Chinese Journal of Tissue Engineering Research ›› 2022, Vol. 26 ›› Issue (27): 4386-4392.doi: 10.12307/2022.872
Previous Articles Next Articles
Application of multicellular construction of vascularized tissue engineered bone in bone repair
Zhao Doudou1, 2, Lin Kaili2
- 1School of Materials Science and Engineering, University of Shanghai for Science and Technology, Shanghai 200093, China; 2Shanghai Ninth People’s Hospital, Shanghai Jiao Tong University School of Medicine, Shanghai 200011, China
-
Received:2021-04-02Accepted:2021-06-10Online:2022-09-28Published:2022-03-12 -
Contact:Lin Kaili, Professor, Doctoral supervisor, Shanghai Ninth People’s Hospital, Shanghai Jiao Tong University School of Medicine, Shanghai 200011, China -
About author:Zhao Doudou, Master candidate, School of Materials Science and Engineering, University of Shanghai for Science and Technology, Shanghai 200093, China; Shanghai Ninth People’s Hospital, Shanghai Jiao Tong University School of Medicine, Shanghai 200011, China -
Supported by:National Key Research and Development Program, No. 2018YFB1105605 (to LKL); National Natural Science Foundation of China, No. 81871490 (to LKL)
CLC Number:
Cite this article
Zhao Doudou, Lin Kaili. Application of multicellular construction of vascularized tissue engineered bone in bone repair[J]. Chinese Journal of Tissue Engineering Research, 2022, 26(27): 4386-4392.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
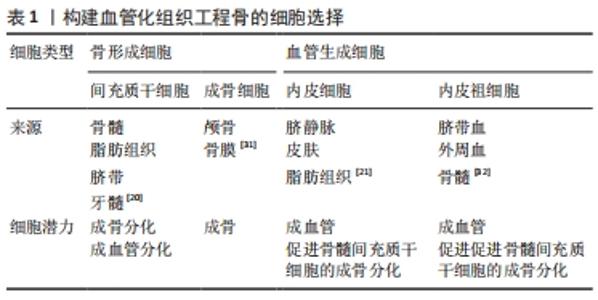
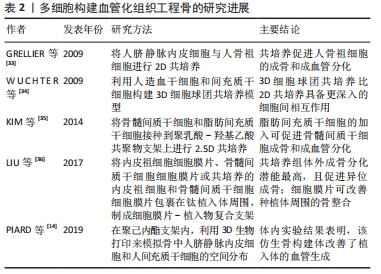
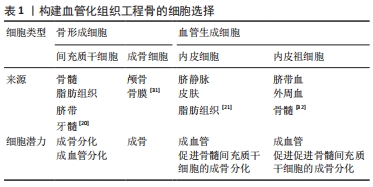
2.1 细胞的选择 在多细胞构建的血管化组织工程骨中,细胞类型、细胞比例和培养基比例极为重要。对于基于细胞的骨组织工程构建体而言,可以从多种解剖部位获取且具有强大的自我更新能力和分化为成骨细胞能力的细胞来源是首选的[19]。迄今为止,最常用的骨形成细胞和血管生成细胞的类型分别为间充质干细胞/脂肪干细胞/成骨细胞和内皮细胞/内皮祖细胞。目前间充质干细胞的来源主要包括:骨髓、脂肪组织、脐带和牙髓4种[20]。脂肪间质干细胞是在脂肪组织中发现的成体干细胞[21]。与骨髓间充质干细胞类似,脂肪间充质干细胞具有分化为脂肪细胞、软骨细胞、成骨细胞、成肌细胞和神经细胞的潜能[22-24],脂肪间质干细胞是组织工程中理想的种子细胞之一,与骨髓间充质干细胞相比,脂肪间质干细胞来源丰富、获取简单和增殖率高[25]。对于血管生成细胞的选择,人脐静脉内皮细胞相比于内皮祖细胞具备成本低、特征明显、寿命长及更快的增殖速率等优点[26],因此,人脐静脉内皮细胞仍是骨组织工程研究中使用最多的细胞类型。由于其成熟分化特性,人脐静脉内皮细胞不能进一步应用于临床[27],但内皮祖细胞等替代细胞也已被探索。此外,间充质干细胞同时分化为成骨细胞和血管生成细胞类型可以使细胞获取过程更容易[28]。 最佳的细胞比例仍在研究中,对于一些多细胞共培养体系,如骨髓间质干细胞/人脐静脉内皮细胞和脂肪间质干细胞/人脐静脉内皮细胞。据报道,1∶1的比例对骨生成和血管生成都是最佳的[29]。虽然从生物学角度来看,骨组织中成骨细胞和内皮细胞的比例是5∶1[30];但1∶1的比例可以在骨形成细胞使用较少的情况下获得最佳的成骨和血管生成能力[31-36]。见表1、表2。"
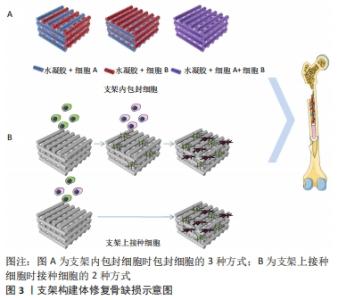
2.2 基于支架的多细胞组织工程骨 目前,用于骨组织工程支架的材料主要有水凝胶、天然聚合物、合成聚合物、生物活性陶瓷材料及其复合材料等。单纯材料支架缺乏骨再生能力,为了设计具有更好的骨生物相容性的替代品,组织工程技术制备结合细胞或生长因子的三维支架,来诱导骨组织再生[37-38]。骨组织工程支架的目的是模拟天然骨细胞外基质的生物学功能和结构,为细胞的黏附、增殖和分化提供一个三维环境。通常,理想的3D支架是由生物相容性、生物可降解材料组成的,其力学性能与天然骨组织相似。从设计上看,支架并不是永久性植入体,它将促进细胞沉积细胞外基质,并随着时间的推移取代支架结构。支架的三维结构应该是高度多孔的,并具有相互连通的开放多空网络结构,以促进营养输送,细胞增殖、分化、迁移和组织血管化等骨组织再生的关键因素,为细胞的生长和功能提供了合适的微环境。目前制备多孔组织工程支架的方法有模板法、发泡法、冻干技术、静电纺丝技术、3D打印技术和4D打印技术等[39-40]。 支架作为载体与细胞或生长因子结合,诱导组织再生。支架材料的性质决定了细胞在支架上的结合方式。细胞与材料的结合方式有2种,一是将细胞接种在支架材料表面;二是直接将细胞包封在支架材料内,见图3。"
