Chinese Journal of Tissue Engineering Research ›› 2022, Vol. 26 ›› Issue (32): 5242-5247.doi: 10.12307/2022.921
Long non-coding RNA and osteoporosis
Kang Teng1, Lan Fengjun2, Qin Hao2, Liu Gang1, 2
- 1Department of Orthopedics, the Affiliated Hospital of Guizhou Medical University, Guiyang 550004, Guizhou Province, China; 2School of Clinical Medicine, Guizhou Medical University, Guiyang 550004, Guizhou Province, China
-
Received:2021-11-25Accepted:2022-01-29Online:2022-11-18Published:2022-05-14 -
Contact:Liu Gang, MD, Chief physician, Department of Orthopedics, the Affiliated Hospital of Guizhou Medical University, Guiyang 550004, Guizhou Province, China; School of Clinical Medicine, Guizhou Medical University, Guiyang 550004, Guizhou Province, China -
About author:Kang Teng, Master candidate, Department of Orthopedics, the Affiliated Hospital of Guizhou Medical University, Guiyang 550004, Guizhou Province, China -
Supported by:the Basic Research Program of Science and Technology Bureau of Guizhou Province, No. J-2019-41 (to LG)
CLC Number:
Cite this article
Kang Teng, Lan Fengjun, Qin Hao, Liu Gang. Long non-coding RNA and osteoporosis[J]. Chinese Journal of Tissue Engineering Research, 2022, 26(32): 5242-5247.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
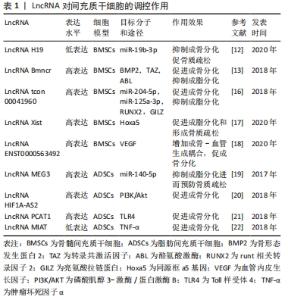
2.1 LncRNA对间充质干细胞的调控作用 间充质干细胞具有多向分化和无限增殖潜能,包括胚胎干细胞、骨髓间充质干细胞、脂肪间充质干细胞、滑膜间充质干细胞等[7]。间充质干细胞在特定诱导条件下可以分化为成骨细胞、软骨细胞、脂肪细胞、支持造血的基质细胞等多种组织细胞[8]。间充质干细胞已广泛应用于组织工程和再生医学领域,目前,LncRNA通过调控骨髓间充质干细胞和脂肪间充质干细胞在骨质疏松症发生机制中的研究相对较多。 2.1.1 LncRNA对骨髓间充质干细胞的调控作用 骨髓间充质干细胞向成骨细胞谱系分化是预防骨质疏松发生和形成的基础,骨髓间充质干细胞分化受到来自细胞外环境的机械信号和分子信号等多种信号的精确调控,主要体现在转录和转录后水平[9-10]。近年来研究证实,LncRNA已成为骨髓间充质干细胞成骨过程中有前途的新型调节因子。ZHANG等[11]采用人全转录组芯片对人骨髓间充质干细胞成骨分化过程中编码和非编码转录本表达谱和功能网络分析进行了研究,发现其中785个LncRNA显著上调,623个LncRNA显著下调,这揭示了LncRNA在骨髓间充质干细胞定向分化方向中可能起着非同一般的调节作用。XIAO LING等[12]研究发现LncRNA H19在绝经后骨质疏松症患者中明显下调,而低表达H19则可以通过上调miR-19b-3p水平抑制骨髓间充质干细胞的增殖和成骨分化,从而加剧骨质疏松,这揭示了H19在绝经后骨质疏松症中的重要作用。除此之外,LncRNA Bmncr还可以通过调节细胞外基质蛋白、纤维调节蛋白和激活骨形态发生蛋白2(BMP2)信号通路来调控骨髓间充质干细胞的成骨生态位,并作为支架促进转录共激活因子TAZ和ABL酪氨酸激酶的相互作用,转录共激活因子TAZ可参与Runt 相关转录因子2(runt-related transcription factor 2,RUNX2)/过氧化物酶体增殖物激活受体γ(peroxisome proliferator-activated receptor γ,PPARγ)转录复合物的组装,从而促进骨髓间充质干细胞成骨分化和抑制其成脂分化[13]。骨髓间充质干细胞在一定的诱导条件下也能够分化为脂肪细胞,而脂肪的形成与骨形成呈反比关系,成骨和成脂分化之间的不平衡容易引发骨质疏松症,所以抑制PPARγ等成脂因子可能有利于骨髓间充质干细胞向成骨细胞分化而不是向脂肪细胞谱系分化[14]。亮氨酸拉链蛋白(GILZ)是主要成脂因子PPARγ2的抑制剂,可以通过抑制PPARγ2启动子上的CCAAT/增强子结合蛋白(C/EBP)元件参与抑制PPARγ2启动子的转录[15]。SHANG等[16]发现过表达LncRNA tcon00041960不仅靶向调控miR-204-5p增加成骨因子RUNX2的表达,促进成骨分化,并且还可以靶向调控miR-125a-3p增加亮氨酸拉链蛋白的水平,由此阻止骨髓间充质干细胞向脂肪分化,这可能有助于延缓骨质疏松的进程。人同源框a5(Hoxa5)基因能促进骨髓间充质干细胞向脂肪细胞分化,最近研究表明,LncRNA Xist可能通过增加人同源框a5的表达水平形成衰老性骨质疏松[17]。此外,LncRNA ENST0000563492还可以通过增加骨髓间充质干细胞成骨分化过程中血管内皮生长因子的表达,改善成骨-血管生成耦合过程,在体内促进骨髓间充质干细胞成骨预防骨质疏松[18]。此类有关骨质疏松症的研究还有很多,这些结果增加了对LncRNA功能网络的认识,其能通过影响骨髓间充质干细胞的生物学特性来调控骨质疏松症疾病。 