Chinese Journal of Tissue Engineering Research ›› 2022, Vol. 26 ›› Issue (34): 5504-5511.doi: 10.12307/2022.463
Previous Articles Next Articles
Application of manganese in biomaterials: current situation and problems
Zhang Chang1, Yu Zhengwen1, 2
- 1School of Stomatology, Zunyi Medical University, Zunyi 563099, Guizhou Province, China; 2Key Laboratory of Oral Disease Research, Guizhou Institution of Higher Education, Zunyi 563099, Guizhou Province, China
-
Received:2021-04-30Accepted:2021-05-22Online:2022-12-08Published:2022-04-15 -
Contact:Yu Zhengwen, Associate professor, Master’s supervisor, School of Stomatology, Zunyi Medical University, Zunyi 563099, Guizhou Province, China; Key Laboratory of Oral Disease Research, Guizhou Institution of Higher Education, Zunyi 563099, Guizhou Province, China -
About author:Zhang Chang, Master candidate, School of Stomatology, Zunyi Medical University, Zunyi 563099, Guizhou Province, China -
Supported by:National Key Research and Development Program Project of the Ministry of Science and Technology, No. 2016YFC1102800 (to YZW); Talent Base of Medical Biomaterials Research of Guizhou Province, No. QRLF[2018]3 (to YZW); Projects of Zunyi Science and Technology Bureau and Hospital of Stomatology, Zunyi Medical University, No. ZSKHSZ(2018)240 (to YZW); Academic Talents and Innovation Program of Zunyi Medical University, No. QKHPTRC[2017]5733-057 (to YZW)
CLC Number:
Cite this article
Zhang Chang, Yu Zhengwen. Application of manganese in biomaterials: current situation and problems[J]. Chinese Journal of Tissue Engineering Research, 2022, 26(34): 5504-5511.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
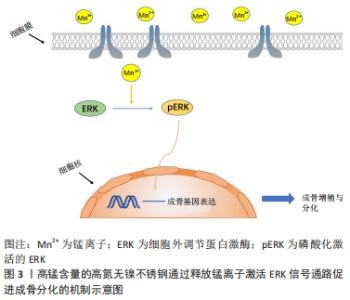
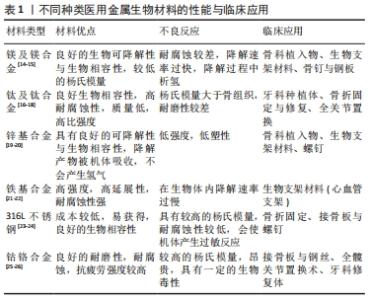
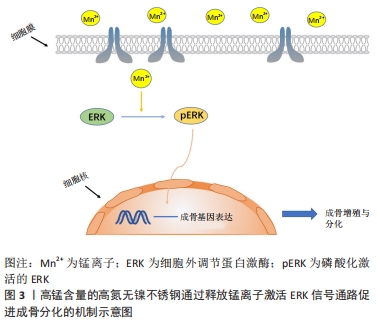
然而,不同种类的生物合金材料在实际临床应用中尚存在一些不足之处,比如镁和镁合金在人体内腐蚀降解速率较快,与人体血液和体液等交互作用后发生腐蚀降解行为,释放出大量的氢气,不利于骨组织的愈合;铁及铁基金属生物材料的体内降解速率过慢,植入材料需要二次取出易造成术区的二次损伤;传统的Ti-6Al-4V医用钛合金有较高的弹性模量会导致新生骨组织骨质疏松,同时植入人体经过腐蚀降解后会释放出有毒的铝、钒离子,易造成植入区域炎症反应;锌及锌合金具有较低的强度和塑性,较差的力学性能限制了它在临床的应用[27-29]。为了改善上述合金的不足之处,由于锰元素具有以下3种优点:①锰元素是人体内必需的微量元素,具有良好的生物相容性;②添加到合金中能有效改善合金的机械强度和耐腐蚀性;③具有良好的成骨作用,可促进细胞增殖、黏附、扩散并调节骨代谢。因此,将锰元素引入到合金材料中是一种改善合金材料生物和力学性能的有效策略。 锰元素引入到金属生物材料中主要包括以下2种方式,引入金属材料中和制备表面涂层。 (1)通过合金化将锰元素引入金属生物材料:镁基与锌基医用金属材料作为新型可降解吸收金属植入修复材料,具有良好的骨诱导性和生物相容性,已逐渐成为可降解吸收金属植入修复材料研究领域的热点。但纯镁具有腐蚀速率过快的问题,并在体内腐蚀过程中会产生大量的氢气,不利于损伤骨组织的愈合[30]。 YANG等[31]将锰元素添加到镁基质中,引起合金的腐蚀电位增加与晶粒细化,抗压强度从纯镁的47.1 MPa增加到 Mg-2Mn合金的64.2 MPa,此外镁锰合金经过腐蚀处理后的析氢量明显低于纯镁,锰元素的加入使得镁合金的机械性能和耐腐蚀性能都得到加强。同样,有研究表明含锰镁合金生物材料在热挤压变形过程中动态析出大量细小弥散分布的第二相Mn单质颗粒,显著细化材料微观组织结构,优化了材料晶粒取向分布,进而显著改善了材料的室温强度和塑性[32-33]。锰元素的添加使得镁合金的耐腐蚀性得到提高并且增强了合金的强度,从而使得合金的生物相容性得到提升。 为了改善纯锌的低强度和脆性,JIA等[34]将少量的锰元素添加到Zn中,制备的Zn-Mn合金获得了极高的伸长率[(83.96±2.36)%],显著提高了锌合金的延展性。与纯锌相比,锌锰合金改善了细胞相容性,体外细胞实验证实添加锰元素后的锌合金可促进成骨前体细胞(MC3T3-E1)碱性磷酸酶和成骨相关基因的表达,提高了成骨活性。在体内实验中,SUN等[35]将Zn-Mg-Mn合金螺钉植入兔的膝关节用于韧带重建固定,与钛合金相比,合金螺钉显著促进了新骨的形成并使部分肌腱矿化,促进了肌腱与骨的融合。所以锰元素的加入改善了锌合金的力学性能,并提高了锌基金属生物材料的成骨活性。 铁基金属生物材料与镁基、锌基金属生物材料的不同之处在于它在体内极难降解,手术取出易造成术区的二次损伤,它常被用于骨科内固定材料。同时铁基金属生物材料具有良好的磁学性能会影响患者的磁共振成像。