Chinese Journal of Tissue Engineering Research ›› 2022, Vol. 26 ›› Issue (18): 2874-2880.doi: 10.12307/2022.697
Previous Articles Next Articles
Application of micro-CT in rabbit tibial osteomyelitis modeling
Li Yufan1, 2, Lin Mingyue1, 2, Wang Chenxin1, 2, Zhang Rui1, 2, Li Yubao1, 2, Chen Li1, Zou Qin1, 2
- 1Analysis and Testing Center, 2Nano-biomaterials Research Center, Sichuan University, Chengdu 610064, Sichuan Province, China
-
Received:2021-08-27Accepted:2021-09-30Online:2022-06-28Published:2022-01-30 -
Contact:Chen Li, PhD, Experimentalist, Analysis and Testing Center, Sichuan University, Chengdu 610064, Sichuan Province, China Zou Qin, PhD, Associate researcher, Analysis and Testing Center, Sichuan University, Chengdu 610064, Sichuan Province, China; Nano-biomaterials Research Center, Sichuan University, Chengdu 610064, Sichuan Province, China -
About author:Li Yufan, Master candidate, Analysis and Testing Center, Nano-biomaterials Research Center, Sichuan University, Chengdu 610064, Sichuan Province, China -
Supported by:the Major Science and Technology Project of Sichuan Province, No. 2020ZDZX0008 (to ZQ); Chengdu Municipal Science and Technology Project, No. 2019-YF05-00020-SN (to ZQ)
CLC Number:
Cite this article
Li Yufan, Lin Mingyue, Wang Chenxin, Zhang Rui, Li Yubao, Chen Li, Zou Qin. Application of micro-CT in rabbit tibial osteomyelitis modeling[J]. Chinese Journal of Tissue Engineering Research, 2022, 26(18): 2874-2880.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
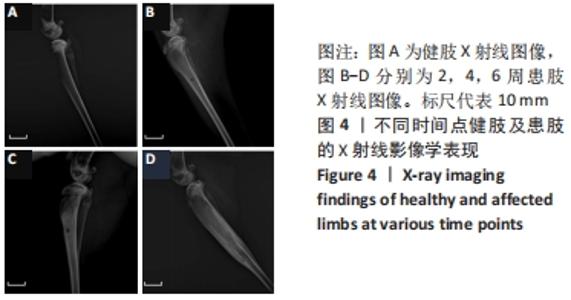
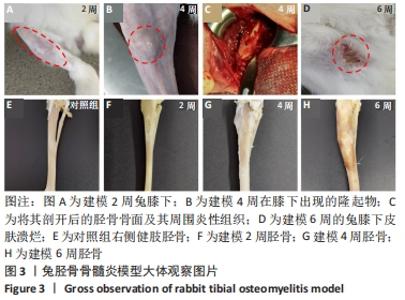
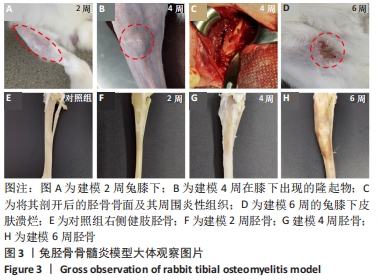
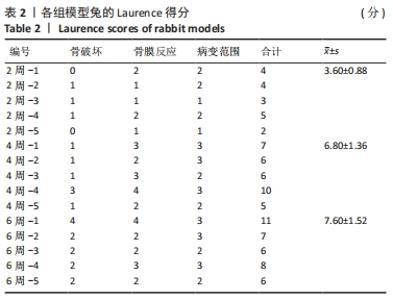
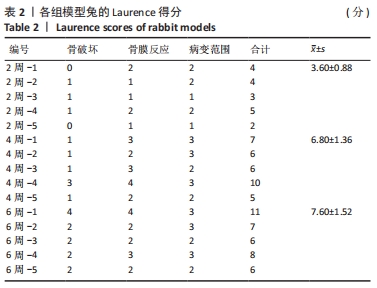
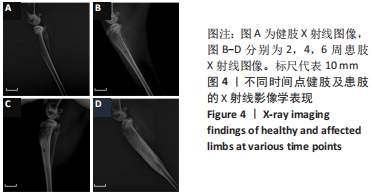