2.1.2 LncRNA对脂肪间充质干细胞的调控作用 脂肪间充质干细胞具有分化为成骨细胞和脂肪细胞等多种细胞系的能力,因此具有治疗骨质疏松的潜力。目前,在脂肪间充质干细胞多向分化中LncRNA的研究还很有限。LI等[19]研究发现LncRNA MEG3可以通过靶向调控miR-140-5p抑制人脂肪间充质干细胞成脂分化进而抑制骨质疏松症的发生。LncRNA HIF1A-AS2可海绵化miR-665,从而上调白细胞介素6激活磷酸肌醇3-激酶/蛋白激酶B(phosphatidylinositol 3-kinase/protein kinase B,PI3K/Akt)信号通路,加速脂肪间充质干细胞的成骨过程[20]。在脂肪间充质干细胞成骨分化中lncRNA PCAT1和Toll样受体4表达水平上调,LncRNA PCAT1负调控miR-145-5p,通过激活Toll样受体信号通路,间接上调Toll样受体4(TLR4)的表达,促进人脂肪间充质干细胞成骨分化[21]。JIN等[22]发现在人脂肪间充质干细胞成骨分化过程中LncRNA MIAT的表达明显下调,体外敲除LncRNA MIAT可显著促进人脂肪间充质干细胞成骨分化,其机制可能是敲低LncRNA MIAT可以抑制肿瘤坏死因子α(TNF-α)水平进而促进脂肪间充质干细胞成骨分化。 综上所述,LncRNA可以通过调节骨髓间充质干细胞、脂肪间充质干细胞向成骨方向分化和抑制其成脂分化来参与骨质疏松症的防治,见表1。 "
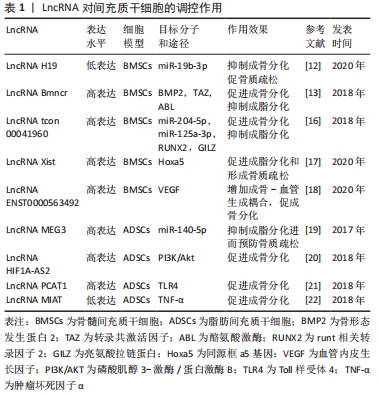
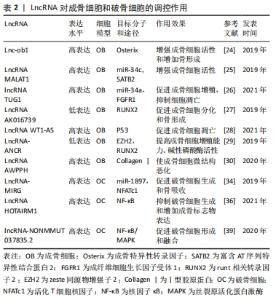
2.2 LncRNA对成骨细胞的调控作用 成骨细胞主要来源于间充质干细胞,并合成复杂的细胞外基质,包括骨钙素、碱性磷酸酶和Ⅰ型胶原蛋白等,随后沉积并矿化形成新骨[23],调节和影响骨的形成和重建过程。成骨细胞数量减少和功能降低均会使骨密度下降,导致骨质疏松症的发生。因此,骨质疏松症等代谢性骨病最重要的预防和治疗方式便是诱导间充质干细胞向成骨细胞分化和增加成骨细胞的活力。大量的研究表明,LncRNA在成骨细胞的增殖、分化、凋亡中发挥着重要的调节作用。 LncRNA根据功能在成骨细胞中可大致分为两类:①促进成骨,如Sun等[24]发现 Lnc-ob1在成骨细胞中表达丰富,在成骨形成过程中表达上调,并通过上调成骨特异性转录因子Osterix调节小鼠成骨细胞活性和骨形成率,在成骨细胞中特异性过表达Lnc-ob1可以抵抗卵巢切除诱导的小鼠骨质疏松症,这也许是应用成骨细胞治疗骨质疏松症的药物靶点。Yang等[25]研究发现骨髓间充质干细胞来源外泌体LncRNA MALAT1能够通过与miR-34c结合,促进成骨细胞中富含AT序列特异性结合蛋白2(SATB2)蛋白的表达,从而增强骨质疏松小鼠成骨细胞的活性,而沉默LncRNA MALAT1则降低了成骨细胞的活性和矿化结节的数量。WANG等[26]发现机械负荷的增加可以促进骨形成,在流体剪切应力作用下,lncRNA TUG1可以上调成纤维细胞生长因子受体1(FGFR1)的表达,从而促进成骨细胞增殖,抑制成骨细胞凋亡。②抑制成骨,YIN等[27]研究发现一种新型的LncRNA AK016739,敲低LncRNA AK016739可以上调成骨因子RUNX2的表达,从而促进转录因子TCF7/LEF1的表达和活性来促进成骨细胞分化和骨形成。LncRNA WT1-AS在骨质疏松患者血浆中表达上调,过表达LncRNA WT1-AS可导致凋亡基因p53表达升高,而P53是人体一种重要的抑癌基因,与细胞周期的调控、DNA的修复、细胞分化、细胞凋亡等重要的生物学功能相关,二者相互作用共同调控成骨细胞凋亡[28],LncRNA-ANCR在绝经后骨质疏松症患者中表达增加,沉默LncRNA-ANCR可减少其与zeste同源物增强子2 (EZH2)的结合,增加RUNX2基因的表达,显著提高成骨细胞增殖、碱性磷酸酶活性,从而使成骨细胞增殖能力更高,钙沉积更稳定[29]。Ⅰ型胶原α1与α2的比例(2∶1)是维持正常骨微结构所必需的,此比例的改变会引起胶原同质三聚体的形成和骨骼微结构的恶化,过表达LncRNA AWPPH导致成骨细胞中Ⅰ型胶原α1下调而Ⅰ型胶原α2上调,使Ⅰ型胶原α1与α2的比值低于2∶1,从而致使骨质疏松症的发生[30]。 2.3 LncRNA对破骨细胞的调控作用 骨质疏松症的发生与破骨细胞的活性增强息息相关,破骨细胞是起源于单核/巨噬细胞前体细胞的多核细胞,其可分泌盐酸和蛋白水解酶,形成骨吸收陷窝,病理状态下其可溶解骨矿物质并消化骨基质导致骨质疏松和骨破坏[31]。NF-κB配体受体激活因子(receptor activator of nuclear factor kappa B-ligand,RANKL)和巨噬细胞集落刺激因子(M-CSF)是单核/巨噬细胞前体细胞向破骨细胞分化的2个重要的调控因子,RANKL受体(RANK)是位于破骨细胞表面的肿瘤坏死因子受体超家族成员,RANKL通过结合并激活其受体RANK来促进破骨细胞分化[32]。研究表明,雌激素缺乏、免疫学疾病和恶性骨紊乱能够激活RANKL/RANK通路,从而促进破骨细胞分化,最终导致骨质疏松症的发生[33]。LncRNA可以通过RANKL/RANK通路调节破骨细胞的增殖、分化、凋亡,从而调控骨质疏松症。LING等[34]发现LncRNA-MIRG在破骨细胞中表达上调,其可以作为分子海绵通过调节miR-1897对活化T细胞核因子(NFATc1)的抑制作用,促进破骨细胞生成和骨吸收,从而导致骨质疏松症。NF-κB家族包括p50、p52、RelA(p65)、RelB和c-Rel,抑制NF-κB信号通路需要抑制IKBs蛋白,包括IKBα、IKBβ和IKBε[35]。LncRNA HOTAIRM1 可以通过抑制p65和IKBα的磷酸化,减弱 RANKL 介导的磷酸化p65和IKBα的增强,表明 LncRNA HOTAIRM1 能够通过灭活 NF-κB通路促进成骨分化和抑制破骨细胞分化,预防骨质疏松症[36]。骨保护素是RANKL的天然拮抗剂,可抑制RANKL与其受体RANK的结合,从而阻断破骨细胞前体向成熟破骨细胞分化[37],CHE等[38]首次发现RANKL可以诱导LncRNA MALAT1的表达,LncRNA MALAT1敲低可逆转RANKL诱导的细胞生长抑制和细胞周期阻滞,并且还可以上调骨保护素的表达进而促进成骨分化。lncRNA-NONMMUT037835.2可抑制破骨细胞分化,而下调lncRNA-NONMMUT037835.2促进破骨细胞形成和融合,进一步的研究表明,lncRNA-NOMMUT037835.2可能通过负向调节RANK表达,抑制NF-κB/MAPK信号通路来调节破骨细胞的形成[39]。 以上这些研究结果都有助于更好地理解 LncRNA与成骨细胞和破骨细胞相互作用在骨质疏松症形成中的分子机制,见表2。 "
