Chinese Journal of Tissue Engineering Research ›› 2023, Vol. 27 ›› Issue (8): 1193-1198.doi: 10.12307/2023.138
Previous Articles Next Articles
Calcium phosphate cement/poly(lactic-co-glycolic acid) degradation products promote osteoclast differentiation of mouse monocytes
Long Guiyue1, Li Dongdong1, Liao Hongbing2
- 1Guangxi Key Laboratory of Oral and Maxillofacial Restoration and Reconstruction, Guangxi Cranio-Maxillofacial Malformation Clinical Research Center, Guangxi Health Commission Key laboratory of prevention and treatment for oral infectious diseases, 2Department of Prosthetics, School of Stomatology, Guangxi Medical University, Nanning 530021, Guangxi Zhuang Autonomous Region, China
-
Received:2022-03-09Accepted:2022-05-14Online:2023-03-18Published:2022-07-27 -
Contact:Liao Hongbing, MD, Professor, Department of Prosthetics, School of Stomatology, Guangxi Medical University, Nanning 530021, Guangxi Zhuang Autonomous Region, China -
About author:Long Guiyue, Master candidate, Guangxi Key Laboratory of Oral and Maxillofacial Restoration and Reconstruction, Guangxi Cranio-Maxillofacial Malformation Clinical Research Center, Guangxi Key Laboratory of Oral and Maxillofacial Surgery, Medical Experimental Center of Guangxi Medical University, School of Stomatology, Guangxi Medical University, Nanning 530021, Guangxi Zhuang Autonomous Region, China -
Supported by:National Natural Science Foundation of China, No. 81860201, 81560190 (to LHB)
CLC Number:
Cite this article
Long Guiyue, Li Dongdong, Liao Hongbing. Calcium phosphate cement/poly(lactic-co-glycolic acid) degradation products promote osteoclast differentiation of mouse monocytes[J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1193-1198.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
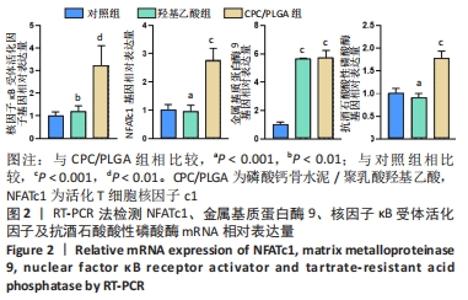
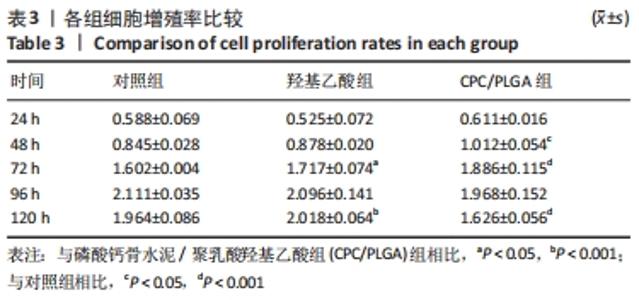
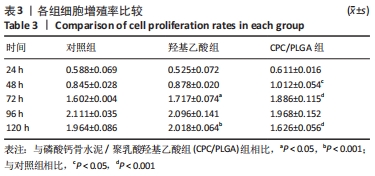
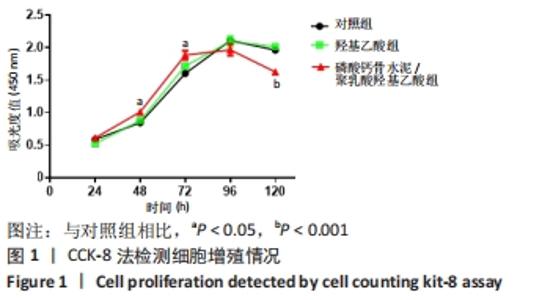
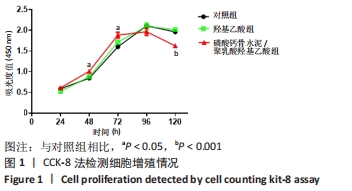
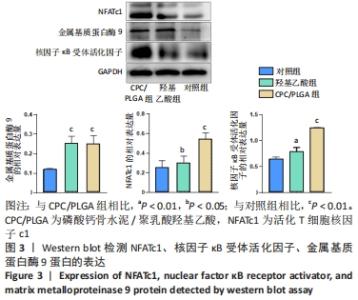
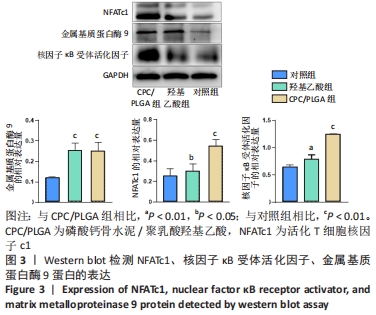
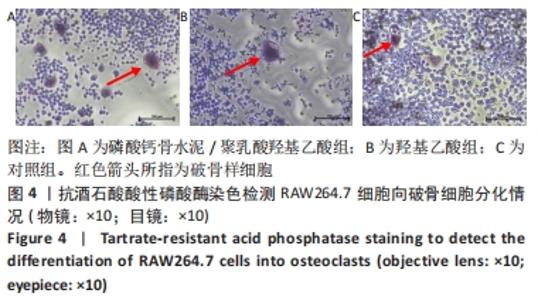
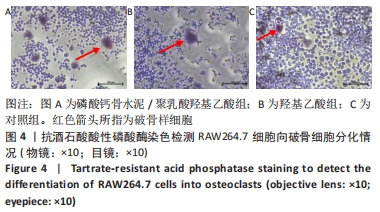
各组细胞经过48 h潜伏后进入对数生长期,48 h时,CPC/PLGA组细胞数比对照组多,差异有显著性意义(P < 0.05);72 h时,CPC/PLGA组的细胞数明显高于对照组及羟基乙酸组,差异有显著性意义(P < 0.05);72 h后,细胞增长速率减慢;在96 h时,细胞数量达到顶峰;120 h时,各组细胞数量下降,CPC/PLGA组较其他两组显著降低(P < 0.001)。 2.2 RT-PCR检测结果 CPC/PLGA组NFATc1、核因子κB受体活化因子、抗酒石酸酸性磷酸酶 mRNA的表达水平较对照组和羟基乙酸组升高,差异有显著性意义(P < 0.01);羟基乙酸组和对照组的NFATc1、核因子κB受体活化因子、抗酒石酸酸性磷酸酶的mRNA表达无差异;羟基乙酸组、CPC/PLGA组的金属基质蛋白酶9 mRNA表达水平显著高于对照组,差异有显著性意义(P < 0.001),见图2。 "