于是DARGUSCH等[36]研究了Fe-35Mn合金,研究表明Fe-35Mn合金的腐蚀速率较纯铁提高了近5倍,具备优异的腐蚀降解性能。此外,材料的体外细胞活性实验研究表明,Fe-35Mn合金显著促进了细胞的增殖;不仅如此,Fe-35Mn合金的晶体学结构为奥氏体结构,表现出良好的反磁性,极大地提高了生物材料的磁共振成像兼容性。同时CARLUCCIO等[37]也发现该材料的腐蚀降解速率明显高于纯铁,体外细胞生物学实验研究表明,基于3D打印技术制备的多孔Fe-35Mn可降解生物支架材料具备更为优异的生物相容性和成骨能力,是较为理想的可降解吸收植入修复材料。相较于纯铁,锰元素的引入加快了铁基生物材料的降解速率,改善了合金的成像性能,使其具有了更好的应用价值。同样锰元素引入到钛合金中也取得了良好的效果,传统的Ti-6Al-4V医用钛合金生物材料因其较高的弹性模量,易导致新生骨组织骨质疏松。同时,植入材料与宿主交互作用后发生腐蚀降解行为,释放出铝离子、钒离子等金属离子,易造成植入区域炎症反应,不利于损伤骨组织的愈合[38-39]。 医用植入材料中无Al和V的高强度新型钛合金在近几年中得到了极大关注[40-44]。ALSHAMMARI等[40]研究表明Ti-Mn合金具有良好的机械性能可以用来替代传统的钛合金。SANTOS等[43]研究了不同含量Mn(质量分数6%-18%)的Ti-Mn合金的机械性能和生物学性能。研究结果显示,当合金中Mn元素的质量分数为9%时,材料的弹性模量最低为94 GPa,同临床上使用较多的医用Ti-6AL-4V合金110 GPa相比,其弹性模量更加接近人体骨组织的弹性模量,且材料在室温轴向拉伸试验研究中具备十分优异的拉伸性能。同时当Ti-Mn(Mn质量分数<13%)时具有良好的细胞增殖能力,Ti-Mn合金具备较为优异的生物相容性,是较为理想的金属植入材料。然而值得注意的是当合金中锰的质量分数高于18%时会有锰中毒的风险[43-44]。 锰的引入改善了钛及钛合金的力学性能和生物学相容性,使其变为了新型无生物毒性的医用钛合金,让它的应用变得更加广泛。与钛及钛合金相比,生物医用不锈钢有着同样的缺点,目前临床应用较广泛的316L不锈钢在植入人体后,与宿主交互作用后发生腐蚀并释放出金属镍、铬等有毒金属离子,引起术区组织过敏与炎症反应,不利于损伤骨组织的愈合[45]。YU等[46]制备了高锰含量的高氮无镍不锈钢(high nitrogen nickel-free stainless steel,HNS),具有更高的抗生物腐蚀性和表面亲水性,通过上调整合素α1和整合素β1的基因表达,促进了大鼠骨髓间充质干细胞的黏附与扩散。同时锰离子的释放增强了大鼠骨髓间充质干细胞的成骨细胞相关基因的表达水平,促进了碱性磷酸酶的活性和骨钙素蛋白表达,结果表明HNS可以诱导大鼠骨髓间充质干细胞的成骨分化并促进植入物的骨整合,说明在体内实验中,HNS促进了骨-种植体界面的新骨形成并增强了骨结合强度,表明该植入物在早期稳定性和长期骨结合等方面更具优势,见图3。 "

(2)锰离子通过金属表面改性技术制备表面涂层。金属表面改性技术是改善金属生物材料性能的低成本且最简单有效的方法,可以增强材料的抗腐蚀性能、生物相容性和骨结合能力[47]。YU等[48]对医用钛合金生物材料进行表面改性后形成致密的含锰涂层,随着涂层中Mn离子的缓慢释放促进了细胞增殖,同时通过研究发现表面修饰后的医用钛合金材料促进了成骨细胞的黏附、扩散、增殖和分化并对大肠杆菌具有良好的抗菌能力,是较为理想的口腔种植修复植入材料。ANANTH等[49]采用电沉积工艺在316L不锈钢表面制备具有生物活性的锰羟基磷灰石/氧化锌双分子涂层(Mn-HAp/ZnO涂层),同316L不锈钢相比,包覆Mn-HAp/ZnO表面活性涂层的316L不锈钢材料经模拟无菌溶液浸泡后,材料的腐蚀降解速率显著降低,有效避免了材料因腐蚀降解释放的有毒金属离子导致术区组织过敏或炎性反应,进而改善了材料的骨整合效应。 综上所述,医用金属生物材料在结合锰元素后可以有效提高材料的机械性能、生物相容性和抗菌性,同时增强了间充质干细胞的体外成骨分化进而增强了材料的骨整合效应,进一步改善了医用金属生物材料的性能,是很有前途的医用金属材料研制方向。 2.2 锰元素在骨组织工程材料中的应用 骨组织工程材料主要用于修复骨缺损,目前修复骨缺损的方法主要是自体与异体骨移植,然而它们的缺点也比较明显,自体骨移植需要从患者体内取得需要进行二次手术,这会导致供体部位的损伤增加手术风险。而异体骨移植会导致免疫排斥反应并且材料来源比较短缺,这些缺点都限制了它们在临床中的应用。骨组织工程材料正好弥补了以上的缺点,常用的材料主要包括生物陶瓷材料(如羟基磷灰石及磷酸三钙等)、高分子有机合成材料(主要包括聚乳酸、聚羟基乙酸和聚乳酸羟基乙酸等)以及天然高分子材料(如甲壳素及其衍生物、纤维蛋白和胶原)等[50]。这些材料拥有良好的抗压强度、生物相容性和骨整合效果,而锰离子的引入对此类材料进行了改性加强了这些效果,使其成为更为优异的植入修复材料。 锰离子将其引入骨组织工程材料中具有以下几个方面的优势: (1) 锰离子改善了生物陶瓷支架材料的力学性能。NAWAZ等[51]通过研究涂布于45S5生物活性玻璃支架表面的明胶/锰掺杂生物活性纳米颗粒(Mn-containing mesoporous bioactive glass nanoparticles,Mn-MBGNs)生物涂层的力学性能和生物相容性,发现明胶/Mn-MBGNs生物活性涂层显著增强了支架材料的抗弯强度。体外细胞实验研究显示明胶/Mn-MBGNs生物活性涂层具备良好的细胞活性,有利于成骨细胞的增殖。 (2) 锰离子赋予了材料一定的抗菌作用。RAU等[52]研究发现掺杂了锰离子的β-磷酸三钙材料显著抑制了金黄色葡萄球菌、大肠杆菌及铜绿假单胞菌等5种细菌的生长,同时对细胞免疫毒性作用,表明添加了锰离子的磷酸三钙具有一定的抗菌作用,可以预防植入材料在人体内相关感染的发生。 (3) 锰离子可以进一步增强材料诱导成骨的作用。大量的研究也表明,同羟基磷灰石相比较,含锰羟基磷灰石具有更为优异的生物相容性,并显著增强细胞黏附和增殖能力,以及植入材料与骨组织之间的骨整合能力[53-55]。BARRIONI等[56]采用溶胶凝胶工艺在58S生物活性玻璃中掺杂金属锰,结果显示含锰58S生物活性玻璃显著刺激骨髓源性干细胞成骨分化与矿化。体外细胞活性实验研究也表明,含锰58S生物活性玻璃无细胞毒性并促进成骨相关基因和蛋白的高表达。不仅如此,含锰生物活性玻璃经模拟体液浸泡后在其表面形成羟基磷灰石层,经体外细胞实验研究发现材料具有良好的体外细胞活性[57]。WU等[58]在β-磷酸三钙生物陶瓷中添加金属锰,结果显示低浓度的锰离子可以刺激骨髓间充质干细胞的增殖,因此,可通过锰离子与β-磷酸三钙的结合控制锰离子的释放浓度来改善生物陶瓷的成骨作用。YU等[59]将锰离子引入胶原蛋白-羟基磷灰石中,锰的掺入进一步增强了层状支架的成骨作用,促进了骨髓间充质干细胞的体外成骨分化,同时该新型功能性支架可以负载新生骨髓细胞实现体内骨再生。