Chinese Journal of Tissue Engineering Research ›› 2022, Vol. 26 ›› Issue (13): 2093-2098.doi: 10.12307/2022.339
Previous Articles Next Articles
Function and hot spot of adipose-derived stem cell exosomes in tissue regeneration and repair
Li Jianyi, Liu Zhiyuan, Deng Chengliang
- Department of Plastic Surgery, Affiliated Hospital of Zunyi Medical University, Zunyi 563003, Guizhou Province, China
-
Received:2020-12-01Revised:2020-12-19Accepted:2021-01-27Online:2022-05-08Published:2021-12-20 -
Contact:Deng Chengliang, MD, Master’s supervisor, Chief physician, Department of Plastic Surgery, Affiliated Hospital of Zunyi Medical University, Zunyi 563003, Guizhou Province, China -
About author:Li Jianyi, Master candidate, Department of Plastic Surgery, Affiliated Hospital of Zunyi Medical University, Zunyi 563003, Guizhou Province, China -
Supported by:the National Natural Science Foundation of China, No. 81801921 (to DCL); the Master's Startup Fund of Affiliated Hospital of Zunyi Medical College, No. (2018)34 (to LZY)
CLC Number:
Cite this article
Li Jianyi, Liu Zhiyuan, Deng Chengliang. Function and hot spot of adipose-derived stem cell exosomes in tissue regeneration and repair[J]. Chinese Journal of Tissue Engineering Research, 2022, 26(13): 2093-2098.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks

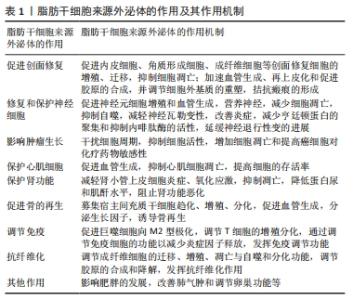
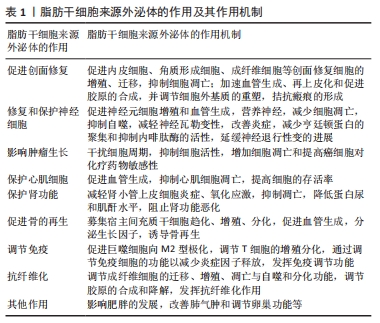
2.1 促进皮肤创面修复 皮肤创面愈合是机体复杂而有序的自我修复过程,涉及细胞增殖与迁移、血管生成和细胞外基质沉积与重塑。CHOI等[2]体外研究发现脂肪干细胞来源外泌体可通过上调Ⅰ型胶原、角质细胞生长因子、弹性蛋白的miRNAs表达,下调NPM1、PDCD4、CCL5和NUP62等衰老相关基因的miRNAs表达,刺激人真皮成纤维细胞增殖与迁移,且呈剂量依赖性。另有研究发现,脂肪干细胞来源外泌体可激活Wnt/β-catenin信号通路促进角质形成细胞增殖与迁移并抑制其凋亡,在创面修复的增殖期发挥积极作用[13]。 此外,脂肪干细胞来源外泌体也可通过促进血管生成而加快创面愈合。脂肪干细胞来源外泌体可刺激内皮细胞增殖、迁移,减少细胞凋亡而加快促进血管生成[15]。研究发现,脂肪干细胞来源外泌体中的miR-423-5p可靶向下调人脐静脉内皮细胞中Sufu的表达,促进内皮细胞的增殖、迁移和加速血管生成[16]。 脂肪干细胞来源外泌体治疗14 d后糖尿病大鼠足溃疡面积显著缩小,作用机制可能是其促进创面肉芽组织和血管生成,增加各种生长因子表达,降低炎症和减少氧化应激,同时过表达Nrf2的脂肪干细胞来源外泌体可增强上述作用[14]。类似地,mmu_circ_0000250修饰的脂肪干细胞来源外泌体可通过调控miR-128-3p/SIRT1信号通路激活自噬,减少内皮细胞凋亡,加快创面愈合[17]。