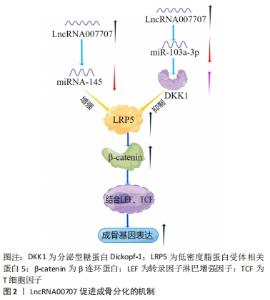
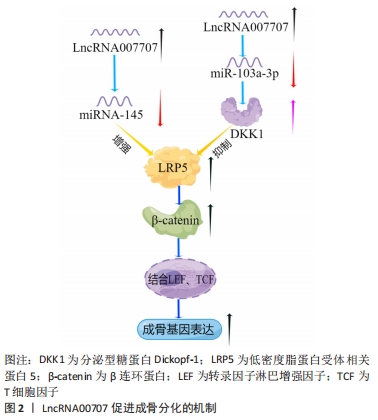
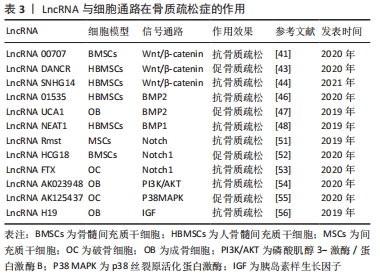
2.4.1 LncRNA与Wnt/β-catenin信号通路 在经典的Wnt/β-catenin信号通路中,Wnt蛋白与卷曲蛋白(Frizzled)的氨基端和低密度脂蛋白受体相关蛋白5/6(LRP5/6)结合,抑制β-catenin磷酸化,导致细胞内的β-catenin水平升高并进入细胞核与转录因子淋巴增强因子(lymphoid enhance factor,LEF)/T细胞因子(T-cell Factor,TCF)结合,最终诱导相关成骨靶基因的表达[40]。LncRNA能够通过Wnt/β-catenin信号通路发挥其骨代谢的调控作用,如LncRNA00707通过靶向调控miR-145上调LRP5的表达,激活Wnt/β-catenin通路促进人骨髓间充质干细胞的成骨分化[41]。上调的LncRNA00707同时也可以通过靶向miR-103a-3p增加DKK1的水平,DKK1是一种有效的LRP5/6拮抗剂,可抑制Wnt信号通路,从而抑制成骨相关基因的表达,见图2[42]。WANG等[43]发现在骨髓间充质干细胞成骨分化过程中LncRNA DANCR的表达水平降低,而CTNNB1的mRNA和蛋白水平均升高,CTNNB1编码β-catenin的基因,从而激活Wnt/β-catenin信号通路促进成骨分化,预防骨质疏松症。LIU等[44]报道了在骨质疏松症患者来源骨髓间充质干细胞中LncRNA SNHG14表达显著下调,过表达LncRNA SNHG14促进成骨分化, 其可能是通过下调Wnt1诱导信号通路蛋白2(WISP2)轴加速了骨质疏松症的进展。"
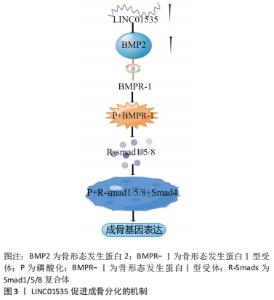
2.4.2 LncRNA与骨形态发生蛋白信号通路 骨形态发生蛋白是一类重要的分泌型骨诱导蛋白,属于转化生长因子超家族的一员,骨形态发生蛋白在许多细胞调控过程中发挥重要作用,包括骨和软骨细胞的分化和形成[45]。迄今为止,已在哺乳动物身上发现超过20多种骨形态发生蛋白配体,其中骨形态发生蛋白1,2,4,6,7,9在间充质干细胞分化中研究较为广泛,且不同的配体对受体的亲和性不同[45]。LINC01535能够增加骨形态发生蛋白2的表达水平,骨形态发生蛋白2配体可以与Ⅱ型受体结合,进而磷酸化Ⅰ型受体,Ⅰ型受体磷酸化下游的R-Smads(Smad1/5/8复合体),细胞质中磷酸化的R-Smads与Smad4结合形成转录复合物,转位进入核内上调成骨相关基因表达,见图3[46]。低水平的LncRNA UCA1能够上调成骨细胞中骨形态发生蛋白2、Smad1/5/8和Runx2蛋白的表达,从而促进成骨细胞的增殖、分化[47]。在骨质疏松症患者中骨形态发生蛋白1相对低表达,骨形态发生蛋白1被证明是miR-29b-3p的靶mRNA,LncRNA NEAT1可调控miR-29b-3p-BMP1轴促进成骨分化,进而防治骨质疏松[48]。"

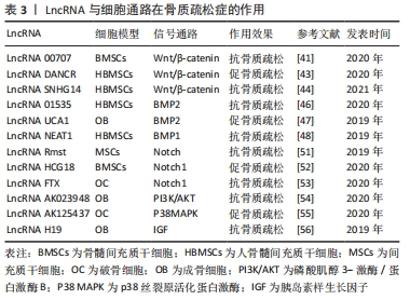
2.4.3 LncRNA与Notch信号通路 Notch是调节细胞生命进程的保守信号受体,在调节细胞增殖、分化、迁移及凋亡等方面具有重要作用[49]。哺乳动物中Notch受体有4种亚型,配体分别为Delta1,3,4及Jagged1,2,当Notch受体与配体结合后,分别在TAGE及γ-分泌酶的作用下水解释放Notch胞内活性片段(NICD),NICD向细胞核内转移激活并启动下游靶基因的表达[50]。LncRNA Rmst通过拮抗Notch信号通路,在BMP9/Smad诱导的间充质干细胞成骨分化中发挥重要的中介作用,沉默LncRNA Rmst可下调Notch受体和配体的表达,并抑制了骨形态发生蛋白9诱导的成骨、成软骨和成脂分化,而激活的Notch突变体可有效逆转这一过程[51]。LncRNA HCG18在骨质疏松症患者中表达升高,LncRNA HCG18促进NOTCH1的表达从而抑制骨髓间充质干细胞的成骨分化,慢病毒介导HCG18敲低可缓解后肢去负荷小鼠诱导的骨量丢失[52]。YU等[53]研究发现骨质疏松症患者骨和血清样本中的LncRNA FTX表达受到抑制,而miR-137表达则显著升高,下调的LncRNA FTX通过靶向miR-137抑制Notch1信号通路的激活,促进破骨细胞的形成,抑制成骨分化。 2.4.4 LncRNA与PI3K/AKT、P38MAPK、IGF信号通路 PI3K/AKT是参与调控细胞增殖、死亡和存活的重要信号通路,LncRNA AK023948可通过PI3K/AKT信号通路调节雌激素缺乏相关性骨质疏松大鼠成骨细胞AKT磷酸化水平,从而调节成骨细胞增殖[54]。MAPK信号通路在骨质疏松症骨代谢异常中起重要调节作用,LncRNA AK125437通过激活MAPK信号通路促进破骨细胞的增殖和分化,最终加剧骨质疏松症[55]。胰岛素样生长因子1是生长发育中的重要因子,与矿化相关基因如碱性磷酸酶、骨钙蛋白、Ⅰ型胶原蛋白的表达密切相关,LncRNA H19可以减轻miR-185-5p对胰岛素样生长因子1的抑制,从而增强成骨细胞基质矿化[56]。此外,在某些信号通路如Hedgehogs通路中LncRNA的作用尚未得到研究。 以上研究发现,多种信号通路共同介导和影响骨质疏松,见表3,并且这些信号通路不仅仅是单一的发挥作用,而是与其他信号通路共同协作组成复杂的调控网络。常见的细胞信号通路包括Wnt/β-catenin、BMP、Notch、PI3K/AKT、P38 MAPK、IGF等。LncRNA的调节作用将引起上述细胞通路的激活或抑制,从而改变靶基因表达来参与骨质疏松症的进程。 "