建模组实验动物出现膝下局部肿胀,日活动频率显著降低,步姿偏跛。造模术后2周,膝下局部脓肿,见图3A,取样可见有骨膜反应,骨组织未见明显肿胀,见图3F。术后4周,膝下脓肿隆起明显,见图3B,取样后可见造模处周围有黄色黏稠脓性液体及粒状脓性组织,见图3C,骨肿胀明显并出现炎性反应,见图3G。术后6周,膝下皮肤溃烂,致病菌已从骨组织内流出,导致周围软组织较重的感染,窦道形成同时有渗出物自窦道流出,流出物色黄,稍黏稠,内部组织暴露,见图3D,大面积骨质增生腓骨与胫骨出现粘连迹象,见图3H。对照组胫骨骨面光滑,无肿胀隆起,见图3E。 2.3 X射线影像学观察结果 由X射线片可见,对照组骨连续性好,骨皮质光滑完整,无骨破坏及骨膜反应,见图4A。建模组术后2周,摄胫骨侧位片,可见轻微骨膜反应,骨破坏不明显,见图4B。术后4周,胫骨骨膜反应与骨破坏明显,病变范围向骨窗周围扩张,见图4C。术后6周,胫骨明显肿胀,骨膜反应加剧,骨密度降低,骨皮质变薄,骨溶解,骨干畸形和疑似软组织炎性阴影,见图4D。实验兔Laurence评分显示,2周最高 5 分,最低 2 分,平均(3.60±0.88)分;4周最高10分,最低5分,平均(6.80±1.36)分;6周最高11分,最低6分,平均(7.60±1.52)分,见表2。平均值渐升说明炎症随时间逐渐加剧,而标准偏差增加说明不同动物对炎症的耐受程度不同,受个体差异影响较大。"
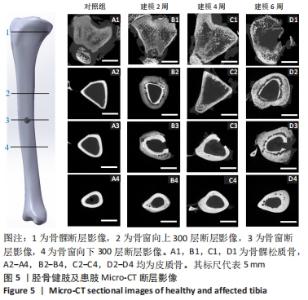
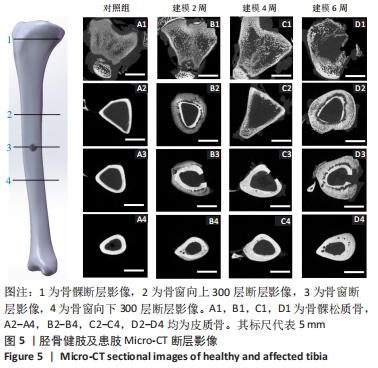
2.4 Micro-CT分析结果 2.4.1 Micro-CT影像分析结果 由Micro-CT断层影像可见,对照组胫骨影像解剖结构清晰可见,对比度和锐利度良好,未出现明显伪影和噪声,骨髁松质骨均匀,骨小梁结构连续,厚度分布均匀,呈现规则的海绵网状结构,见图5A1,皮质骨形貌完整,无异常的肿胀或缺损,见图5A2-4。建模2周,骨髁松质骨骨质增生,骨小梁结构连续性改变,少量骨小梁断裂,见图5B1,皮质骨出现空洞,见图5B2-4,骨质破坏,骨窗及骨窗上部骨质疏松明显。建模4周,骨髁松质骨部分骨小梁变细,断裂,大部分骨小梁的连接消失,见图5C1,骨窗及上下层皮质骨对比对照组发生明显肿胀,见图5C2-4。建模6周,骨髁松质骨部分骨小梁断裂破碎,连续性破坏,结构较为散乱,见图5D1。皮质骨变薄有缺损空洞形成,骨外面有广泛性的层状高密度影,骨膜反应明显,见图5D2-4。"
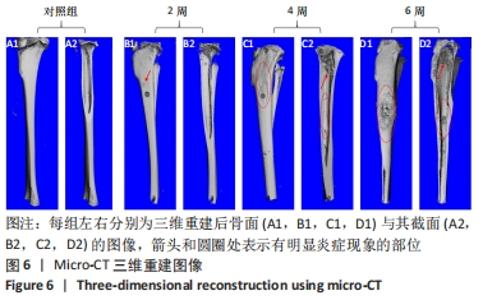
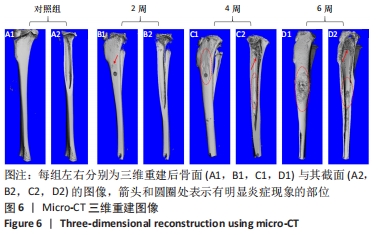
2.4.2 三维重建结果 对实验兔胫骨进行三维重建可见,对照组骨面光滑,无隆起或增生,无死骨形成,骨松质与骨密质可清晰区分,内部可见对照组骨内膜完整光滑,骨髁松质骨均匀,骨小梁结构清晰,无骨质破坏,见图6A。由2周后的重建图像可见,造模处周围有轻微骨膜反应,见图6B1→,肿胀不明显,内部无明显病变,骨髁处松质骨有少量破损断裂,见图6B2。建模4周,骨窗周围可见明显的骨质增生,见图6C1○,肿胀明显;内部图像显示松质骨结构连续性破坏,大量骨小梁断裂破损,向骨窗处增生,见图6C2→,骨膜有炎症反应,见图6C2○。建模6周,骨窗周围有大量死骨形成且骨膜反应剧烈,整体明显肿胀,骨质增生严重,见图6D1○;内部松质骨结构严重破坏变形,骨小梁大面积破损缺失,见图6D2→,且炎症已蔓延至整根胫骨,骨内膜有显著损伤,皮质骨增生且出现空隙,骨关节面破损,见图6D2○。"
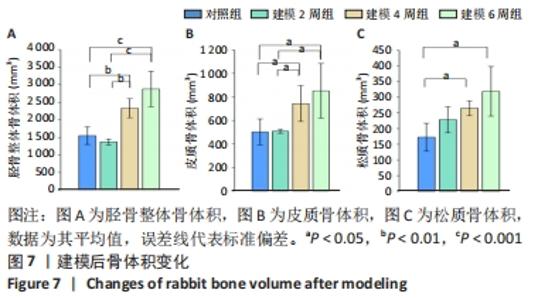
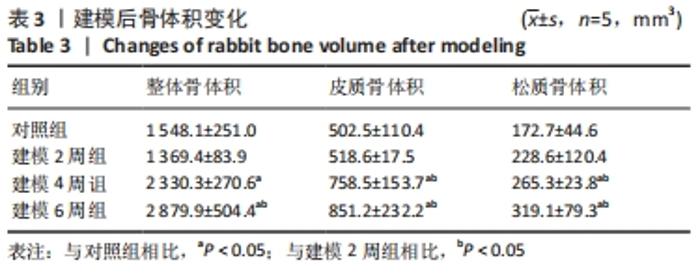
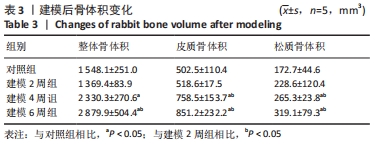
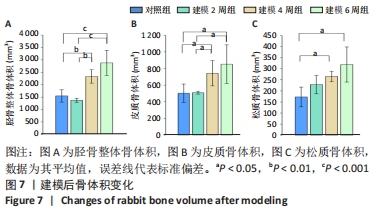
骨体积是衡量骨骼形态的重要测量值,可以衡量骨骼的发育情况,与骨骼的力学性能也息息相关。随建模时间推移,胫骨整体骨体积均有所增加。对照组的整体骨体积为(1 548.1±251.0) mm3,建模2周的胫骨整体骨体积与对照组相比没有明显差异;建模4周胫骨有明显肿胀,整体骨体积相比对照组明显增大至(2 330.3±270.6) mm3(P < 0.01);建模6周,整体骨体积明显高于建模2周组和对照组(P < 0.001),见图7A。 皮质骨体积对照组和建模2周组相比无明显变化;而4周和6周皮质骨肿胀都较为明显,与2周建模和对照组相比差异均有显著性意义(P < 0.05),见图7B。 但由此次实验中的CT断层图像可见,骨骺线上下方松质骨均受炎症影响产生了较大变化,因此将其上下方的松质骨均纳入计算范围。在选取感兴趣区时,骨骺线下方仅选择骨小梁部分,而上方的松质骨与其表面较薄的皮质骨难以区分,因此也纳入了体积的统计。此次计算的松质骨体积是所有松质骨以及骨骺线上方骨表面直至髁间隆起处表层皮质骨的总体积。对照组松质骨骨体积为(172.7±44.6) mm3,2周松质骨体积略有增长至(228.6±120.4) mm3但与对照组相比差异无显著性意义。4周和6周松质骨体积与2周建模、对照组相比差异均有显著性意义(P < 0.05),见图7C。"
