Chinese Journal of Tissue Engineering Research ›› 2023, Vol. 27 ›› Issue (10): 1560-1566.doi: 10.12307/2023.302
Previous Articles Next Articles
Effects of high mobility group box 1 and ERK1/2 pathway on autophagy of human periodontal ligament cells under tensile stress
Zhang Min1, 2, Bai Shulin3, Li Shenghong1, 2, Fan Zhibo1, 2, Xie Yijia1, 2, Xu Xiaomei1, 2
- 1Department of Orthodontics, Affiliated Stomatology Hospital of Southwest Medical University, Luzhou 646000, Sichuan Province, China; 2Oral & Maxillofacial Reconstruction and Regeneration Laboratory, Southwest Medical University, Luzhou 646000, Sichuan Province, China; 3Department of Orthodontics, First People’s Hospital of Neijiang, Neijiang 641000, Sichuan Province, China
-
Received:2022-04-16Accepted:2022-06-02Online:2023-04-08Published:2022-09-08 -
Contact:Xu Xiaomei, MD, Associate professor, Master’s supervisor, Department of Orthodontics, Affiliated Stomatology Hospital of Southwest Medical University, Luzhou 646000, Sichuan Province, China; Oral & Maxillofacial Reconstruction and Regeneration Laboratory, Southwest Medical University, Luzhou 646000, Sichuan Province, China -
About author:Zhang Min, Master candidate, Physician, Department of Orthodontics, Affiliated Stomatology Hospital of Southwest Medical University, Luzhou 646000, Sichuan Province, China; Oral & Maxillofacial Reconstruction and Regeneration Laboratory, Southwest Medical University, Luzhou 646000, Sichuan Province, China -
Supported by:Scientific Research Project of Sichuan Provincial Department of Science and Technology, No. 2021YJ0151 (to XXM); Scientific Research Project of Southwest Medical University, No. 2021LZMS019 (to XXM)
CLC Number:
Cite this article
Zhang Min, Bai Shulin, Li Shenghong, Fan Zhibo, Xie Yijia, Xu Xiaomei. Effects of high mobility group box 1 and ERK1/2 pathway on autophagy of human periodontal ligament cells under tensile stress[J]. Chinese Journal of Tissue Engineering Research, 2023, 27(10): 1560-1566.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
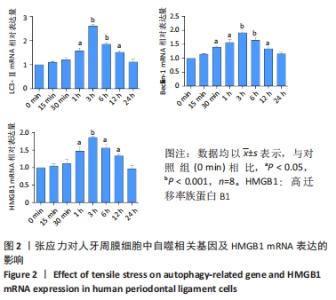
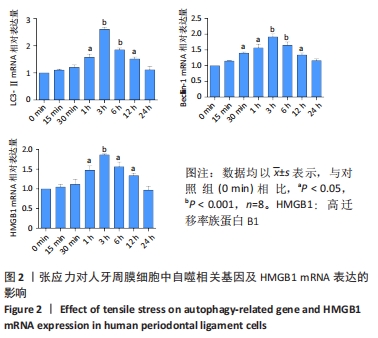
2.1 适宜强度的张应力可诱导人牙周膜细胞内自噬的发生和上调HMGB1的表达 第3-5代人牙周膜细胞施加不同时间的周期性牵张力后,采用RT-qPCR检测LC3-Ⅱ、Beclin-1、HMGB1的表达,结果可见随着张应力加载时间的延长,LC3-Ⅱ和Beclin-1及HMGB1 mRNA表达均呈现先增加后降低的趋势。LC3-Ⅱ、Beclin-1表达量在3 h时达到高峰(P < 0.001),随后开始下降,加力24 h其表达量下降至与对照组相比无明显差异(P > 0.05)。HMGB1 mRNA表达在张应力加载1 h后开始增加(P < 0.05),于3 h后达到高峰(P < 0.001),随后开始下降,同样加力24 h其表达量与对照组相比无差异,见图2。"
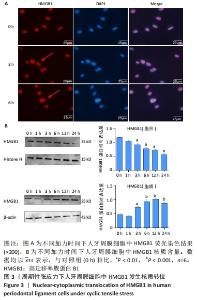
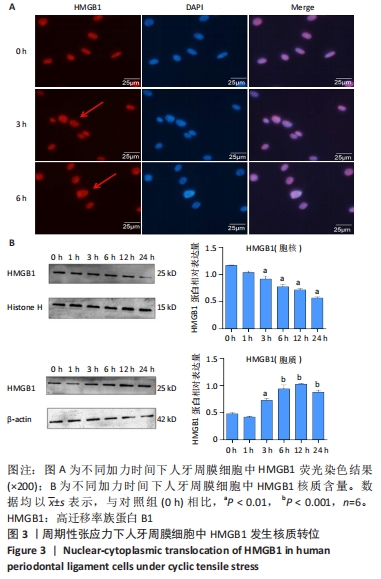
