Chinese Journal of Tissue Engineering Research ›› 2022, Vol. 26 ›› Issue (32): 5217-5222.doi: 10.12307/2022.917
Previous Articles Next Articles
Autophagy and inflammation-induced alveolar bone metabolism
Song Na1, Liu Guanjuan1, Luo Shanshan1, Huo Hua1, Cheng Yuting1, Hong Wei2, Liao Jian1
- 1School of Stomatology/Stomatological Hospital of Guizhou Medical University, Guiyang 550004, Guizhou Province, China; 2Key Laboratory of Molecular Biology, Guizhou Medical University, Guiyang 550004, Guizhou Province, China
-
Received:2021-11-29Accepted:2022-01-22Online:2022-11-18Published:2022-05-14 -
Contact:Liao Jian, MD, Professor, Chief physician, Master’s and doctoral supervisor, School of Stomatology/Stomatological Hospital of Guizhou Medical University, Guiyang 550004, Guizhou Province, China -
About author:Song Na, Master candidate, School of Stomatology/Stomatological Hospital of Guizhou Medical University, Guiyang 550004, Guizhou Province, China -
Supported by:the National Natural Science Foundation of China, No. 82060207 (to LJ); Guizhou Provincial Health Commission Science and Technology Foundation, No. gzwkj2022-165 (to LJ)
CLC Number:
Cite this article
Song Na, Liu Guanjuan, Luo Shanshan, Huo Hua, Cheng Yuting, Hong Wei, Liao Jian. Autophagy and inflammation-induced alveolar bone metabolism[J]. Chinese Journal of Tissue Engineering Research, 2022, 26(32): 5217-5222.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
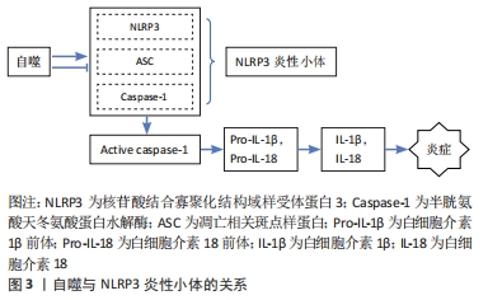
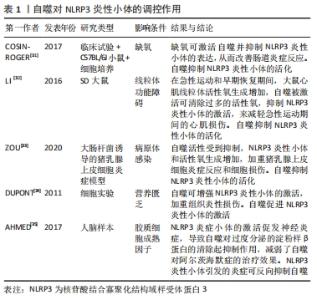
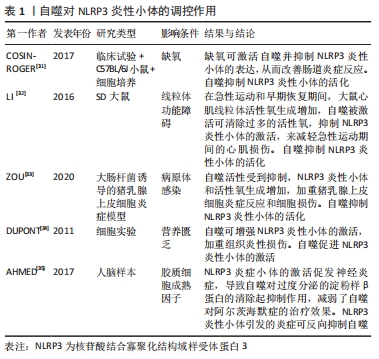
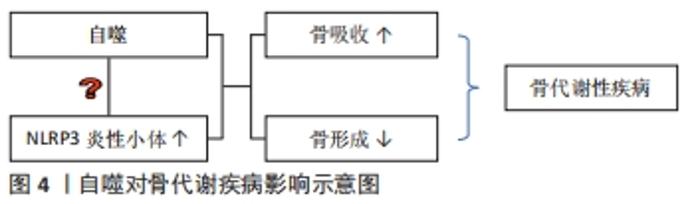
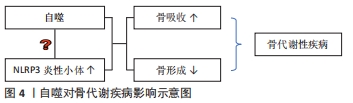
2.1 自噬对成骨细胞、破骨细胞的影响 2.1.1 自噬对成骨细胞的影响 自噬对维持骨组织稳态有着重要的作用[9],可直接调节成骨细胞和破骨细胞比例,从而维持骨组织的正常骨改建。成骨细胞是骨组织中骨形成的关键细胞,参与骨基质的合成、分泌和矿化[10],在牙槽骨改建过程中发挥重要作用。近年来,已有研究发现,自噬与成骨细胞密切相关,且参与成骨细胞的矿化过程及骨组织稳态[11]。但自噬活性水平降低直接扰乱骨重建过程中的动态平衡,且与多种骨性疾病的发生和发展有关,如骨质疏松[2]。NOLLET等[11]研究发现,自噬相关基因(BECN-1、ATG7、LC3)敲除导致成骨细胞在矿化过程中的矿化能力降低,进而骨形成不足。自噬活性能够促进成骨细胞分化,分泌骨基质,并调节骨改建,但是过度或长期的自噬活性会造成骨量流失及骨强度降低[12]。有相关体外细胞实验研究报道表明,低剂量的地塞米松类抗炎药物不仅可适度地激活细胞自噬,而且对成骨细胞也具有一定的免疫保护作用,而高剂量的地塞米松有可能会过度激活细胞自噬,加速成骨细胞的凋亡 [13-14]。 2.1.2 自噬对破骨细胞的影响 破骨细胞数量可以代表骨吸收活性 [15]。炎症的发生使得破骨细胞分化增加,这也是导致骨吸收的主要因素。炎症微环境给破骨细胞的形成和活化提供一个舒适的栖息地,造成过度炎症反应及牙槽骨吸收[16-17]。自噬相关基因在炎性环境中也显著表达,且破骨细胞的活性及骨吸收能力增强[18]。SUL等[19]研究者利用脂多糖刺激巨噬细胞建立炎症细胞模型,结果发现破骨细胞数量增多,且微管相关蛋白1轻链的表达也增多,而使用自噬特异性抑制剂3-甲基腺嘌呤可显著降低破骨细胞数量和骨吸收能力。自噬相关基因Atg5、Atg7、Atg4B和LC3对调节自噬功能有着独立的作用,与破骨细胞形成皱褶边缘、破骨分泌功能和破骨细胞骨吸收功能密切相关,且主要负责参与破骨细胞的形成及对骨质的吸收;而自噬相关基因缺陷会引起破骨细胞形态变化和功能障碍[20]。