PINA等[60]在丝素蛋白/β-磷酸三钙高分子生物材料中掺杂锰元素后,得到新型可降解吸收骨再生支架材料,研究发现该生物材料增强了成骨细胞的成骨分化能力,同时碱性磷酸酶的活性显著增强,表明该新型可降解吸收骨再生支架材料具有良好的诱导细胞成骨分化能力,能够促进形成新生骨组织,从而有利于损伤骨组织的愈合。因此,将锰离子掺入到骨组织工程材料中为其功能化的改造及更好地应用于临床开辟了新的方向。 2.3 锰元素在药物递送及肿瘤治疗中的应用 药物递送系统主要是由药物和负载药物的载体组成复合物,靶向、按需递送药物至治疗部位。生物医用载体材料的种类十分广泛,主要分为天然载体和合成载体,天然载体主要有高分子可降解壳聚糖、聚乳酸-羟基乙酸共聚物、天然凝胶及纤维素等,合成药物载体主要有纳米粒子和水凝胶等多种形态的复合物[61-62]。而纳米材料作为目前常用的生物载体,是由颗粒尺寸在1-100 nm的晶体构成,与其他载体相比具有良好的稳定性同时有着良好的生物相容性,在负载了药物后可以通过控制自身的降解速度,从而控制负载药物的释放,并且能够通过温度、pH值及光响应等作用实现药物的靶向释放,主要有聚合物胶束、金纳米颗粒、介孔二氧化硅及纳米脂质体等[63]。 当锰离子掺入到纳米粒子中时,形成了新型的药物递送系统,将其引入生物载体中进一步改善了该类载体的性能。 (1)引入锰离子促进了药物的控释,同时锰是一种顺磁性的金属离子,可以作为现有Gd3+对比剂的替代品,有着更小的毒性,并增强特异性的核磁成像性能用于肿瘤成像并监测化学动力治疗过程。WANG等[64]将金纳米棒(gold nanorods,Au NRs)与多烯紫杉醇(docetaxel,DTX)负载到聚乳酸-羟基乙酸(polylactic acid hydroxy acetic acid,PLGA)上,在其表面涂覆二氧化锰纳米薄膜构建PLGA/Au NR/DTX@MnO2药物输送系统,研究显示该药物递送系统释放的金属锰离子显著增强了磁共振成像效果,促进了肿瘤区域的药物控释,在肿瘤治疗中具有良好的应用前景。同样,REN等[65]也发现用空心锰钴氧化物纳米颗粒(hollow manganese/cobalt oxide nanoparticles,H-MCONPs)包埋抗肿瘤药物,在磁共振成像实验中可以准确定位肿瘤位置,实现体内药物的靶向释放,对肿瘤有良好的抑制作用并表现出优异的磁共振成像效果,研究表明H-MCONPs是较为理想的靶向抗肿瘤药物递送载体和磁共振造影剂。XI等[66]使用聚多巴胺(polydopamine,PDA)修饰PLGA,并在其表面配位金属锰离子,得到Mn2+-PDA@DOX/PLGA共聚物复合纳米粒子,结果显示配位锰离子显著改善了磁共振成像性能,同时Mn2+-PDA@DOX/PLGA共聚物复合纳米粒子表现出良好的生物相容性和优异的靶向治疗效果。 (2)锰离子使药物递送系统对pH值及光反应有较强的灵敏度,加强了光热/光动力治疗。LIN等[67]利用二氧化锰包覆的介孔二氧化硅纳米粒子(MS@MnO2 NPs)负载治疗药物,二氧化锰薄膜保护层可以防止治疗药物在未到达治疗部位时就提前释放,在治疗过程中二氧化锰可以对肿瘤区域表达过量的谷胱甘肽进行消耗,可以减少其对羟基自由基的消耗,增强化学动力治疗的效果。此外,释放出来的Mn2+可以催化肿瘤区域表达过量的过氧化氢为羟基自由基用于化学动力治疗,同时可用于肿瘤部位成像并监测治疗过程。MONTHA等[68]使用PLGA包裹锰锌铁氧体磁性纳米颗粒,合成DOX-PLGA@CS@Mn0.9Zn0.1Fe2O4共聚物纳米复合颗粒,结果显示该共聚物纳米复合颗粒可持续释放药物靶向抑制癌细胞扩散,具有较强的抗癌活性和灵敏的酸碱响应速度,是较为理想的靶向抗癌药物载体。YANG等[69]使用空心二氧化锰(hollow manganese dioxide,H-MnO2)纳米生物材料修饰聚乙二醇(polyethylene glycol,PEG),负载光动力试剂氯e6与阿霉素后合成了H-MnO2-PEG/C&D纳米药物递送系统。研究显示该药物递送系统具有灵敏的酸碱响应和优异的肿瘤特异性核磁成像性能,并且在酸性肿瘤微环境下可靶向释放药物并增强光动力化学治疗效果。 (3) 锰离子可诱导抗菌作用。已有研究表明Mn3O4纳米颗粒(Mn3O4 NPs)具有良好的生物学特性和较低的毒性,发现它对金黄色葡萄球菌和白色念珠菌有着一定的抑制作用。同时,Mn3O4 NPs对A549肺癌和MCF-7乳腺癌具有显著的抗癌活性[70]。而PACKIRISAMY等[71]同样也发现用溶胶-凝胶法合成的氧化锰-壳聚糖纳米复合材料(Mn3O4-CS)对大肠杆菌和伤寒沙门氏菌等革兰阴性细菌有着优异的抑制作用。ATIF等[72]研究了锰掺杂的氧化铈纳米复合物,锰掺杂的二氧化铈纳米复合材料对金黄色葡萄球菌、大肠杆菌及铜绿假单胞菌具有良好的抑制作用。同时,9% Mn掺杂的二氧化铈降低了MCF-7乳腺癌细胞的活力,表明它有潜力成为一种优良的纳米药物递送载体。因此,近年来的研究表明锰离子在药物递送及肿瘤治疗领域有着重要的应用,掺锰纳米药物不仅降低了对正常组织的毒副作用还赋予了一定的抗菌作用,而且加强了对药物的控释效果,使得药物能够在肿瘤部位有效释放,从而提高了治疗效果。所以掺锰纳米药物对于药物递送及肿瘤治疗具有巨大的应用前景。 表2展示了近几年来锰元素在生物材料中的研究脉络,阐述了锰元素在医用金属材料、骨组织工程材料、药物递送及肿瘤治疗等领域的研究现状。 "

| [1] 江权.锰的存在及应用[J].中国锰业,2001,19(3):37-39. [2] 卫潇茗,王晨光,张睿,等.锰离子作为免疫调节剂的发现及应用展望[J].中国细胞生物学学报,2020,42(10):1721-1731. [3] HORNING KJ, CAITO SW, TIPPS KG, et al. Manganese is essential for neuronal health. Annu Rev Nutr. 2015;35:71-108. [4] HREHA J, WEY A, CUNNINGHAM C, et al. Local manganese chloride treatmentaccelerates fracture healing in a rat model. J Orthop Res. 2015;33(1):122-130. [5] LI L, YANG X. The essential element manganese, oxidative stress, and metabolic diseases: links and interactions. Oxid Med Cell Longev. 2018; 2018:7580707. [6] 郭书含,宋平平,陈曾曾,等.人骨髓间充质干细胞脑内移植治疗锰中毒模型的可行性[J].中国组织工程研究,2018,22(9):1402-1406. [7] LÜTHEN F, BULNHEIM U, MÜLLER PD, et al. Influence of manganese ions on cellular behavior of human osteoblasts in vitro. Biomol Eng. 2007;24(5):531-536. [8] PARK JW, KIM YJ, JANG JH. Surface characteristics and in vitro biocompatibility of a manganese-containing titanium oxide surface. Applied Surface Science. 