2.2 周期性张应力下人牙周膜细胞中HMGB1发生核质转位 张应力作用不同时间进行HMGB1免疫荧光染色,在高倍镜(×200)下观察可见未加载张应力时HMGB1主要位于细胞核内,胞质未见分布;张应力加载3 h后,胞核、胞质中均可见红染的HMGB1;张应力加载6 h后,胞质中HMGB1分布进一步增加,见图3A。 为进一步确认人牙周膜细胞中HMGB1定位随张应力加载时间的关系,在0,1,3,6,12,24 h张应力加载完成后分别提取胞核胞浆蛋白,采用Western blot分别定量检测胞核胞浆中HMGB1的表达,结果可见:在胞核中,加力3 h时HMGB1表达随张应力加载逐渐减少(P < 0.01),加力6 h时胞核中HMGB1表达进一步减少(P < 0.01),但12 h与6 h时相比未有明显减少(P > 0.05),而后加力24 h HMGB1表达进一步减少(P < 0.001);在胞浆中,HMGB1表达呈现逐渐增加的趋势,加力3 h时HMGB1表达开始增加(P < 0.01),12 h和24 h胞浆中HMGB1表达虽与对照组相比有明显增加(P < 0.001),但与6 h时相比未有明显增加,见图3B。从蛋白层面上证实了随张应力加载HMGB1出现了由胞核到胞质的转位。 "
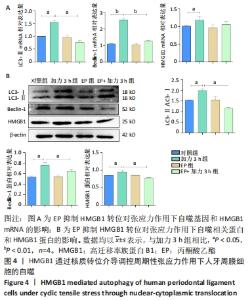
2.3 HMGB1通过核质转位参与周期性张应力作用下人牙周膜细胞的自噬诱导 为验证HMGB1通过核质转位参与调控自噬,使用7.5 mmol/L丙酮酸乙酯(HMGB1胞质转位抑制剂)预处理细胞24 h后加力3 h,通过RT-qPCR,Western blot观察加力状态下自噬变化情况。 RT-qPCR和Western blot结果显示:丙酮酸乙酯组LC3-Ⅱ、Beclin-1和HMGB1表达与对照组相比均无明显差异,LC3-Ⅱ和Beclin-1在其他各组表达趋势类似。结果显示:与对照组相比,加力3 h时LC3-Ⅱ、 Beclin-1表达增加(P < 0.05)。丙酮酸乙酯+加力3 h组LC3-Ⅱ和Beclin-1表达较加力3 h组明显减少(P < 0.05),可见抑制HMGB1转位可下调张应力引起的自噬。同时,作者还发现,HMGB1在加力3 h组表达有增加(P < 0.05),抑制转位后,丙酮酸乙酯+加力3 h组HMGB1 mRNA表达与加力3 h组相比均无明显差异(P > 0.05),但其蛋白的表达减少(P < 0.05),见图4。 "
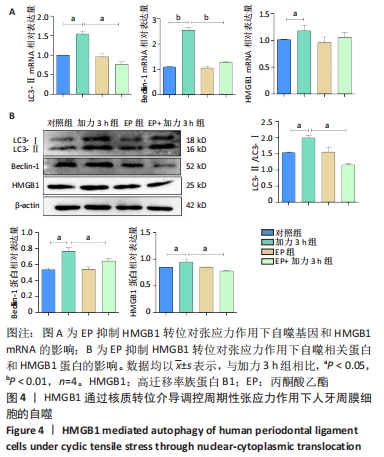
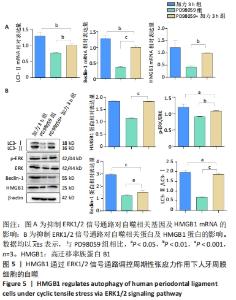
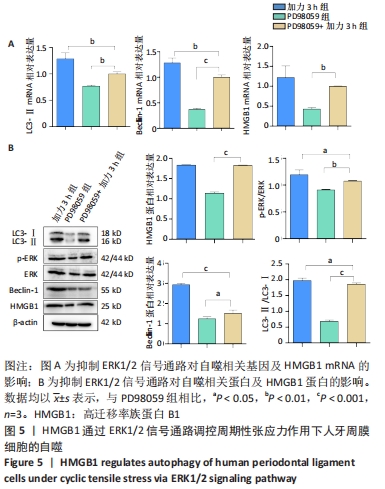
2.4 HMGB1通过ERK1/2通路参与调控张应力作用下人牙周膜细胞的自噬 采用10 μmol/L PD98059抑制ERK1/2信号通路,通过RT-qPCR,Western blot观察张应力下自噬变化情况。结果显示:与张应力加载3 h相比,PD98059+3 h组LC3-Ⅱ、Beclin-1 mRNA及蛋白的表达均减少;而单纯抑制ERK1/2通路,HMGB1的表达未见明显变化(P > 0.05),这表明抑制ERK1/2通路并不会影响加力状态下HMGB1的增加。此外,与PD98059组相比,加力3 h可逆转PD98059导致的自噬下调。结果证实了抑制ERK1/2信号通路后,加载张应力条件下HMGB1导致的自噬受到抑制,见图5。"
| [1] UHLIR R, MAYO V, LIN PH, et al. Biomechanical characterization of the periodontal ligament: Orthodontic tooth movement. Angle Orthod. 2017;87(2):183-192. [2] LI Y, JACOX LA, LITTLE SH, et al. Orthodontic tooth movement: The biology and clinical implications. Kaohsiung J Med Sci. 2018;34(4):207-214. [3] 尹圆圆,马华钰,李昕怡,等.小鼠正畸牙移动中牙周组织自噬相关基因表达的初步研究[J].国际口腔医学杂志,2020,47(6):627-634. [4] LIU Y, AI Y, SUN X, et al. Interleukin-20 Acts as a Promotor of Osteoclastogenesis and Orthodontic Tooth Movement. Stem Cells Int. 2021;2021:5539962. [5] DUTRA EH, NANDA R, YADAV S. Bone Response of Loaded Periodontal Ligament. Curr Osteoporos Rep. 2016;14(6):280-283. [6] LIN J, HUANG J, ZHANG Z, et al. Periodontal ligament cells under mechanical force regulate local immune homeostasis by modulating Th17/Treg cell differentiation. Clin Oral Investig. 2022;26(4):3747-3764. [7] JIN Y, DING L, DING Z, et al. Tensile force-induced PDGF-BB/PDGFRβ signals in periodontal ligament fibroblasts activate JAK2/STAT3 for orthodontic tooth movement. Sci Rep. 2020;10(1):11269. [8] CASSIDY LD, NARITA M. Autophagy at the intersection of aging, senescence, and cancer. Mol Oncol. 2022. doi: 10.1002/1878-0261. 13269. Online ahead of print. [9] 陈辛玲,王生兰.细胞自噬过程、通路、调控及其与肺动脉高压的多重相关性[J].