研究发现,Atg5,Atg7和 LC3基因缺失后,破骨细胞的组织蛋白酶K分泌能力和皱褶结构形成能力减弱[20-21]。在骨生长和骨重建过程中,溶酶体对骨质的更新起着重要作用。破骨细胞的溶酶体酶能释放到细胞外,分解和消除陈旧的骨基质,这是骨质更新的一个重要步骤。而自噬体与溶酶体融合形成自噬溶酶体,参与骨质的形成与更新[22]。因此,在病理情况下,可能因过度的自噬水平增加破骨细胞活性及骨吸收;而正常情况下,自噬参与破骨细胞的分化、成熟,以对骨质进行更新。 2.2 自噬与炎症反应之间的关系 NLRP3 属于 NOD 样受体(nucleotide-binding oligomerization domain-like receptors,NLRs)家族成员,与凋亡相关斑点样蛋白(apoptosis-associated speck-like protein containing CARD,ASC)和半胱氨酸的天冬氨酸蛋白水解酶1(cysteinyl aspartate specific proteinase1,caspase-1)组装成NLRP3炎症小体,且在炎症中发挥着重要的作用[23]。CHEN等[24]对NLRP3基因敲除小鼠及其野生型同窝出生的小鼠诱导牙周炎模型,结果发现NLRP3基因敲除小鼠破骨细胞前体数量、破骨细胞分化和牙槽骨丢失均明显较野生型小鼠减少,表明NLRP3可通过促进破骨细胞分化来调节牙周炎小鼠牙槽骨骨量丢失。此外,SAITOH等[25]首次在细胞实验上证明了自噬对炎症小体的激活有影响。NLRP3炎性小体可通过病原相关分子模式(pathogen associated molecular patterns,PAMPs)和损伤相关分子模式(damage associated molecular patterns,DAMPs)来激活,促使白细胞介素1β和白细胞介素18等促炎因子的活化、成熟和分泌。损伤相关分子模式在诱发炎性反应时,能够激活细胞内部自噬的产生,而自噬的产生可以抑制炎性反应[26]。研究表明,自噬可清除细胞内的损伤相关分子模式、NLRP3炎性小体成分和细胞因子等炎性小体激活剂来减少炎性小体的活化和炎症反应;自噬缺失或功能不全可能会直接导致过度的炎症反应及NLRP3炎性小体活化型疾病[27]。已有研究表明,自噬与NLRP3炎症小体之间的相互作用可以维持机体和组织内部的平衡[28-29]。 由于自噬与固有性和适应性免疫系统密切相关,NLRP3是机体内一种能够防御各类病原细菌和微生物发展的固有性和适应性免疫系统的主要组成部分。因此,自噬与NLRP3密切相关。自噬与NLRP3炎性小体之间的相互作用关系极其复杂,其不仅可以抑制NLRP3炎性小体,且在某些情况下,自噬还能促进NLRP3炎性小体,而NLRP3炎性小体也可以反过来影响自噬[30]。众多研究发现,在缺氧、线粒体功能障碍、病原体感染等条件下自噬对NLRP3炎性小体起抑制作用。 COSIN-ROGER等 [31]研究发现,缺氧时可通过激活自噬来抑制NLRP3和mTOR的结合,从而对肠道炎症起到改善作用。LI等[32]发现,在急性运动期间,由于线粒体功能障碍,活性氧增加,而线粒体活性氧的积累可激活自噬活性,清除过多活性氧生成,抑制NLRP3炎性小体的激活,减轻急性运动期间的心肌损伤。ZOU等 [33]研究者在体外建立大肠杆菌诱导的猪乳腺上皮细胞炎症模型,发现自噬活性受到抑制,NLRP3炎性小体和活性氧生成增加,加重猪乳腺上皮细胞炎症反应和细胞损伤。而在营养缺乏等条件下自噬对NLRP3炎性小体起促进作用。DUPONT等 [34]研究发现,细胞在饥饿条件下,自噬可增强NLRP3炎性小体的激活,促使caspase-1活化,释放白细胞介素1β、白细胞介素18,从而加重组织炎性损伤。此外,NLRP3炎性小体对自噬也存在影响。AHMED等 [35]在阿尔茨海默病中研究发现,NLRP3炎症小体的激活促发神经炎症,可能与胶质细胞成熟因子放大NLRP3炎性小体引起的炎症反应有关,进而导致自噬对过度分泌的淀粉样β蛋白的清除起抑制作用,减弱了自噬对阿尔茨海默症的治疗效果。因此,自噬对NLRP3 炎症小体具有正、负向调控作用,且某些条件的改变其所产生的调控作用也不同。自噬对NLRP3炎性小体的调控作用见图3,表1。 "

2.3.1 自噬对根尖周炎的影响 慢性根尖周炎在发病初期时,根管感染的病原菌和细菌产物诱导牙周膜细胞产生各种炎症递质[36]。而这些炎性递质在根尖周炎周围建立各种网络联系,从而促进根尖周围骨组织的吸收[37]。研究发现,在大鼠根尖周炎模型中,NLRP3、Caspase-1、白细胞介素1β的表达量显著增加,从而加重根尖区炎症反应及牙槽骨吸收[38]。NLRP3炎性小体的组装及caspase-1的活化会促使白细胞介素1β和白细胞介素18的成熟和分泌。而自噬可以解除已聚合的炎性小体结构,从而有助于抑制炎症反应[39]。PARK等[40] 研究发现,抑制自噬可显著增加白细胞介素1β的释放和NLRP3炎性小体的形成。目前,自噬被认为与大鼠根尖周病变的发病机制有关[41]。ZHU等[42]通过收集临床中的根尖囊肿及根尖肉芽标本进行检测分析,发现自噬相关标记物LC3在根尖周病变中显著表达。HUANG等[43]研究者对15例根尖囊肿和21例根尖肉芽肿及17例健康牙髓组织进行检测,结果发现在根尖周病变中白细胞介素1β水平显著高于正常牙髓组织;根尖周病变中缺氧相关蛋白(PAMPK和BNIP3、HIF-1α)和自噬相关蛋白(BECN1、ATG5-ATG12、LC3II/I)的表达显著增高。说明自噬可能在根尖病变的发生和维持中起潜在的致病作用。此外,马金宝等[44]利用脂多糖刺激牙髓细胞引起炎症反应,结果发现随着脂多糖刺激时间的延长,在炎性牙髓细胞中Ⅱ型LC3B表达增高,提示用脂多糖诱导的牙髓炎自噬活力增强,而48 h比24 h表达减弱,说明此时自噬趋于平稳并进入蛋白降解阶段,这可能是牙髓炎及根尖周炎中自噬被调动以抵抗机体受到的炎性刺激,自噬途径异常,导致牙髓炎、根尖周炎发生发展。自噬在应激、饥饿或缺氧条件下的能量供应以及生理细胞内稳态中起着关键作用[45]。但过度的炎症反应导致炎症性根尖囊肿和根尖肉芽肿中存在大量自噬,使自噬过度激活,周围骨组织功能受损。因而,还需进一步研究NLRP3炎症小体是否参与根尖周炎介导的自噬过程。自噬在根尖周病变中的实验研究及其结果汇总,见表2。"