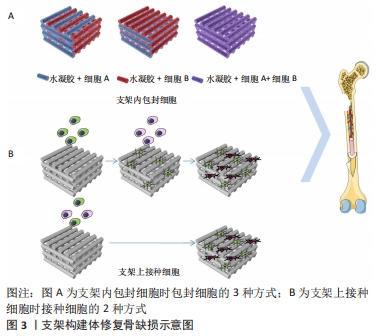
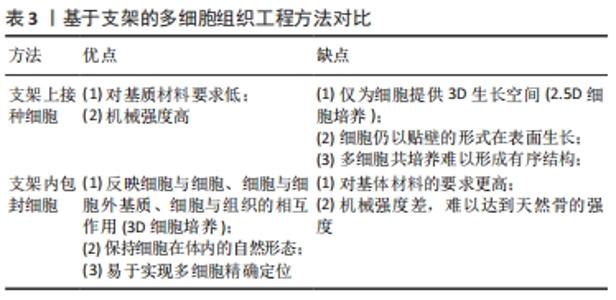
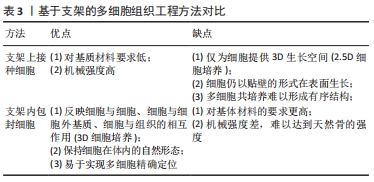
2.2.1 支架上接种细胞 骨组织工程支架材料的基本要求是:材料既要能促进细胞外基质的沉积,还要能促进细胞黏附和迁移。但细胞的黏附常受材料的表面粗糙度、亲疏水性和缺乏可识别生化结合位点的限制,在选择支架材料时应选择非免疫原性的生物材料。此外,通过与其他材料(如明胶和壳聚糖)或蛋白质(如纤维粘连蛋白)复合[41-43],或通过表面修饰(如表面形貌修饰、蛋白质吸附、矿物涂层、官能团嵌套和生物大分子固定[44])等来改善细胞在支架上的黏附和迁移情况。相比于在支架内包封细胞的方法,在支架表面接种细胞可根据需要,调整各种细胞的接种时间,但其细胞接种部位不可控,不能很好地模拟天然骨组织中的细胞分布。 有研究将人脐静脉内皮细胞和人成骨细胞按4∶1的比例同时接种在发泡法制备的硅酸钙骨水泥支架上,与仅接种人成骨细胞组做比,共培养组的血管内皮生长因子和Ⅰ型胶原以及成骨标志物碱性磷酸酶、骨钙蛋白和Runx2的表达明显提高;共培养42 d后,CD31免疫荧光染色显示共培养组有丰富的微毛细血管样结构,表明在大孔硅酸钙骨水泥支架上同时接种人脐静脉内皮细胞和人成骨细胞具有良好的血管生成能力,并能促进骨生成[45]。有研究制备了肝素修饰的聚己内酯-明胶电纺纤维支架,并在支架上以1∶1接种脂肪来源间充质干细胞和人脐静脉内皮细胞[46],发现在共培养条件下,CD31表达增强,且人脐静脉内皮细胞呈健康的鹅卵石状,表明与间充质干细胞共培养有助于血管化。双向诱导人脐血间充质干细胞分别使其分化为成骨细胞和内皮细胞,发现在磷酸三钙支架上以3∶1接种成骨分化和成管分化的人脐血间充质干细胞与接种未分化人脐血间充质干细胞组相比,修复大鼠临界尺寸颅骨缺损的能力显著增强[47]。 2.2.2 支架内包封细胞 相比于在支架上接种细胞,将细胞包封在支架材料内的方法对支架材料要求更高。首先,考虑到高物质交换率是包封细胞的重要条件,因为气体(O2和CO2)、营养物质和代谢产物的不断交换对细胞的存活和增殖至关重要;其次,适当的机械性能、高保水率也是支架材料的必备条件。水凝胶材料因可模拟天然细胞外基质的物理和生化特性而得到了广泛研究[48]。水凝胶是一种由亲水聚合物链组成的三维分子网络,在富含水的环境中具有高达其干质量1 000倍的吸水能力。目前,常用的水凝胶有海藻酸盐基水凝胶、透明质酸基水凝胶、壳聚糖基水凝胶、明胶 [49]、甲基丙烯酸改性明胶(GelMA)基水凝胶和聚乙二醇基水凝胶。近年来,出现了多种水凝胶改性方法,如点击化学、凝胶机制和复合纳米生物材料等,实现了水凝胶物化性质的有效控制。因此,骨组织工程中常将水凝胶作为包封细胞的载体,并利用生物3D打印技术实现对多种细胞的精准定位,模拟天然骨中的细胞分布。 有研究采用生物3D打印技术制备得到具有可灌注血管腔的类骨组织构建体[48-50],构建体的中心纤维由快速降解的GelMA(GelMA-low)包封人脐静脉内皮细胞构成,中心纤维的周围打印复合了硅酸盐纳米片的生物墨水来诱导骨髓间充质干细胞成骨;此外,为了促进血管延伸,在诱导成骨部分使用了血管内皮生长因子梯度功能化的GelMA,结果表明,这种策略适用于制造大尺寸带血管蒂的骨组织结构。最近一项研究利用同轴生物3D打印将小鼠成骨细胞系(MC3T3-E1)和人脐静脉内皮细胞的生物墨水分别从同轴针头的外部(与GelMA混合)和内部(与明胶混合)同时打印出来[51],得到的大尺寸组织由芯鞘纤维组成,当芯纤维溶解形成通道时,人脐静脉内皮细胞自动沉积并黏附在通道上,通过这种方法构建了长期培养(≥20 d)的三维负载细胞的血管化骨组织构建体。 水凝胶缺乏骨组织工程所需的机械强度。将机械强度高的生物相容性材料(如聚己内酯、聚乳酸-羟基乙基共聚物等)与水凝胶生物墨水进行复合,可以提高整个构建体的机械强度。PIARD等[51-52]利用基于挤压的生物3D打印技术来模拟天然骨中的空间分布。通过沿着聚己内酯基体支架共打印纤维蛋白水凝胶独立包封人脐静脉内皮细胞和人间充质干细胞的生物墨水,构建了双相骨样支架,该结构的抗压力学性能处于皮质骨范围内。仿生支架的血管生成标志物的基因表达显著上调。将双相仿生支架植入大鼠皮下,组织学分析显示,3D打印骨样支架的血管数量显著增多。有文献报道:在多巴胺修饰的硅酸钙-聚己内酯支架的间隙分别打印海藻酸盐基水凝胶包封人脐静脉内皮细胞和间充质干细胞的生物墨水构建了复合骨组织工程支架,该复合支架与多细胞共培养相结合,不仅能够改善构建体的力学性能,还能够促进成骨和血管生成[45]。见表3。"
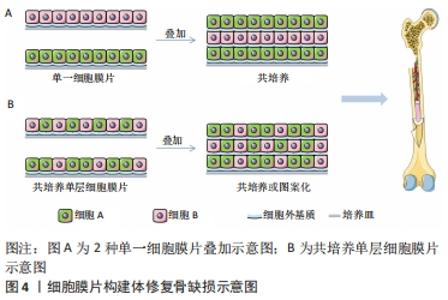
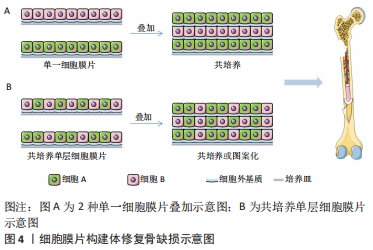
2.3 基于细胞膜片的多细胞组织工程骨 传统的组织工程技术采用胰蛋白酶消化的方法收集种子细胞,其存在细胞利用率低、细胞完整性差及细胞活性低等缺点。细胞膜片技术不存在这些问题。细胞膜片是一种膜质细胞载体,从培养皿上物理剥离来获得扩增的细胞,该技术的优点是保留了丰富的细胞外基质和表面蛋白,避免了细胞的流失、提高了细胞利用率、保留了整个信号通信网络、促进细胞与原生组织的黏附,形成具有较强可塑性和良好机械强度的三维组织结构。 将多层细胞膜片叠加制备出的三维组织能够达到高细胞密度。但超过一定厚度的3D高细胞密度膜片工程组织在植入体内后,往往会导致内部细胞因缺氧而坏死[54],构成组织的细胞的生存能力将直接影响组织的功能。在构建组织工程骨时,构建体的血管化也极为重要,因此,改善骨组织工程细胞膜片结构的血管化成为研究的重点。该方面的研究往往涉及到骨形成细胞与内皮细胞共培养的策略,其优势明显好于单一种类细胞培养获得的细胞膜片。细胞膜片共培养技术能够模拟生理条件,因此保留了多种细胞间的旁分泌效应,使得细胞膜片的功能得到改善或保持与体内组织相似[55]。利用多细胞共培养细胞膜片对移植体进行预血管化,可以为膜片中间部位提供营养供给,同时解决移植后的血供问题,从而提高移植细胞膜片的成活率[56]。