| [1] LI H, XIAO Z, QUARLES LD, et al. Osteoporosis: Mechanism, Molecular Target and Current Status on Drug Development. Curr Med Chem. 2021;28(8):1489-1507. [2] EL HAGE C, HALLIT S, AKEL M, et al. Osteoporosis awareness and health beliefs among Lebanese women aged 40 years and above. Osteoporos Int. 2019;30(4):771-786. [3] JIN F, LI J, ZHANG YB, et al. A functional motif of long noncoding RNA Nron against osteoporosis. Nat Commun. 2021;12(1):3319. [4] CHARLES RICHARD JL, EICHHORN PJA. Platforms for Investigating LncRNA Functions. SLAS Technol. 2018;23(6):493-506. [5] LI D, YANG C, YIN C, et al. LncRNA, Important Player in Bone Development and Disease. Endocr Metab Immune Disord Drug Targets. 2020;20(1):50-66. [6] GUO Q, GUO Q, XIAO Y, et al. Regulation of bone marrow mesenchymal stem cell fate by long non-coding RNA. Bone. 2020;141:115617. [7] LI WY, XIONG H. Study on MSCs homing and its research on osteodiseases. Zhongguo Gu Shang. 2020;33(7):689-692. [8] BAKER N, BOYETTE LB, TUAN RS. Characterization of bone marrow-derived mesenchymal stem cells in aging. Bone. 2015;70:37-47. [9] CHEN Q, SHOU P, ZHENG C, et al. Fate decision of mesenchymal stem cells: adipocytes or osteoblasts? Cell Death Differ. 2016;23(7): 1128-1139. [10] YI S, YU M, YANG S, et al. Tcf12, A Member of Basic Helix-Loop-Helix Transcription Factors, Mediates Bone Marrow Mesenchymal Stem Cell Osteogenic Differentiation In Vitro and In Vivo. Stem Cells. 2017; 35(2):386-397. [11] ZHANG W, DONG R, DIAO S, et al. Differential long noncoding RNA/mRNA expression profiling and functional network analysis during osteogenic differentiation of human bone marrow mesenchymal stem cells. Stem Cell Res Ther. 2017;8(1):30. [12] XIAOLING G, SHUAIBIN L, KAILU L. MicroRNA-19b-3p promotes cell proliferation and osteogenic differentiation of BMSCs by interacting with lncRNA H19. BMC Med Genet. 2020;21(1):11. [13] LI CJ, XIAO Y, YANG M, et al. Long noncoding RNA Bmncr regulates mesenchymal stem cell fate during skeletal aging. J Clin Invest. 2018; 128(12):5251-5266. [14] GUO J, REN R, YAO X, et al. PKM2 suppresses osteogenesis and facilitates adipogenesis by regulating β-catenin signaling and mitochondrial fusion and fission. Aging (Albany NY). 2020;12(4):3976-3992. [15] MOTA DE SÁ P, RICHARD AJ, Hang H, et al. Transcriptional Regulation of Adipogenesis. Compr Physiol. 2017;7(2):635-674. [16] SHANG G, WANG Y, XU Y, et al. Long non-coding RNA TCONS_00041960 enhances osteogenesis and inhibits adipogenesis of rat bone marrow mesenchymal stem cell by targeting miR-204-5p and miR-125a-3p. J Cell Physiol. 2018;233(8):6041-6051. [17] CHEN S, LI Y, ZHI S, et al. lncRNA Xist Regulates Osteoblast Differentiation by Sponging miR-19a-3p in Aging-induced Osteoporosis. Aging Dis. 2020;11(5):1058-1068. [18] OUYANG Z, TAN T, ZHANG X, et al. LncRNA ENST00000563492 promoting the osteogenesis-angiogenesis coupling process in bone mesenchymal stem cells (BMSCs) by functions as a ceRNA for miR-205-5p. Cell Death Dis. 2020;11(6):486. [19] LI Z, JIN C, CHEN S, et al. Long non-coding RNA MEG3 inhibits adipogenesis and promotes osteogenesis of human adipose-derived mesenchymal stem cells via miR-140-5p. Mol Cell Biochem. 