中国组织工程研究,2021,25(2):311-316. [10] DUAN R, XIE H, LIU ZZ. The Role of Autophagy in Osteoarthritis. Front Cell Dev Biol. 2020;8:608388. [11] ZHANG Y, WANG H, LI F, et al. Inhibitory effects of Dulcitol on rat C6 glioma by regulating autophagy pathway. Nat Prod Res. 2020;34(10): 1437-1441. [12] ZHANG HL, ZHU YM, ZHOU XY. Coordination of Autophagy and Other Cellular Activities. Adv Exp Med Biol. 2019;1206:697-727. [13] 蒋玉坤,胡芝爱,关禹哲,等.应力诱导自噬的机械转导过程研究进展[J].四川大学学报(医学版),2021,52(6):929-935. [14] MEMMERT S, NOGUEIRA AVB, DAMANAKI A, et al. Regulation of the autophagy-marker Sequestosome 1 in periodontal cells and tissues by biomechanical loading. Regulation des Autophagie-Markers Sequestosom 1 durch biomechanische Kräfte in parodontalen Zellen und Geweben. J Orofac Orthop. 2020;81(1):10-21. [15] MEMMERT S, DAMANAKI A, WEYKOPF B, et al. Autophagy in periodontal ligament fibroblasts under biomechanical loading. Cell Tissue Res. 2019;378(3):499-511. [16] YANG H, WANG H, ANDERSSON U. Targeting Inflammation Driven by HMGB1. Front Immunol. 2020;11:484. [17] 周冬梅.高迁移率组蛋白1相关自噬在支气管哮喘中的作用研究[D].沈阳:中国医科大学,2018. [18] HATAYAMA K, STONESTREET BS. High mobility group box-1 protein as a therapeutic target in perinatal hypoxic-ischemic brain injury. Neural Regen Res. 2021;16(10):2006-2007. [19] FOGLIO E, PELLEGRINI L, GERMANI A, et al. HMGB1-mediated apoptosis and autophagy in ischemic heart diseases. Vasc Biol. 2019; 1(1):H89-H96. [20] QU L, CHEN C, CHEN Y, et al. High-Mobility Group Box 1 (HMGB1) and Autophagy in Acute Lung Injury (ALI): A Review. Med Sci Monit. 2019;25:1828-1837. [21] XU T, JIANG L, WANG Z. The progression of HMGB1-induced autophagy in cancer biology. Onco Targets Ther. 2018;12:365-377. [22] ZOU Y, XU L, LIN H. Stress overload-induced periodontal remodelling coupled with changes in high mobility group protein B1 during tooth movement: an in-vivo study. Eur J Oral Sci. 2019;127(5):396-407. [23] WOLF M, LOSSDÖRFER S, ABUDUWALI N, et al. Potential role of high mobility group box protein 1 and intermittent PTH (1-34) in periodontal tissue repair following orthodontic tooth movement in rats. Clin Oral Investig. 2013;17(3):989-997. [24] NIE F, ZHANG W, CUI Q, et al. Kaempferol promotes proliferation and osteogenic differentiation of periodontal ligament stem cells via Wnt/β-catenin signaling pathway. Life Sci. 2020;258:118143. [25] XIANG W, JIANG T, HAO X, et al. Primary cilia and autophagy interaction is involved in mechanical stress mediated cartilage development via ERK/mTOR axis. Life Sci. 2019;218:308-313. [26] XI X, ZHAO Y, LIU H, et al. Nrf2 activation is involved in osteogenic differentiation of periodontal ligament stem cells under cyclic mechanical stretch. Exp