此外,当目标细胞黏附较弱时,共培养有利于细胞膜片的剥离;与具有强大收缩力的细胞共培养,可以稳定有效地剥离细胞膜片[36]。 有研究强调了利用培养细胞膜片构建功能性组织结构的重要性,并报道了发现了一种温度敏感培养皿,能够快速分离培养的细胞膜片[57]。细胞膜片的剥离方法主要包括基于温度响应型培养皿、基于表面改性培养皿以及无需进行表面改性的方法,如采用超声剥离、磁性纳米颗粒剥离、纤维蛋白基质涂层剥离等。目前,基于温度响应培养皿剥离的方法应用最为广泛。细胞膜片共培养技术可分为以下2种:一是将2种不同的单培养细胞膜片堆叠在一起;二是利用多种细胞构建每一层细胞膜片。 2.3.1 两种单一细胞膜片叠加 两种单一细胞膜片相叠加可以通过简单地改变片层的垂直位置来实现细胞位置的调控。 PANDUWAWALA等[58]探讨了牙周韧带干细胞(periodontal ligament stem cells,PDLSCs)和人脐静脉内皮细胞构建细胞膜片的体内牙周再生潜能。首先,将PDLSCs、人脐静脉内皮细胞及两者共培养的细胞接种于温度敏感培养皿上,并制备了3种完整的细胞膜片;然后,将细胞膜片进行组合,即PDLSCs-PDLSCs-PDLSCs、PDLSCs-人脐静脉内皮细胞-PDLSCs、Co-culture-Co-culture-Co-culture,再分别包裹在准备好的人牙根上,并植入免疫缺陷小鼠皮下。组织学观察显示,植入2,4,8周后,种植根周围可见牙周韧带样组织排列,含人脐静脉内皮细胞组可见血管腔;植入第4周和第8周的免疫染色显示多细胞组牙周韧带再生、骨水泥形成和骨形成更明显。无论是Co-culture-Co-culture-Co-culture组还是PDLSCs-人脐静脉内皮细胞-PDLSCs组,宿主微血管系统与植入体血管腔结构都能有效整合并促进体内牙周韧带样组织、牙骨质和骨质的形成。有研究也曾以几种不同的方式将人源内皮细胞引入分层组织,并比较了内皮细胞在3D组织不同位置的行为。人类脐静脉内皮细胞、人真皮成纤维细胞及其混合物制备完整的细胞膜片从低温反应培养皿中获得。利用细胞膜片收集装置将3种细胞膜片按不同顺序堆叠,形态学分析结果显示,所有堆叠结构均形成了人脐静脉内皮细胞构成的前血管网络;共聚焦显微镜观察表明,前血管网络形成了类似于天然微血管的管状结构。这些结果表明,内皮细胞在3D组织中的初级定位可能是血管形成的关键[59]。 2.3.2 共培养单层细胞膜片 将共培养的融合细胞从培养表面分离,就可以制得共培养单层细胞膜片。接种细胞的方法也有很多种,主要包括一次性接种多种细胞、不同时间点接种不同种细胞及利用图案化的培养表面来制备图案化的共培养单层细胞膜片等[60],见图4。"

邢飞等 [61]将外周血间充质干细胞和外周血内皮细胞以1∶1比例混合并进行成膜诱导,并与仅含单一细胞膜片进行对比,发现共培养细胞膜片较单一细胞膜片组更厚,细胞密度更大;培养第10天,共培养细胞膜片组的碱性磷酸酶、骨钙蛋白及血管内皮生长因子表达量均显著高于单一细胞膜片组。研究结果证实了共培养细胞膜片比传统的单一细胞膜片具有更多的细胞层数和细胞外基质,成骨分化能力也更强。 周震等[62]分别在低氧和常氧条件下接种骨髓间充质干细胞,培养4周后再接种人脐静脉内皮细胞制得共培养单层细胞膜片,两种条件下均有血管样结构形成,且低氧条件下细胞膜片得血管样结构更长更密、血管内皮生长因子表达水平也更高。这表明骨髓间充质干细胞和人脐静脉内皮细胞共培养在细胞膜片血管化过程中起关键作用,且低氧条件下骨髓间充质干细胞产生得细胞外基质增多为人脐静脉内皮细胞的增殖、迁移和血管形成提供更优的微环境,从而提高牙槽骨缺损修复效果、促进牙周组织再生。XU等[63]利用细胞膜片技术制造带血管蒂的组织工程骨,他们将骨髓间充质干细胞来源的内皮细胞接种到未分化的骨髓间充质干细胞细胞层,共培养3 d后,内皮细胞迁移并重新排列形成管腔,得到预血管化的细胞膜片;另外,通过诱导高密度骨髓间充质干细胞膜片成骨分化得到成骨细胞膜片;将预血管化的细胞膜片堆叠在成骨细胞膜片上,制备无支架的骨再生构建体;最后,将具备成血管和成骨潜能的无支架结构植入大鼠的临界尺寸颅骨缺损。大鼠临界尺寸颅骨缺损修复结果证实了预血管化组有更多的功能性灌注血管和新骨形成。 近年来,已利用以下策略来构建血管化细胞膜片骨组织植入体:将骨形成细胞与血管生成细胞膜片共培养、将血管生成细胞膜片与其他类型细胞膜片堆叠以及在细胞膜片构建体中插入血管束等。但作为大体积骨缺损植入体其力学强度明显不足,因此,在利用细胞膜片共培养技术构建血管化骨构建体时还需结合增强力学强度的方法。"

| [1] BOEHRNSEN F, PETRA M, MAREIKE N, et al. Cotransplantation of mesenchymal stromal cells and endothelial cells on calcium carbonate and hydroxylapatite scaffolds in vivo. J Craniomaxillofac Surg. 2021; 49(3):238-245. [2] SHI R, HUANG YL, MA C, et al. Current advances for bone regeneration based on tissue engineering strategies. Front Med. 2019;13(2):160-188. [3] OWAKI T, SHIMIZU T, YAMATO M, et al. Cell sheet engineering for regenerative medicine:current challenges and strategies. Biotechnol J. 2014;9(7):904-914. [4] SAFINSHA S, MUBARAK AM. Composite scaffolds in tissue engineering. Mater Today Proc. 2020;24:2318-2329. [5] GREEN H, KEHINDE O, THOMAS J. Growth of cultured human epidermal cells into multiple epithelia suitable for grafting. Proc Natl Acad Sci U S A. 1979;76(11):5665-5668. [6] YORUKOGLU CA, KITER EA, AKKAYA