2011;258(2):977-985. [9] HALL ER, SLACK RJ. The effect of divalent metal cations on the αv integrin binding site is ligand and integrin specific. Biomed Pharmacother. 2019; 110:362-370. [10] VORUP-JENSEN T, WALDRON TT, ASTROF N, et al. The connection between metal ion affinity and ligand affinity in integrin I domains. Biochimica et Biophysica Acta. 2007;1774(9):1148-1155. [11] SHARMA N, JANDAIK S, KUMAR S, et al. Synthesis, characterisation and antimicrobial activity of manganese- and iron-doped zinc oxide nanoparticles. J Exp Nanosci. 2015;11(1):54-71. [12] GU Y, ZHANG J, ZHANG X, et al. Three-dimensional printed mg-doped β-tcp bone tissue engineering scaffolds: effects of magnesium ion concentration on osteogenesis and angiogenesis in vitro. Tissue Eng Regen Med. 2019;16(4):415-429. [13] YANG K, ZHOU C, FAN H, et al. Bio-functional design, application and trends in metallic biomaterials. Int J Mol Sci. 2017;19(1):24. [14] ZARTNER P, CESNJEVAR R, SINGER H, et al. First successful implantation of a biodegradable metal stent into the left pulmonary artery of a preterm baby. Catheter Cardiovasc Interv. 2005;66(4):590-594. [15] LI Y, WANG L, CHEN S, et al. Biodegradable magnesium alloy stents as a treatment for vein graft restenosis. Yonsei Med J. 2019;60(5):429-439. [16] OTTRIA L, LAURITANO D, ANDREASI BASSI M, et al. Mechanical, chemical and biological aspects of titanium and titanium alloys in implant dentistry. J Biol Regul Homeost Agents. 2018;32(2 Suppl 1): 81-90. [17] LI Y, DING Y, MUNIR K, et al. Novel β-Ti35Zr28Nb alloy scaffolds manufactured using selective laser melting for bone implant applications. Acta Biomater. 2019;87:273-284. [18] APOSTU D, LUCACIU O, BERCE C, et al. Current methods of preventing aseptic loosening and improving osseointegration of titanium implants in cementless total hip arthroplasty: a review. J Int Med Res. 2018;46(6):2104-2119. [19] SU Y, COCKERILL I, WANG Y, et al. Zinc-based biomaterials for regeneration and therapy. Trends Biotechnol. 2019;37(4):428-441. [20] MOSTAED E, SIKORA-JASINSKA M, DRELICH JW, et al. Zinc-based alloys for degradable vascular stent applications. Acta Biomater. 2018;71: 1-23. [21] FRANCIS A, YANG Y, VIRTANEN S, et al. Iron and iron-based alloys for temporary cardiovascular applications. J Mater Sci Mater Med. 2015; 26(3):138. [22] QI Y, QI H, HE Y, et al. Strategy of metal-polymer composite stent to accelerate biodegradation of iron-based biomaterials. ACS Appl Mater Interfaces. 2018;10(1):182-192. [23] DÍAZ M, SEVILLA P, GALÁN AM, et al. Evaluation of ion release, cytotoxicity, and platelet adhesion of electrochemical anodized 316 L stainless steel cardiovascular stents. J Biomed Mater Res B Appl Biomater. 2008;87(2):555-561. [24] HU H, CUI R, MEI L, et al. Cytocompatibility and bone-formation potential of se-coated 316L stainless steel with nano-pit arrays. J Biomed Nanotechnol. 