Chinese Journal of Tissue Engineering Research ›› 2022, Vol. 26 ›› Issue (23): 3691-3699.doi: 10.12307/2022.669
Previous Articles Next Articles
Interleukin-1 alpha induces osteoclast activation and bone loss
Yang Ruijuan1, Li Yangyang1, Cai Ruiyan1, Liu Huibin1, Guo Chun1, 2
- 1Henan Key Laboratory of Neural Regeneration, First Affiliated Hospital of Xinxiang Medical University, Weihui 453100, Henan Province, China; 2Department of Orthopedics, First Affiliated Hospital of Xinxiang Medical University, Weihui 453100, Henan Province, China
-
Received:2021-09-20Accepted:2021-11-29Online:2022-08-18Published:2022-02-11 -
Contact:Guo Chun, MD, Professor, Henan Key Laboratory of Neural Regeneration, First Affiliated Hospital of Xinxiang Medical University, Weihui 453100, Henan Province, China; Department of Orthopedics, First Affiliated Hospital of Xinxiang Medical University, Weihui 453100, Henan Province, China -
About author:Yang Ruijuan, Master, Henan Key Laboratory of Neural Regeneration, First Affiliated Hospital of Xinxiang Medical University, Weihui 453100, Henan Province, China -
Supported by:the National Natural Science Foundation of China, No. 81672190 (to GC); the Project of Health Commission of Henan Province, No. SB201903013 (to GC); the Project of Science and Technology Department of Henan Province, No. 182102310260 (to GC); the Youth Foundation of the First Affiliated Hospital of Xinxiang Medical University, No. QN-2020-B11 (to YRJ)
CLC Number:
Cite this article
Yang Ruijuan, Li Yangyang, Cai Ruiyan, Liu Huibin, Guo Chun. Interleukin-1 alpha induces osteoclast activation and bone loss[J]. Chinese Journal of Tissue Engineering Research, 2022, 26(23): 3691-3699.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
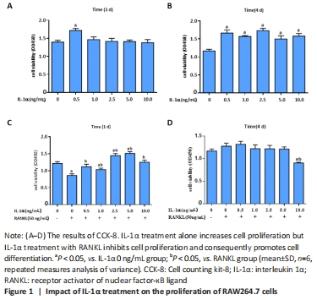
IL-1α increases cell viability in RAW264.7 cells but reduces cell viability in differentiated osteoclasts Results from cell counting kit-8 assays showed that 0.5 ng/mL IL-1α increased the cell viability of RAW264.7 cells compared with the other groups at 1 day (P < 0.05; Figure 1A), and IL-1α increased the cell viability of RAW264.7 cells compared with the non-treated culture at 4 days (P < 0.05; Figure 1B). To assess the role of IL-1α in