2017; 433(1-2):51-60. [20] WU R, RUAN J, SUN Y, et al. Long non-coding RNA HIF1A-AS2 facilitates adipose-derived stem cells (ASCs) osteogenic differentiation through miR-665/IL6 axis via PI3K/Akt signaling pathway. Stem Cell Res Ther. 2018;9(1):348. [21] YU L, QU H, YU Y, et al. LncRNA-PCAT1 targeting miR-145-5p promotes TLR4-associated osteogenic differentiation of adipose-derived stem cells. J Cell Mol Med. 2018;22(12):6134-6147. [22] JIN C, ZHENG Y, HUANG Y, et al. Long non-coding RNA MIAT knockdown promotes osteogenic differentiation of human adipose-derived stem cells. Cell Biol Int. 2017;41(1):33-41. [23] KIM JM, LIN C, STAVRE Z, et al. Osteoblast-Osteoclast Communication and Bone Homeostasis. Cells. 2020;9(9):2073. [24] SUN Y, CAI M, ZHONG J, et al. The long noncoding RNA lnc-ob1 facilitates bone formation by upregulating Osterix in osteoblasts. Nat Metab. 2019;1(4):485-496. [25] YANG X, YANG J, LEI P, et al. LncRNA MALAT1 shuttled by bone marrow-derived mesenchymal stem cells-secreted exosomes alleviates osteoporosis through mediating microRNA-34c/SATB2 axis. Aging (Albany NY). 2019;11(20):8777-8791. [26] WANG X, HE J, WANG H, et al. Fluid shear stress regulates osteoblast proliferation and apoptosis via the lncRNA TUG1/miR-34a/FGFR1 axis. J Cell Mol Med. 2021;25(18):8734-8747. [27] YIN C, TIAN Y, YU Y, et al. A novel long noncoding RNA AK016739 inhibits osteoblast differentiation and bone formation. J Cell Physiol. 2019;234(7):11524-11536. [28] WANG C, XIE Q, SUN W, et al. lncRNA WT1-AS is upregulated in osteoporosis and regulates the apoptosis of osteoblasts by interacting with p53. Exp Ther Med. 2021;22(1):734. [29] CAI N, LI C, WANG F. Silencing of LncRNA-ANCR Promotes the Osteogenesis of Osteoblast Cells in Postmenopausal Osteoporosis via Targeting EZH2 and RUNX2. Yonsei Med J. 2019;60(8):751-759. [30] QIAN G, YU Y, DONG Y, et al. LncRNA AWPPH is downregulated in osteoporosis and regulates type I collagen α1 and α2 ratio. Arch Physiol Biochem. 2020:1-5. doi: 10.1080/13813455.2020.1767150. Online ahead of print. [31] CHEN RS, ZHANG XB, ZHU XT, et al. LncRNA Bmncr alleviates the progression of osteoporosis by inhibiting RANML-induced osteoclast differentiation. Eur Rev Med Pharmacol Sci. 2019;23(21):9199-9206. [32] UDAGAWA N, KOIDE M, NAKAMURA M, et al. Osteoclast differentiation by RANKL and OPG signaling pathways. J Bone Miner Metab. 2021; 39(1):19-26. [33] HE Y, CHEN Y. The potential role of lncRNAs in osteoporosis. J Bone Miner Metab. 