2018;14(4):716-724. [25] MADL AK, LIONG M, KOVOCHICH M, et al. Toxicology of wear particles of cobalt-chromium alloy metal-on-metal hip implants Part I: physicochemical properties in patient and simulator studies. Nanomedicine. 2015;11(5):1201-1215. [26] Wang H, Lim JY. Metal-ceramic bond strength of a cobalt chromium alloy for dental prosthetic restorations with a porous structure using metal 3D printing. Comput Biol Med. 2019;112:103364. [27] RAZAVI M, HUANG Y. Assessment of magnesium-based biomaterials: from bench to clinic. Biomater Sci. 2019;7(6):2241-2263. [28] BEKMURZAYEVA A, DUNCANSON WJ, AZEVEDO HS, et al. Surface modification of stainless steel for biomedical applications: revisiting a century-old material. Mater Sci Eng C Mater Biol Appl. 2018;93:1073-1089. [29] KIRADZHIYSKA DD, MANTCHEVA RD. Overview of biocompatible materials and their use in medicine. Folia Med (Plovdiv). 2019;61(1): 34-40. [30] STAIGER MP, PIETAK AM, HUADMAI J, et al. Magnesium and its alloys as orthopedic biomaterials: a review. Biomaterials. 2006;27(9): 1728-1734. [31] YANG Y, WU P, WANG Q, et al. The Enhancement of Mg Corrosion Resistance by Alloying Mn and Laser-Melting. Materials (Basel). 2016; 9(4):216. [32] YU Z, TANG A, WANG Q, et al. High strength and superior ductility of an ultra-fine grained magnesium–manganese alloy. Mater Sci Eng A. 2015;648:202-207. [33] YU Z, TANG A, HE J, et al. Effect of high content of manganese on microstructure, texture and mechanical properties of magnesium alloy. Materials Characterization. 2018;136:310-317. [34] JIA B, YANG H, HAN Y, et al. In vitro and in vivo studies of Zn-Mn biodegradable metals designed for orthopedic applications. Acta Biomater. 2020;108:358-372. [35] SUN J, ZHANG X, SHI ZZ, et al. Development of a high-strength Zn-Mn-Mg alloy for ligament reconstruction fixation. Acta Biomater. 2021;119: 485-498. [36] DARGUSCH MS, DEHGHAN-MANSHADI A, SHAHBAZI M, et al. Exploring the role of manganese on the microstructure, mechanical properties, biodegradability, and biocompatibility of porous iron-based scaffolds. ACS Biomater Sci Eng. 2019;5(4): 1686-1702. [37] CARLUCCIO D, XU C, VENEZUELA J, et al. Additively manufactured iron-manganese for biodegradable porous load-bearing bone scaffold applications. Acta Biomater. 2020;103:346-360. [38] COSTA BC, TOKUHARA CK, ROCHA LA, et al. Vanadium ionic species from degradation of Ti-6Al-4V metallic implants: in vitro cytotoxicity and speciation evaluation. Mater Sci Eng C Mater Biol Appl. 2019;96: 730-739. [39] HARUN WSW, KAMARIAH MSIN, MUHAMAD N, et al. A review of powder additive manufacturing processes for metallic biomaterials. Powder Technol. 2018;327:128-151. [40] ALSHAMMARI Y, YANG F, BOLZONI L. Mechanical properties and microstructure of Ti-Mn alloys produced via powder metallurgy for biomedical applications. J Mech Behav Biomed Mater. 