Chinese Journal of Tissue Engineering Research ›› 2023, Vol. 27 ›› Issue (14): 2242-2248.doi: 10.12307/2023.180
Previous Articles Next Articles
Advances in exercise modulation of mitochondrial function to improve myocardial ischemia-reperfusion injury
Tan Xuefeng1, Ding Zhimin2, Guo Chenggen1, Sun Pu1
- 1School of Physical Education and Sports, Beijing Normal University, Beijing 100875, China; 2School of Rehabilitation Medicine, Henan University of Chinese Medicine, Zhengzhou 450046, Henan Province, China
-
Received:2022-05-12Accepted:2022-06-10Online:2023-05-18Published:2022-09-30 -
Contact:Sun Pu, Professor, Doctoral supervisor, School of Physical Education and Sports, Beijing Normal University, Beijing 100875, China -
About author:Tan Xuefeng, Master candidate, School of Physical Education and Sports, Beijing Normal University, Beijing 100875, China -
Supported by:Beijing Social Science Foundation in 2020, No. 20YTB012 (to SP)
CLC Number:
Cite this article
Tan Xuefeng, Ding Zhimin, Guo Chenggen, Sun Pu. Advances in exercise modulation of mitochondrial function to improve myocardial ischemia-reperfusion injury[J]. Chinese Journal of Tissue Engineering Research, 2023, 27(14): 2242-2248.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
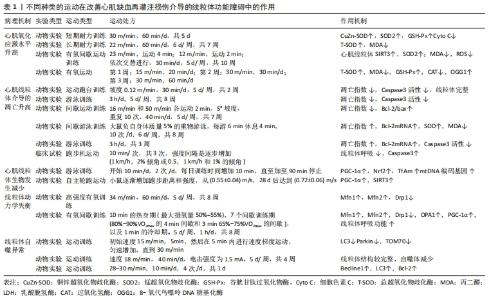
2.1.1 MIRI后心肌细胞线粒体氧化应激增加 氧化应激是生物对于机体内产生和积累的氧自由基没有与之相匹配的解毒能力所引起的一种现象[14],其主要是活性氧形成酶和非酶抗氧化剂之间不平衡的结果。活性氧是一种对于机体具有极大危害的物质,它能够对细胞膜表面的脂质及蛋白质和核酸等生物大分子发生极强的氧化作用,从而使其丧失生理功能。研究表明,无论是在MIRI过程中还是在心肌梗死后的重塑期间,氧化应激都会显著导致心脏损伤,而活性氧是导致缺血再灌注损伤的特殊驱动因素[15]。 在正常的生理条件下,线粒体能够及时清除由于过量的活性氧积累而造成的生物氧化损伤,为机体提供有效的抗氧化作用。然而,研究发现当缺血后重新恢复时,氧气大量进入会加剧活性氧的生成,破坏线粒体DNA(mitochondrial DNA,mtDNA)、蛋白质、脂质以及影响线粒体中电子传递链的正常运转,最终导致线粒体功能障碍[16]。WANG等[17]利用血浆同型半胱氨酸(HCY)处理的动物模型和缺氧后再供氧建立了MIRI损伤模型以评估线粒体功能和氧化应激,结果发现,同型半胱氨酸通过刺激活性氧产生能够诱导MIRI损伤中的线粒体功能障碍和氧化应激,证明了活性氧诱导的线粒体功能障碍是心肌细胞再灌注期间损伤的重要因素。另有研究发现,修复mtDNA可以提高线粒体抗氧化能力,减少心肌梗死面积,改善心脏功能[18]。此外,CHEN等[19]发现,在缺血期间对复合物Ⅰ处的电子传递链进行可逆阻断可保护电子传递链并减少线粒体损伤和心脏损伤。综上所述,活性氧介导的线粒体中的DNA、蛋白质、脂质的被破坏以及呼吸链的非正常运转与MIRI引起的心肌细胞损伤和坏死有着直接的联系。 2.1.2 MIRI后线粒体介导的细胞过度凋亡 细胞内的基因及其产物会对机体细胞进行一系列的生理性调控,细胞在此过程中所发生的有序的死亡过程被称为细胞凋亡,又可称之为细胞程序性死亡,该过程不涉及炎性反应[20]。细胞凋亡是MIRI后的必然结果,而线粒体在心肌细胞凋亡中扮演着重要角色[21],且线粒体介导的细胞过度凋亡的机制中,线粒体膜通透性的变化是主要因素[22]。有研究表示,在MIRI后,线粒体功能受到影响,其中,线粒体发生分解作用,使得线粒体膜电位失衡进而诱导线粒体通透性转换孔(mitochondrial permeability transition pore,mPTP)的开放,同时线粒体内的心磷脂发生过氧化,从而使细胞色素C(Cytochrome C)经mPTP释放至线粒体外,引发心肌细胞凋亡[23]。FANG等[24]在一项药理研究中发现,鞘氨醇1-磷酸通过抑制mPTP的开放,维持心肌线粒体完整性,显著降低心肌梗死的大小和细胞凋亡。同样,WANG等[25]在实验研究中也证实了药物对心肌细胞的保护作用是通过保护线粒体和抑制线粒体凋亡途径实现的。综上可见,MIRI后的细胞凋亡与mPTP的打开有着直接的联系,且与心肌中的线粒体功能密不可分。 Bcl-2家族蛋白是控制线粒体相关的凋亡因子释放的主要调节因子。研究表明,与空白对照组相比,MIRI模型组Bcl-2/Bax及其mRNA的表达水平显著降低,而裂解的半胱天冬酶3(caspase-3)及其mRNA在心肌细胞中的表达水平显著升高,心肌细胞凋亡增加[26]。一项药理学研究表明,黄芪甲苷对心肌细胞的保护作用可能是通过上调Bcl-2的表达、促进Bcl-2易位到线粒体、抑制mPTP开放、减少细胞色素C释放、抑制细胞凋亡来实现的[27]。同样,在YUAN等[28]的动物实验中发现,抑制细胞质中caspase-3可以降低MIRI期间的心肌能量代谢和细胞凋亡,从而达到了对心肌细胞和线粒体的保护。综上所述,MIRI与Bcl-2家族蛋白参与的线粒体介导的细胞过度凋亡有关,同时调控Bcl-2家族蛋白的表达也是缓解MIRI后心肌细胞损伤的关键。 2.1.3 MIRI后线粒体质量控制体系失调 线粒体可通过融合/裂变以及线粒体自噬等自身修复机制维持线粒体各项生理功能的正常进行,这些机制被称为“线粒体质量控制”,是细胞防御线粒体受损的重要机制[29]。