Cell Res. 2021;403(2):112598. [27] TSUGE A, NODA K, NAKAMURA Y. Early tissue reaction in the tension zone of PDL during orthodontic tooth movement. Arch Oral Biol. 2016; 65:17-25. [28] XI X, LI Z, LIU H, et al. Nrf2 Activation Is Involved in Cyclic Mechanical Stress-Stimulated Osteogenic Differentiation in Periodontal Ligament Stem Cells via PI3K/Akt Signaling and HO1-SOD2 Interaction. Front Cell Dev Biol. 2022;9:816000. [29] WANG Y, DU C, WAN W, et al. shRNA knockdown of integrin-linked kinase on hPDLCs migration, proliferation, and apoptosis under cyclic tensile stress. Oral Dis. 2020;26(8):1747-1754. [30] LI S, LI Q, ZHU Y, et al. GDF15 induced by compressive force contributes to osteoclast differentiation in human periodontal ligament cells. Exp Cell Res. 2020;387(1):111745. [31] WEI W, AN Y, AN Y, et al. Activation of autophagy in periodontal ligament mesenchymal stem cells promotes angiogenesis in periodontitis. J Periodontol. 2018;89(6):718-727. [32] DU L, LI Y, LIU W. Maresin 1 regulates autophagy and inflammation in human periodontal ligament cells through glycogen synthase kinase-3β/β-catenin pathway under inflammatory conditions. Arch Oral Biol. 2018;87:242-247. [33] RUSSELL RC, GUAN KL. The multifaceted role of autophagy in cancer. EMBO J. 2022 May 10:e110031. doi: 10.15252/embj.2021110031. Online ahead of print. [34] DIKIC I, ELAZAR Z. Mechanism and medical implications of mammalian autophagy. Nat Rev Mol Cell Biol. 2018;19(6):349-364. [35] WANG K. Autophagy and apoptosis in liver injury. Cell Cycle. 2015; 14(11):1631-1642. [36] HOSAKOTE YM, BRASIER AR, CASOLA A, et al. Respiratory Syncytial Virus Infection Triggers Epithelial HMGB1 Release as a Damage-Associated Molecular Pattern Promoting a Monocytic Inflammatory Response. J Virol. 2016;90(21):9618-9631. [37] YU H, ZHOU W, ZHONG Z, et al. High-mobility group box chromosomal protein-1 deletion alleviates osteoporosis in OVX rat model via suppressing the osteoclastogenesis and inflammation. J Orthop Surg Res. 2022;17(1):232. [38] ZHU K, ZHU X, LIU S, et al. Glycyrrhizin Attenuates Hypoxic-Ischemic Brain Damage by Inhibiting Ferroptosis and Neuroinflammation in Neonatal Rats via the HMGB1/GPX4 Pathway. Oxid Med Cell Longev. 2022;2022:8438528. [39] CHEN R, KANG R, TANG D. The mechanism of HMGB1 secretion and release. Exp Mol Med. 2022;54(2):91-102. [40] LI J, ZHOU W, MAO Q, et al. HMGB1 Promotes Resistance to Doxorubicin in Human Hepatocellular Carcinoma Cells by Inducing Autophagy via the AMPK/mTOR Signaling Pathway. Front Oncol. 