脂肪干细胞来源外泌体还可以通过促进血管生成、减少细胞凋亡和减轻缺血再灌注损伤,从而提高皮瓣成活率[18-19]。另外脂肪干细胞来源外泌体可以诱导脂肪再生,增加脂肪的保留率,促进胶原的合成[20]。 脂肪干细胞来源外泌体还可调节细胞外基质的沉积与重塑,拮抗瘢痕形成。脂肪干细胞来源外泌体可通过激活PI3K/Akt信号通路调控Ⅲ型胶原/Ⅰ型胶原、转化生长因子β3/转化生长因子β1和基质金属蛋白酶3/基质金属蛋白酶抑制剂1的比例,抑制成纤维细胞向肌成纤维细胞分化,重塑细胞外基质,以拮抗瘢痕形成[21]。此外,脂肪干细胞来源外泌体可促进体外培养的成纤维细胞增殖、迁移和胶原合成,即增加了N-cadherin、cyclin-1、PCNA和Ⅰ型胶原、Ⅲ型胶原的表达,同时脂肪干细胞来源外泌体在小鼠创面愈合早期可增加胶原合成以促进创面愈合,而在愈合后期则抑制胶原合成以减少瘢痕形成[11]。 以上研究表明脂肪干细胞来源外泌体可作用于创面愈合过程中的各种参与细胞,如角质形成细胞、成纤维细胞和内皮细胞等,促进其增殖与迁移、抑制凋亡;促进创面血管生成;调节细胞外基质的沉积与重塑,从而加速创面愈合并减少瘢痕形成。 2.2 修复神经系统疾病 脂肪干细胞来源外泌体在神经损伤领域的治疗机制可能与其促进神经元细胞增殖、血管生成、营养神经有关。研究发现,脂肪干细胞通过外泌体将与神经再生相关的mRNAs和miRNAs转移到神经元细胞而在体外显著促进轴突生长[22]。YANG等[23]研究证实脂肪干细胞来源外泌体在体外可促进血管生成,而过表达miR-181b-5p的脂肪干细胞来源外泌体可增强这一作用,其主要机制为脂肪干细胞来源外泌体中的miR-181b-5p靶向抑制TRPM7的表达而提高脑微血管内皮细胞的血管生成功能,这为脑卒中的治疗提供了理论依据。脂肪干细胞来源外泌体通过减少细胞凋亡、抑制其自噬、减轻神经瓦勒变性和炎症而促进周围神经损伤后再生修复[24]。 脂肪干细胞来源外泌体对神经退行性疾病也有明显的保护作用。亨廷顿病是由突变的亨廷顿蛋白聚集引起的遗传性神经退行性疾病,LEE等[25]研究通过体外模型发现脂肪干细胞来源外泌体可减少亨廷顿蛋白的聚集、改善线粒体功能障碍和降低细胞凋亡率。同时,该研究团队还发现脂肪干细胞来源外泌体可以改善肌萎缩侧索硬化症的病理改变。另外,阿尔茨海默病是一种以β-淀粉样肽产生与清除失衡导致β-淀粉样肽在脑内异常积聚为病理特征的神经退行性疾病,Neprilysin(NEP)是脑内参与β-淀粉样肽水解与清除的关键降解酶,研究发现脂肪干细胞来源外泌体具有较高的NEP酶活性,可降低神经细胞系N2a的胞内β-淀粉样肽水平从而减少β-淀粉样肽在大脑中积聚[26]。以上这些提示了脂肪干细胞来源外泌体对神经损伤与神经退行性疾病具有一定的治疗潜力。 2.3 影响肿瘤生长 脂肪干细胞来源外泌体通过干扰细胞周期、抑制细胞活性、增加细胞凋亡和提高癌细胞对化疗药物敏感性而发挥抗肿瘤作用。REZA等[27]研究显示脂肪干细胞来源外泌体处理后的卵巢癌细胞体外活力降低,作用机制可能是脂肪干细胞来源外泌体增强氧化应激诱导的线粒体凋亡信号,上调促凋亡蛋白Bax、CASP9、CASP3表达,下调抗凋亡蛋白Bcl-2表达,从而促进肿瘤细胞凋亡,同时其可抑制细胞增殖导致细胞周期阻滞。该团队进一步研究发现当卵巢癌A2780细胞用胰蛋白酶消化后的脂肪干细胞来源外泌体裂解物处理后抑制了细胞增殖,而用经RNA酶消化的脂肪干细胞来源外泌体裂解物处理后细胞增殖并未被抑制,提示脂肪干细胞来源外泌体中携带的RNA可能是抑制肿瘤细胞增殖的主要活性物质。SEO等[28]发现携带miR-503-3P的脂肪干细胞来源外泌体可通过调节肿瘤干细胞干性而抑制肿瘤细胞的生长。 此外,脂肪干细胞来源外泌体可作为载体传递特定miRNA,增强肝癌细胞对化疗药物的敏感性。有学者发现富集miR-199a和miR-122的脂肪干细胞来源外泌体与化疗药物联合治疗肝癌,可抑制肝癌细胞增殖、诱导凋亡和阻滞细胞DNA复制,有效提高了肝癌细胞对化疗药物的敏感性而抑制肿瘤进展,然而有意思的是,单用脂肪干细胞来源外泌体对肝癌的生长没有影响,可能与脂肪干细胞来源外泌体中特定miRNA浓度太低有关[29]。KO等[30]的研究也表明了脂肪干细胞来源外泌体具有明显的肿瘤抑制作用,在小鼠模型中用脂肪干细胞来源外泌体处理后肝细胞癌的体积明显缩小,表观扩散系数增高,发挥抗肿瘤效应。 2.4 影响心血管疾病 脂肪干细胞来源外泌体通过促进血管生成、抑制心肌细胞凋亡,对心肌缺血性疾病具有保护作用。