S, et al. A Concise review on the use of mesenchymal stem cells in cell sheet-based tissue engineering with special emphasis on bone tissue regeneration. Stem Cells Int. 2017; 2017:2374161. [7] GALLICO G, COMPTON C, KEHINDE O, et al. Cultured epithelial autografts for giant congenital nevi. Plast Reconstr Surg. 1990;84:1-9. [8] IWASAKI K, WASHIO K, MEINZER W, et al. Application of cell-sheet engineering for new formation of cementum around dental implants. Heliyon. 2019;5:e01991. [9] ORCIANILM M, MILENA F, ROBERTO DP, et al. Biofabrication and bone tissue regeneration: cell source, approaches, and challenges. Front Bioeng Biotechnol. 2017;5:17-32. [10] YE LIN P, KIWON P, JAE MIN C. 3D-bioprinting strategies based on in situ bone-healing mechanism for vascularized bone tissue engineering. Micromachines. 2021;12(3):287-310. [11] GUREL PG, TORUN KOSE G, HASIRCI V. Influence of co-culture on osteogenesis and angiogenesis of bone marrow mesenchymal stem cells and aortic endothelial cells. Microvasc Res. 2016;108:1-9. [12] TAKAHISA A, CHI CHUN P, ALEXANDER S, et al. Vascularized bone-mimetic hydrogel constructs by 3d bioprinting to promote osteogenesis and angiogenesis. Int J Mol Sci. 2019;20(5):1096-1201. [13] SILVA AS, SANTOS LF, MENDES MC, et al. Multi-layer pre-vascularized magnetic cell sheets for bone regeneration. Biomaterials. 2020;231: 119664. [14] PIARDA C, JEYARAMA A, YI L, et al. 3D printed HUVECs/MSCs cocultures impact cellular interactions and angiogenesis depending on cell-cell distance. Biomaterials. 2019;222:119423. [15] AKBARI A, JABBARI N, SHARIFI R, et al. Free and hydrogel encapsulated exosome-based therapies in regenerative medicine. Life Sci. 2020; 249:1-12. [16] WENG YM, WANG ZF, SUN JW, et al. Engineering of axially vascularized bone tissue using natural coral scaffold and osteogenic bone marrow mesenchymal stem cell sheets. Arch Biochem Biophys. 2021;50:1-8. [17] TORRESA AL, BIDARRA SJ, VASCONCELOS DP, et al. Microvascular engineering: dynamic changes in microgel-entrapped vascular cells correlates with higher vasculogenic/angiogenic potential. Biofabrication. 2020;228:119554. [18] MA JL, SANNE K, YANG F, et al. Concise review:cell-based strategies in bone tissue engineering and regenerative medicine. Stem Cells Transl Med. 2014;3(1):98-107. [19] DIOMEDE F, MARCONI GD, FONTICOLI L, et al. Functional relationship between osteogenesis and angiogenesis in tissue regeneration. Int J Mol Sci. 2020;21(9):32-42. [20] AMLER AK, THOMAS A, TUZUNER S, et al. 3D bioprinting of tissue-specific osteoblasts and endothelial cells to model the human jawbone. Sci Rep. 2021;11(1):4876. [21] ZHANG HL, ZHOU YL, ZHANG W, et al. Construction of vascularized tissue-engineered bone with a double-cell sheet complex. Acta Biomater. 