研究表明,调节线粒体质量控制体系可以纠正线粒体代谢紊乱,减少心肌缺血再灌注损伤[30]。 线粒体生物发生是一个严格的调控过程,当细胞能量需求超过线粒体的ATP产生时,便会诱发线粒体生物发生。在此过程中,调节因子便会驱动mtDNA复制和转录,并在翻译因子的作用下翻译成蛋白,逐步进入线粒体外膜、膜间隙、线粒体内膜或基质,最后引起线粒体网络扩张,以满足机体的能量代谢需求[31]。正常生理条件下,8-氧代鸟嘌呤DNA糖基化酶与MutY DNA糖基化酶协同作用,以保持mtDNA的完整性。体内及体外实验研究表明,缺血再灌注损伤增加了8-氧代鸟嘌呤DNA糖基化酶含量,降低mtDNA含量和mtDNA转录水平,并导致心肌细胞线粒体功能障碍[32],作者通过进一步药理实验研究发现,番茄红素对mtDNA的保护作用与线粒体活性氧产生的减少和线粒体转录因子A的稳定有关。QI等[33]通过实时荧光定量PCR和免疫印迹分析对受损心肌细胞的线粒体生物发生相关的相关参数进行观察后发现,损伤后过氧化物酶体增殖物激活受体γ共激活因子1α(peroxisome proliferator-activated receptor γ coactivator-1α,PGC-1α)、核因子红系2相关因子2(nuclear factor-erythroid 2-Related Factor 2,Nrf2)、线粒体转录因子A均有所下降,与此同时新生物标志物沉默调节蛋白3(recombinant Sirtuin 3,Sirt3)的mRNA水平也在损伤后明显降低。此外,SHANMUGAM等[34]通过实验研究也得出,MIRI与线粒体结构的丧失和线粒体生物发生的减少有关。而促进线粒体生物发生、抑制线粒体氧化应激,可以增强对心肌细胞的保护作用[33]。可见,促进线粒体生物发生是治疗MIRI的有希望的靶点。 线粒体动力学是线粒体融合/裂变的动态变化过程,且与Ca2+超载、mPTP打开、氧化应激产生活性氧等有着密切联系。其中,线粒体的融合过程受动力相关蛋白GTPase的调控,而线粒体裂变的过程受动力相关蛋白1(dynamin-related protein 1,Drp1)的调控。线粒体融合发生在线粒体轻微受损的情况下,通过对内质网-线粒体连接的增强,可以使线粒体的网状结构更加稳定;线粒体通过分裂进行增殖,线粒体分裂能够促进产生活性氧,使得线粒体发生自噬,这两种作用能够维持心肌细胞中的线粒体稳态,是心脏能量代谢的重要来源[35]。研究发现,心肌缺血再灌注导致线粒体融合蛋白1/2(Mitofusin1/2,Mfn1/2)减少,而在心肌缺血期间和再灌注开始时,通过抑制线粒体功能障碍和动态失衡可以对心脏进行保护,有效减少梗死大小并改善心脏功能[36]。同样,YANG等[37]在药理学研究中发现,盐酸戊乙奎醚通过对MIRI中氧化应激诱导的线粒体动力学不平衡进行改善,减慢细胞的凋亡速率,对心肌损伤起到了有效的保护。 线粒体动力学在调节细胞正常能量代谢中发挥关键作用。研究表明,缺血再灌注导致心肌ATP明显降低[38]。ZHOU等[39]在一项细胞实验中发现,增加线粒体融合并抑制线粒体裂变,可以改善线粒体能量代谢障碍,促进ATP的产生,从而实现对心肌的保护作用。同样,WANG等[25]的实验研究中也发现,通过减轻线粒体功能障碍,可以有效促进ATP的产生,帮助大鼠MIRI后心脏功能的恢复。综上可见,线粒体动力学失衡及其介导的能量代谢障碍与MIRI有着密切联系,同时也是有希望的治疗靶点。 线粒体自噬作为一种选择性自噬形式,通过靶向需要清除的功能失调的线粒体来维持细胞的动态平衡。线粒体自噬的前提条件是线粒体发生裂变,该过程与线粒体动力学密切相关[40]。ZHU等[41]在研究中也印证了以上观点,抑制线粒体裂变可以有效抑制线粒体自噬。 当线粒体受损失时提高其自噬水平可维持有效的细胞稳态,但过度可能会引起线粒体过度清除,使线粒体出现功能障碍,最终导致细胞死亡[42]。可见,线粒体自噬是一把“双刃剑”。研究表明,缺血再灌注抑制了线粒体融合及自噬,而上调线粒体自噬可以实现对心肌的保护[43]。然而,XIAO等[44]通过实验研究表明,电针预处理通过抑制由mTORC1-ULK1-FUNDC1途径介导的线粒体自噬来保护心肌免受缺血再灌注损伤。综上可见,MIRI与线粒体自噬有关,如何调控MIRI中线粒体自噬水平的“正常化”对病程的康复具有重要意义。 2.2 运动改善受损心肌细胞线粒体功能障碍帮助MIRI恢复的研究 如表1所示,不同种类的运动已被证实在调节线粒体功能障碍从而帮助MIRI中具有重要意义,具体表现在降低MIRI后的氧化应激水平、抑制线粒体介导的细胞凋亡以及改善线粒体的质量控制障碍。 "


2.2.1 运动可降低MIRI后的氧化应激增加 运动一直被认为是预防和治疗心血管疾病的有效手段,不同种类的运动在抑制MIRI后氧化应激水平中的作用已被许多研究所证实。RANJBAR等[45]在研究中发现,与MIRI模型组小鼠相比,10周的抗阻运动可以显著减轻心肌细胞缺血再灌注损伤,并指出其机制可能与抗阻运动抑制线粒体氧化应激水平有关。早期研究显示,3-5 d持续的耐力训练通过防止线粒体功能障碍导致的活性氧过度释放,增加线粒体抗氧化剂超氧化物歧化酶(superoxide dismutase,SOD),谷胱甘肽过氧化物酶(GSH-Px)等的活性,保护了心脏线粒体免受MIRI后的氧化损伤,从而实现其心肌保护作用[46]。随后许思毛等[47]通过进一步研究发现,长期较大强度的耐力运动预处理能够减轻MIRI后心肌锰超氧化物歧化酶(SOD2)蛋白活性的下降程度,提高活性氧代谢水平,从而形成对MIRI后氧化损伤的心脏保护效应。此外,研究发现,间歇性有氧运动训练能够有效强化心肌线粒体抗氧化酶系统中的关键因子沉默调节蛋白3(SIRT3)和超氧化物歧化酶(SOD2)的表达,从而降低缺血后心肌中氧自由基的产生,促进损伤心肌的恢复[48]。同样,RUI等[49]通过研究发现,早期有氧运动训练通过部分调节8-氧代鸟嘌呤DNA糖基化酶等抗氧化相关蛋白和线粒体相关蛋白的表达来保护缺血后心肌的损伤。综上表明,抗阻运动与有氧运动均可增强MIRI后心肌细胞中线粒体的抗氧化酶活性与蛋白活性,减少线粒体活性氧的释放,继而减轻线粒体功能障碍造成的氧化应激水平升高。 2.2.2 运动可以抑制MIRI后线粒体介导的细胞凋亡 MIRI后由线粒体介导的心肌细胞凋亡的发生可以通过运动进行抑制[50],多项研究已证实不同种类的运动在抑制MIRI后线粒体介导的细胞凋亡的作用。于新彦等[51]在研究中发现,与MIRI模型组小鼠相比,2周的有氧运动可有效抑制心肌组织中caspase-3的活性从而抑制MIRI后心肌细胞凋亡和促进心肌功能的恢复。与此研究类似,ZHANG等[52]通过动物实验研究发现,8周的有氧运动对缺血再灌注损伤的心肌发挥保护作用,部分是通过抗心肌细胞凋亡实现的。然而,目前尚未有横向研究对比有氧训练在抑制MIRI后心肌凋亡中的量效关系。