LUO等[31]研究发现脂肪干细胞来源外泌体可降低心肌细胞炎症因子和纤维化相关蛋白的表达水平,刺激血管内皮细胞迁移和血管生成,而体内实验发现其可抑制心肌细胞凋亡和降低心肌细胞炎症水平,促进心肌细胞增殖,进一步研究发现过表达miR-126的脂肪干细胞来源外泌体可增强上述作用,作用机制可能是脂肪干细胞来源外泌体内的miR-126直接靶向调控Spred1和PI3KR2的表达,增强血管内皮生长因子信号通路刺激血管生成,从而保护心肌细胞。 脂肪干细胞来源外泌体还可以直接激活相关信号通路,抑制心肌细胞凋亡[32],或通过减轻氧化应激以减少细胞凋亡,对心肌细胞产生保护作用[33]。研究发现,脂肪干细胞来源外泌体激活Wnt/β-catenin信号通路,上调抗凋亡蛋白Bcl-2并下调促凋亡蛋白Bax、Caspase3的表达,从而抑制心肌细胞凋亡,提高心肌细胞存活率,保护心肌功能[34]。LIU等[35]研究表明,心房颤动可降低心肌细胞miR-320d的表达,促进心肌细胞凋亡,降低细胞存活率,而转染miR-320d的脂肪干细胞来源外泌体可逆转房颤对心肌的损害作用,作用机制可能是miR-320d直接靶向调控STAT3信号通路,减少细胞凋亡并提高细胞存活率,保护心肌细胞。 2.5 影响肾脏疾病 脂肪干细胞来源外泌体对急性肾缺血等引起的急性肾损伤具有保护作用。脂肪干细胞来源外泌体可通过SIRT1途径对急性肾损伤产生保护作用[36]。LIN等[37]建立小鼠肾脏缺血再灌注模型,分别静脉注射脂肪干细胞、脂肪干细胞来源外泌体、脂肪干细胞+脂肪干细胞来源外泌体,72 h后发现均能减轻肾缺血再灌注损伤,脂肪干细胞组效果优于脂肪干细胞来源外泌体组,脂肪干细胞+脂肪干细胞来源外泌体组效果最好,其机制为脂肪干细胞来源外泌体减轻肾小管上皮细胞炎症、氧化应激,抑制凋亡,降低蛋白尿和肌酐水平,阻止肾功能恶化。ZHU等[38]发现脂肪干细胞来源外泌体通过上调Sox9表达促进肾小管上皮细胞再生,抑制转化生长因子β1诱导的肾小管上皮细胞向纤维化表型转化,减轻急性肾损伤后期的肾纤维化,阻断急性肾损伤向慢性肾病转变。此外,脂肪干细胞来源外泌体还可通过上调足细胞miR-486的表达,抑制足细胞Smad1/mTOR信号通路以促进自噬和抑制足细胞凋亡,从而明显改善糖尿病肾病的症状[39]。类似地,脂肪干细胞来源外泌体还可以抑制细胞凋亡和系膜组织增生而对糖尿病肾病起到保护肾功能的作用[40]。 2.6 促进骨再生 脂肪干细胞通过旁分泌外泌体参与骨再生修复过程。研究发现,脂肪干细胞来源外泌体具有促成骨效应,促进脂肪干细胞向成骨细胞分化[41-42]。类似地,过表达miR-130a-3p的脂肪干细胞来源外泌体可以增强脂肪干细胞的成骨分化,降低miR-130a-3p的靶标SIRT7蛋白和mRNA水平,并上调Wnt信号通路相关蛋白,即外泌体miR-130a-3p可以通过介导SIRT7/Wnt/β-catenin轴来促进脂肪干细胞的成骨分化[43]。脂肪干细胞来源外泌体联合聚乳酸-羟基乙酸共聚物支架移植6周后,在小鼠颅骨缺损模型可看到明显的新生骨形成,其机制可能与脂肪干细胞来源外泌体减少成骨细胞凋亡,募集宿主间充质干细胞趋化、增殖、分化,促血管生成有关,而且聚乳酸-羟基乙酸共聚物支架缓慢控释脂肪干细胞来源外泌体,可延长脂肪干细胞来源外泌体作用时间,提高脂肪干细胞来源外泌体移植的疗效[44]。CHEN等[45]进一步研究发现相对于单纯的脂肪干细胞来源外泌体结合水凝胶移植,富集miR-375的脂肪干细胞来源外泌体结合水凝胶移植后可出现更多的新生骨,机制可能为miR-375是骨再生过程中的关键调节因子,过表达miR-375的脂肪干细胞来源外泌体通过水凝胶缓慢控释于受损区并靶向下调胰岛素样生长因子结合蛋白3来诱导成骨效应。 2.7 调控免疫 脂肪干细胞来源外泌体通过调节免疫细胞以及炎症因子释放,发挥免疫调节功能。有学者发现脂肪干细胞来源外泌体通过携带有活性的STAT3反式激活精氨酸酶1而诱导巨噬细胞向抗炎的M2表型转化,从而减轻炎症和胰岛素抵抗,发挥调节免疫和代谢稳态的作用[46]。另一项研究发现,脂肪干细胞来源外泌体以剂量依赖的方式促进巨噬细胞极化成M2样表型,降低促炎细胞因子水平,增加促血管生成因子而促进内皮细胞的增殖、迁移和管状形成,从而加快血管的生成;脂肪干细胞来源外泌体中的miR-21及其集落刺激因子1的正反馈可能参与了巨噬细胞极化。此外,脂肪干细胞来源外泌体还可以通过调节T细胞功能而发挥免疫调节功能[47]。研究表明,携带miR-10a的脂肪干细胞来源外泌体可促进原始CD4T细胞分化为Th17细胞和Treg细胞,降低Th1应答,减少炎症因子的产生[48]。