differentiated osteoclasts, RAW264.7 cells were incubated in the indicated concentration of IL-1α supplemented with RANKL (50 ng/mL) for 1 and 4 days. The cell viability of RANKL treated-RAW264.7 cells was increased by IL-1α treatment after 1 day (P < 0.05; Figure 1C). By contrast, cell viability was found to be significantly decreased after 4 days with 10 ng/mL IL-1α and RANKL treatment compared with cells treated without IL-1α or incubated with other doses of IL-1α and RANKL (P < 0.05; Figure 1D). These results showed that IL-1α at 10 ng/mL in RAW264.7 cells may promote RANKL-induced the formation of multinuclear osteoclasts. "
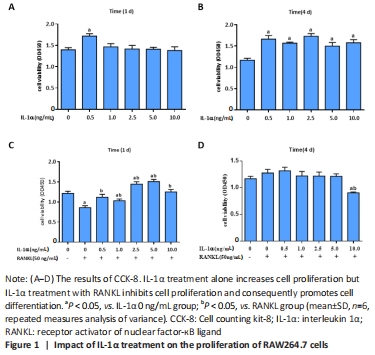
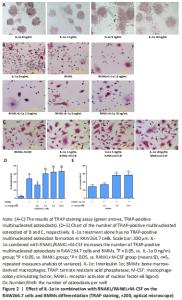
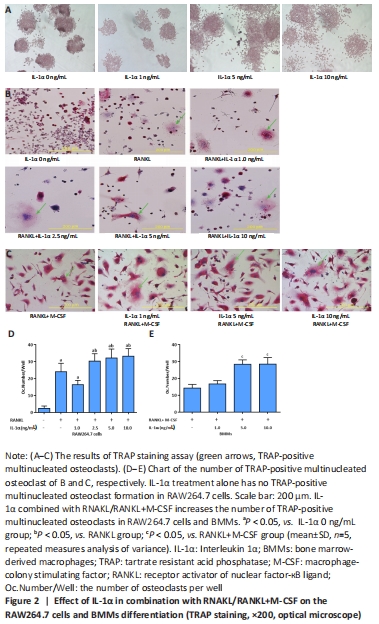
IL-1α promotes osteoclastogenesis in vitro To demonstrate the role of IL-1α in osteoclastogenesis, RAW264.7 cells were incubated in the varied dosages of IL-1α alone or supplemented with 50 ng/mL RANKL for 4 days. In addition, BMMs were incubated with varied dosages of IL-1α supplemented with 30 ng/mL M-CSF and 50 ng/mL RANKL for 7 days. TRAP assay showed that IL-1α treatment alone has no effect on osteoclast formation in RAW264.7 cells; however, the combination of IL-1α and RANKL except for IL-1α 1.0 ng/mL+RANKL treatment appreciably increased the population of osteoclasts compared with the RANKL treatment alone in RAW264.7 cells (P < 0.05; Figure 2A, B and D). Similarly, the combination of IL-1α and RANKL+M-CSF except for IL-1α 1.0 ng/mL+RANKL+M-CSF treatment increased the population of osteoclasts compared with the RANKL+M-CSF treatment in BMMs (P < 0.05; Figure 2C and E)."
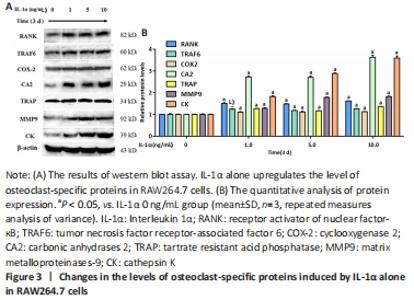
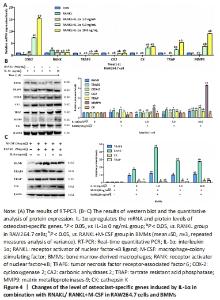
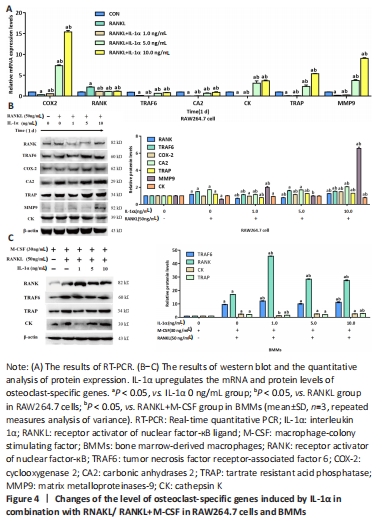
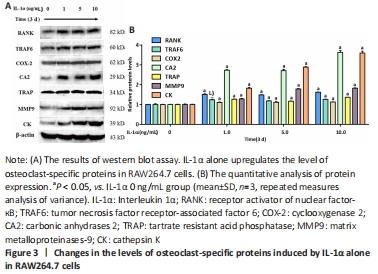
IL-1α upregulates the expression of osteoclastogenesis-related genes Osteoclast-related markers, including RANK, MMP9, CK, COX-2, TRAF6, TRAP, and CA2, are pivotal for the differentiation of osteoclasts. The western blot results showed that IL-1α treatment significantly upregulated the expression of osteoclast-specific proteins including RANK, TRAF6, COX-2, CA2, TRAP, MMP9, and CK in RAW264.7 cells (P < 0.05; Figure 3)."