2021;39(3):341-352. [34] LING L, HU HL, LIU KY, et al. Long noncoding RNA MIRG induces osteoclastogenesis and bone resorption in osteoporosis through negative regulation of miR-1897. Eur Rev Med Pharmacol Sci. 2019; 23(23):10195-10203. [35] BOYCE BF, XIU Y, LI J, et al. NF-kappaB-Mediated Regulation of Osteoclastogenesis. Endocrinol Metab (Seoul). 2015;30(1):35-44. [36] REN Y, ZHANG K, WANG J, et al. HOTAIRM1 promotes osteogenic differentiation and alleviates osteoclast differentiation by inactivating the NF-κB pathway. Acta Biochim Biophys Sin (Shanghai). 2021;53(2): 201-211. [37] YASUDA H. Discovery of the RANKL/RANK/OPG system. J Bone Miner Metab. 2021;39(1):2-11. [38] CHE W, DONG Y, QUAN HB. RANKL inhibits cell proliferation by regulating MALAT1 expression in a human osteoblastic cell line hFOB 1.19. Cell Mol Biol (Noisy-le-grand). 2015;61(1):7-14. [39] CHANG Y, YU D, CHU W, et al. LncRNA expression profiles and the negative regulation of lncRNA-NOMMUT037835.2 in osteoclastogenesis. Bone. 2020;130:115072. [40] LI VS, NG SS, BOERSEMA PJ, et al. Wnt signaling through inhibition of β-catenin degradation in an intact Axin1 complex. Cell. 2012;149(6): 1245-1256. [41] CAI WL, ZENG W, LIU HH, et al. LncRNA LINC00707 promotes osteogenic differentiation of hBMSCs through the Wnt/β-catenin pathway activated by LINC00707/miR-145/LRP5 axis. Eur Rev Med Pharmacol Sci. 2020;24(1):18-28. [42] LIU J, WU M, FENG G, et al. Downregulation of LINC00707 promotes osteogenic differentiation of human bone marrow‑derived mesenchymal stem cells by regulating DKK1 via targeting miR‑103a‑3p. Int J Mol Med. 2020;46(3):1029-1038. [43] WANG CG, HU YH, SU SL, et al. LncRNA DANCR and miR-320a suppressed osteogenic differentiation in osteoporosis by directly inhibiting the Wnt/β-catenin signaling pathway. Exp Mol Med. 2020; 52(8):1310-1325. [44] LIU ZH, QI DD, LI X, et al. LncRNA SNHG14 promotes osteogenic differentiation of human bone marrow-derived mesenchymal stem cells via regulating miR-185-5p/WISP2 axis. J Biol Regul Homeost Agents. 2021;35(2):605-615. [45] LOWERY JW, ROSEN V. The BMP Pathway and Its Inhibitors in the Skeleton. Physiol Rev. 2018;98(4):2431-2452. [46] ZHAO Y, CHEN Y, HU X, et al. lncRNA LINC01535 upregulates BMP2 expression levels to promote osteogenic differentiation via sponging miR‑3619‑5p. Mol Med Rep. 2020;22(6):5428-5435. [47] ZHANG RF, LIU JW, YU SP, et al. LncRNA UCA1 affects osteoblast proliferation and differentiation by regulating BMP-2 expression. Eur Rev Med Pharmacol Sci. 2019;23(16):6774-6782. [48] ZHANG Y, CHEN B, LI D, et al. LncRNA NEAT1/miR-29b-3p/BMP1 axis promotes osteogenic differentiation in human bone marrow-derived mesenchymal stem cells. Pathol Res Pract. 