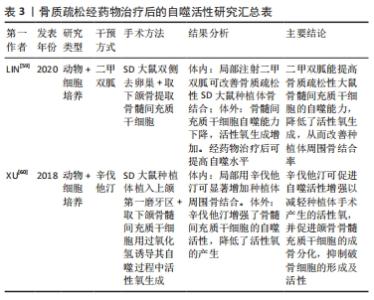
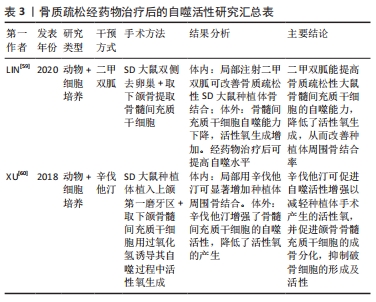
2.3.2 自噬对牙周炎的影响 牙龈卟啉单胞菌是引起慢性牙周炎较为常见的致病菌,其释放的毒性产物造成牙周组织破坏,同时还可以诱导炎性因子的释放,引起炎症反应,进而加重牙周组织破坏[46]。研究发现,牙龈卟啉单胞菌(P. gingivalis)能够利用其引发的细胞自噬来实现其在宿主细胞中的生存和增殖,从而逃避宿主的免疫监视,造成牙周组织破坏[47]。LEE等[48]发现,牙龈卟啉单胞菌可内化于牙龈上皮细胞并定植于口腔黏膜中,将感染的牙龈上皮细胞连续切片及其断层重建,结果发现牙龈卟啉单胞菌定植在富含内质网的双层膜自噬体空泡中,其数量的增加呈时间依赖性;而通过3-甲基腺嘌呤或siRNA-ATG5抑制细胞自噬,结果发现在感染24 h后,牙龈卟啉单胞菌的存活率显著下降,结果表明,牙龈卟啉单胞菌感染人牙龈上皮后,可能是利用自噬空泡中的蛋白质等物质生存和不断复制增殖,来逃避宿主的防御反应,进而对组织造成破坏。而PARK等[40] 研究发现,利用牙龈卟啉单胞菌处理人急性单核白血病细胞来源巨噬细胞后,可激活巨噬细胞中的自噬活性水平,减少牙龈卟啉单胞菌数量,使用自噬抑制剂后,牙龈卟啉单胞菌的存活率显著增加,巨噬细胞对牙龈卟啉单胞菌清除作用明显减弱,说明自噬对牙龈卟啉单胞菌具有清除作用,从而保护牙周骨质健康。此外,NLRP3炎性小体参与机体固有免疫反应,其活化后可促进成熟的白细胞介素1β分泌,抵抗某些病原体损伤,而过度的NLRP3炎性小体激活会导致各种炎性疾病的发生[27,49]。近年来,研究发现自噬依赖于炎性小体传感器的存在,阻断自噬会增强炎性小体的活性,激活自噬会降低其活性,进而抑制caspase-1的激活,使白细胞介素1β、白细胞介素18等促炎细胞因子的释放减少[39,50]。白细胞介素1β是牙周病发病机制中主要的促炎细胞因子[51],其可以使免疫炎症细胞聚集、蛋白酶的产生和释放、破骨细胞的发育成熟和活化,从而促进牙周炎症和组织破坏[52]。研究发现,自噬可以抑制白细胞介素1β的分泌,以减轻炎症反应,缓解疾病发展[53]。因此,牙龈卟啉单胞菌对自噬的调节也可能与NLRP3炎性小体介导的细胞途径有关。 2.3.3 自噬对种植体骨结合的影响 充足的骨量和健康的骨质能使种植体与骨界面之间紧密结合并易获得初期稳定性[54]。系统性骨代谢障碍不利于种植体获得初期稳定性。目前,骨质疏松症是常见的与年龄相关的骨代谢性疾病。研究表明大约有10.9%的骨质疏松患者在种植体植入初期因牙槽骨骨结构不良、骨量不足导致种植失败[51]。自噬参与调节骨代谢水平,对口腔种植体周围骨结合存在影响。正常情况下,机体中骨组织细胞可以利用自噬调控清除损坏的细胞器和大分子物质,使细胞保持正常的功能,进而使衰老对骨骼细胞的功能影响减少至最小化[55]。因此,自噬对维持人类机体骨代谢起着重要作用,但自噬水平失调会导致骨质疏松等疾病的发生[56-57]。研究发现,在老年人群中,随着年龄的增长,骨细胞自噬活性降低,可能导致年龄相关骨质疏松症的骨质流失[58]。LIN等[59]通过体内外研究发现,骨质疏松状态下种植体骨结合过程中骨髓间充质干细胞的自噬能力下降,活性氧生成量明显增加,种植体骨周围的成骨相关分子表达(Runx2、OCN、ALP)显著降低,而经50 mmol/L二甲双胍治疗后,自噬表达水平增高,活性氧生成减少,骨形成标记物增加,说明二甲双胍可提高自噬表达水平,从而促进种植体周围新骨生成,改善骨微结构,提高种植体骨结合。XU等[60]体内外研究发现,辛伐他汀能有效增强骨髓间充质干细胞的自噬水平,减少活性氧生成,促进成骨细胞分化,加速种植体骨结合。因此,自噬水平升高,活性氧生成减少,抑制炎症反应,促进骨的形成,从而增加种植体周围骨结合率[61]。此外,XU等[62]研究发现,在雌激素缺乏诱导的骨质疏松性疾病中,NLRP3炎性小体的表达显著,可能原因是随着年龄的增大,自噬功能受到抑制,NLRP3炎性小体被激活,使机体处于炎症状态,加重骨组织破坏[63]。自噬与NLRP3炎性小体具有双向调控作用。那么,如何通过调控自噬,进而抑制NLRP3炎性小体活性,提高种植体周围骨结合率,是值得研究的课题。骨质疏松经药物治疗后的自噬活性研究汇总,见表3。"
| [1] DIKIC I, ELAZAR Z. Mechanism and medical implications of mammalian autophagy. Nat Rev Mol Cell Biol. 2018;19(6):349-364. [2] YIN X, ZHOU C, LI J, et al. Autophagy in bone homeostasis and the onset of osteoporosis. Bone Res. 2019;7:28. [3] RUBINSZTEIN DC, MARIñO G, KROEMER G. Autophagy and aging. Cell. 2011;146(5):682-695. [4] TAN YQ, ZHANG J, ZHOU G. Autophagy and its implication in human oral diseases. Autophagy. 