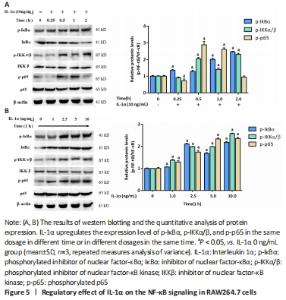
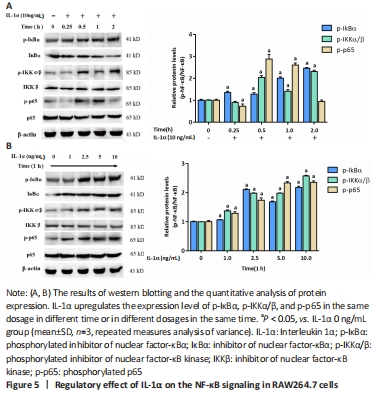
IL-1α activates the NF-κB pathway Since the activation of NF-κB signaling has positive effects on osteoclastogenesis, we evaluated whether IL-1α regulates the NF-κB pathway. The western blot results showed that IL-1α enhanced the expression of NF-κB signal proteins including p-IκB α, p-IKK α/β, and p-p65 (P < 0.05; Figure 5)."
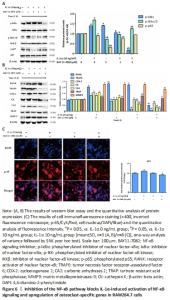
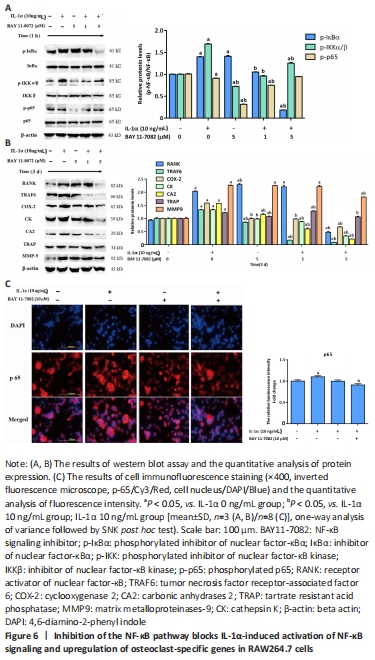
Inhibition of NF-κB signaling was found to reverse the IL-1α-induced enhancements in the NF-κB signaling proteins (p-IκB α, p-IKK α/β, p-p65) and osteoclast-specific proteins (RANK, TRAF6, COX-2, CA2, TRAP, MMP9, CK) in RAW264.7 cells (P < 0.05; Figure 6A and B). The immunofluorescence assay results showed that RAW264.7 cells with IL-1α treatment exhibited strong fluorescent intensity of p65 staining; however, this change was abolished by BAY11-7082 (P < 0.05; Figure 6C)."

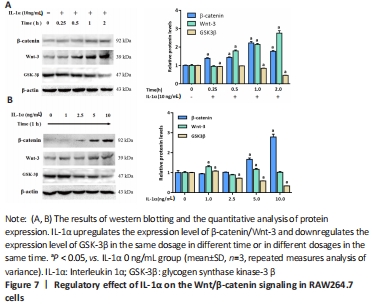
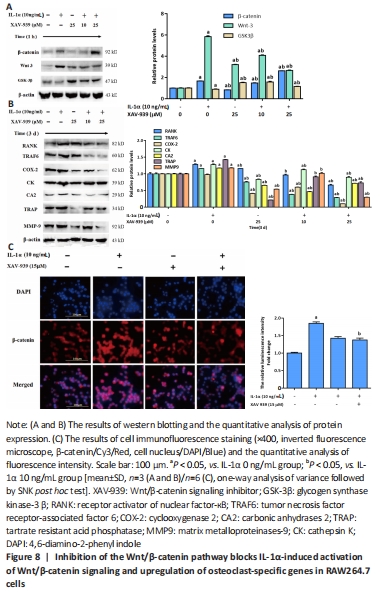
IL-1α activates the Wnt/β-catenin pathway in RAW264.7 cells Since Wnt signaling is momentous for the pre-osteoclast differentiation into osteoclasts[23-24], we investigated whether IL-1 α changes this pathway. In RAW264.7 cells, IL-1α treatment improved Wnt3 and β-catenin expressions and reduced GSK3β expression in dose- and time-dependent manners, as measured by western blot assay (P < 0.05; Figure 7)."