2019;91:391-397. [41] KIM JW, HWANG MJ, HAN MK, et al. Effect of manganese on the microstructure, mechanical properties and corrosion behavior of titanium alloys. Mater Chem Phys. 2016;180:341-348. [42] ZHANG F, BURKEL E. Novel titanium manganese alloys and their macroporous foams for biomedical applications prepared by field assisted sintering. Biomed Eng. 2011:203-224. [43] SANTOS PF, NIINOMI M, CHO K, et al. Microstructures, mechanical properties and cytotoxicity of low cost beta Ti-Mn alloys for biomedical applications. Acta Biomater. 2015;26:366-376. [44] SANTOS PF, NIINOMI M, LIU H, et al. Fabrication of low-cost beta-type Ti-Mn alloys for biomedical applications by metal injection molding process and their mechanical properties. J Mech Behav Biomed Mater. 2016;59:497-507. [45] MATUSIEWICZ H. Potential release of in vivo trace metals from metallic medical implants in the human body: from ions to nanoparticles--a systematic analytical review. Acta Biomater. 2014;10(6):2379-2403. [46] YU Y, DING T, XUE Y, et al. Osteoinduction and long-term osseointegration promoted by combined effects of nitrogen and manganese elements in high nitrogen nickel-free stainless steel. J Mater Chem B. 2016;4(4):801-812. [47] ASRI RIM, HARUN WSW, SAMYKANO M, et al. Corrosion and surface modification on biocompatible metals: a review. Mater Sci Eng C Mater Biol Appl. 2017;77:1261-1274. [48] YU L, TIAN Y, QIAO Y, et al. Mn-containing titanium surface with favorable osteogenic and antimicrobial functions synthesized by PIII&D. Colloids Surf B Biointerfaces. 2017;152:376-384. [49] ANANTH KP, SUN J, BAI J. An innovative approach to manganese-substituted hydroxyapatite coating on Zinc oxide-coated 316L SS for implant application. Int J Mol Sci. 2018;19(8):2340. [50] 廖欣宇,王福科,王国梁.骨组织工程支架的进展与挑战[J].中国组织工程研究,2021,25(28):4553-4560. [51] NAWAZ Q, UR REHMAN MA, ROETHER JA, et al. Bioactive glass based scaffolds incorporating gelatin/manganese doped mesoporous bioactive glass nanoparticle coating. Ceram Int. 2019;45(12):14608-14613. [52] RAU JV, FADEEVA IV, FOMIN AS, et al. Sic Parvis magna: manganese-substituted tricalcium phosphate and its biophysical properties. ACS Biomater Sci Eng. 2019;5(12):6632-6644. [53] MUTHUSAMY S, MAHENDIRAN B, SAMPATH S, et al. Hydroxyapatite nanophases augmented with selenium and manganese ions for bone regeneration: physiochemical, microstructural and biological characterization. Mater Sci Eng C Mater Biol Appl. 2021;126:112149. [54] LALA S, GHOSH M, DAS PK, et al. Mechanical preparation of nanocrystalline biocompatible single-phase Mn-doped A-type carbonated hydroxyapatite (A-cHAp): effect of Mn doping on microstructure. Dalton Trans. 2015;44(46):20087-20097. [55] PARK SY, CHOE DH. Bone-like apatite formation on the plasma electrolytic oxidation-treated Ti-6Al-4V alloy in solution containing Si and Mg Ions. J Nanosci Nanotechnol. 