Chinese Journal of Tissue Engineering Research ›› 2022, Vol. 26 ›› Issue (34): 5518-5524.doi: 10.12307/2022.464
Previous Articles Next Articles
Advantages and application strategies of hyaluronic acid scaffold in cartilage repair engineering
Liu Hui, Liu Aifeng, Zhang Yu, Zhang Chao
- Department of Orthopedics and Traumatology, First Teaching Hospital of Tianjin University of Traditional Chinese Medicine; Regional TCM Bone Injury Diagnosis and Treatment Center of State Administration of Traditional Chinese Medicine, Tianjin 300381, China
-
Received:2021-06-01Accepted:2021-07-08Online:2022-12-08Published:2022-04-16 -
Contact:Liu Aifeng, Chief physician, Department of Orthopedics and Traumatology, First Teaching Hospital of Tianjin University of Traditional Chinese Medicine; Regional TCM Bone Injury Diagnosis and Treatment Center of State Administration of Traditional Chinese Medicine, Tianjin 300381, China -
About author:Liu Hui, Master candidate, Department of Orthopedics and Traumatology, First Teaching Hospital of Tianjin University of Traditional Chinese Medicine; Regional TCM Bone Injury Diagnosis and Treatment Center of State Administration of Traditional Chinese Medicine, Tianjin 300381, China -
Supported by:National Natural Science Foundation of China, No. 81873316 (to LAF)
CLC Number:
Cite this article
Liu Hui, Liu Aifeng, Zhang Yu, Zhang Chao. Advantages and application strategies of hyaluronic acid scaffold in cartilage repair engineering[J]. Chinese Journal of Tissue Engineering Research, 2022, 26(34): 5518-5524.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
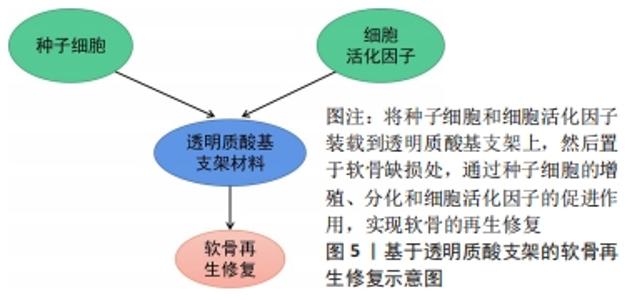
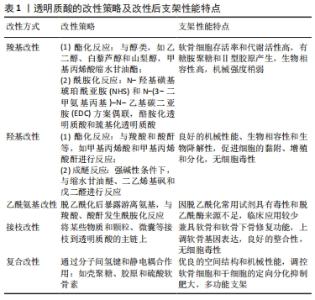
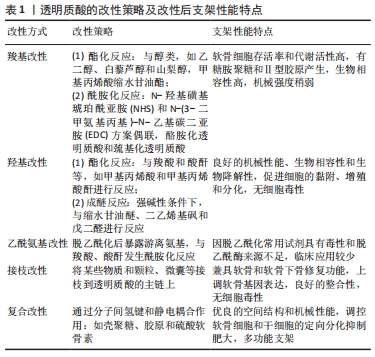
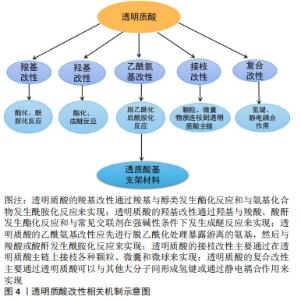
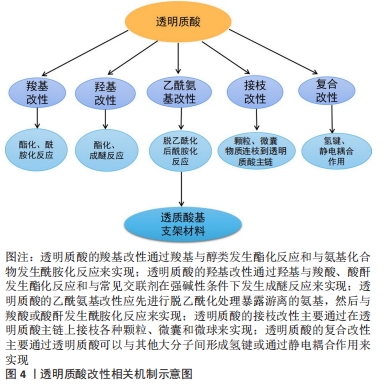
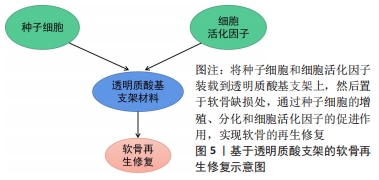
2.2 羧基改性 羧基是透明质酸上重要的活性官能团,也是透明质酸带负电属性的原因所在。透明质酸不仅可以与醇类发生酯化反应,还可以与氨基化合物发生反应形成酰胺键,此外透明质酸中的羧基经活化后可以与自身的羟基进行交联反应,形成不含交联剂的凝胶[34-35]。 2.2.1 酯化反应 HSIEH等[36]使用甲基丙烯酸缩水甘油酯对透明质酸进行了改性,并将其渗透到由甲氧基聚乙二醇-聚己内酯(mPEG-PCL)与羟基磷灰石以2∶1的质量比通过熔融沉积模型逐层混合形成的支架上,然后对猪的膝关节软骨缺损进行修复实验,并用CT跟踪修复的过程,结果发现实验组猪的软骨缺损处生长着不同的组织,对照组软骨缺损处则完全被再生组织覆盖,通过苏木精和曙红染色,发现实验组的软骨部分成熟,对照组的软骨则全都是纤维软骨,同时发现残留在软骨缺损处的支架会影响愈合过程,需重新考虑支架的设计。HE等[37]利用甲基丙烯酸缩水甘油酯和透明质酸在-20 ℃制成了一种拥有弹性、大孔和高度互连的网状低温凝胶,该凝胶在软骨缺损处可以为软骨细胞提供高存活率和高代谢活性的微环境,培养15 d后软骨细胞表现出较强的细胞增殖和代谢,软骨细胞外基质有糖胺聚糖和Ⅱ型胶原的产生,软骨细胞得到了良好的表型。但有学者在对其机械强度进行无侧限压缩实验时发现,经甲基丙烯酸甘油酯改性的透明质酸存在机械强度不足等缺点[38]。 白藜芦醇(反式-3,5,40-三羟基二苯乙烯)是一种天然的植物防御素,SHEU等[39]制备和表征了氧化型透明质酸/白藜芦醇(Oxi-透明质酸/Res)支架,细胞活力和细胞毒性实验表明,Oxi-透明质酸/Res支架具有良好的生物相容性,不仅可以上调Ⅱ型胶原、糖胺聚糖和环氧化酶9基因的表达,下调白细胞介素1β、MMP-1、MMP-3及MMP13基因的表达,还能减轻脂多糖诱导的炎症和软骨细胞损伤,是一种潜在的治疗软骨缺损的合适细胞载体。LIU等[40]使用乙二醇对氧化的透明质酸进行改性后再与壳聚糖合成水凝胶,通过不同浓度的软骨细胞外基质颗粒对该水凝胶进行功能化,发现该水凝胶可以为间充质干细胞的增殖提供优良的微环境,并且有较高水平的糖胺聚糖和Ⅱ型胶原的生成,小鼠体内软骨缺损得到了成熟组织的修复,并且与周围正常组织具有优异的整合性。LI等[41]开发了一种聚乙二醇改性的透明质酸支架,然后利用从人关节镜冲洗液中得到的间充质干细胞对大鼠的软骨缺损进行修复,结果显示随着培养时间的延长,水凝胶中间充质干细胞的数目显著增加,明显高于对照组,培养8周后软骨缺损得到了全层的修复,表面光滑,为透明软骨修复。MONGKHON等[42]发现山梨醇/透明质酸水凝胶在减轻软骨损伤所涉及的细胞的凋亡及改善骨关节炎方面具有积极作用。 