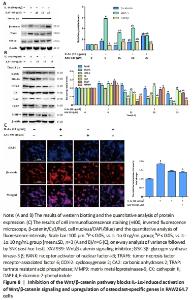
Subsequently, inhibition of the Wnt/beta-catenin signaling attenuated the potentiating effects of IL-1α on the level of Wnt3, β-catenin and osteoclast-specific proteins (RANK, TRAF6, COX-2, CA2, TRAP, MMP9, CK) in RAW264.7 cells (P < 0.05; Figure 8A and B). In addition, the blocking of the Wnt pathway also reversed the downregulation of GSK3β induced by IL-1α (Figure 8A). The level of β-catenin was also tested by immunofluorescence in IL-1α-induced RAW264.7 cells. The results showed that the cells administered with IL-1α exhibited strong fluorescent intensity of β-catenin staining, but this effect was reversed by XAV-939 (P < 0.05; Figure 8C)."
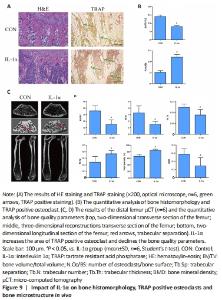
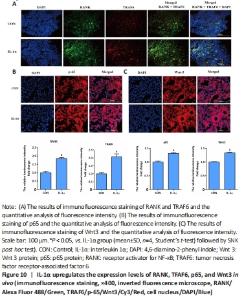
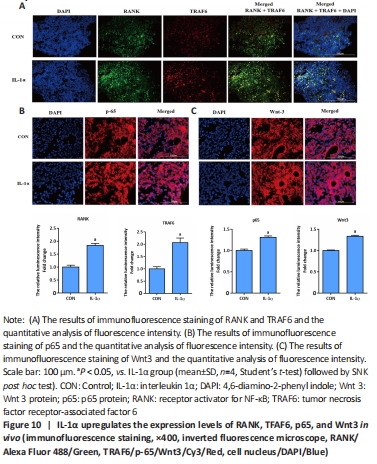
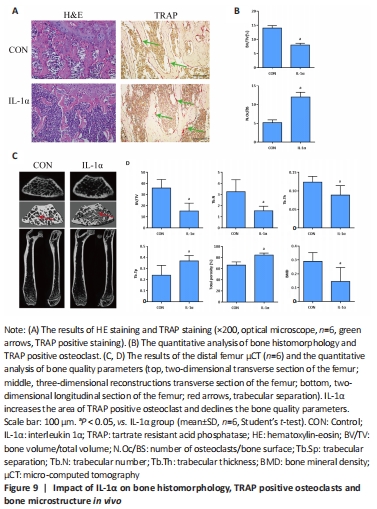
IL-1α induces bone loss in vivo We evaluated the impact of IL-1α on bone loss and osteoclast activity in mice. Results from hematoxylin-eosin staining and TRAP staining demonstrated that IL-1α injection markedly declined bone structure and increased TRAP positive area in the distal femurs of mice (P < 0.05; Figure 9A and B). The μCT quantitative analysis of bone parameters found that IL-1α treatment significantly decreased BMD, Tb.Th, Tb.N, and BV/TV while increasing Tb.Sp and total porosity (%) in vivo (P < 0.05; Figure 9C and D). Moreover, immunofluorescence analysis of the femur exhibited that IL-1α significantly enhanced the expression of osteoclast-specific genes (RANK, TRAF6) and signaling components (p65, Wnt3) compared with those in control mice (P < 0.05; Figure 10). IL-1α induced osteoclast activation and bone loss in mice."