2021; 11:739145. [41] MENG Q, PU L, QI M, et al. Laminar shear stress inhibits inflammation by activating autophagy in human aortic endothelial cells through HMGB1 nuclear translocation. Commun Biol. 2022;5(1):425. [42] ANDERSSON U, YANG H, HARRIS H. High-mobility group box 1 protein (HMGB1) operates as an alarmin outside as well as inside cells. Semin Immunol. 2018;38:40-48. [43] Zhao M, Zhang Y, Jiang Y, et al. YAP promotes autophagy and progression of gliomas via upregulating HMGB1. J Exp Clin Cancer Res. 2021;40(1):99. [44] LEI T, HUANG J, XIE F, et al. HMGB1-mediated autophagy promotes gefitinib resistance in human non-small cell lung cancer. Acta Biochim Biophys Sin (Shanghai). 2022;54(4). doi: 10.3724/abbs.2022023. Online ahead of print. [45] RAUCCI A, DI MAGGIO S, SCAVELLO F, et al. The Janus face of HMGB1 in heart disease: a necessary update. Cell Mol Life Sci. 2019;76(2):211-229. [46] TANG D, KANG R, ZEH HJ 3RD, et al. High-mobility group box 1, oxidative stress, and disease. Antioxid Redox Signal. 2011;14(7):1315-1335. [47] YIN H, YANG X, GU W, et al. HMGB1-mediated autophagy attenuates gemcitabine-induced apoptosis in bladder cancer cells involving JNK and ERK activation. Oncotarget. 2017;8(42):71642-71656. [48] ZHU L, HUANG G, SHENG J, et al. High-mobility group box 1 induces neuron autophagy in a rat spinal root avulsion model. Neuroscience. 2016;315:286-295. [49] MA C, CHEN H, ZHANG S, et al. Exosomal and extracellular HMGB1 have opposite effects on SASH1 expression in rat astrocytes and glioma C6 cells. Biochem Biophys Res Commun. 2019;518(2):325-330. [50] KIM YH, KWAK MS, LEE B, et al. Secretory autophagy machinery and vesicular trafficking are involved in HMGB1 secretion. Autophagy. 2021;17(9):2345-2362. [51] AOYAGI H, YAMASHIRO K, HIRATA-YOSHIHARA C, et al. HMGB1-induced inflammatory response promotes bone healing in murine tooth extraction socket. J Cell Biochem. 2018;119(7):5481-5490. |
| [1] | Nie Chenchen, Su Kaiqi, Gao Jing, Fan Yongfu, Ruan Xiaodi, Yuan Jie, Duan Zhaoyuan, Feng Xiaodong. The regulatory role of circular RNAs in cerebral ischemia-reperfusion injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1286-1291. |
| [2] | Wu Yujie, Wan Xiaofang, Wei Mianxing, Peng Shiyuan, Xu Xiaomei. Correlation between autophagy and the Hippo-YAP protein pathway in periodental ligament cells on the pressure side of a mouse model of orthodontic tooth movement [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(5): 683-689. |
| [3] | Li Long, Li Guangdi, Shi Hao, Deng Keqi. Circular RNA as a competing endogenous RNA is involved in the regulation of osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(5): 751-757. |
| [4] | Li Zhichao, Tan Guoqing, Su Hui, Xu Zhanwang, Xue Haipeng. Regulatory role of non-coding RNAs as potential therapeutic targets in spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(5): 758-764. |