CHO等[49]建立小鼠特应性皮炎模型,通过静脉或皮下注射脂肪干细胞来源外泌体,28 d后发现肥大细胞浸润明显减轻,同时临床评分、血清IgE水平、外周血嗜酸粒细胞数和白细胞介素4、白细胞介素23、白细胞介素31和肿瘤坏死因子α等多种炎性细胞因子水平均降低,结果提示脂肪干细胞来源外泌体具有治疗小鼠特应性皮炎的潜力。 2.8 抗纤维化 脂肪干细胞来源外泌体可通过调节成纤维细胞的迁移、增殖、凋亡和分化功能发挥抗纤维化作用。QU等[50] 发现富集于脂肪干细胞来源外泌体中的miR-181-5p可抑制STAT3/Bcl-2/Beclin信号通路激活细胞自噬,抑制转化生长因子β1诱导的肝星状细胞纤维化基因的表达,下调Ⅰ型胶原、波形蛋白、α-平滑肌肌动蛋白和纤维连接蛋白的表达,减轻肝损伤并抑制肝纤维化。李洪超等[51]研究也发现脂肪干细胞来源外泌体通过减少胶原纤维、平滑肌动蛋白和基质金属蛋白酶抑制剂1合成,增加基质金属蛋白酶9的合成来改善四氯化碳诱导的肝纤维化。另有研究发现,脂肪干细胞来源外泌体含有促血管生成的miRNA(如miR-126、miR-130a和miR-132)和一些抗纤维化的miRNA(miR-let7b和miR-let7c),并以剂量依赖性的方式促进内皮细胞增殖,减少海绵体纤维化,改善糖尿病大鼠的勃起功能障碍[52]。CHEN等[53]研究也得出了类似的结论,并且证实了脂肪干细胞减轻糖尿病大鼠勃起功能障碍的效果主要是通过其外泌体起作用的。脂肪干细胞来源外泌体也可通过调节胶原纤维的生成与代谢改善压力性尿失禁。脂肪干细胞来源外泌体能增加尿道周围阴道壁组织成纤维细胞中Ⅰ型胶原、基质金属蛋白酶抑制剂1和基质金属蛋白酶抑制剂3的表达水平,下调基质金属蛋白酶1和基质金属蛋白酶2水平,增加压力性尿失禁患者阴道成纤维细胞胶原合成和减少胶原降解,增加尿道中的横纹肌纤维和周围神经纤维,对压力性尿失禁有治疗作用[54]。另一项研究发现过表达 miR-34a的脂肪干细胞来源外泌体可通过下调TGF-β/Smad信号通路抑制增生性瘢痕成纤维细胞增殖,并促进其凋亡,从而发挥抗瘢痕作用[55]。 2.9 其他作用 研究发现,过表达miR-27的脂肪干细胞来源外泌体可抑制白色脂肪的棕色化,从而影响肥胖的发展[56]。脂肪干细胞来源纳米囊泡可通过增加成纤维细胞生长因子2而抑制肺气肿的发展[57]。此外,脂肪干细胞来源外泌体可以有效减少肩袖撕裂肌肉的萎缩和变性,改善肌肉再生和生物力学性能[58]。脂肪干细胞来源外泌体对早产卵巢功能不全具有一定的治疗作用,即经过脂肪干细胞来源外泌体治疗后可改善小鼠的卵巢功能,提高激素水平,增加卵泡数量,其主要机制是脂肪干细胞来源外泌体通过激活SMAD来降低凋亡基因的表达,抑制细胞凋亡和提高细胞的增殖能力,从而改善卵巢功能不全早产儿的卵巢功能[59]。类似地,脂肪干细胞来源外泌体还可促进子宫粘连大鼠的子宫内膜再生和生育能力恢复[60]。"

| [1] FANG Y, ZHANG Y, ZHOU J, et al. Adipose-derived mesenchymal stem cell exosomes: a novel pathway for tissues repair. Cell Tissue Bank. 2019;20(2):153-161. [2] CHOI EW, SEO MK, WOO EY, et al. Exosomes from human adipose-derived stem cells promote proliferation and migration of skin fibroblasts. Exp Dermatol. 2018;27(10):1170-1172. [3] AKERS JC, GONDA D, KIM R, et al. Biogenesis of extracellular vesicles (EV): exosomes, microvesicles, retrovirus-like vesicles, and apoptotic bodies. J Neurooncol. 2013;113(1):1-11. [4] LI P, KASLAN M, LEE SH, et al. Progress in Exosome Isolation Techniques. Theranostics. 