2.2.2 酰胺化反应 利用羧基可发生酰胺化反应,JIN等[43]开发了一种修复骨关节炎软骨损伤的可注射透明质酸水凝胶,酪胺与透明质酸采用N-羟基磺基琥珀酰亚胺(NHS)和N-(3-二甲氨基丙基)-N-乙基碳二亚胺(EDC)方案偶联,即先将透明质酸钠溶于乙磺酸缓冲盐水中,制成体积分数1%的透明质酸溶液,在搅拌时入N-羟基磺基琥珀酰亚胺和N-(3-二甲氨基丙基)-N-乙基碳二亚胺,盐酸酪胺与透明质酸的摩尔比为1∶1,进而与表没食子儿茶素没食子酸酯物理混合制得注射用水凝胶,通过研究发现,该水凝胶可保护软骨细胞免受促炎因子白细胞介素1β的侵袭,在体外可诱导软骨再生。ZHANG等[44]也制备了一种Ⅰ型胶原-酪胺(Col-TA)和透明质酸-酪胺(HA-TA)组成的可注射水凝胶体系(Col-HA),结果发现Col-HA水凝胶具有优异的生物相容性,在体外能够为骨髓间充质干细胞提供良好的生长和软骨分化的微环境,是一种理想的软骨再生凝胶。有研究者将血小板裂解物作为细胞活化因子接入到透明质酸酪胺水凝胶中,观察了该水凝胶对人骨髓间充质干细胞的贴壁、存活和软骨定向分化的影响,见图5,结果表明该水凝胶在体外可诱导软骨样细胞外基质沉积,随着时间的推移,Ⅱ型胶原和蛋白多糖逐渐增多,新的细胞外基质在凝胶降解的同时沉积,最终形成坚韧致密的基质[45]。"
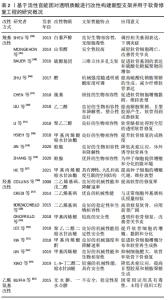
采用动态共价化学交联方法,ZHU等[46]使酰肼改性的类弹性蛋白(ELP-HYD)和醛改性的透明质酸(HA-ALD)通过动态腙键形成ELP-HA水凝胶的机械强度可随透明质酸浓度的改变而变化,可用于制备不同机械性能需求的支架;此外,研究者将软骨细胞包裹在该水凝胶中,发现随着透明质酸浓度的增加,15个软骨标志物基因的表达呈剂量依赖性增加,细胞外基质硫酸化糖胺聚糖的沉积也随之增加,能够最大限度减少不良纤维软骨的表达。 巯基化透明质酸通过2个自由巯基衍生的二硫键自交联形成,CHEN等[47]制备了一种体外构建组织工程软骨的注射用Ⅰ型胶原蛋白与巯基化透明质酸自交联共混水凝胶(HA-SH/Col),研究发现该水凝胶具有极佳的生物相容性,其包裹的软骨细胞能够保持软骨细胞表型,并且分泌大量基质,促进软骨细胞黏附、增殖和软骨化分化。BAUER等[48]开发了用于软骨修复的多孔新型交联透明质酸支架,该支架由透明质酸与含有硫醇基团的连接物进行衍生化,在pH值为7.1-7.3的去离子水溶液中硫醇基团逐渐形成二硫键,最终制得了三维立体多孔支架,该支架能够促进软骨特异性基因的表达和硫酸化糖胺聚糖的增加,软骨细胞分化指数明显改善,增强了软骨形成的表型,同时也促进了软骨的固位。 2.3 羟基改性 羟基是透明质酸的另一重要可修饰官能团,尤其是伯羟基,可以与含有羧酸和酸酐等活性基团的物质发生酯化反应,同时羟基可以在强碱性条件下与常见的交联剂发生成醚反应来实现透明质酸的交联改性[49-50]。 2.3.1 酯化反应 目前常见的改性物质有甲基丙烯酸和甲基丙烯酸酐等,XIA等[51]利用3D打印技术开发了一种光固化的可用于软骨修复的透明质酸水凝胶冻干支架,在制备该支架时,先利用3D打印技术精确控制透明质酸水凝胶的外观形状和内部孔结构,然后在该水凝胶体系中引入甲基丙烯酸酐和光引发剂,使水凝胶固化,结果发现采用冷冻干燥技术后,支架的力学性能得到了进一步提高,缓解了降解速率,其内部也观察到了适合细胞分布、黏附和增殖的孔隙结构,体外实验也证明该支架成功地再生出具有典型陷窝结构和软骨特异性细胞外基质的成熟软骨。甲基丙烯酸酯化透明质酸(mHA)已被证明通过激活关键的细胞信号通路来刺激软骨形成,LIN等[52]将甲基丙烯酸明胶(mGL)和mHA分别合成含有不同比例mHA的支架,以确定支架生物力学性能和软骨细胞增殖、分化能力最佳的mHA比例,结果表明当MGL/MHA的比例为9∶1(%,质量/体积)时,该支架具有最佳的压缩模量,人骨髓间充质干细胞的肥大程度最低,糖胺聚糖含量最高,体外实验也证明该比例支架植入12周后,兔股骨髁软骨缺损模型处的软骨和软骨下骨均出现再生,具有较高的软骨修复能力。ONOFRILLO等[53]也开发了由mGL和mHA合成的支架,该支架可以负载从骨关节炎患者髌下脂肪垫中获得的间充质干细胞,对软骨缺损进行透明样软骨修复,基于该支架的原位软骨缺损修复对软骨损伤和骨关节炎患者的定制化软骨修复具有重要意义。 2.3.2 成醚反应 在强碱性条件下,羟基优于羧基发生去质子化而形成具有强亲核性氧负离子,通过亲核加成形成醚键。XIAO等[54]用1,4-丁二醇二缩水甘油醚制备了透明质酸钠支架,在小型猪全层软骨缺损模型中评估了该支架的安全性和有效性,结果显示该支架具有较强的压缩模量和抗压能力,实验组Ⅱ型胶原和糖胺聚糖较对照组显著增加,血液学检查发现实验组所有常规血液学因子都在正常生理范围内,证实该支架植入是安全的,没有任何异物反应或炎症,然而组织学检查指出修复后的软骨组织不同于正常软骨,为纤维软骨。LEE等[55]以聚乙二醇二缩水甘油醚为交联剂制备透明质酸水凝胶,然后用鞣酸处理该水凝胶,鞣酸与聚乙二醇二缩水甘油醚之间强大的氢键既加强了该支架的机械性能,同时支持软骨细胞的黏附、增殖和分化,未有任何细胞毒性迹象出现;此外得益于鞣酸对透明质酸酶的抑制作用,该支架在体内表现出良好的稳定性,其降解速率也明显降低。 COLLINS等[56]使用二乙烯基砜和戊二醛交联剂对透明质酸进行改性,发现经二乙烯基砜和戊二醛改性后的水凝胶在机械性能和降解速率等方面都有明显改善。CREDI等[57]以二乙烯基砜为交联剂对透明质酸改性,研究二乙烯基砜含量对透明质酸水凝胶机械性能的影响,结果发现通过改变二乙烯基砜含量,可以制备杨氏模量在0.03-37.00 kPa范围的水凝胶,有趣的是,正常的细胞外基质的杨氏模量恰好在该范围内。BORZACCHIELLO等[58]通过二乙烯基砜对透明质酸进行改性,得到的水凝胶支架经检测无任何交联剂残留,该研究发现,通过增加透明质酸浓度和降低透明质酸/二乙烯基砜重量比可以改善该支架的机械强度,体外实验也证明该支架对酶降解表现出良好的稳定性以及生物相容性,是一种有前途的软骨组织工程支架。 2.4 乙酰氨基改性 乙酰氨基的改性主要是通过脱乙酰化来完成,透明质酸经脱乙酰化后将产生两性离子多糖,主要由β-1,4葡萄糖醛酸和β-1,3氨基葡萄糖的重复二糖单元组成[59]。乙酰氨基经脱乙酰化后暴露出的游离氨基可以作为改性的靶点,与羧酸或酸酐等发生酰胺化反应,甚至可以与自身的羧基发生交联产生新的凝胶支架[60]。透明质酸的脱乙酰化主要通过化学方法和酶裂解法实现[61],对于化学脱乙酰化,通常在强碱或酸性或反应温度在55-105 ℃的条件下进行,此条件下脱乙酰化会伴随着透明质酸的降解,目前常用的试剂有无水肼和一水合肼,虽然这些试剂在一定程度上限制了透明质酸在化学脱乙酰化时的降解,但这些试剂的毒性使得该方法并不能用于组织工程[62];酶裂解脱乙酰化则允许在温和的条件下进行,并且不会发生透明质酸的降解,而对于这种酶的来源及提取,研究者进行了积极探索,仍处于初期的实验阶段,未真正应用于组织工程中的软骨修复。基于透明质酸中活性官能团对透明质酸进行改性构建新型支架并用于软骨修复工程的研究汇总,见表2。"