| [1] ROODMAN GD. Advances in bone biology: the osteoclast. Endocr Rev. 1996;17:308-332. [2] HE S, ZHOU Q, LUO B, et al. Chloroquine and 3-methyladenine attenuates periodontal inflammation and bone loss in experimental periodontitis. Inflammation. 2020;43(1):220-230. [3] TEITELBAUM SL. Bone resorption by osteoclasts. Science. 2000;289:1504-1508. [4] KITAURA H, MARAHLEH A, OHORI F, et al. Osteocyte-related cytokines regulate osteoclast formation and bone resorption. Int J Mol Sci. 2020;21(14):5169. [5] YASUDA H. Discovery of the RANKL/RANK/OPG system. J Bone Miner Metab. 2021;39(1):2-11. [6] YAMAZAKI M, KAWAI M, MIYAGAWA K, et al. Interleukin-1-induced acute bone resorption facilitates the secretion of fibroblast growth factor 23 into the circulation. J Bone Miner Metab. 2015;33(3):342-354. [7] GUO C, YANG XG, WANG F, et al. IL-1α induces apoptosis and inhibits the osteoblast differentiation of MC3T3-E1 cells through the JNK and p38 MAPK pathways. Int J Mol Med. 2016; 38:319-327. [8] MENG M, CHEN Y, CHEN X, et al. IL-1α Regulates osteogenesis and osteoclastic activity of dental follicle cells through JNK and p38 MAPK pathways. Stem Cells Dev. 2020;29(24):1552-1566. [9] BRODZIKOWSKA A, GÓRSKA R, KOWALSKI J. Interleukin-1 genotype in periodontitis. Arch Immunol Ther Exp. 2019;67(6):367-373. [10] VINCENT TL. IL-1 in osteoarthritis: time for a critical review of the literature. F1000Res. 2019;8:934. [11] DE MARTINIS M, GINALDI L, SIRUFO MM, et al. Alarmins in osteoporosis, RAGE, IL-1, and IL-33 pathways: a literature review. Medicina (Kaunas). 2020;56(3):138. [12] KHAZIM K, AZULAY EE, KRISTAL B, et al. Interleukin 1 gene polymorphism and susceptibility to disease. Immunol Rev. 2018;281(1):40-56. [13] SAAD K, ABDALLAH AM, ABDEL-RAHMAN AA, et al. Polymorphism of interleukin-1β and interleukin-1 receptor antagonist genes in children with autism spectrum disorders. Prog Neuropsychopharmacol Biol Psychiatry. 2020;103:109999. [14] KIM JM, LIN C, STAVRE Z, et al. Osteoblast-osteoclast communication and bone homeostasis. Cells. 2020;9(9):2073. [15] THUMMURI D, GUNTUKU L, CHALLA VS, et al. Abietic acid attenuates RANKL induced osteoclastogenesis and inflammation associated osteolysis by inhibiting the NF-KB and MAPK signaling. J Cell Physiol. 2019; 234:443-453. [16] Kim K, Kim JH, Kim I, et al. TRIM38 regulates NF-κB activation through TAB2 degradation in osteoclast and osteoblast differentiation. Bone. 2018;113:17-28. [17] JIA ZK, LI HY, LIANG YL, et al. Monomeric C-reactive protein binds and neutralizes receptor activator of nf-κb ligand-induced osteoclast differentiation. Front Immunol. 2018;9:234. [18] NAKAMURA I, JIMI E. Regulation of osteoclast differentiation and function by interleukin-1. Vitam Horm. 2006;74:357-370. [19] WEIVODA MM, RUAN M, HACHFELD CM, et al. Wnt Signaling Inhibits Osteoclast Differentiation by Activating Canonical and Noncanonical cAMP/PKA Pathways. J Bone Miner Res. 2016;31:65-75. [20] MAEDA K, KOBAYASHI Y, KOIDE M, et al. The regulation of bone metabolism and disorders by Wnt signaling. Int J Mol Sci. 2019;20(22):5525. [21] SUI X, DENG S, LIU M, et al. Constitutive activation of β-catenin in differentiated osteoclasts induces bone loss in mice. Cell Physiol Biochem. 2018;48:2091-2102. [22] YOSHIDA Y, YAMASAKI S, OI K, et al. IL-1β Enhances Wnt Signal by Inhibiting DKK1. Inflammation. 2018;41(5):1945-1954. [23] YANG L, WANG Z, ZOU C, et al. Ubiquitin-specific protease 49 attenuates IL-1β-induced rat primary chondrocyte apoptosis by facilitating Axin deubiquitination and subsequent Wnt/β-catenin signaling cascade inhibition. Mol Cell Biochem. 