2018;18(2):1439-1441. [56] BARRIONI BR, NORRIS E, LI S, et al. Osteogenic potential of sol-gel bioactive glasses containing manganese. J Mater Sci Mater Med. 2019; 30(7):86. [57] BARRIONI BR, OLIVEIRA AC, DE FÁTIMA LEITE M, et al. Sol–gel-derived manganese-releasing bioactive glass as a therapeutic approach for bone tissue engineering. J Mater Sci. 2017;52(15):8904-8927. [58] WU T, SHI H, LIANG Y, et al. Improving osteogenesis of calcium phosphate bone cement by incorporating with manganese doped β-tricalcium phosphate. Mater Sci Eng C Mater Biol Appl. 2020;109: 110481. [59] YU L, ROWE DW, PERERA IP, et al. Intrafibrillar mineralized collagen-hydroxyapatite-based scaffolds for bone regeneration. ACS Appl Mater Interfaces. 2020;12(16):18235-18249. [60] PINA S, CANADAS RF, JIMÉNEZ G, et al. Biofunctional ionic-doped calcium phosphates:silk fibroin composites for bone tissue engineering scaffolding. Cells Tissues Organs. 2017;204(3-4):150-163. [61] 詹世平,苗宏雨,王景昌,等.生物医用材料用于药物递送系统的研究进展[J].功能材料,2019,50(9):9056-9062. [62] 陈璐扬,赵瑾,龙丽霞,等.肿瘤免疫治疗中的生物医用载体[J].化学进展,2017,29(10):1195-1205. [63] WANG J, HU X, XIANG D. Nanoparticle drug delivery systems: an excellent carrier for tumor peptide vaccines. Drug Deliv. 2018;25(1): 1319-1327. [64] WANG L, LI D, HAO Y, et al. Gold nanorod-based poly (lactic-co-glycolic acid) with manganese dioxide core-shell structured multifunctional nanoplatform for cancer theranostic applications. Int J Nanomedicine. 2017;12:3059-3075. [65] REN Q, YANG K, ZOU R, et al. Biodegradable hollow manganese/cobalt oxide nanoparticles for tumor theranostics. Nanoscale. 2019; 11(47):23021-23026. [66] XI J, DA L, YANG C, et al. Mn2+-coordinated PDA@DOX/PLGA nanoparticles as a smart theranostic agent for synergistic chemo-photothermal tumor therapy. Int J Nanomedicine. 2017;12:3331-3345. [67] LIN LS, SONG J, SONG L, et al. Simultaneous fenton-like ion delivery and glutathione depletion by mno2-based nanoagent to enhance chemodynamic therapy. Angew Chem Int Ed Engl. 2018;57(18): 4902-4906. [68] MONTHA W, MANEEPRAKORN W, BUATONG N, et al. Synthesis of doxorubicin-PLGA loaded chitosan stabilized (Mn, Zn) Fe2O4 nanoparticles: biological activity and pH-responsive drug release. Mater Sci Eng C Mater Biol Appl. 2016;59:235-240. [69] YANG G, XU L, CHAO Y, et al. Hollow MnO2 as a tumor-microenvironment-responsive biodegradable nano-platform for combination therapy favoring antitumor immune responses. Nat Commun. 2017;8(1):902. [70] SHAIK MR, SYED R, ADIL SF, et al. Mn3O4 nanoparticles:Synthesis, characterization and their antimicrobial and anticancer activity against A549 and MCF-7 cell lines. Saudi J Biol Sci. 2021;28(2):1196-1202. [71] PACKIRISAMY RG, GOVINDASAMY C, SANMUGAM A, et al. Synthesis and antibacterial properties of novel ZnMn2O4-chitosan nanocomposites. Nanomaterials (Basel). 2019;9(11):1589. [72] ATIF M, IQBAL S, FAKHAR-E-ALAM M, et al. Manganese-doped cerium oxide nanocomposite induced photodynamic therapy in MCF-7 cancer cells and antibacterial activity. Biomed Res Int. 2019;2019:7156828. |
| [1] | Jin Tao, Liu Lin, Zhu Xiaoyan, Shi Yucong, Niu Jianxiong, Zhang Tongtong, Wu Shujin, Yang Qingshan. Osteoarthritis and mitochondrial abnormalities [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1452-1458. |