| [1] KARARGYRIS O, POLYZOIS VD, KARABINAS P, et al. Papineau debridement, Ilizarov bone transport, and negative-pressure wound closure for septic bone defects of the tibia. Eur J Orthop Surg Traumatol. 2014; 24(6): 1013-1017. [2] LIAO H, WALBOOMERS XF, HABRAKEN WJ, et al. Injectable calcium phosphate cement with PLGA, gelatin and PTMC microspheres in a rabbit femoral defect. Acta Biomater. 2011;7(4):1752-1759. [3] YAO J, LIU Z, MA W, et al. Three-Dimensional Coating of SF/PLGA Coaxial Nanofiber Membranes on Surfaces of Calcium Phosphate Cement for Enhanced Bone Regeneration. ACS Biomater Sci Eng. 2020;6(5):2970-2984. [4] ZHANG M, MATINLINNA JP, TSOI J KH, et al. Recent developments in biomaterials for long-bone segmental defect reconstruction: A narrative overview. J Orthop Translat. 2020;22:26-33. [5] LODOSO-TORRECILLA I, VAN DEN BEUCKEN J, JANSEN JA. Calcium phosphate cements: Optimization toward biodegradability. Acta Biomater. 2021;119:6985. [6] BAUER T W, MUSCHLER GF. Bone graft materials. An overview of the basic science. Clin Orthop Relat Res. 2000;(371):10-27. [7] CAMPANA V, MILANO G, PAGANO E, et al. Bone substitutes in orthopaedic surgery: from basic science to clinical practice. J Mater Sci Mater Med. 2014;25(10):2445-2461. [8] CUI X, HUANG C, CHEN Z, et al. Hyaluronic acid facilitates bone repair effects of calcium phosphate cement by accelerating osteogenic expression. Bioact Mater. 2021;6(11):3801-3811. [9] MASTROGIACOMO M, CORSI A, FRANCIOSO E, et al. Reconstruction of extensive long bone defects in sheep using resorbable bioceramics based on silicon stabilized tricalcium phosphate. Tissue Eng. 2006;12(5): 1261-1273. [10] KUMARI A, YADAV SK, YADAV SC. Biodegradable polymeric nanoparticles based drug delivery systems. Colloids Surf B Biointerfaces. 2010;75(1): 1-18. [11] WU L, DING J. In vitro degradation of three-dimensional porous poly (D,L-lactide-co-glycolide) scaffolds for tissue engineering. Biomaterials. 2004;25(27):5821-5830. [12] ZHAO D, ZHU T, LI J, et al. Poly(lactic-co-glycolic acid)-based composite bone-substitute materials. Bioact Mater. 2021;6(2):346-360. [13] AN J, LIAO H, KUCKO NW, et al. Long-term evaluation of the degradation behavior of three apatite-forming calcium phosphate cements. J Biomed Mater Res A. 2016;104(5):1072-1081. [14] HABRAKEN WJ, LIAO HB, ZHANG Z, et al. In vivo degradation of calcium phosphate cement incorporated into biodegradable microspheres. Acta Biomater. 2010;6(6):2200-2211. [15] MANTOVANI A, BISWAS SK, GALDIERO MR, et al. Macrophage plasticity and polarization in tissue repair and remodelling. J Pathol. 2013;229(2): 176-185. [16] UDAGAWA N, KOIDE M, NAKAMURA M, et al. Osteoclast differentiation by RANKL and OPG signaling pathways. J Bone Miner Metab. 2021;39(1):19-26. [17] MARTIN TJ, SIMS NA. RANKL/OPG; Critical role in bone physiology. Rev Endocr Metab Disord. 2015;16(2):131-139. [18] WENISCH S, STAHL JP, HORAS U, et al. In vivo mechanisms of hydroxyapatite ceramic degradation by osteoclasts: fine structural microscopy. J Biomed Mater Res A. 2003;67(3):713-718. [19] WENISCH S, STAHL JP, HORAS U, et al. Ultrastructural examination of cell-mediated degradation of a calcium phosphate ceramic. Unfallchirurg. 