2020;474(1-2):263-275. [24] MARINO S, LOGAN JG, MELLIS D, et al. Generation and culture of osteoclasts. Bonekey Rep. 2014;3:570. [25] CHEN X, ZHI X, PAN P, et al. Matrine prevents bone loss in ovariectomized mice by inhibiting RANKL-induced osteoclastogenesis. FASEB J. 2017;31:4855-4865. [26] LIVAK KJ, SCHMITTGEN TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods. 2001;25:402-408. [27] MANTOVANI A, DINARELLO CA, MOLGORA M, et al. Interleukin-1 and related cytokines in the regulation of inflammation and immunity. Immunity. 2019;50(4):778-795. [28] KANY S, VOLLRATH JT, RELJA B. Cytokines in inflammatory disease. Int J Mol Sci. 2019;20(23):6008. [29] JENEI-LANZL Z, MEURER A, ZAUCKE F. Interleukin-1β signaling in osteoarthritis-chondrocytes in focus. Cell Signal. 2019;53:212-223. [30] EBE Y, NAKAMURA T, HASEGAWA-NAKAMURA K, et al. Effect of interleukin-1β on bone morphogenetic protein-9-induced osteoblastic differentiation of human periodontal ligament fibroblasts. Eur J Oral Sci. 2021;129(4):e12792. [31] ZHAO Y, CAI B, ZHU W, et al. IL-1 receptor antagonist protects the osteogenesis capability of gingival-derived stem/progenitor cells under inflammatory microenvironment induced by Porphyromonas gingivalis lipopolysaccharides. Stem Cells Int. 2021;2021:6638575. [32] KIM JH, JIN HM, KIM K, et al. The mechanism of osteoclast differentiation induced by IL-1. J Immunol. 2009;183:1862-1870. [33] YUKATA K, XIE C, LI TF, et al. Teriparatide (human PTH1-34) compensates for impaired fracture healing in COX-2 deficient mice. Bone. 2018;110:150-159. [34] LEE YM, FUJIKADO N, MANAKA H, et al. IL-1 plays an important role in the bone metabolism under physiological conditions. Int Immunol. 2010;22:805-816. [35] BOYLE WJ, SIMONET WS, LACEY DL. Osteoclast differentiation and activation. Nature. 2003;423(6937):337-342. [36] BOYCE BF. Advances in the regulation of osteoclasts and osteoclast functions. J Dent Res. 2013;92(10):860-867. [37] Cheng SC, Huang WC, S Pang JH, et al. Quercetin inhibits the production of il-1β-induced inflammatory cytokines and chemokines in ARPE-19 cells via the MAPK and NF-κB signaling pathways. Int J Mol Sci. 2019;20(12):2957. [38] ILCHOVSKA DD, BARROW DM. An Overview of the NF-kB mechanism of pathophysiology in rheumatoid arthritis, investigation of the NF-kB ligand RANKL and related nutritional interventions. Autoimmun Rev. 2021;20(2):102741. [39] SHAO J, DING Z, PENG J, et al. MiR-146a-5p promotes IL-1β-induced chondrocyte apoptosis through the TRAF6-mediated NF-kB pathway. Inflamm Res. 2020;69(6):619-630. [40] APPELMAN-DIJKSTRA NM, PAPAPOULOS SE. Clinical advantages and disadvantages of anabolic bone therapies targeting the WNT pathway. Nat Rev Endocrinol. 2018;14(10):605-623. [41] YANG B, LI S, CHEN Z, et al. Amyloid β peptide promotes bone formation by regulating Wnt/β-catenin signaling and the OPG/RANKL/RANK system. FASEB J. 2020;34(3):3583-3593. [42] AMIRHOSSEINI M, MADSEN RV, ESCOTT KJ, et al. GSK-3β inhibition suppresses instability-induced osteolysis by a dual action on osteoblast and osteoclast differentiation. J Cell Physiol. 2018;233(3):2398-2408. [43] KOBAYASHI Y, UEHARA S, KOIDE M. Regulations of osteoclast formation and function by Wnt signals. Clin Calcium. 2019;29(3):309-315. [44] SUI Y, LIU Z, PARK SH, et al. IKKβ is a β-catenin kinase that regulates mesenchymal stem cell differentiation. JCI Insight. 2018;3(2):e96660. [45] LIU XZ, FAN J, QI K, et al. Dishevelled2 promotes apoptosis and inhibits inflammatory cytokine secretion in rheumatoid arthritis fibroblast-like synoviocytes through crosstalk with the NF-κB pathway. Oncotarget. 2017;8(8):12649-12663. |