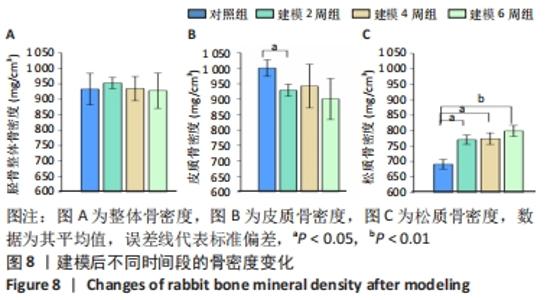
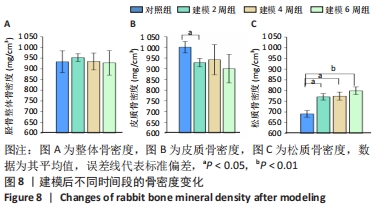
整体骨骨密度随建模时间变化不明显,约为930 mg/cm3。通过对皮质骨骨密度分析可见,建模组的皮质骨骨密度与对照组(1 000.4±26.4) mg/cm3相比,差异均有显著性意义(P < 0.05);但随着建模时间的延长,皮质骨骨密度无明显变化,分别为(928.3±19.5) mg/cm3(2周)、(941.8±70.5) mg/cm3(4周)以及(900.1±66.6) mg/cm3(6周)。松质骨在建模后的骨密度均明显高于对照组(691.2±55.2) mg/cm3(P < 0.05),但随建模时间的延长各组之间无明显变化,分别为(770.5±14.9) mg/cm3(2周)、(774.0±18.7) mg/cm3 (4周),直至6周时的(799.0±17.8) mg/cm3,见图8。"
| [1] 黄金亮, 唐辉, 徐永清. 骨髓炎流行病学[J]. 国际骨科学杂志,2011, 32(2):26-27. [2] AGAJA SB, AYORINDE RO. Chronic osteomyelitis in Ilorin, Nigeria. S Afr J Surg . 2008;46(4):116-118. [3] 吴二俊, 张树明. 慢性骨髓炎的治疗进展[J]. 临床医药实践,2016, 25(10):780-785. [4] DELEO FR, CHAMBERS HF. Reemergence of antibiotic-resistant Staphylococcus aureus in the genomics era. J Clin Invest. 2009;119(9): 2464. [5] KUBES JN, FRIDKIN SK. Factors affecting the geographic variability of antibiotic-resistant healthcare-associated infections in the United States using the CDC Antibiotic Resistance Patient Safety Atlas. Infect Control Hosp Epidemiol. 2019;40(5):597-599. [6] MONACO M, PIMENTEL DE ARAUJO F, CRUCIANI M, et al. Worldwide Epidemiology and Antibiotic Resistance of Staphylococcus aureus//BAGNOLI F, RAPPUOLI R, GRANDI G. Staphylococcus aureus: Microbiology, Pathology, Immunology, Therapy and Prophylaxis. Cham; Springer International Publishing. 2017:21-56. [7] SALGADO-PABÓN W, SCHLIEVERT PM. Models matter: the search for an effective Staphylococcus aureus vaccine. Nat Rev Microbiol. 2014; 12(8):585-591. [8] HORST SA, HOERR V, BEINEKE A, et al. A Novel Mouse Model of Staphylococcus aureus Chronic Osteomyelitis That Closely Mimics the Human Infection: An Integrated View of Disease Pathogenesis. Am J Pathol. 