2018;77:212-227. [22] PRZEKORAL A, VANDROVCOVA M, TRAVNICKOVA M, et al. Evaluation of the potential of chitosan/ beta-1,3-glucan/hydroxyapatite material as a scaffold for living bone graft production in vitro by comparison of ADSC and BMDSC behaviour on its surface. Biomed Mater. 2017; 12(1):015030. [23] WANG Z, HAN L, SUN T, et al. Construction of tissue-engineered bone with differentiated osteoblasts from adipose-derived stem cell and coral scaffolds at an ectopic site. Br J Oral Maxillofac Surg. 2020;59:46-51. [24] STORTI G, SCIOLI MG, KIM BS, et al. Adipose-derived stem cells in bone tissue engineering: useful tools with new applications. Stem Cells Int. 2019; 2019:3673857. [25] ZHANG JH, WEHRLE E, ADAMEK PA, et al. Optimization of mechanical stiffness and cell density of 3D bioprinted cell-laden scaffolds improves extracellular matrix mineralization and cellular organization for bone tissue engineering. Acta Biomater. 2020;11:307-322. [26] TROUNSON A, DEWITT ND. Pluripotent stem cells progressing to the clinic. Nat Rev Mol Cell Biol. 2016;17(3):194-200. [27] RONG Q, Li SY, ZHOU Y, et al. A novel method to improve the osteogenesis capacity of hUCMSCs with dual-directional pre-induction under screened co-culture conditions. Cell Prolif. 2020;53(2):e12740. [28] MUTSCHALL H, WINKLER S, WEISBACH V, et al. Bone tissue engineering using adipose-derived stem cells and endothelial cells: effects of the cell ratio. J Cell Mol Med. 2020;24:7034-7043. [29] SHEN QH, FAN XT, JIANG ML, et al. Inhibiting expression of Cxcl9 promotes angiogenesis in MSCs-HUVECs coculture. Arch Biochem Biophys. 2019;675:108-119. [30] COLLINS MN, REN G, YOUNG K, et al. Scaffold Fabrication Technologies and Structure/Function Properties in Bone Tissue Engineering. Adv Funct Mater. 2021;12:47309-47319. [31] MA J, BOTH SK, YANG F, et al. Concise review: cell-based strategies in bone tissue engineering and regenerative medicine. Stem Cells Transl Med. 2014;3:98-107. [32] URBICH C, DIMMELER S. Endothelial progenitor cells functional characterization. Trends Cardiovasc Med. 2004;14:318-322. [33] GRELLIER M, TOJAIS NF, BOURGET C, et al. Role of vascular endothelial growth factor in the communication between human osteoprogenitors and endothelial cells. J Cell Biochem. 2009;106:390-398. [34] WUCHTER PMD, SAFFRICH R, LUDWIG A, et al. Cellular interaction between human mesenchymal stem cells and hematopoietic stem cells in 2D-and 3D-culture-systems. Blood. 2009;114(22):1442. [35] KIM K, PARK S, IM G. Osteogenic differentiation and angiogenesis with cocultured adipose-derived stromal cells and bone marrow stromal cells. Biomaterials. 