| [5] | Wang Huanhuan, Wang Qing, Tang Peng, Zhang Rui, Min Hongwei. Effects of extracorporeal shock wave on the proliferation and autophagy of chondrocytes from osteoarthritis rats [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(2): 252-257. |
| [6] | Wang Yaxin, Lin Yican, Guo Yinuo, Zhang Bo, Ma Yuying, He Xiaoping. Methods, technologies and future of Chinese medicine modulation of cellular autophagy in the treatment of spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(13): 2116-2123. |
| [7] | Xiao Hao, Liu Jing, Zhou Jun. Research progress of pulsed electromagnetic field in the treatment of postmenopausal osteoporosis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1266-1271. |
| [8] | Cui Xing, Sun Xiaoqi, Zheng Wei, Ma Dexin. Huangqin Decoction regulates autophagy to intervene with intestinal acute graft-versus-host disease in mice [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1057-1062. |
| [9] | Xu Yixin, Wang Yixin, Li Yongming. Autophagy level of the mandible in nasal obstruction rats [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(35): 5633-5638. |
| [10] | Fan Jilin, Zhu Tingting, Tian Xiaoling, Liu Sijia, Su Jing, Zhang Shiliang. Effect and mechanism of non-coding RNA regulating autophagy in myocardial ischemia-reperfusion injury [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(35): 5716-5723. |
| [11] | Li Maoxue, Ding Yi, Guo Shujuan. Understanding of periodontal biomaterials based on the concept of bone homeostasis control and the effects on bone formation [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(34): 5562-5568. |
| [12] | Li Hongyuan, Yang Liu, Jin Xianhui. Relationship between hypoxia-inducible factors and bone homeostasis disorders [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(33): 5393-5399. |
| [13] | Li Jian, Guan Tianmin, Zhu Ye. Mechanics analysis of sacral lumbarization based on finite element method [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(33): 5249-5253. |
| [14] | Ma Hailong, Zhao Zhenqun, Liu Wanlin, Sun Jun. Microtubule-associated protein 1 light chain 3 is involved in the occurrence and development of steroid-induced avascular necrosis of the femoral head in a rabbit model [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(32): 5167-5172. |
| [15] | Song Na, Liu Guanjuan, Luo Shanshan, Huo Hua, Cheng Yuting, Hong Wei, Liao Jian. Autophagy and inflammation-induced alveolar bone metabolism [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(32): 5217-5222. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||