早期研究在探讨间歇运动预处理对心脏缺血再灌注的作用机制中发现,长时间的间歇运动和一次间歇运动均能影响Bcl-2和bax的表达,使Bcl-2/bax增加,抑制线粒体介导的心肌细胞凋亡,改善心肌再灌注损伤[53]。刘志梅等[54]进一步研究发现,间歇游泳训练对MIRI大鼠的心肌损伤具有保护作用,具体体现在8周的间歇游泳能够增强MIRI大鼠Bcl-2 mRNA及蛋白的表达,提高心肌细胞8-氧代鸟嘌呤DNA糖基化酶活性,减少丙二醛生成,从而降低心肌细胞凋亡指数以达到对心肌的保护作用。此外,LAI等[55]针对游泳运动研究发现,游泳运动可降低肿瘤坏死因子α水平,抑制caspase-3激活,增强Bcl-2蛋白的表达,从而减少了心肌中凋亡细胞的数量,并提出游泳运动可以作为预防心肌损伤的有效方式。尽管有许多动物实验研究均证明了运动训练及运动预处理在保护心肌中的有效性,然而,一项随机对照试验显示,与单纯进行冠状动脉搭桥术的患者相比,术前24 h进行一次适度运动反而使心肌细胞线粒体呼吸水平降低,且凋亡相关标志物Caspase3转录水平增加1.5倍[56],这表明运动预处理并未在MIRI中发挥积极的保护作用。另有研究发现,抗阻运动对MIRI后细胞凋亡的抑制作用不佳,抗阻运动在改变梗死的大小和细胞凋亡上与对照组相比无统计学意义,并提示未来需要进一步的研究[57]。以上研究表明,运动及运动预处理可能通过调节线粒体凋亡途径发挥其心肌保护作用,未来需开展更多高质量的研究进一步证实其有效性。 2.2.3 运动可改善MIRI后线粒体的质量控制障碍 MIRI后心肌内线粒体出现的线生物发生减弱、粒体动力学失衡、自噬活性降低以及能量代谢障碍等导致心肌受损。因此,如何增强线粒体生物发生,维持心肌中线粒体的分裂融合平衡,提高线粒体自噬活性以及调节能量代谢障碍对改善MIRI后心肌中线粒体质量控制障碍有着举足轻重的作用。 线粒体生物发生是改善神经元能量代谢,维护mtDNA,平衡机体内环境的重要途径。mtDNA受损是造成MIRI后心肌细胞线粒体生物发生障碍的重要原因之一。TAO等[58]研究发现,游泳训练可以使mtDNA复制和转录的适应性增加,激活PGC-1α信号传导,增强线粒体生物发生,改善心肌细胞的能量代谢,减轻急性的心肌损伤。同样,在BUDIONO等[59]也在研究中发现,与MIRI模型组小鼠相比,跑步运动有效改善了大鼠mtDNA复制和转录,上调了沉默调节蛋白3、PCG-1α等线粒体生物发生相关基因的表达,最终发挥其神经保护作用,但研究也进一步发现,运动对心脏的保护作用并非剂量依赖性的。此外,研究发现,不仅在动物模型上发生了此类现象,在健康运动员的血浆中也发现,运动会增加mtDNA复制和转录以及线粒体呼吸链的活动[60]。由此可见,运动调节心肌线粒体完整性和生物发生,对于再灌注后心肌细胞的损伤具有一定的缓解作用,然而,对于运动训练周期的制定仍有待深入研究。 有研究表示,与MIRI模型组相比,8周的有氧运动能促使大鼠的心肌梗死大小与Drp1降低,从而诱导线粒体裂变的调节以达到对心脏的保护[61]。同样,MISHRA等[62]也在研究中得出有氧训练可以诱导大鼠Bax和Drp1蛋白降低,并且抑制了Caspase-3活化,从而促使大鼠线粒体功能得到改善以达到对MIRI后心脏的保护。此外,JIANG等[63]在研究中发现,急性心肌缺血会导致不良的线粒体网络动力学(高分裂低融合),较低线粒体呼吸水平,有氧间歇训练可以促进视神经萎缩症蛋白1(optic atrophy1,OPA1)等线粒体融合蛋白的表达,抑制Drp1的表达,改善线粒体能量代谢,促进心肌重塑。综上可见,平衡线粒体动力学在治疗MIRI中十分关键,这一结论也在MUI等[64]的研究中被证实。 运动作为一种临床可操作性强且不良反应较小的康复方法,可控制MIRI后心肌自噬表达,有效缓解心肌线粒体损伤,促进心肌功能恢复。有研究表示,运动预处理通过调节PTEN诱导的激酶1(PINK1)介导的线粒体自噬途径清除受损线粒体,发挥其对缺血缺氧心肌的保护作用[65]。同样,唐强等[66]通过实验研究发现,MIRI导致心肌细胞及线粒体损伤严重,而运动与处理组大鼠心肌细胞损伤轻微,线粒体结构较为完整,自噬体减少,这表明运动预处理可以通过抑制线粒体自噬水平缓解心肌损伤。与此相反,LIU等[67]在研究中发现,晚期运动预处理可以激活线粒体自噬,并且激活的自噬部分参与对缺血缺氧的心肌保护。此外,李宏玉等[68]还得出,与MIRI模型组相比,运动预处理可上调自噬相关蛋白LC3-Ⅰ,抑制Beclin 1、LC3-Ⅱ的表达,从而将线粒体自噬控制在适度水平。以上研究表明,运动可以调节适度的线粒体自噬,缓解心肌缺血再灌注损伤。 "

| [1] BOATENG S, SANBORN T. Acute myocardial infarction. Dis Mon. 2013; 59(3):83-96. [2] FERNANDEZ RICO C, KONATE K, JOSSE E, et al. Therapeutic Peptides to Treat Myocardial Ischemia-Reperfusion Injury. Front Cardiovasc Med. 2022;9:792885. [3] 田野.活性氧在急性心肌梗死心肌再灌注损伤中的作用和机制[J].临床心血管病杂志,2017,33(7):611-614. [4] WANG J, TOAN S, ZHOU H. Mitochondrial quality control in cardiac microvascular ischemia-reperfusion injury: New insights into the mechanisms and therapeutic potentials. Pharmacol Res. 2020;156: 104771. [5] XU T, DING W, AO X, et al. ARC regulates programmed necrosis and myocardial ischemia/reperfusion injury through the inhibition of mPTP opening. Redox Biol. 2019;20:414-426. [6] KURIAN GA, RAJAGOPAL R, VEDANTHAM S, et al. The Role of Oxidative Stress in Myocardial Ischemia and Reperfusion Injury and Remodeling: Revisited. Oxid Med Cell Longev. 2016;2016:1656450. [7] ZHOU M, YU Y, LUO X, et al. Myocardial Ischemia-Reperfusion Injury: Therapeutics from a Mitochondria-Centric Perspective. Cardiology. 