2012;181(4):1206-1214. [9] HASSANI BESHELI N, MOTTAGHITALAB F, ESLAMI M, et al. Sustainable Release of Vancomycin from Silk Fibroin Nanoparticles for Treating Severe Bone Infection in Rat Tibia Osteomyelitis Model. ACS Biomater Sci Eng. 2017;9(6):5128-5138. [10] 鲁锋,严雪征,马术友,等. 加替沙星-聚癸二酸酐局部缓释系统预防兔骨髓炎[J]. 中国组织工程研究,2014,18(21):3355-3360. [11] 刘飞,崔宇韬,刘贺. 局部抗生素递送系统治疗骨髓炎的优势与问题[J]. 中国组织工程研究,2021,25(4):128-134. [12] CLARK DP, BADEA CT. Advances in micro-CT imaging of small animals. Physica Medica. 2021;88(6):175-192. [13] 郭磊, 赵宏斌. 显微CT在骨科研究中的应用优势[J]. 昆明医学院学报,2012,33(S1):274-276. [14] 李亮亮. 兔慢性骨髓炎模型的建立及效果评价[J]. 安徽医药,2015, 19(12):2299-2301. [15] 高翠芳, 李晓泉. 5%鱼肝油酸钠对慢性骨髓炎造模的影响[J]. 中医正骨,2002,14(9):12-13+63-64. [16] HORN J, SCHLEGEL U, KRETTEK C, et al. Infection resistance of unreamed solid, hollow slotted and cannulated intramedullary nails : an in-vivo experimental comparison. J Orthop Res. 2010;23(4):810-815. [17] DAHNERS LE, FUNDERBURK CH. Gentamicin-loaded plaster of Paris as a treatment of experimental osteomyelitis in rabbits. Clin Orthop Relat Res. 1987;(219):278-282. [18] 王军, 毕龙, 白建萍, 等. 显微CT与组织切片技术在骨形态计量研究中的比较[J]. 中国矫形外科杂志,2017,223(5):381-384. [19] 郭磊, 赵宏斌. 显微CT在骨科研究中的应用优势[J]. 昆明医科大学学报,2012,33(S1):274-276. [20] 卞阳阳, 武子荃, 顾运涛, 等. 创伤后慢性骨髓炎的治疗进展[J]. 海南医学,2018,29(22):124-127. [21] 袁承杰, 翁蔚宗, 周启荣, 等. 骨髓炎骨缺损动物模型构建研究进展[J]. 中华骨与关节外科杂志,2016,9(1):87-90+83. [22] 李鼎鹏, 谢兴文, 汪付田, 等. 基于文献回顾分析动物骨髓炎模型制备方法的特点研究[J]. 中药药理与临床,2019,35(5):159-163. [23] PERLMAN RL. Mouse Models of Human Disease: An Evolutionary Perspective. Evol Med Public Health. 2016;2016(1):170-176. [24] PEARCE AI, RICHARDS RG, MILZ S, et al. Animal models for implant biomaterial research in bone: a review. Eur Cells Mater. 2007;13:1. [25] NORDEN CW, MYEROWITZ RL, KELETI E. Experimental osteomyelitis due to Staphylococcus aureus or Pseudomonas aeruginosa: a radiographic-pathological correlative analysis. Br J Exp Pathol. 