2017;13(2):225-236. [5] CHEN X, WANG Z, DUAN N, et al. Osteoblast-osteoclast interactions. Connect Tissue Res. 2018;59(2):99-107. [6] 孙友强,马超,梁萌梦,等.自噬在骨细胞中重要作用的最新研究进展:骨相关细胞活性和骨代谢[J].中国组织工程研究,2022, 26(2):276-282. [7] WANG L, HAUENSTEIN AV. The NLRP3 inflammasome: Mechanism of action, role in disease and therapies. Mol Aspects Med. 2020;76: 100889. [8] LI Y, LING J, JIANG Q. Inflammasomes in Alveolar Bone Loss. Front Immunol. 2021;12:691013. [9] FLORENCIO-SILVA R, SASSO GR, SIMÕES MJ, et al. Osteoporosis and autophagy: What is the relationship? Rev Assoc Med Bras (1992). 2017; 63(2):173-179. [10] HUANG L, JIN P, LIN X, et al. Beneficial effects of sulfonamide‑based gallates on osteoblasts in vitro. Mol Med Rep. 2017;15(3):1149-1156. [11] NOLLET M, SANTUCCI-DARMANIN S, BREUIL V, et al. Autophagy in osteoblasts is involved in mineralization and bone homeostasis. Autophagy. 2014;10(11):1965-1977. [12] 连璐雅,朱海华.自噬对炎症性牙槽骨吸收作用的研究进展[J].中国临床新医学,2020,13(8):833-836. [13] LIU W, ZHAO Z, NA Y, et al. Dexamethasone-induced production of reactive oxygen species promotes apoptosis via endoplasmic reticulum stress and autophagy in MC3T3-E1 cells. Int J Mol Med. 2018;41(4): 2028-2036. [14] ZHANG S, LIU Y, LIANG Q. Low-dose dexamethasone affects osteoblast viability by inducing autophagy via intracellular ROS. Mol Med Rep. 2018;17(3):4307-4316. [15] WANG L, ZHANG H, DONG M, et al. Role of the Btk-PLCγ2 Signaling Pathway in the Bone Destruction of Apical Periodontitis. Mediators Inflamm. 2019;2019:8767529. [16] SONG L, TAN J, WANG Z, et al. Interleukin‑17A facilitates osteoclast differentiation and bone resorption via activation of autophagy in mouse bone marrow macrophages. Mol Med Rep. 2019;19(6):4743-4752. [17] 杨彩会,刘启成,董明,等.丝氨酸/苏氨酸蛋白激酶促进慢性根尖周炎模型小鼠的骨破坏[J].中国组织工程研究,2021,25(23): 3654-3659. [18] LI RF, CHEN G, REN JG, et al. The adaptor protein p62 is involved in RANKL-induced autophagy and osteoclastogenesis. J Histochem Cytochem. 2014;62(12):879-888. [19] SUL OJ, PARK HJ, SON HJ, et al. Lipopolysaccharide (LPS)-Induced Autophagy Is Responsible for Enhanced Osteoclastogenesis. Mol Cells. 2017;40(11):880-887. [20] DESELM CJ, MILLER BC, ZOU W, et al. Autophagy proteins regulate the secretory component of osteoclastic bone resorption. Dev Cell. 2011;21(5):966-974. [21] LIN NY, CHEN CW, KAGWIRIA R, et al. Inactivation of autophagy ameliorates glucocorticoid-induced and ovariectomy-induced bone loss. Ann Rheum Dis. 2016;75(6):1203-1210. [22] 杨钰粒,徐亦文,安蒂,等.自噬在破骨细胞中的调控作用[J].