2017;7(3):789-804. [5] HONG P, YANG H, WU Y, et al. The functions and clinical application potential of exosomes derived from adipose mesenchymal stem cells: a comprehensive review. Stem Cell Res Ther. 2019;10(1):242. [6] COOPER DR, WANG C, PATEL R, et al. Human Adipose-Derived Stem Cell Conditioned Media and Exosomes Containing MALAT1 Promote Human Dermal Fibroblast Migration and Ischemic Wound Healing. Adv Wound Care (New Rochelle). 2018;7(9):299-308. [7] WANG C, WANG M, XU T, et al. Engineering Bioactive Self-Healing Antibacterial Exosomes Hydrogel for Promoting Chronic Diabetic Wound Healing and Complete Skin Regeneration. Theranostics. 2019; 9(1):65-76. [8] VALADI H, EKSTRÖM K, BOSSIOS A, et al. Exosome-mediated transfer of mRNAs and microRNAs is a novel mechanism of genetic exchange between cells. Nat Cell Biol. 2007;9(6):654-659. [9] QIU H, LIU S, WU K, et al. Prospective application of exosomes derived from adipose-derived stem cells in skin wound healing: A review. J Cosmet Dermatol. 2020;19(3):574-581. [10] MAAS SLN, BREAKEFIELD XO, WEAVER AM. Extracellular Vesicles: Unique Intercellular Delivery Vehicles. Trends Cell Biol. 2017;27(3): 172-188. [11] HU L, WANG J, ZHOU X, et al. Exosomes derived from human adipose mensenchymal stem cells accelerates cutaneous wound healing via optimizing the characteristics of fibroblasts. Sci Rep. 2016;6:32993. [12] 郝海珍,郭铁,余丹.脂肪间充质干细胞来源外泌体对脑缺血再灌注大鼠神经元凋亡及炎症因子影响[J].第三军医大学学报,2019, 41(17):1656-1665. [13] MA T, FU B, YANG X, et al. Adipose mesenchymal stem cell-derived exosomes promote cell proliferation, migration, and inhibit cell apoptosis via Wnt/β-catenin signaling in cutaneous wound healing. J Cell Biochem. 2019;120(6):10847-10854. [14] LI X, XIE X, LIAN W, et al. Exosomes from adipose-derived stem cells overexpressing Nrf2 accelerate cutaneous wound healing by promoting vascularization in a diabetic foot ulcer rat model. Exp Mol Med. 2018; 50(4):1-14. [15] HAN Y, REN J, BAI Y, et al. Exosomes from hypoxia-treated human adipose-derived mesenchymal stem cells enhance angiogenesis through VEGF/VEGF-R. Int J Biochem Cell Biol. 2019;109:59-68. [16] XU F, XIANG Q, HUANG J, et al. Exosomal