1980;61(4):451. [26] 闻一新, 周田华, 徐永清, 等. 兔胫骨金黄色葡萄球菌慢性骨髓炎模型的制备[J]. 实用骨科杂志,2013,19(11):1001-1003. [27] POULTSIDES LA, PAPATHEODOROU LK, KARACHALIOS TS, et al. Novel model for studying hematogenous infection in an experimental setting of implant-related infection by a community-acquired methicillin-resistant S. aureus Strain. J Orthop Res. 2008;26(10):1355-1362. [28] 褚立涛, 郭国栋, 刘刚, 等. 兔胫骨创伤后骨髓炎动物模型的建立[J]. 医学研究生学报,2012,25(11):1160-1162. [29] 李彦豪,李树新. 鱼肝油酸钠动脉栓塞的实验研究[J]. 中华放射学杂志,1987,21(6):357-360. [30] 滕加文,梁学振,李梁.骨髓炎兔胫骨动物模型的建立与评价[J]. 中国现代药物应用,2017,11(16):194-197. [31] 平小芳.骨髓炎的影像学诊断[J]. 中外医学研究,2010,8(19):91. [32] FREEMAN TA, PATEL P, PARVIZI J, et al. Micro-CT analysis with multiple thresholds allows detection of bone formation and resorption during ultrasound-treated fracture healing. J Orthop Res. 2010;27(5):673-679. [33] 李高团.不典型骨髓炎的X线,CT和MRI影像特点比较[J]. 深圳中西医结合杂志,2020,30(6):65-67. [34] 邹梦晨,胡萍,罗祥蓉,等.糖尿病足骨髓炎的诊疗进展[J]. 国际内分泌代谢杂志,2018,38(4):257-261. [35] 雷霆,刘傥,禹晓东,等.显微CT在骨科学中的应用[J].中国矫形外科杂志,2010,18(8):652-653. [36] NASSER A, AZIMI T, OSTADMOHAMMADI S, et al. A comprehensive review of bacterial osteomyelitis with emphasis on Staphylococcus aureus. Microb Pathog. 2020;148:104431. [37] ANA B, MM C, PJ D, et al. Staphylococcus aureus versus neutrophil: Scrutiny of ancient combat - ScienceDirect. Microb Pathog. 2019;131: 259-269. [38] DYM H, ZEIDAN J. Microbiology of Acute and Chronic Osteomyelitis and Antibiotic Treatment. Dent Clin North Am. 2017;61(2):271-282. [39] SACHA F, JEAN-PHILIPPE R, CéDRIC B, et al. Human Monocyte-Derived Osteoclasts Are Targeted by Staphylococcal Pore-Forming Toxins and Superantigens. Plos One. 2016;11(3):e0150693. [40] 杜虎羽, 刘君, 郭志坚, 等. 应用Masquelet技术治疗跟骨骨髓炎的临床研究[J]. 中国矫形外科杂志,2017,25(15):1435-1437. [41] 彭江, 汪爱媛, 孙明学, 等. Micro-CT在松质骨结构研究中的应用[J]. 中国矫形外科杂志,2005,13(11):859-861. [42] 王法琪, 严亚波, 温鑫鑫, 等. 显微CT分辨率对松质骨微观结构及生物力学测量的影响[J]. 现代生物医学进展,2015,15(10):1877-1880. [43] 周佳雁, 蒋晓红, 王龙. 以脓毒症首诊的2型糖尿病合并脊柱化脓性骨髓炎一例分析[J]. 临床误诊误治,2018,31(1):44-46. |