2014;35:4792-4804. [36] LIU H, ZHOU W, REN N, et al. Cell sheets of co-cultured endothelial progenitor cells and mesenchymal stromal cells promote osseointegration in irradiated rat bone. Sci Rep. 2017;7(1):3038-3050. [37] ROMANAZZO S,MOLLEY TG, NEMEC S, et al. Synthetic bone‐like structures through omnidirectional ceramic bioprinting in cell suspensions. Adv Funct Mater. 2021;31(13):1-13. [38] MOBARAKI M, GHAFFARI M, YAZDANPANAH A, et al. Bioinks and Bioprinting: a Focused Review. Bioprinting. 2020;18:e00080. [39] GEORGOPOULOU A, PAPADOGIANNIS F, BATSALI A, et al. Chitosan/gelatin scaffolds support bone regeneration. J Mater Sci Mater Med. 2018;29(5):59-72. [40] SINGHL R, EITLERL D, MORELLE R, et al. Optimization of cell seeding on electrospun PCL-silk fibroin scaffolds. Eur Polym J. 2020;134:109838-109849. [41] LIVERANI L, RAFFEL N, FATTAHI A, et al. Electrospun patterned porous scaffolds for the support of ovarian follicles growth:a feasibility study. Sci Rep. 2019;9(1):1150-1164. [42] LIVERANI L, LACINA J, JUDITH AR, et al. Incorporation of bioactive glass nanoparticles in electrospun PCL/chitosan fibers by using benign solvents. Bioact Mater. 2018;3(1):55-63. [43] THEIN HW, HOCKIN HK. Prevascularization of a gas-foaming macroporouscalcium phosphate cement scaffold via coculture of human umbilical vein endothelial cells and osteoblasts. Tissue Eng Part A. 2013;19(15-16):1675-1685. [44] SUN P, ZHANG QQ, NIE W, et al. Biodegradable mesoporous silica nanocarrier bearing angiogenic QK peptide and dexamethasone for accelerating angiogenesis in bone regeneration. ACS Biomater Sci Eng. 2019;5(12):6766-6778. [45] LIVERANI L, KILLIAN MS, BOCCACCINI AR. Fibronectin functionalized electrospun fibers by using benign solvents: best way to achieve effective functionalization. Front Bioeng Biotechnol. 2019;7(68):1-9. [46] JOSHI A, ZHE X, IKEGAMI Y, et al. Co-culture of mesenchymal stem cells and human umbilical vein endothelial cells on heparinized polycaprolactone/gelatin co-spun nanofibers for improved endothelium remodeling. Int J Biol Macromol. 2020;151:186-192. [47] JANITHA M, UNAGOLLA AC, UNAGOLLA J. Hydrogel-based 3D bioprinting: a comprehensive review on cell-laden hydrogels, bioink formulations, and future perspectives. Appl Mater Today. 2020;18:1-22. [48] BYAMBAA B, ANNABI N, YUE K, et al. Bioprinted osteogenic and vasculogenic patterns for engineering 3D bone tissue. Adv Healthc Mater. 2017;6(16):1-15. [49] SHAO L, GAO Q, XIE CQ, et al. Directly coaxial 3D bioprinting of large-scale vascularized tissue constructs. Biofabrication. 2020;12(3):035014. [50] MOBARAKI M, GHAFFARI M, YAZDANPANAH A, et al. Bioinks and bioprinting: a focused review. Bioprinting. 2020;18:e00080. [51] PIARD CM, YU C, FIAHER JOHN PY. Cell-laden 3D printed scaffolds for bone tissue engineering. Clin Rev Bone Miner Metab. 