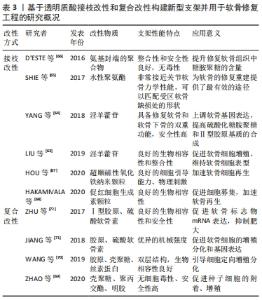
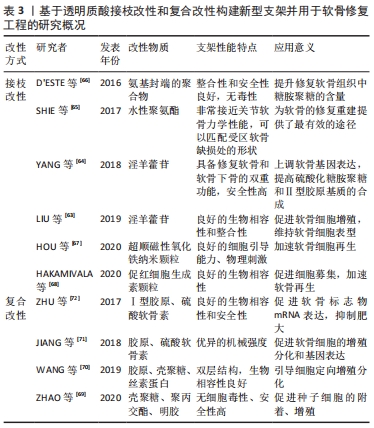
2.5 接枝改性 目前对于透明质酸的接枝改性的策略主要是通过将某些小分子物质、大分子聚合物以及包裹细胞活化因子的微球颗粒接枝到透明质酸主链上来实现的。 淫羊藿苷是从淫羊藿中分离得到的黄酮类成分,具有多种药理活性,LIU等[63]将淫羊藿苷先合成了具有良好细胞相容性和生物活性的硫代淫羊藿苷,然后通过胶原分子的自组装和巯基间二硫键的形成,制备了硫代化淫羊藿苷功能化透明质酸/胶原水凝胶支架,该支架不仅能促进软骨细胞的增殖、维持软骨细胞的表型、增强软骨外细胞基质的分泌,同时可以与周围正常组织达到良好整合。YANG等[64]通过研究也发现,将淫羊藿苷接枝到透明质酸和胶原水凝胶上,成软骨基因的表达水平明显上调,硫酸化糖胺聚糖和Ⅱ型胶原基质的合成显著增强,同时Runx2、碱性磷酸酶和骨钙蛋白等成骨基因的表达在早期也有显著上调,该支架具备修复软骨和软骨下骨的双重功能,是一种具有广泛应用前景的新型支架。 SHIE等[65]报道了一种以透明质酸为载体的水性聚氨酯共聚物支架,该支架是一种非常接近关节软骨力学性能的支架,同时借助3D打印技术,可以对该支架的三维结构进行设计以匹配受区软骨缺损处的形状,为软骨组织的修复与重建提供了最有效的途径。有研究者将N-异丙基丙烯酰胺和N,N-二甲基甲酰胺通过自由基聚合法合成了氨基封端的聚合物(PNIPAM-NH2),并将该聚合物与透明质酸的羧基通过共价接枝,得到了一种刷状共聚物HPN支架,在兔软骨缺损模型中评估HPN支架的安全性和软骨修复反应,结果发现在软骨缺损处形成了富含硫糖胺聚糖的纤维软骨组织,该组织与正常软骨组织具有良好的整合性,同时没有观察到局部或全身急性或亚急性毒性的迹象[66]。 将细胞活化因子包裹到微球囊或颗粒中再接枝到支架上,可以显著改善其释放速度。HOU等[67]提出了透明质酸接枝两亲性明胶微囊的仿生软骨细胞外基质环境,通过在明胶微囊壳中加入超顺磁性氧化铁纳米颗粒,显示出良好的细胞引导能力,而且还诱导了静磁场和磁致剪应力对软骨的两种物理刺激,加速了软骨细胞的再生。HAKAMIVALA等[68]将负载促红细胞生成素的颗粒接枝到透明质酸上,研究表明该支架具有良好的生物相容性,在促进内源性祖细胞募集的同时实现了软骨形成。 2.6 复合改性 透明质酸分子中的羧基、羟基和乙酰氨基等活性官能团可以通过形成分子间氢键的方式与其它聚合物制备新型支架,是实现透明质酸复合改性的一种方式;透明质酸是一种阴离子聚多糖,因此透明质酸可以与阳离子聚合物通过静电作用耦合到一起,是实现透明质酸复合改性的又一常用方式。 近年来,电纺纳米纤维因其在天然细胞外基质中模拟胶原纳米纤维网络的形态而被广泛研究作为促进细胞生物学活性的支架材料。ZHAO等[69]通过静电纺丝将透明质酸、壳聚糖、聚丙交酯和明胶合成了纤维网状支架,经过研究发现,所制备的支架都无细胞毒性,能够促进种子细胞的附着、扩散和增殖,以上聚合物在调节软骨形成方面表现出显著的协同作用。 多种聚合物合成支架在其空间结构设计和理化性质的需求上可以有更多的选择性,WANG等[70]开发了一种双因子定向多孔结构仿生软骨之间,该支架的表层由胶原蛋白、壳聚糖和透明质酸钠制成,由胶原蛋白、壳聚糖和丝素蛋白制备了微管阵列结构的过渡层,同时在表层和过渡层之间加入了含核生成素的聚乳酸微球和含转化生长因子β1的多聚赖氨酸-肝素钠纳米粒,扫描电镜显示该双层复合支架具有类似天然软骨的双重结构,同时软骨缺损模型修复研究表明该支架能引导骨髓间充质干细胞定向增殖和分化,新生软骨组织与周围组织达到良好整合,形态与正常软骨基本一致。JIANG等[71]合成了一种胶原、硫酸软骨素和透明质酸三相水凝胶,然后利用兔的自体软骨细胞进行软骨缺损修复,细胞活性分析和体外生化评价显示,软骨细胞在透明质酸三相水凝胶支架中的细胞增殖、糖胺聚糖分泌和软骨特异性标志物基因的表达均明显高于单纯胶原支架,该支架构建的体内异位软骨组织显示出典型的透明软骨特征,具有更接近天然软骨的形态学和组织学特征以及更大的机械强度,ZHU等[72]通过研究发现,由Ⅰ型胶原、透明质酸和硫酸软骨素合成的复合支架不仅能使软骨形成标志物的mRNA表达上调,同时可以抑制软骨细胞肥大标志物的表达,作者推测透明质酸水凝胶中的Ⅰ型胶原和硫酸软骨素等这些额外的细胞外基质成分可能通过协同作用进一步调节人间充质干细胞的软骨生成和肥大。基于透明质酸接枝改性和复合改性构建新型支架并用于软骨修复工程的研究汇总,见表3。"
| [1] ULRICH-VINTHER M, MALONEY MD, SCHWARZ EM, et al. Articular cartilage biology. J Am Acad Orthop Surg. 2003;11(6):421-430. [2] MOBASHERI A, MATTA C, ZAKANY R, et al. Chondrosenescence: definition, hallmarks and potential role in the pathogenesis of osteoarthritis. Maturitas. 2015;80(3):237-244. [3] GOBBI A, DALLO I, KUMAR V. Editorial commentary: biological cartilage repair technique-an “effective, accessible, and safe” surgical solution for an old difficult biological problem. Arthroscopy. 2020;36(3):859-861. [4] HORKAY F. Interactions of cartilage extracellular matrix macromolecules. J Polym Sci B Polym Phys. 2012;50(24):1699-1705. [5] WANG AT, ZHANG QF, WANG NX, et al. Cocktail of hyaluronic acid and human amniotic mesenchymal cells effectively repairs cartilage injuries in sodium iodoacetate-induced osteoarthritis rats. Front Bioeng Biotechnol. 2020;8:87. [6] 付力伟,杨振,李浩,等.亲和肽在软骨组织工程中应用的新策略和问题[J].中国组织工程研究,2021,25(16):2569-2574. [7] OLSON A, GRAVER A, GRANDE D. Scaffolds for articular cartilage repair. J Long Term Eff Med Implants. 