生命的化学,2016,36(3):354-358. [23] SCHRODER K, TSCHOPP J. The inflammasomes. Cell. 2010;140(6):821-832. [24] CHEN Y, YANG Q, LV C, et al. NLRP3 regulates alveolar bone loss in ligature-induced periodontitis by promoting osteoclastic differentiation. Cell Prolif. 2021;54(2):e12973. [25] SAITOH T, FUJITA N, JANG MH, et al. Loss of the autophagy protein Atg16L1 enhances endotoxin-induced IL-1beta production. Nature. 2008;456(7219):264-268. [26] LEVINE B, MIZUSHIMA N, VIRGIN HW. Autophagy in immunity and inflammation. Nature. 2011;469(7330):323-335. [27] BIASIZZO M, KOPITAR-JERALA N. Interplay Between NLRP3 Inflammasome and Autophagy. Front Immunol. 2020;11:591803. [28] NETEA-MAIER RT, PLANTINGA TS, VAN DE VEERDONK FL, et al. Modulation of inflammation by autophagy: Consequences for human disease. Autophagy. 2016;12(2):245-260. [29] ZHONG Z, SANCHEZ-LOPEZ E, KARIN M. Autophagy, NLRP3 inflammasome and auto-inflammatory/immune diseases. Clin Exp Rheumatol. 2016;34(4 Suppl 98):12-16. [30] CAO Z, WANG Y, LONG Z, et al. Interaction between autophagy and the NLRP3 inflammasome. Acta Biochim Biophys Sin (Shanghai). 2019; 51(11):1087-1095. [31] COSIN-ROGER J, SIMMEN S, MELHEM H, et al. Hypoxia ameliorates intestinal inflammation through NLRP3/mTOR downregulation and autophagy activation. Nat Commun. 2017;8(1):98. [32] LI H, MIAO W, MA J, et al. Acute Exercise-Induced Mitochondrial Stress Triggers an Inflammatory Response in the Myocardium via NLRP3 Inflammasome Activation with Mitophagy. Oxid Med Cell Longev. 2016; 2016:1987149. [33] ZOU YJ, XU JJ, WANG X, et al. Lactobacillus johnsonii L531 Ameliorates Escherichia coli-Induced Cell Damage via Inhibiting NLRP3 Inflammasome Activity and Promoting ATG5/ATG16L1-Mediated Autophagy in Porcine Mammary Epithelial Cells. Vet Sci. 2020;7(3):112. [34] DUPONT N, JIANG S, PILLI M, et al. Autophagy-based unconventional secretory pathway for extracellular delivery of IL-1β. EMBO J. 2011; 30(23):4701-4711. [35] AHMED ME, IYER S, THANGAVEL R, et al. Co-Localization of Glia Maturation Factor with NLRP3 Inflammasome and Autophagosome Markers in Human Alzheimer’s Disease Brain. J Alzheimers Dis. 2017; 60(3):1143-1160. [36] ZHANG J, HUANG X, LU B, et al. Can apical periodontitis affect serum levels of CRP, IL-2, and IL-6 as well as induce pathological changes in remote organs. Clin Oral Investig. 2016;20(7):1617-1624. [37] JAKOVLJEVIC A, MILETIC M, NIKOLIC N, et al. Notch signaling pathway mediates alveolar bone resorption in apical periodontitis. Med Hypotheses. 