2015;13(4):245-255. [52] PIARD C, BAKER H, Kamalitdinov T, et al. Bioprinted osteon-like scaffolds enhance in vivo neovascularization. Biofabrication. 2019; 11(2):025013. [53] CHEN YW, SHEN YF, HO CC, et al. Osteogenic and angiogenic potentials of the cell-laden hydrogel/mussel-inspired calcium silicate complex hierarchical porous scaffold fabricated by 3D bioprinting. Mater Sci Eng C Mater Biol Appl. 2018;91:679-687. [54] SAKAGUCHI K, SHIMIZU T, OKANO T. Construction of three-dimensional vascularized cardiac tissue with cell sheet engineering. J Control Release. 2015;205:83-88. [55] KWON OH, KIKUCHI A, YAMATO M, et al. Rapid cell sheet detachment from poly[N-isopropylacrylamide]-grafted porous cell culture membranes. J Biomed Mater Res. 2015;50:82-89. [56] SAKAI Y, KOIKE M, HASEGAWA H, et al. Rapid fabricating technique for multi-layered human hepatic cell sheets by forceful contraction of the fibroblast monolayer. PLoS One. 2019;8(7):e70970. [57] PANDUWAWALA CP, ZHAN X, DISSANAYAKA WL, et al. In vivo periodontal tissue regeneration by periodontal ligament stem cells and endothelial cells in three-dimensional cell sheet constructs. J Periodontal Res. 2017;52(3):408-418. [58] IMASHIRO C, SHIMIZU T. Fundamental Technologies and Recent Advances of Cell-Sheet-Based Tissue Engineering. Int J Mol Sci. 2021; 22(1):425-443. [59] LU YZ, ZHANG WJ, WANG J, et al. Recent advances in cell sheet technology for bone and cartilage regeneration:from preparation to application. Int J Oral Sci. 2019;11(2):17-30. [60] HIRONOBU T, TERUO O. Cell sheet-based tissue engineering for organizing anisotropic tissue constructs produced using microfabricated thermoresponsive substrates. Adv Healthc Mater. 2016;4(16):2388-2407. [61] 邢飞,段鑫,刘明,等.外周血来源间充质干细胞/内皮祖细胞复合细胞膜片的构建及其生物学特性初步研究[J].中国修复重建外科杂志, 2020,34(1):109-115. [62] 周震,王亚敏,任飞,等.骨髓间充质干细胞膜片和脐静脉内皮细胞共培养模型的构建及鉴定[J].中国组织工程研究,2019,23(25): 3951-3955. [63] XU M, LI JD, LIU XN, et al. Fabrication of vascularized and scaffold-free bone tissue using endothelial and osteogenic cells differentiated from bone marrow derived mesenchymal stem cells. Tissue Cell. 2019;61: 21-29. |
| [1] | Zhang Lichuang, Xu Hao, Ma Yinghui, Xiong Mengting, Han Haihui, Bao Jiamin, Zhai Weitao, Liang Qianqian. Mechanism and prospects of regulating lymphatic reflux function in the treatment of rheumatoid arthritis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1459-1466. |
| [2] | Li Huo, Wang Peng, Gao Jianming, Jiang Haoran, Lu Xiaobo, Peng Jiang. Relationship between revascularization and internal microstructure changes in osteonecrosis of the femoral head [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1323-1328. |