miR-423-5p mediates the proangiogenic activity of human adipose-derived stem cells by targeting Sufu. Stem Cell Res Ther. 2019;10(1):106. [17] SHI R, JIN Y, HU W, et al. Exosomes derived from mmu_circ_0000250-modified adipose-derived mesenchymal stem cells promote wound healing in diabetic mice by inducing miR-128-3p/SIRT1-mediated autophagy. Am J Physiol Cell Physiol. 2020;318(5):C848-C856. [18] 胡玄,易阳艳,朱元正,等.脂肪干细胞来源外泌体促进大鼠皮瓣移植后血管新生的研究[J].中国修复重建外科杂志,2019,33(12): 1560-1565. [19] BAI Y, HAN YD, YAN XL, et al. Adipose mesenchymal stem cell-derived exosomes stimulated by hydrogen peroxide enhanced skin flap recovery in ischemia-reperfusion injury. Biochem Biophys Res Commun. 2018;500(2):310-317. [20] CHEN B, CAI J, WEI Y, et al. Exosomes Are Comparable to Source Adipose Stem Cells in Fat Graft Retention with Up-Regulating Early Inflammation and Angiogenesis. Plast Reconstr Surg. 2019;144(5):816e-827e. [21] WANG L, HU L, ZHOU X, et al. Exosomes secreted by human adipose mesenchymal stem cells promote scarless cutaneous repair by regulating extracellular matrix remodelling. Sci Rep. 2017;7(1):13321. [22] CHING RC, WIBERG M, KINGHAM PJ. Schwann cell-like differentiated adipose stem cells promote neurite outgrowth via secreted exosomes and RNA transfer. Stem Cell Res Ther. 2018;9(1):266. [23] YANG Y, CAI Y, ZHANG Y, et al. Exosomes Secreted by Adipose-Derived Stem Cells Contribute to Angiogenesis of Brain Microvascular Endothelial Cells Following Oxygen-Glucose Deprivation In Vitro Through MicroRNA-181b/TRPM7 Axis. J Mol Neurosci. 2018;65(1):74-83. [24] 尹刚,刘蔡钺,林耀发,等.脂肪干细胞来源外泌体对周围神经损伤后再生作用的实验研究[J].干细胞与组织工程,2018,32(12): 1592-1596. [25] LEE M, LIU T, IM W, et al. Exosomes from adipose-derived stem cells ameliorate phenotype of Huntington’s disease in vitro model. Eur J Neurosci. 