2019;124:87-90. [38] WANG L, JIN H, YE D, et al. Enterococcus faecalis Lipoteichoic Acid-induced NLRP3 Inflammasome via the Activation of the Nuclear Factor Kappa B Pathway. J Endod. 2016;42(7):1093-1100. [38] SHI CS, SHENDEROV K, HUANG NN, et al. Activation of autophagy by inflammatory signals limits IL-1β production by targeting ubiquitinated inflammasomes for destruction. Nat Immunol. 2012;13(3):255-263. [40] PARK MH, JEONG SY, NA HS, et al. Porphyromonas gingivalis induces autophagy in THP-1-derived macrophages. Mol Oral Microbiol. 2017; 32(1):48-59. [41] LAI EH, HONG CY, KOK SH, et al. Simvastatin alleviates the progression of periapical lesions by modulating autophagy and apoptosis in osteoblasts. J Endod. 2012;38(6):757-763. [42] ZHU L, YANG J, ZHANG J, et al. The presence of autophagy in human periapical lesions. J Endod. 2013;39(11):1379-1384. [43] HUANG HY, WANG WC, LIN PY, et al. The roles of autophagy and hypoxia in human inflammatory periapical lesions. Int Endod J. 2018; 51 Suppl 2:e125-e145. [44] 马金宝,王倩,董明,等.人牙髓炎及根尖周炎病损组织中自噬相关蛋白微管相关蛋白1轻链3B的表达[J].中国组织工程研究,2019, 23(15):2364-2368. [45] ALVAREZ VE, KOSEC G, SANT’ANNA C, et al. Autophagy is involved in nutritional stress response and differentiation in Trypanosoma cruzi. J Biol Chem. 2008;283(6):3454-3464. [46] MYSAK J, PODZIMEK S, SOMMEROVA P, et al. Porphyromonas gingivalis: major periodontopathic pathogen overview. J Immunol Res. 2014; 2014:476068. [47] 赵妍,于阳,寇育荣.牙龈卟啉单胞菌调控细胞自噬的分子机制研究进展[J].华西口腔医学杂志,2017,35(6):654-658. [48] LEE K, ROBERTS JS, CHOI CH, et al. Porphyromonas gingivalis traffics into endoplasmic reticulum-rich-autophagosomes for successful survival in human gingival epithelial cells. Virulence. 2018;9(1):845-859. [49] LU A, WU H. Structural mechanisms of inflammasome assembly. FEBS J. 2015;282(3):435-444. [50] MATSUZAWA-ISHIMOTO Y, HWANG S, CADWELL K. Autophagy and Inflammation. Annu Rev Immunol. 2018;36:73-101. [51] ARAL K, MILWARD MR, KAPILA Y, et al. Inflammasomes and their regulation in periodontal disease: A review. J Periodontal Res. 2020; 55(4):473-487. [52] MEMMERT S, NOGUEIRA AVB, DAMANAKI A, et al. Damage-regulated autophagy modulator 1 in oral inflammation and infection. Clin Oral Investig. 2018;22(8):2933-2941. [53] VAN DER BURGH R, NIJHUIS L, PERVOLARAKI K, et al. Defects in mitochondrial clearance predispose human monocytes to interleukin-1β hypersecretion. J Biol Chem. 2014;289(8):5000-5012. [54] PALASKAR JN, JOSHI N, SHAH PM, et al. Influence of different implant placement techniques to improve primary implant stability in low-density bone: A systematic review. J Indian Prosthodont Soc. 2020; 20(1):11-16. [55] JILKA RL, O’BRIEN CA. The Role of Osteocytes in Age-Related Bone Loss. Curr Osteoporos Rep. 2016;14(1):16-25. [56] SHAPIRO IM, LAYFIELD R, LOTZ M, et al. Boning up on autophagy: the role of autophagy in skeletal biology. Autophagy. 