2012;22(3):219-227. [8] FU N, DONG T, MENG A, et al. Research progress of the types and preparation techniques of scaffold materials in cartilage tissue engineering. Curr Stem Cell Res Ther. 2018;13(7):583-590. [9] ZASLAV K, MCADAMS T, SCOPP J, et al. New frontiers for cartilage repair and protection. Cartilage. 2012;3(1 Suppl):77S-86S. [10] SIEGEL MG, LUBOWITZ JH, BRAND JC, et al. Articular cartilage restoration requires cells, scaffolds, growth factors, and mechanical stimulation. Arthroscopy. 2021; 37(5):1359-1360. [11] KALKAN R, NWEKWO CW, ADALI T. The use of scaffolds in cartilage regeneration. Crit Rev Eukaryot Gene Expr. 2018;28(4):343-348. [12] MAKRIS EA, GOMOLL AH, MALIZOS KN, et al. Repair and tissue engineering techniques for articular cartilage. Nat Rev Rheumatol. 2015;11(1):21-34. [13] SHA’BAN M, AHMAD RADZI MA. Scaffolds for cartilage regeneration: to use or not to use? Adv Exp Med Biol. 2020;1249:97-114. [14] SACHLOS E, CZERNUSZKA JT. Making tissue engineering scaffolds work. Review: the application of solid freeform fabrication technology to the production of tissue engineering scaffolds. Eur Cell Mater. 2003;5:29-39. [15] FRASER JR, LAURENT TC, LAURENT UB. Hyaluronan: its nature, distribution, functions and turnover. J Intern Med. 1997;242(1):27-33. [16] ABBRUZZESE F, BASOLI F, COSTANTINI M, et al. Hyaluronan: an overview. J Biol Regul Homeost Agents. 2017 Dec 27;31(4 Suppl 2):9-22. [17] LAURENT TC, LAURENT UB, FRASER JR. The structure and function of hyaluronan: an overview. Immunol Cell Biol. 1996;74(2):A1-A7. [18] ABATANGELO G, VINDIGNI V, AVRUSCIO G, et al. Hyaluronic acid: redefining its role. Cells. 2020;9(7):1743. [19] BOWMAN S, AWAD ME, HAMRICK MW, et al. Recent advances in hyaluronic acid based therapy for osteoarthritis. Clin Transl Med. 2018;7(1):6. [20] OLIVEIRA I, CARVALHO AL, RADHOUANI H, et al. Promising biomolecules. Adv Exp Med Biol. 2018;1059:189-205. [21] FALLACARA A, BALDINI E, MANFREDINI S, et al. Hyaluronic acid in the third millennium. Polymers (Basel). 2018;10(7):701. [22] BRUN P, ZAVAN B, VINDIGNI V, et al. In vitro response of osteoarthritic chondrocytes and fibroblast-like synoviocytes to a 500-730 kDa hyaluronan amide derivative. J Biomed Mater Res B Appl Biomater. 2012;100(8):2073-2081. [23] VETRANO M, RANIERI D, NANNI M, et al. Hyaluronic acid (HA), platelet-rich plasm and extracorporeal shock wave therapy (ESWT) promote human chondrocyte regeneration in vitro and ESWT-mediated increase of CD44 expression enhances their susceptibility to HA treatment. PLoS One. 2019;14(6):e0218740. [24] KATOH S, YOSHIOKA H, SENTHILKUMAR R, et al. Enhanced expression of hyaluronic acid in osteoarthritis-affected knee-cartilage chondrocytes during three-dimensional in vitro culture in a hyaluronic-acid-retaining polymer scaffold. Knee. 2021;29:365-373. [25] WANG T, LAI JH, YANG F. Effects of hydrogel stiffness and extracellular compositions on modulating cartilage regeneration by mixed populations of stem cells and chondrocytes in vivo. Tissue Eng Part A. 2016;22(23-24):1348-1356. [26] 张晓鸥,吕旸,毛华,等.