2016;44(4):2114-2119. [26] KATSUDA T, TSUCHIYA R, KOSAKA N, et al. Human adipose tissue-derived mesenchymal stem cells secrete functional neprilysin-bound exosomes. Sci Rep. 2013;3:1197. [27] REZA AMMT, CHOI YJ, YASUDA H, et al. Human adipose mesenchymal stem cell-derived exosomal-miRNAs are critical factors for inducing anti-proliferation signalling to A2780 and SKOV-3 ovarian cancer cells. Sci Rep. 2016;6:38498. [28] SEO M, KIM SM, WOO EY, et al. Stemness-Attenuating miR-503-3p as a Paracrine Factor to Regulate Growth of Cancer Stem Cells. Stem Cells Int. 2018;2018:4851949. [29] LOU G, CHEN L, XIA C, et al. MiR-199a-modified exosomes from adipose tissue-derived mesenchymal stem cells improve hepatocellular carcinoma chemosensitivity through mTOR pathway. J Exp Clin Cancer Res. 2020;39(1):4. [30] KO SF, YIP HK, ZHEN YY, et al. Adipose-Derived Mesenchymal Stem Cell Exosomes Suppress Hepatocellular Carcinoma Growth in a Rat Model: Apparent Diffusion Coefficient, Natural Killer T-Cell Responses, and Histopathological Features. Stem Cells Int. 2015;2015:853506. [31] LUO Q, GUO D, LIU G, et al. Exosomes from MiR-126-Overexpressing Adscs Are Therapeutic in Relieving Acute Myocardial Ischaemic Injury. Cell Physiol Biochem. 2017;44(6):2105-2116. [32] 梁政,钟剑锋,吴源聪,等.脂肪间充质干细胞来源外泌体对缺氧/复氧诱导的心肌细胞凋亡的影响[J].中国医药导报,2020,17(5): 4,7,22,197. [33] 夏海兵.脂肪间充质干细胞外泌体缓解阿霉素所致急性心肌损伤的研究[D].蚌埠:蚌埠医学院,2019. [34] CUI X, HE Z, LIANG Z, et al. Exosomes From Adipose-derived Mesenchymal Stem Cells Protect the Myocardium Against Ischemia/Reperfusion Injury Through Wnt/β-Catenin Signaling Pathway. J Cardiovasc Pharmacol. 2017;70(4):225-231. [35] LIU L, ZHANG H, MAO H, et al. Exosomal miR-320d derived from adipose tissue-derived MSCs inhibits apoptosis in cardiomyocytes with atrial fibrillation (AF). Artif Cells Nanomed Biotechnol. 2019;47(1): 3976-3984. [36] GAO F, ZUO B, WANG Y, et al. Protective function of exosomes from adipose tissue-derived mesenchymal stem cells in acute kidney injury through SIRT1 pathway. Life Sci. 2020;255:117719. [37] LIN KC, YIP HK, SHAO PL, et al. Combination of adipose-derived mesenchymal stem cells (ADMSC) and ADMSC-derived exosomes for protecting kidney from acute ischemia-reperfusion injury. Int J Cardiol. 