| [1] | Wei Guoqiang, Li Yunfeng, Wang Yi, Niu Xiaofen, Che Lifang, Wang Haiyan, Li Zhijun, Shi Guopeng, Bai Ling, Mo Kai, Zhang Chenchen, Xu Yangyang, Li Xiaohe. Biomechanical analysis of non-uniform material femur under different loads [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1318-1322. |
| [2] | Wang Baojuan, Zheng Shuguang, Zhang Qi, Li Tianyang. Miao medicine fumigation can delay extracellular matrix destruction in a rabbit model of knee osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1180-1186. |
| [3] | Zhu Chan, Han Xuke, Yao Chengjiao, Zhang Qiang, Liu Jing, Shao Ming. Acupuncture for Parkinson’s disease: an insight into the action mechanism in animal experiments [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1272-1277. |
| [4] | Wang Xinmin, Liu Fei, Xu Jie, Bai Yuxi, Lü Jian. Core decompression combined with dental pulp stem cells in the treatment of steroid-associated femoral head necrosis in rabbits [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1074-1079. |
| [5] | Yi Xinrong, Jia Fuquan, He Xin, Zhang Shaojie, Ren Xiaoyan, Li Zhijun. Establishment of cervical bone age equation for male adolescents aged 8-16 years old in Hohhot based on thin-slice CT [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(6): 954-958. |
| [6] | Yuan Jing, Sun Xiaohu, Chen Hui, Qiao Yongjie, Wang Lixin. Digital measurement and analysis of the distal femur in adults with secondary knee valgus deformity [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(6): 881-885. |
| [7] | Wang Shaoling, Wang Yanxue, Zheng Yaochao, Yu Shaojun, Ma Chao, Wu Shaoling. Feasibility of ultrasound-guided intra-articular injection in rabbit hip joint [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(5): 657-662. |