2019;215(3):525-531. [49] KOPAN R, ILAGAN MX. The canonical Notch signaling pathway: unfolding the activation mechanism. Cell. 2009;137(2):216-233. [50] ZANOTTI S, CANALIS E. Notch and the skeleton. Mol Cell Biol. 2010; 30(4):886-896. [51] ZHANG Z, LIU J, ZENG Z, et al. lncRNA Rmst acts as an important mediator of BMP9-induced osteogenic differentiation of mesenchymal stem cells (MSCs) by antagonizing Notch-targeting microRNAs. Aging (Albany NY). 2019;11(24):12476-12496. [52] CHE M, GONG W, ZHAO Y, et al. Long noncoding RNA HCG18 inhibits the differentiation of human bone marrow-derived mesenchymal stem cells in osteoporosis by targeting miR-30a-5p/NOTCH1 axis. Mol Med. 2020;26(1):106. [53] YU Y, YAO P, WANG Z, et al. Down-regulation of FTX promotes the differentiation of osteoclasts in osteoporosis through the Notch1 signaling pathway by targeting miR-137. BMC Musculoskelet Disord. 2020;21(1):456. [54] WANG H, ZHAO W, TIAN QJ, et al. Effect of lncRNA AK023948 on rats with postmenopausal osteoporosis via PI3K/AKT signaling pathway. Eur Rev Med Pharmacol Sci. 2020;24(5):2181-2188. [55] WANG H, LI YK, CUI M, et al. Effect of lncRNA AK125437 on postmenopausal osteoporosis rats via MAPK pathway. Eur Rev Med Pharmacol Sci. 2020;24(5):2173-2180. [56] WU Y, JIANG Y, LIU Q, et al. lncRNA H19 promotes matrix mineralization through up-regulating IGF1 by sponging miR-185-5p in osteoblasts. BMC Mol Cell Biol. 2019;20(1):48. [57] MURACA M, CAPPARIELLO A. The Role of Extracellular Vesicles (EVs) in the Epigenetic Regulation of Bone Metabolism and Osteoporosis. Int J Mol Sci. 2020;21(22):8682. [58] YANG Y, YUJIAO W, FANG W, et al. The roles of miRNA, lncRNA and circRNA in the development of osteoporosis. Biol Res. 2020;53(1):40. [59] YU L, LIU Y. circRNA_0016624 could sponge miR-98 to regulate BMP2 expression in postmenopausal osteoporosis. Biochem Biophys Res Commun. 2019;516(2):546-550. |
| [1] | Li Xiaomin, Tian Xiangdong, Tan Yetong, Zhu Guangyu, Wang Rongtian, Wang Jian, Xue Zhipeng, Ma Sheng, Hu Yuanyi, Huang Ye, Ding Tiansong. Changes of lower limb force line and knee function after high tibial osteotomy in osteoporotic medial ventricular knee osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1325-1329. |
| [2] | Dang Yi, Du Chengyan, Yao Honglin, Yuan Nenghua, Cao Jin, Xiong Shan, Zhang Dingmei, Wang Xin. Hormonal osteonecrosis and oxidative stress [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1469-1476. |
| [3] | Jiang Xiaocheng, Shi Lu, Wang Yinbin, Li Qiujiang, Xi Chuangzhen, Ma Zefeng, Cai Lijun. Systematical evaluation of bone fusion rate after interbody fusion in patients with osteoporosis and lumbar degenerative disease treated with teriparatide [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1427-1433. |
| [4] | Sun Jiajia, Zhu Haidi, Lu Yun, Zhang Kai. Comparison of bone metabolism markers between type 2 diabetes mellitus and non-type 2 diabetes mellitus patients with hip fracture [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1156-1160. |