透明质酸支架材料:应用研究与产品转化前景[J].中国组织工程研究,2018,22(2):294-302. [27] 刘效仿,张健,侯蕾,等.透明质酸促进关节软骨缺损修复的实验研究[J].中国矫形外科杂志,2015,23(24):2271-2276. [28] CHEN J, YANG J, WANG L, et al. Modified hyaluronic acid hydrogels with chemical groups that facilitate adhesion to host tissues enhance cartilage regeneration. Bioact Mater. 2020;6(6):1689-1698. [29] LOPEZ-RUIZ E, JIMENEZ G, ÁLVAREZ DE CIENFUEGOS L, et al. Advances of hyaluronic acid in stem cell therapy and tissue engineering, including current clinical trials. Eur Cell Mater. 2019;37:186-213. [30] GUPTA RC, LALL R, SRIVASTAVA A, et al. Hyaluronic acid: molecular mechanisms and therapeutic trajectory. Front Vet Sci. 2019;6:192. [31] BULPITT P, AESCHLIMANN D. New strategy for chemical modification of hyaluronic acid: preparation of functionalized derivatives and their use in the formation of novel biocompatible hydrogels. J Biomed Mater Res. 1999;47(2):152-169. [32] KIM IL, MAUCK RL, BURDICK JA. Hydrogel design for cartilage tissue engineering: a case study with hyaluronic acid. Biomaterials. 2011;32(34):8771-8782. [33] 魏健,刘建建,黄思玲,等.透明质酸交联衍生物的研究进展[J].高分子材料科学与工程,2017,33(10):183-190. [34] BENCHERIF SA, SRINIVASAN A, HORKAY F, et al. Influence of the degree of methacrylation on hyaluronic acid hydrogels properties. Biomaterials. 2008; 29(12):1739-1749. [35] TIWARI S, BAHADUR P. Modified hyaluronic acid based materials for biomedical applications. Int J Biol Macromol. 2019;121:556-571. [36] HSIEH YH, SHEN BY, WANG YH, et al. Healing of osteochondral defects implanted with biomimetic scaffolds of poly(ε-caprolactone)/hydroxyapatite and glycidyl-methacrylate-modified hyaluronic acid in a minipig. Int J Mol Sci. 2018;19(4): 1125. [37] HE T, LI B, COLOMBANI T, et al. Hyaluronic acid-based shape-memory cryogel scaffolds for focal cartilage defect repair. Tissue Eng Part A. 2021;27(11-12):748-760. [38] WANG T, LAI JH, HAN LH, et al. Modulating stem cell-chondrocyte interactions for cartilage repair using combinatorial extracellular matrix-containing hydrogels. J Mater Chem B. 2016;4(47):7641-7650. [39] SHEU SY, CHEN WS, SUN JS, et al. Biological characterization of oxidized hyaluronic acid/resveratrol hydrogel for cartilage tissue engineering. J Biomed Mater Res A. 2013;101(12):3457-3466. [40] LIU C, LIU D, WANG Y, et al. Glycol chitosan/oxidized hyaluronic acid hydrogels functionalized with cartilage extracellular matrix particles and incorporating BMSCs for cartilage repair. Artif Cells Nanomed Biotechnol. 2018;46(sup1):721-732. [41] LI J, HUANG Y, SONG J, et al. Cartilage regeneration using arthroscopic flushing fluid-derived mesenchymal stem cells encapsulated in a one-step rapid cross-linked hydrogel. Acta Biomater. 