2003;106(5):387-391. [20] 覃媛. 磷酸钙骨水泥/聚乳酸-羟基乙酸复合物凝固初期对 RAW264.7 细胞 RANK 因子表达的影响[D]. 南宁: 广西医科大学,2015. [21] 曲任飞,麦昱颖,陈晓薇,等.小鼠单核细胞破骨向分化与乳酸浓度的关系[J].中国组织工程研究,2020,24(14):2177-2183. [22] WALSH NC, CROTTI TN, GOLDRING SR, et al. Rheumatic diseases: the effects of inflammation on bone. Immunol Rev. 2005;208:228-251. [23] FELIX LANAO RP, LEEUWENBURGH SC, WOLKE JG, et al. Bone response to fast-degrading, injectable calcium phosphate cements containing PLGA microparticles. Biomaterials. 2011;32(34):8839-8847. [24] SUH KS, CHON S, JUNG WW, et al. Effects of methylglyoxal on RANKL-induced osteoclast differentiation in RAW264.7cells. Chem Biol Interact. 2018;296:18-25. [25] SUN X, KANG Y, BAO J, et al. Modeling vascularized bone regeneration within a porous biodegradable CaP scaffold loaded with growth factors. Biomaterials. 2013;34(21):4971-4981. [26] FELIX LANAO RP, LEEUWENBURGH SC, WOLKE JG, et al. In vitro degradation rate of apatitic calcium phosphate cement with incorporated PLGA microspheres. Acta Biomater. 2011;7(9):3459-3468. [27] LU T, HE F, YE J. Physicochemical Properties, In Vitro Degradation, and Biocompatibility of Calcium Phosphate Cement Incorporating Poly(lactic-co-glycolic acid) Particles with Different Morphologies: A Comparative Study. ACS Omega. 2021;6(12):8322-8331. [28] ASAGIRI M, TAKAYANAGI H. The molecular understanding of osteoclast differentiation. Bone. 2007;40(2):251-264. [29] SHARMA A, SHARMA L, GOYAL R. Molecular Signaling Pathways and Essential Metabolic Elements in Bone Remodeling: An Implication of Therapeutic Targets for Bone Diseases. Curr Drug Targets. 2021;22(1):77-104. [30] WANG J, ZHENG H, MA R. Natural Occurring Compounds Inhibit Osteoclastogenesis via Targeting NFATc1-related Signaling Pathways. Curr Drug Targets. 2020;21(4):358-364. [31] ZHANG K, BARRAGAN-ADJEMIAN C, YE L, et al. E11/gp38 selective expression in osteocytes: regulation by mechanical strain and role in dendrite elongation. Mol Cell Biol. 2006;26(12):4539-4552. [32] Brown EM, Pollak M, Seidman CE, et al. Calcium-ion-sensing cell-surface receptors. N Engl J Med. 1995;333(4):234-240. [33] ZAIDI M, ADEBANJO OA, MOONGA BS, et al. Emerging insights into the role of calcium ions in osteoclast regulation. J Bone Miner Res. 1999; 14(5):669-674. [34] DENTON RM. Regulation of mitochondrial dehydrogenases by calcium ions. Biochim Biophys Acta. 2009;1787(11):1309-1316. [35] ZAIDI M, DATTA HK, PATCHELL A, et al. ‘Calcium-activated’ intracellular calcium elevation: a novel mechanism of osteoclast regulation. Biochem Biophys Res Commun. 1989;163(3):1461-1465. [36] ARIOKA M, TAKAHASHI-YANAGA F, TATSUMOTO N, et al. Inorganic phosphate-induced impairment of osteoclast cell-cell fusion by the inhibition of AP-1-mediated DC-STAMP expression. Biochem Biophys Res Commun. 