2021;146(6):781-92. [8] WU Y, LENG Y, MENG Q, et al. Suppression of Excessive Histone Deacetylases Activity in Diabetic Hearts Attenuates Myocardial Ischemia/Reperfusion Injury via Mitochondria Apoptosis Pathway. J Diabetes Res. 2017;2017:8208065. [9] MUI D, ZHANG Y. Mitochondrial scenario: roles of mitochondrial dynamics in acute myocardial ischemia/reperfusion injury. J Recept Signal Transduct Res. 2021;41(1):1-5. [10] YANG M, LINN BS, ZHANG Y, et,al. Mitophagy and mitochondrial integrity in cardiac ischemia-reperfusion injury. Biochim Biophys Acta Mol Basis Dis. 2019;1865(9):2293-2302. [11] TAO L, BEI Y, LIN S, et al. Exercise Training Protects Against Acute Myocardial Infarction via Improving Myocardial Energy Metabolism and Mitochondrial Biogenesis. Cell Physiol Biochem. 2015;37(1):162-175. [12] CORONADO M, FAJARDO G, NGUYEN K, et al. Physiological Mitochondrial Fragmentation Is a Normal Cardiac Adaptation to Increased Energy Demand. Circ Res. 2018;122(2):282-295. [13] KUZNETSOV AV, JAVADOV S, MARGREITER R, et al. The Role of Mitochondria in the Mechanisms of Cardiac Ischemia-Reperfusion Injury. Antioxidants (Basel). 2019;8(10):454. [14] PIZZINO G, IRRERA N, CUCINOTTA M, et al. Oxidative Stress: Harms and Benefits for Human Health. Oxid Med Cell Longev. 2017;2017:8416763. [15] BUGGER H, PFEIL K. Mitochondrial ROS in myocardial ischemia reperfusion and remodeling. Biochim Biophys Acta Mol Basis Dis. 2020; 1866(7):165768. [16] ANDREADOU I, SCHULZ R, PAPAPETROPOULOS A, et al. The role of mitochondrial reactive oxygen species, NO and H(2) S in ischaemia/reperfusion injury and cardioprotection. J Cell Mol Med. 2020;24(12): 6510-6522. [17] WANG L, NIU H, ZHANG J. Homocysteine induces mitochondrial dysfunction and oxidative stress in myocardial ischemia/reperfusion injury through stimulating ROS production and the ERK1/2 signaling pathway. Exp Ther Med. 2020;20(2):938-944. [18] BRADLEY JM, LI Z, ORGAN CL, et al. A novel mtDNA repair fusion protein attenuates maladaptive remodeling and preserves cardiac function in heart failure. Am J Physiol Heart Circ Physiol. 2018;314(2):H311-h321. [19] CHEN Q, YOUNUS M, THOMPSON J, et al. Intermediary metabolism and fatty acid oxidation: novel targets of electron transport chain-driven injury during ischemia and reperfusion. Am J Physiol Heart Circ Physiol. 2018;314(4):H787-h795 [20] XU X, LAI Y, HUA ZC. Apoptosis and apoptotic body: disease message and therapeutic target potentials. Biosci Rep. 2019;39(1):BSR20180992. [21] LI Y, LIU X. Novel insights into the role of mitochondrial fusion and fission in cardiomyocyte apoptosis induced by ischemia/reperfusion. J Cell Physiol. 2018;233(8):5589-5597. [22] GRANCARA S, OHKUBO S, ARTICO M, et al. Milestones and recent discoveries on cell death mediated by mitochondria and their interactions with biologically active amines. Amino Acids. 