2018;79:202-215. [42] MONGKHON JM, THACH M, SHI Q, et al. Sorbitol-modified hyaluronic acid reduces oxidative stress, apoptosis and mediators of inflammation and catabolism in human osteoarthritic chondrocytes. Inflamm Res. 2014;63(8):691-701. [43] JIN Y, KOH RH, KIM SH, et al. Injectable anti-inflammatory hyaluronic acid hydrogel for osteoarthritic cartilage repair. Mater Sci Eng C Mater Biol Appl. 2020;115:111096. [44] ZHANG Y, CAO Y, ZHAO H, et al. An injectable BMSC-laden enzyme-catalyzed crosslinking collagen-hyaluronic acid hydrogel for cartilage repair and regeneration. J Mater Chem B. 2020;8(19):4237-4244. [45] JOOYBAR E, ABDEKHODAIE MJ, ALVI M, et al. An injectable platelet lysate-hyaluronic acid hydrogel supports cellular activities and induces chondrogenesis of encapsulated mesenchymal stem cells. Acta Biomater. 2019;83:233-244. [46] ZHU D, WANG H, TRINH P, et al. Elastin-like protein-hyaluronic acid (ELP-HA) hydrogels with decoupled mechanical and biochemical cues for cartilage regeneration. Biomaterials. 2017;127:132-140. [47] CHEN Y, SUI J, WANG Q, et al. Injectable self-crosslinking HA-SH/Col I blend hydrogels for in vitro construction of engineered cartilage. Carbohydr Polym. 2018;190:57-66. [48] BAUER C, BERGER M, BAUMGARTNER RR, et al. A novel cross-linked hyaluronic acid porous scaffold for cartilage repair: an in vitro study with osteoarthritic chondrocytes. Cartilage. 2016;7(3):265-273. [49] KHUNMANEE S, JEONG Y, PARK H. Crosslinking method of hyaluronic-based hydrogel for biomedical applications. J Tissue Eng. 2017;8:2041731417726464. [50] FAKHARI A, BERKLAND C. Applications and emerging trends of hyaluronic acid in tissue engineering, as a dermal filler and in osteoarthritis treatment. Acta Biomater. 2013;9(7):7081-7092. [51] XIA H, ZHAO D, ZHU H, et al. Lyophilized scaffolds fabricated from 3D-printed photocurable natural hydrogel for cartilage regeneration. ACS Appl Mater Interfaces. 2018;10(37):31704-31715. [52] LIN H, BECK AM, SHIMOMURA K, et al. Optimization of photocrosslinked gelatin/hyaluronic acid hybrid scaffold for the repair of cartilage defect. J Tissue Eng Regen Med. 2019;13(8):1418-1429. [53] ONOFRILLO C, DUCHI S, O’CONNELL CD, et al. Biofabrication of human articular cartilage: a path towards the development of a clinical treatment. Biofabrication. 2018;10(4):045006. [54] XIAO SP, TANG LS, CHEN JY, et al. Effect of cross-linked hyaluronate scaffold on cartilage repair: an in vivo study. Orthop Surg. 2019;11(4):679-689. [55] LEE HY, HWANG CH, KIM HE, et al. Enhancement of bio-stability and mechanical properties of hyaluronic acid hydrogels by tannic acid treatment. Carbohydr Polym. 2018;186:290-298. [56] COLLINS MN, BIRKINSHAW C. Physical properties of crosslinked hyaluronic acid hydrogels. J Mater Sci Mater Med. 2008;19(11):3335-3343. [57] CREDI C, BIELLA S, DE MARCO C, et al. Fine tuning and measurement of mechanical properties of crosslinked hyaluronic acid hydrogels as biomimetic scaffold coating in regenerative medicine. J Mech Behav Biomed Mater. 2014;29:309-316. [58] BORZACCHIELLO A, RUSSO L, MALLE BM, et al. Hyaluronic acid based hydrogels for regenerative medicine applications. Biomed Res Int. 2015;2015: 871218. [59] ZHANG W, MU H, DONG D, et al. Alteration in immune responses toward N-deacetylation of hyaluronic acid. Glycobiology. 