2017;493(1):9-13. [37] MOZAR A, HAREN N, CHASSERAUD M, et al. High extracellular inorganic phosphate concentration inhibits RANK-RANKL signaling in osteoclast-like cells. J Cell Physiol. 2008;215(1):47-54. [38] 赵崇莹. 可注射磷酸钙骨水泥凝固初期钙磷离子的变化对成骨细胞的影响[D]. 南宁: 广西医科大学,2013. [39] 梁翠薇. 磷酸钙骨水泥/聚乳酸-羟基乙酸复合微球对成骨细胞 RANKL 和 OPG 因子表达的影响[D]. 南宁: 广西医科大学,2016. [40] DEZEREGA A, MADRID S, MUNDI V, et al. Pro-oxidant status and matrix metalloproteinases in apical lesions and gingival crevicular fluid as potential biomarkers for asymptomatic apical periodontitis and endodontic treatment response. J Inflamm (Lond). 2012;9(1):8. [41] MONDAL S, ADHIKARI N, BANERJEE S, et al. Matrix metalloproteinase-9 (MMP-9) and its inhibitors in cancer: A minireview. Eur J Med Chem. 2020;194:112260. [42] GUO J, ZENG X, MIAO J, et al. MiRNA-218 regulates osteoclast differentiation and inflammation response in periodontitis rats through Mmp9. Cell Microbiol. 2019;21(4):e12979. [43] SHIN Y, GHATE NB, MOON B, et al. DNMT and HDAC inhibitors modulate MMP-9-dependent H3 N-terminal tail proteolysis and osteoclastogenesis. Epigenetics Chromatin. 2019;12(1):25. [44] KIM Y, KIM J, LEE H, et al. Tetracycline Analogs Inhibit Osteoclast Differentiation by Suppressing MMP-9-Mediated Histone H3 Cleavage. Int J Mol Sci. 2019;20(16):4038. [45] PAULA-SILVA F WG, ARNEZ MFM, DE CAMPOS CHAVES LAMARQUE G, et al. Osteoclast formation, inflammation, and matrix metalloproteinase-9 are downregulated in bone repair following root canal treatment in dogs teeth. Clin Oral Investig. 2021;25(7):4699-4707. |
| [1] | Lin Shi, Yuan Jiayao, Lin Xiancan, Yang Binbin, Wu Jianjun, Dongzhi Zhuoma, Tang Zijia, Yang Zhijie, Wan Lei, Huang Hongxing. Diagnostic value of peripheral blood interferon-gamma and monocyte chemoattractant protein-1 in postmenopausal osteoporosis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(2): 165-170. |
| [2] | Liu Ke, Fan Haixia, Wang Hong, Cheng Huanzhi, Geng Haixia. Expression and significance of collagen fiber and matrix metalloproteinase-9 during orthodontic root resorption in rats [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(27): 4288-4292. |
| [3] | Li Kun, An Jie, Dong Suge, Cui Lili, Liu Lei, Li Tingting. Effects of different fimA genotypes of Porphyromonas gingivalis on the secretion related proteins and cytokines in human umbilical vein endothelial cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(19): 3043-3047. |
| [4] | Bi Qingwei, Liu Chengpu, Li Yan, Zhao Wenwen, Han Mei. Structure analysis of platelet-rich fibrin derived from two centrifugation procedures [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3534-3539. |