| [1] | Wang Jing, Xiong Shan, Cao Jin, Feng Linwei, Wang Xin. Role and mechanism of interleukin-3 in bone metabolism [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1260-1265. |
| [2] | Xiao Hao, Liu Jing, Zhou Jun. Research progress of pulsed electromagnetic field in the treatment of postmenopausal osteoporosis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1266-1271. |
| [3] | Hu Weifan, Zheng Li, Li Dadi, Sun Yang, Zhao Fengchao. Overexpression of miR-25 downregulates titanium particle-induced osteoclast differentiation through the NFATc1 signaling pathway [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(5): 682-687. |
| [4] | Shan Shuai, Dong Yi, Liu Jialin, Han Xiangzhen, He Huiyu. Effects of red deer horn powder extract on the biological behavior of bone marrow derived macrophages [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(24): 3840-3845. |
| [5] | Liu Qiqi, Liu Min, Yang Jian, Yu Ke. Lipopolysaccharide stimulates the expression of interleukin-6 and nuclear factor kappa B receptor activator ligand in mouse MLO-Y4 osteoblasts [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(20): 3121-3126. |
| [6] | Wang Jiajia, Liu Jie, Wang Min. Establishing a murine model of experimental apical periodontitis induced by Fusobacterium nucleatum [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(2): 176-181. |
| [7] | Wu Saixuan, Zhang Mi, Dong Ming, Lu Ying, Niu Weidong. Regulatory role of Keap1/Nrf2/ARE signaling pathway in bone homeostasis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(2): 271-275. |
| [8] | Sun Youqiang, Ma Chao, Liang Mengmeng, Xin Pengfei, Zhang Hua, Xiang Xiaobing. The pivotal role of autophagy in bone cells: bone-related cell activity and bone metabolism [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(2): 276-282. |
| [9] | Fan Danyang, Fu Runze, Mi Jiajing, Liu Chunyan. Expression and role of cannabinoid receptors during bone remodeling [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(2): 283-288. |
| [10] | Wang Weiwei, Ou Zhixue, Zhang Xiaoyun, Li Shibin, Zhou Yi, Li Tong. Regulatory mechanism of exosomes in signal communication network of steroid induced avascular necrosis of the femoral head repair [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(19): 3056-3064. |
| [11] | Li Yufan, Lin Mingyue, Wang Chenxin, Zhang Rui, Li Yubao, Chen Li, Zou Qin. Application of micro-CT in rabbit tibial osteomyelitis modeling [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(18): 2874-2880. |
| [12] | Wang Jing, Wang Wanyuji, Zhang Yi, Ma Yaping, Wang Xin. Interleukin 6 involved in a series of reaction processes of osteogenesis and bone repair [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(18): 2945-2951. |
| [13] | Cheng Yi, Liu Ting, Guo Yujing, Sun Xiaotong, Bi Lan, Zhang Ronghe. Effect of estrogen on bone remodeling and root resorption during orthodontics [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(17): 2782-2788. |
| [14] | Huang Fayi, Liu Yuhao, Zhou Chi, Huang Haoran, Chen Weijian, He Wei, Wang Haibin. Belamcandin inhibits osteoclast differentiation: its role and mechanism [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(17): 2636-2641. |
| [15] | Tian Xinbao, Xu Jianfeng, Huang Yuan, Lai Zheying, Li Xiaolong, Liu Xiaoli, Lin Ruizhu, Zhu Ning. Internal heat-type acupuncture inhibits osteoblast viability and promotes bone formation in a rat model of steroid-induced avascular necrosis of the femoral head [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(17): 2642-2648. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||