2014;24(12):1334-1342. [60] SEDLACEK J, HERMANNOVA M, MRAZEK J, et al. Insight into the distribution of amino groups along the chain of chemically deacetylated hyaluronan. Carbohydr Polym. 2019;225:115156. [61] SEDLACEK J, HERMANNOVA M, ŠATINSKY D, et al. Current analytical methods for the characterization of N-deacetylated hyaluronan: a critical review. Carbohydr Polym. 2020;249:116720. [62] BUFFA R, ŠEDOVA P, BASARABOVA I, et al. α, β-Unsaturated aldehyde of hyaluronan--Synthesis, analysis and applications. Carbohydr Polym. 2015;134: 293-299. [63] LIU Y, YANG J, LUO Z, et al. Development of an injectable thiolated icariin functionalized collagen/hyaluronic hydrogel to promote cartilage formation in vitro and in vivo. J Mater Chem B. 2019;7(17):2845-2854. [64] YANG J, LIU Y, HE L, et al. Icariin conjugated hyaluronic acid/collagen hydrogel for osteochondral interface restoration. Acta Biomater. 2018;74:156-167. [65] SHIE MY, CHANG WC, WEI LJ, et al. 3D printing of cytocompatible water-based light-cured polyurethane with hyaluronic acid for cartilage tissue engineering applications. Materials (Basel). 2017;10(2):136. [66] D’ESTE M, SPRECHER CM, MILZ S, et al. Evaluation of an injectable thermoresponsive hyaluronan hydrogel in a rabbit osteochondral defect model. J Biomed Mater Res A. 2016;104(6):1469-1478. [67] HOU KT, LIU TY, CHIANG MY, et al. Cartilage tissue-mimetic pellets with multifunctional magnetic hyaluronic acid-graft-amphiphilic gelatin microcapsules for chondrogenic stimulation. polymers (basel). 2020;12(4):785. [68] HAKAMIVALA A, SHUXIN Li, ROBINSON K, et al. Recruitment of endogenous progenitor cells by erythropoietin loaded particles for in situ cartilage regeneration. Bioact Mater. 2020;5(1):142-152. [69] ZHAO W, DU Z, FANG J, et al. Synthetic/natural blended polymer fibrous meshes composed of polylactide, gelatin and glycosaminoglycan for cartilage repair. J Biomater Sci Polym Ed. 2020;31(11):1437-1456. [70] WANG J, WANG Y, SUN X, et al. Biomimetic cartilage scaffold with orientated porous structure of two factors for cartilage repair of knee osteoarthritis. Artif Cells Nanomed Biotechnol. 2019;47(1):1710-1721. [71] JIANG X , LIU J , LIU Q , et al . Therapy for cartilage defects: functional ectopic cartilage constructed by cartilage-simulating collagen, chondroitin sulfate and hyaluronic acid (CCH) hybrid hydrogel with allogeneic chondrocytes. Biomater Sci. 2018;6(6):1616-1626. [72] ZHU M, FENG Q, SUN Y, et al. Effect of cartilaginous matrix components on the chondrogenesis and hypertrophy of mesenchymal stem cells in hyaluronic acid hydrogels. J Biomed Mater Res B Appl Biomater. 2017;105(8):2292-2300. |
| [1] | Yao Xiaoling, Peng Jiancheng, Xu Yuerong, Yang Zhidong, Zhang Shuncong. Variable-angle zero-notch anterior interbody fusion system in the treatment of cervical spondylotic myelopathy: 30-month follow-up [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1377-1382. |