2016;48(10): 2313-2326. [23] ZHOU H, ZHANG Y, HU S, et al. Melatonin protects cardiac microvasculature against ischemia/reperfusion injury via suppression of mitochondrial fission-VDAC1-HK2-mPTP-mitophagy axis. J Pineal Res. 2017;63(1):e12413. [24] FANG R, ZHANG LL, ZHANG LZ, et al. Sphingosine 1-Phosphate Postconditioning Protects Against Myocardial Ischemia/reperfusion Injury in Rats via Mitochondrial Signaling and Akt-Gsk3β Phosphorylation. Arch Med Res. 2017;48(2):147-155. [25] WANG Y, YUAN Y, WANG X, et al. Tilianin Post-Conditioning Attenuates Myocardial Ischemia/Reperfusion Injury via Mitochondrial Protection and Inhibition of Apoptosis. Med Sci Monit. 2017;23:4490-4499. [26] LI J, HU HP, LI Y, et al. Influences of remifentanil on myocardial ischemia-reperfusion injury and the expressions of Bax and Bcl-2 in rats. Eur Rev Med Pharmacol Sci. 2018;22(24):8951-8960. [27] LUO Y, WAN Q, XU M, et al. Nutritional preconditioning induced by astragaloside Ⅳ on isolated hearts and cardiomyocytes against myocardial ischemia injury via improving Bcl-2-mediated mitochondrial function. Chem Biol Interact. 2019;309:108723. [28] YUAN Y, CAO W, HONG Y, et al. Tilianin pretreatment prevents myocardial ischemia-reperfusion injury via preservation of mitochondrial function in rat heart. Phytomedicine. 2017;34:106-114. [29] 张坦,孙易,丁树哲.运动介导AMPK调控线粒体质量控制的机制研究进展[J].中国体育科技,2018,54(6):97-102. [30] JIN Q, LI R, HU N, et al. DUSP1 alleviates cardiac ischemia/reperfusion injury by suppressing the Mff-required mitochondrial fission and Bnip3-related mitophagy via the JNK pathways. Redox Biol. 2018;14:576-587. [31] POPOV LD. Mitochondrial biogenesis: An update. J Cell Mol Med. 2020; 24(9):4892-4899. [32] YUE R, XIA X, JIANG J, et al. Mitochondrial DNA oxidative damage contributes to cardiomyocyte ischemia/reperfusion-injury in rats: cardioprotective role of lycopene. J Cell Physiol. 2015;230(9):2128-2141. [33] QI X, WANG J. Melatonin improves mitochondrial biogenesis through the AMPK/PGC1α pathway to attenuate ischemia/reperfusion-induced myocardial damage. Aging (Albany NY). 2020;12(8):7299-7312. [34] SHANMUGAM K, RAVINDRAN S, KURIAN GA, et al. Fisetin Confers Cardioprotection against Myocardial Ischemia Reperfusion Injury by Suppressing Mitochondrial Oxidative Stress and Mitochondrial Dysfunction and Inhibiting Glycogen Synthase Kinase 3β Activity. Oxid Med Cell Longev. 2018;2018:9173436. [35] DORN GW, 2ND, KITSIS RN. The mitochondrial dynamism-mitophagy-cell death interactome: multiple roles performed by members of a mitochondrial molecular ensemble. Circ Res. 2015;116(1):167-182. [36] MANEECHOTE C, PALEE S, KERDPHOO S, et al. Balancing mitochondrial dynamics via increasing mitochondrial fusion attenuates infarct size and left ventricular dysfunction in rats with cardiac ischemia/reperfusion injury. Clin Sci (Lond). 2019;133(3):497-513. [37] YANG Y, ZHAO L, MA J. Penehyclidine hydrochloride preconditioning provides cardiac protection in a rat model of myocardial ischemia/reperfusion injury via the mechanism of mitochondrial dynamics mechanism. Eur J Pharmacol. 