Wei, Zhao Bin. Application of the interaction between biological scaffolds and macrophages in bone regeneration [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1071-1079. |
| [12] | Li Cheng, Zheng Guoshuang, Kuai Xiandong, Yu Weiting. Alginate scaffold in articular cartilage repair [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1080-1088. |
| [13] | Chen Shisong, Liu Xiaohong, Xu Zhiyun. Current status and prospects of bioprosthetic heart valves [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1096-1102. |
| [14] | Lu Di, Zhang Cheng, Duan Rongquan, Liu Zongxiang. Osteoinductive properties of calcium phosphate ceramic bone repair materials [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1103-1109. |
| [15] | Shi Yehong, Wang Cheng, Chen Shijiu. Early thrombosis and prevention of small-diameter blood vessel prosthesis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1110-1116. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||