| [2] | Zhang Lichuang, Xu Hao, Ma Yinghui, Xiong Mengting, Han Haihui, Bao Jiamin, Zhai Weitao, Liang Qianqian. Mechanism and prospects of regulating lymphatic reflux function in the treatment of rheumatoid arthritis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1459-1466. |
| [3] | Yao Xiaoling, Peng Jiancheng, Xu Yuerong, Yang Zhidong, Zhang Shuncong. Variable-angle zero-notch anterior interbody fusion system in the treatment of cervical spondylotic myelopathy: 30-month follow-up [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1377-1382. |
| [4] | Zhu Chan, Han Xuke, Yao Chengjiao, Zhou Qian, Zhang Qiang, Chen Qiu. Human salivary components and osteoporosis/osteopenia [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1439-1444. |
| [5] | Wang Jing, Xiong Shan, Cao Jin, Feng Linwei, Wang Xin. Role and mechanism of interleukin-3 in bone metabolism [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1260-1265. |
| [6] | Zhu Chan, Han Xuke, Yao Chengjiao, Zhang Qiang, Liu Jing, Shao Ming. Acupuncture for Parkinson’s disease: an insight into the action mechanism in animal experiments [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1272-1277. |
| [7] | Guo Jia, Ding Qionghua, Liu Ze, Lü Siyi, Zhou Quancheng, Gao Yuhua, Bai Chunyu. Biological characteristics and immunoregulation of exosomes derived from mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1093-1101. |
| [8] | Wu Weiyue, Guo Xiaodong, Bao Chongyun. Application of engineered exosomes in bone repair and regeneration [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1102-1106. |
| [9] | Zhou Hongqin, Wu Dandan, Yang Kun, Liu Qi. Exosomes that deliver specific miRNAs can regulate osteogenesis and promote angiogenesis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1107-1112. |
| [10] | Zhang Jinglin, Leng Min, Zhu Boheng, Wang Hong. Mechanism and application of stem cell-derived exosomes in promoting diabetic wound healing [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1113-1118. |
| [11] | Huang Chenwei, Fei Yankang, Zhu Mengmei, Li Penghao, Yu Bing. Important role of glutathione in stemness and regulation of stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1119-1124. |
| [12] | Hui Xiaoshan, Bai Jing, Zhou Siyuan, Wang Jie, Zhang Jinsheng, He Qingyong, Meng Peipei. Theoretical mechanism of traditional Chinese medicine theory on stem cell induced differentiation [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1125-1129. |
| [13] | An Weizheng, He Xiao, Ren Shuai, Liu Jianyu. Potential of muscle-derived stem cells in peripheral nerve regeneration [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1130-1136. |
| [14] | Fan Yiming, Liu Fangyu, Zhang Hongyu, Li Shuai, Wang Yansong. Serial questions about endogenous neural stem cell response in the ependymal zone after spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1137-1142. |
| [15] | Xu Lei, Han Xiaoqiang, Zhang Jintao, Sun Haibiao. Hyaluronic acid around articular chondrocytes: production, transformation and function characteristics [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(5): 768-773. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||