2017;813:130-139. [38] ZINGARELLI B, CUZZOCREA S, ZSENGELLÉR Z, et al. Protection against myocardial ischemia and reperfusion injury by 3-aminobenzamide, an inhibitor of poly (ADP-ribose) synthetase. Cardiovasc Res. 1997;36(2): 205-215. [39] ZHOU XL, WU X, XU QR, et al. Notch1 provides myocardial protection by improving mitochondrial quality control. J Cell Physiol. 2019;234(7): 11835-11841. [40] ONISHI M, YAMANO K, SATO M, et al. Molecular mechanisms and physiological functions of mitophagy. Embo J. 2021;40(3):e104705. [41] ZHU W, LIU F, WANG L, et al. pPolyHb protects myocardial H9C2 cells against ischemia-reperfusion injury by regulating the Pink1-Parkin-mediated mitochondrial autophagy pathway. Artif Cells Nanomed Biotechnol. 2019;47(1):1248-1255. [42] ZHANG X, LI Q, SUN Y, et al. Chondroitin sulfate from sturgeon bone protects rat chondrocytes from hydrogen peroxide-induced damage by maintaining cellular homeostasis through enhanced autophagy. Int J Biol Macromol. 2020;164:2761-8. [43] TANG L, LI YP, HU J, et al. Dexpramipexole attenuates myocardial ischemia/reperfusion injury through upregulation of mitophagy. Eur J Pharmacol. 2021;899:173962. [44] XIAO Y, CHEN W, ZHONG Z, et al. Electroacupuncture preconditioning attenuates myocardial ischemia-reperfusion injury by inhibiting mitophagy mediated by the mTORC1-ULK1-FUNDC1 pathway. Biomed Pharmacother. 2020;127:110148. [45] RANJBAR K, ZARRINKALAM E, SALEHI I, et al. Cardioprotective effect of resistance training and Crataegus oxyacantha extract on ischemia reperfusion–induced oxidative stress in diabetic rats. Biomed Pharmacother. 2018;100:455-460. [46] LEE Y, MIN K, TALBERT EE, et al. Exercise protects cardiac mitochondria against ischemia-reperfusion injury. Med Sci Sports Exerc. 2012;44(3): 397-405. [47] 许思毛,刘书,王第亮,等.较大强度耐力运动预处理对I/R心肌SOD2表达及其活性影响的研究[J].北京体育大学学报,2014,37(11): 73-78 [48] 李丽,仝武军,方立,等.间歇有氧运动训练改善肥胖小鼠缺血后心脏功能恢复[J].心脏杂志,2017,29(3):281-285 [49] RUI F, CAI M, WANG X, et al. Early Aerobic Exercise Combined with Hydrogen-Rich Saline as Preconditioning Protects Myocardial Injury Induced by Acute Myocardial Infarction in Rats. Appl Biochem Biotechnol. 2019;187(3):663-676. [50] FRENCH JP, HAMILTON KL, QUINDRY JC, et al. Exercise-induced protection against myocardial apoptosis and necrosis: MnSOD, calcium-handling proteins, and calpain. FASEB J. 2008;22(8):2862-2871. [51] 于新彦,杨省利.有氧运动训练对大鼠心肌缺血/再灌注损伤致心肌细胞凋亡的作用[J].心脏杂志,2015,27(6):659-661+665. [52] ZHANG KR, LIU HT, ZHANG HF, et al. Long-term aerobic exercise protects the heart against ischemia/reperfusion injury via PI3 kinase-dependent and Akt-mediated mechanism. Apoptosis. 2007;12(9): 1579-1588. [53] 彭峰林,张林,彭玉宇,等. 间歇运动对I/R损伤大鼠心肌细胞凋亡的影响[J].广西师范大学学报(自然科学版),2009,27(4):119-123. [54] 刘志梅,于珊珊,贾佳,等. 间歇游泳训练对大鼠心肌缺血再灌注损伤的保护作用[J]. 新乡医学院学报,2015,32(9):817-820. [55] LAI CC, TANG CY, FU SK, et al. Effects of swimming training on myocardial protection in rats. Biomed Rep. 2022;16(3):1-13. [56] SMENES BT, BÆKKERUD FH, SLAGSVOLD KH, et al. Acute exercise is not cardioprotective and may induce apoptotic signalling in heart surgery: a randomized controlled trial. Interact Cardiovasc Thorac Surg. 2018;27(1):95-101. [57] YOUSEF D, FARH GS, GHIASSIE R, et al. Effect of Resistance Exercise on Cardiac Apoptosis Following of Ischemic/Reperfusion. Journal of Animal Veterinary Advances. 