| [5] | Nie Wei, Liu Weiwei, Liu Dawei, Cui Xiaoxue, Liu Shanhai, Li Xu, Xiao Guangli, Wang Shiwei, Niu Huanyun, Li Ruizhi. Host response of different cross-linked hyaluronic acid composite gels and matrix metalloproteinase-9 expression [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(10): 1557-1562. |
| [6] | Li Peng, Yang Shuye, Zhang Kai, Jia Long, Du Gangqiang, Liu Dong, Zhang Xinjun, Zhang Degang, Wang Zhigang. Influence of different wear particles on human peripheral blood mononuclear cells in vitro [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(6): 894-900. |
| [7] | Zhou Long, Chen Xi, Luo Zong-ping, Yang Hui-lin, He Fan. C57BL/6 mouse bone marrow monocytes: isolation, cultivation, purification and differentiation into osteoclasts [J]. Chinese Journal of Tissue Engineering Research, 2015, 19(6): 940-944. |
| [8] | Zhang Zi-li, Wang Gao-feng, Mei Dao-qi, Lin Peng-fei, Tian Ling. TLR2 expression in peripheral blood mononuclear cells of Henoeh-Schonlein purpura children and its association with immune response [J]. Chinese Journal of Tissue Engineering Research, 2015, 19(45): 7356-7361. |
| [9] | Chen Gao-yang, Chang Fei, Li Rui, Zhang Han-yang, Dong Quan-yu, Jiang Zhen-de, Liu Mao-sheng. Theory and efficacy of stem cells from different sources in the treatment of diabetic foot [J]. Chinese Journal of Tissue Engineering Research, 2015, 19(41): 6718-6724. |
| [10] | Ge Jing, Cheng Bei, Peng Wen, Li Yun-qiao, Zhai Wei . Comparison of transfection efficiency of THP-1 monocytes by different methods [J]. Chinese Journal of Tissue Engineering Research, 2014, 18(38): 6105-6109. |
| [11] | Zhang Ying, Liao Li-qiang, Zhang Xiao-gang. Mononuclear cells promote mesenchymal stem cell migration after myocardial infarction [J]. Chinese Journal of Tissue Engineering Research, 2014, 18(1): 119-124. |
| [12] | Wang Min, Cao Bing-zhen. Glial cell responses and matrix metalloproteinase-9 expression in the brain of chronic vascular dementia rat models [J]. Chinese Journal of Tissue Engineering Research, 2013, 17(7): 1212-1219. |
| [13] | Qiao Wei, Ran Feng, Liu Chang-jian. Isolation, culture and characterization of endothelial progenitor cells from the human peripheral blood [J]. Chinese Journal of Tissue Engineering Research, 2013, 17(36): 6508-6514. |
| [14] | Tang Jie, Chen Tao, Mi Jie, Xu Ai-guo, Wang Yong-de, Zhang Jian, Qi Xiang-qian. Effect of bone marrow mononuclear cell transplantation on angiogenesis and expression of cytokines following myocardial infarction [J]. Chinese Journal of Tissue Engineering Research, 2013, 17(32): 5840-5846. |
| [15] | Guo Chao-feng, Zhang Hong-qi, Gao Qi-le, Tang Ming-xing, Liu Shao-hua, Deng Ang, Wang Yu-xiang, Lu Shi-jin, Li Jin-song, Yin Xin-hua. Serum monocyte chemoattactant protein-1 level and spinal tuberculosis susceptibility [J]. Chinese Journal of Tissue Engineering Research, 2013, 17(30): 5503-5510. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||