| [2] | Zhu Chan, Han Xuke, Yao Chengjiao, Zhou Qian, Zhang Qiang, Chen Qiu. Human salivary components and osteoporosis/osteopenia [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1439-1444. |
| [3] | Jin Tao, Liu Lin, Zhu Xiaoyan, Shi Yucong, Niu Jianxiong, Zhang Tongtong, Wu Shujin, Yang Qingshan. Osteoarthritis and mitochondrial abnormalities [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1452-1458. |
| [4] | Zhang Lichuang, Xu Hao, Ma Yinghui, Xiong Mengting, Han Haihui, Bao Jiamin, Zhai Weitao, Liang Qianqian. Mechanism and prospects of regulating lymphatic reflux function in the treatment of rheumatoid arthritis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1459-1466. |
| [5] | Wang Jing, Xiong Shan, Cao Jin, Feng Linwei, Wang Xin. Role and mechanism of interleukin-3 in bone metabolism [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1260-1265. |
| [6] | Zhu Chan, Han Xuke, Yao Chengjiao, Zhang Qiang, Liu Jing, Shao Ming. Acupuncture for Parkinson’s disease: an insight into the action mechanism in animal experiments [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1272-1277. |
| [7] | Guo Jia, Ding Qionghua, Liu Ze, Lü Siyi, Zhou Quancheng, Gao Yuhua, Bai Chunyu. Biological characteristics and immunoregulation of exosomes derived from mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1093-1101. |
| [8] | Wu Weiyue, Guo Xiaodong, Bao Chongyun. Application of engineered exosomes in bone repair and regeneration [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1102-1106. |
| [9] | Zhou Hongqin, Wu Dandan, Yang Kun, Liu Qi. Exosomes that deliver specific miRNAs can regulate osteogenesis and promote angiogenesis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1107-1112. |
| [10] | Zhang Jinglin, Leng Min, Zhu Boheng, Wang Hong. Mechanism and application of stem cell-derived exosomes in promoting diabetic wound healing [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1113-1118. |
| [11] | Huang Chenwei, Fei Yankang, Zhu Mengmei, Li Penghao, Yu Bing. Important role of glutathione in stemness and regulation of stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1119-1124. |
| [12] | Hui Xiaoshan, Bai Jing, Zhou Siyuan, Wang Jie, Zhang Jinsheng, He Qingyong, Meng Peipei. Theoretical mechanism of traditional Chinese medicine theory on stem cell induced differentiation [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1125-1129. |
| [13] | An Weizheng, He Xiao, Ren Shuai, Liu Jianyu. Potential of muscle-derived stem cells in peripheral nerve regeneration [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1130-1136. |
| [14] | Fan Yiming, Liu Fangyu, Zhang Hongyu, Li Shuai, Wang Yansong. Serial questions about endogenous neural stem cell response in the ependymal zone after spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1137-1142. |
| [15] | Xu Lei, Han Xiaoqiang, Zhang Jintao, Sun Haibiao. Hyaluronic acid around articular chondrocytes: production, transformation and function characteristics [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(5): 768-773. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||