2014;10(1):7-19. [57] GREENHILL C. Bone: Autophagy regulates bone growth in mice. Nat Rev Endocrinol. 2016;12(1):4. [58] CHEN K, YANG YH, JIANG SD, et al. Decreased activity of osteocyte autophagy with aging may contribute to the bone loss in senile population. Histochem Cell Biol. 2014;142(3):285-295. [59] LIN J, XU R, SHEN X, et al. Metformin promotes the osseointegration of titanium implants under osteoporotic conditions by regulating BMSCs autophagy, and osteogenic differentiation. Biochem Biophys Res Commun. 2020;531(2):228-235. [60] XU R, SHI G, XU L, et al. Simvastatin improves oral implant osseointegration via enhanced autophagy and osteogenesis of BMSCs and inhibited osteoclast activity. J Tissue Eng Regen Med. 2018;12(5): 1209-1219. [61] CHEN Y, GUAN M, REN R, et al. Improved Immunoregulation of Ultra-Low-Dose Silver Nanoparticle-Loaded TiO(2) Nanotubes via M2 Macrophage Polarization by Regulating GLUT1 and Autophagy. Int J Nanomedicine. 2020;15:2011-2026. [62] XU L, ZHANG L, WANG Z, et al. Melatonin Suppresses Estrogen Deficiency-Induced Osteoporosis and Promotes Osteoblastogenesis by Inactivating the NLRP3 Inflammasome. Calcif Tissue Int. 2018;103(4): 400-410. [63] SALMINEN A, KAARNIRANTA K, KAUPPINEN A. Inflammaging: disturbed interplay between autophagy and inflammasomes. Aging (Albany NY). 2012;4(3):166-175. |
| [1] | Nong Fuxiang, Jiang Zhixiong, Li Yinghao, Xu Wencong, Shi Zhilan, Luo Hui, Zhang Qinglang, Zhong Shuang, Tang Meiwen. Bone cement augmented proximal femoral nail antirotation for type A3.3 intertrochanteric femoral fracturalysis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(在线): 1-10. |
| [2] | Pan Zhongjie, Qin Zhihong, Zheng Tiejun, Ding Xiaofei, Liao Shijie. Targeting of non-coding RNAs in the pathogenesis of the osteonecrosis of the femoral head [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1441-1447. |
| [3] | Cai Zhihao, Xie Zhaoyong. Femoral neck anteversion measurement assessment: how to establish a unified method and standard [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1448-1454. |
| [4] | Dang Yi, Du Chengyan, Yao Honglin, Yuan Nenghua, Cao Jin, Xiong Shan, Zhang Dingmei, Wang Xin. Hormonal osteonecrosis and oxidative stress [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1469-1476. |