| [3] | Yao Xiaoling, Peng Jiancheng, Xu Yuerong, Yang Zhidong, Zhang Shuncong. Variable-angle zero-notch anterior interbody fusion system in the treatment of cervical spondylotic myelopathy: 30-month follow-up [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1377-1382. |
| [4] | Zhu Chan, Han Xuke, Yao Chengjiao, Zhou Qian, Zhang Qiang, Chen Qiu. Human salivary components and osteoporosis/osteopenia [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1439-1444. |
| [5] | Jin Tao, Liu Lin, Zhu Xiaoyan, Shi Yucong, Niu Jianxiong, Zhang Tongtong, Wu Shujin, Yang Qingshan. Osteoarthritis and mitochondrial abnormalities [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1452-1458. |
| [6] | Wang Jing, Xiong Shan, Cao Jin, Feng Linwei, Wang Xin. Role and mechanism of interleukin-3 in bone metabolism [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1260-1265. |
| [7] | Zhu Chan, Han Xuke, Yao Chengjiao, Zhang Qiang, Liu Jing, Shao Ming. Acupuncture for Parkinson’s disease: an insight into the action mechanism in animal experiments [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1272-1277. |
| [8] | Guo Jia, Ding Qionghua, Liu Ze, Lü Siyi, Zhou Quancheng, Gao Yuhua, Bai Chunyu. Biological characteristics and immunoregulation of exosomes derived from mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1093-1101. |
| [9] | Wu Weiyue, Guo Xiaodong, Bao Chongyun. Application of engineered exosomes in bone repair and regeneration [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1102-1106. |
| [10] | Zhou Hongqin, Wu Dandan, Yang Kun, Liu Qi. Exosomes that deliver specific miRNAs can regulate osteogenesis and promote angiogenesis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1107-1112. |
| [11] | Zhang Jinglin, Leng Min, Zhu Boheng, Wang Hong. Mechanism and application of stem cell-derived exosomes in promoting diabetic wound healing [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1113-1118. |
| [12] | Huang Chenwei, Fei Yankang, Zhu Mengmei, Li Penghao, Yu Bing. Important role of glutathione in stemness and regulation of stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1119-1124. |
| [13] | Hui Xiaoshan, Bai Jing, Zhou Siyuan, Wang Jie, Zhang Jinsheng, He Qingyong, Meng Peipei. Theoretical mechanism of traditional Chinese medicine theory on stem cell induced differentiation [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1125-1129. |
| [14] | An Weizheng, He Xiao, Ren Shuai, Liu Jianyu. Potential of muscle-derived stem cells in peripheral nerve regeneration [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1130-1136. |
| [15] | Fan Yiming, Liu Fangyu, Zhang Hongyu, Li Shuai, Wang Yansong. Serial questions about endogenous neural stem cell response in the ependymal zone after spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1137-1142. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||