| [5] | Long Guiyue, Li Dongdong, Liao Hongbing. Calcium phosphate cement/poly(lactic-co-glycolic acid) degradation products promote osteoclast differentiation of mouse monocytes [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1193-1198. |
| [6] | Huang Linke, Wei Linhua, Jiang Jie, Liu Qian, Chen Weiwei. Effects of estrogen combined with treadmill exercise on bone mass and articular cartilage in ovariectomized mice [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1166-1171. |
| [7] | Yang Zhishan, Tang Zhenglong. YAP/TAZ, a core factor of the Hippo signaling pathway, is involved in bone formation [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1264-1271. |
| [8] | Liu Xiaolin, Mu Xinyue, Ma Ziyu, Liu Shutai, Wang Wenlong, Han Xiaoqian, Dong Zhiheng. Effect of hydrogel-loaded simvastatin microspheres on osteoblast proliferation and differentiation [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 998-1003. |
| [9] | Xue Ting, Zhang Xinri, Kong Xiaomei. Mesenchymal stem cell therapy for pneumoconiosis using nanomaterials combined with multi-modal molecular imaging [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1133-1140. |
| [10] | Liu Wentao, Feng Xingchao, Yang Yi, Bai Shengbin. Effect of M2 macrophage-derived exosomes on osteogenic differentiation of bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 840-845. |
| [11] | Long Yanming, Xie Mengsheng, Huang Jiajie, Xue Wenli, Rong Hui, Li Xiaojie. Casein kinase 2-interaction protein-1 regulates the osteogenic ability of bone marrow mesenchymal stem cells in osteoporosis rats [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 878-882. |
| [12] | Cui Lianxu, Jiang Wenkang, Lu Dahong, Xu Junrong, Liu Xiaocui, Wang Bingyun. Clinical-grade human umbilical cord mesenchymal stem cells affect the improvement of neurological function in rats with traumatic brain injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 835-839. |
| [13] | Li Qicheng, Deng Jin, Fu Xiaoyang, Han Na. Effects of bone marrow mesenchymal stem cells-derived exosomes on hypoxia-treated myoblasts [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 853-859. |
| [14] | Wang Min, Yin Xiushan, Wang Yingxi, Zhang Yan, Zhao Long, Xia Shuyue. Inhalation of bone marrow mesenchymal stem cells-derived exosomes alleviates inflammatory injury in chronic obstructive pulmonary disease [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 827-834. |
| [15] | Qiao Luhui, Ma Ziyu, Guo Haoyu, Hou Yudong. Comparison of puerarin and icariin on the biological properties of mouse preosteoblasts [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 872-877. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||