| [5] | Yang Zhishan, Tang Zhenglong. YAP/TAZ, a core factor of the Hippo signaling pathway, is involved in bone formation [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1264-1271. |
| [6] | Wang Ji, Zhang Min, Yang Zhongya, Zhang Long. A review of physical activity intervention in type 2 diabetes mellitus with sarcopenia [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1272-1277. |
| [7] | Nie Chenchen, Su Kaiqi, Gao Jing, Fan Yongfu, Ruan Xiaodi, Yuan Jie, Duan Zhaoyuan, Feng Xiaodong. The regulatory role of circular RNAs in cerebral ischemia-reperfusion injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1286-1291. |
| [8] | Gao Yu, Han Jiahui, Ge Xin. Immunoinflammatory microenvironment after spinal cord ischemia-reperfusion injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1300-1305. |
| [9] | Sun Jiajia, Zhu Haidi, Lu Yun, Zhang Kai. Comparison of bone metabolism markers between type 2 diabetes mellitus and non-type 2 diabetes mellitus patients with hip fracture [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1156-1160. |
| [10] | Long Guiyue, Li Dongdong, Liao Hongbing. Calcium phosphate cement/poly(lactic-co-glycolic acid) degradation products promote osteoclast differentiation of mouse monocytes [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1193-1198. |
| [11] | Huang Linke, Wei Linhua, Jiang Jie, Liu Qian, Chen Weiwei. Effects of estrogen combined with treadmill exercise on bone mass and articular cartilage in ovariectomized mice [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1166-1171. |
| [12] | Liu Xiaolin, Mu Xinyue, Ma Ziyu, Liu Shutai, Wang Wenlong, Han Xiaoqian, Dong Zhiheng. Effect of hydrogel-loaded simvastatin microspheres on osteoblast proliferation and differentiation [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 998-1003. |
| [13] | Xu Xingxing, Wen Chaoju, Meng Maohua, Wang Qinying, Chen Jingqiao, Dong Qiang. Carbon nanomaterials in oral implant [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1062-1070. |
| [14] | Li Cheng, Zheng Guoshuang, Kuai Xiandong, Yu Weiting. Alginate scaffold in articular cartilage repair [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1080-1088. |
| [15] | Chen Shisong, Liu Xiaohong, Xu Zhiyun. Current status and prospects of bioprosthetic heart valves [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1096-1102. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||