2011;10(19):2561-2566. [58] TAO L, BEI Y, ZHANG H, et al. Exercise Training Protects Against Acute Myocardial Infarction via Improving Myocardial Energy Metabolism and Mitochondrial Biogenesis. Cell Physiol Biochem. 2015;37(1):162-175. [59] BUDIONO BP, HOE L, PEART JN, et al. Effects of voluntary exercise duration on myocardial ischaemic tolerance, kinase signaling and gene expression. Life Sci. 2021;(3):119253. [60] ZANINI G, DE GAETANO A, SELLERI V, et al. Mitochondrial DNA and Exercise: Implications for Health and Injuries in Sports. Cells. 2021; 10(10):2575. [61] GHAHREMANI R, DAMIRCHI A, SALEHI I, et al. Mitochondrial dynamics as an underlying mechanism involved in aerobic exercise training-induced cardioprotection against ischemia-reperfusion injury. Life Sci. 2018;213:102-108. [62] MISHRA J, KWOK WM, FITTS RH, et al. Exercise induced Increase in Hexokinase II itochondria Association Reduces Cardiac Ischemia in eperfusion Injury in Rats. FASEB J. 2020;34(S1):1-1. [63] JIANG HK, WANG YH, SUN L, et al. Aerobic interval training attenuates mitochondrial dysfunction in rats post-myocardial infarction: roles of mitochondrial network dynamics. Int J Mol Sci. 2014;15(4):5304-5322. [64] MUI D, ZHANG Y. Mitochondrial scenario: roles of mitochondrial dynamics in acute myocardial ischemia/reperfusion injury. J Recept Signal Transduct Res. 2021;41(1):1-5. [65] YUAN Y, PAN SS. Parkin Mediates Mitophagy to Participate in Cardioprotection Induced by Late Exercise Preconditioning but Bnip3 Does Not. J Cardiovasc Pharmacol. 2018;71(5):303-316. [66] 唐强,张继瑶,李宏玉,等.基于细胞自噬探讨运动预处理对大鼠心肌缺血再灌注损伤的保护作用[J]. 康复学报,2019,29(5):40-45. [67] LIU H T, PAN SS. Late Exercise Preconditioning Promotes Autophagy against Exhaustive Exercise-Induced Myocardial Injury through the Activation of the AMPK-mTOR-ULK1 Pathway. Biomed Res Int. 2019; 2019:5697380. [68] 李宏玉,张继瑶,张钰,等.运动预处理对心肌缺血再灌注损伤大鼠自噬相关蛋白表达及心肌细胞凋亡的影响[J].中国康复理论与实践,2020,26(8):903-907. |
| [1] | Guo Shuhui, Yang Ye, Jiang Yangyang, Xu Jianwen. Screening and validation of neurogenic bladder miRNA-mRNA regulatory network [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(在线): 1-8. |
| [2] | Fang Xingyan, Tian Zhenli, Zhao Zheyi, Wen Ping, Xie Tingting. Effects of sodium arsenite on human umbilical vein endothelial cell injury and sphingosine kinases 1/sphingosine 1-phosphate signaling axis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(在线): 1-7. |
| [3] | Dang Yi, Du Chengyan, Yao Honglin, Yuan Nenghua, Cao Jin, Xiong Shan, Zhang Dingmei, Wang Xin. Hormonal osteonecrosis and oxidative stress [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1469-1476. |