| [8] | He Junjun, Huang Zeling, Hong Zhenqiang. Interventional effect of Yanghe Decoction on synovial inflammation in a rabbit model of early knee osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(5): 694-699. |
| [9] | Wang Shihui, Cheng Yang, Zhu Yunjie, Cheng Shaodan, Mao Jianying. Effect of arc edge needle-scalpel therapy on inflammatory factors and histomorphology of the frozen shoulder in rabbit models [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(5): 706-711. |
| [10] | Deng Zhaoyang, Yang Zhaohui. Three-dimensional model analysis of tibial plateau fracture lines [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(3): 334-339. |
| [11] | Yang Ruijuan, Li Yangyang, Cai Ruiyan, Liu Huibin, Guo Chun. Interleukin-1 alpha induces osteoclast activation and bone loss [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(23): 3691-3699. |
| [12] | Wang Zhen, Xu Yi, Tu Yihui. Entry point of an intramedullary rod in knee arthroplasty: determined using singular value decomposition method [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(18): 2828-2833. |
| [13] | Yang Xue, Wang Baoqun, Jiang Xiaowen, Zou Shengcan, Ming Jinfa, Lin Shasha. Preparation and properties of biodegradable plant polysaccharide hemostatic microspheres [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(16): 2487-2491. |
| [14] | Li Jinglian, Zhang Hongfei, Yan Jiapeng, Zhou Jiabin, Yu Hao. Lag screw path for fixation of sacroiliac joint dislocation through S1 pedicle [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(15): 2337-2341. |
| [15] | Lin Xiaosheng, Han Linjing, Wu Keliang, Zhang Zhen, Wang Hongbo, Xiao Qinghua, Du Genfa, Zhu Jianzong. Effects of Zishen Jiangu Recipe on bone microstructure and bone mineral density in ovariectomized rats with osteoporosis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(15): 2382-2386. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||