| [4] | Wu Dongzhe, Gao Xiaolin, Li Chuangtao, Wang Hao. Constructing the prediction model of maximal oxygen uptake by back-propagation neural network based on the cardiorespiratory optimal point [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1224-1231. |
| [5] | Wang Ji, Zhang Min, Yang Zhongya, Zhang Long. A review of physical activity intervention in type 2 diabetes mellitus with sarcopenia [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1272-1277. |
| [6] | Nie Chenchen, Su Kaiqi, Gao Jing, Fan Yongfu, Ruan Xiaodi, Yuan Jie, Duan Zhaoyuan, Feng Xiaodong. The regulatory role of circular RNAs in cerebral ischemia-reperfusion injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1286-1291. |
| [7] | Liang Jiaqi, Liu Hengxu, Yang Jinxin, Yang Yi, Deng Xuhui, Tan Mingjian, Luo Jiong. Health benefit relationship between exercise and intestinal bacteria [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1292-1299. |
| [8] | Huang Linke, Wei Linhua, Jiang Jie, Liu Qian, Chen Weiwei. Effects of estrogen combined with treadmill exercise on bone mass and articular cartilage in ovariectomized mice [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1166-1171. |
| [9] | Ruan Ling, Wang Guanghua, Wu Rongping, Jin Zhan, Lyu Zhenqing, Zhang Nan, Li Shoubang. Correlation between exercise intensity and lipid metabolism disorder and oxidative stress in a high-diet rat model [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1149-1155. |
| [10] | Zhang Yan, He Ruibo, Wang Qingbo, Pi Yihua, Lu Chunmin, Xu Chuanyi, Ma Gang, Peng Peng. Effects of aerobic exercises with different load volumes on inflammatory response and insulin signaling pathway of skeletal muscle in obese rats [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1237-1244. |
| [11] | Tian Qinyu, Tian Xinggui, Tian Zhuang, Sui Xiang, Liu Shuyun, Lu Xiaobo, Guo Quanyi. Protection of manganese oxide nanoparticles for bone marrow mesenchymal stem cell spreading against oxidative stress [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 821-826. |
| [12] | Cui Lianxu, Jiang Wenkang, Lu Dahong, Xu Junrong, Liu Xiaocui, Wang Bingyun. Clinical-grade human umbilical cord mesenchymal stem cells affect the improvement of neurological function in rats with traumatic brain injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 835-839. |
| [13] | Li Qicheng, Deng Jin, Fu Xiaoyang, Han Na. Effects of bone marrow mesenchymal stem cells-derived exosomes on hypoxia-treated myoblasts [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 853-859. |
| [14] | Li Yujiao, Su Kunxia. High-intensity endurance exercise influences browning of white adipose tissue in a mouse model of high-fat diet induced obesity [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(5): 707-713. |
| [15] | Zhang Qing, Gao Chunlan, Yu Feifei, Zhang Zhenghao, Ma Fang, Gao Yuan, Li Guizhong, Jiang Yideng, Ma Shengchao. Ephrin A receptor 2 DNA methylation increases in pancreatic beta cell apoptosis induced by homocysteine [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(5): 714-719. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||