2016;216:173-185. [38] ZHU F, CHONG LEE SHIN OLS, PEI G, et al. Adipose-derived mesenchymal stem cells employed exosomes to attenuate AKI-CKD transition through tubular epithelial cell dependent Sox9 activation. Oncotarget. 2017;8(41):70707-70726. [39] JIN J, SHI Y, GONG J, et al. Exosome secreted from adipose-derived stem cells attenuates diabetic nephropathy by promoting autophagy flux and inhibiting apoptosis in podocyte. Stem Cell Res Ther. 2019;10(1):95. [40] 陈晶,刘清岩,张海宁,等.脂肪干细胞来源的外泌体对2型糖尿病肾病大鼠的保护作用[J].解剖科学进展,2020,26(4):369-373. [41] 陈义庆,郭澍,佟爽,等.可注射水凝胶负载脂肪来源干细胞外泌体促进骨再生[J].中国美容整形外科杂志,2019,30(4):206-209. [42] 魏松乔,郭澍,佟爽,等.脂肪干细胞-丝素/壳聚糖支架复合物在外泌体诱导下的体外成骨效应[J].中国医科大学学报,2019,48(10): 891-895. [43] YANG S, GUO S, TONG S, et al. Exosomal miR-130a-3p regulates osteogenic differentiation of Human Adipose-Derived stem cells through mediating SIRT7/Wnt/β-catenin axis. Cell Prolif. 2020;53(10): e12890. [44] LI W, LIU Y, ZHANG P, et al. Tissue-Engineered Bone Immobilized with Human Adipose Stem Cells-Derived Exosomes Promotes Bone Regeneration. ACS Appl Mater Interfaces. 2018;10(6):5240-5254. [45] CHEN S, TANG Y, LIU Y, et al. Exosomes derived from miR-375-overexpressing human adipose mesenchymal stem cells promote bone regeneration. Cell Prolif. 2019;52(5):e12669. [46] ZHAO H, SHANG Q, PAN Z, et al. Exosomes From Adipose-Derived Stem Cells Attenuate Adipose Inflammation and Obesity Through Polarizing M2 Macrophages and Beiging in White Adipose Tissue. Diabetes. 2018; 67(2): 235-247. [47] ZHU D, JOHNSON TK, WANG Y, et al. Macrophage M2 polarization induced by exosomes from adipose-derived stem cells contributes to the exosomal proangiogenic effect on mouse ischemic hindlimb. Stem Cell Res Ther. 2020; 11(1):162. [48] BOLANDI Z, MOKHBERIAN N, EFTEKHARY M, et al. Adipose derived mesenchymal stem cell exosomes loaded with miR-10a promote the differentiation of Th17 and Treg from naive CD4+ T cell. Life Sci. 2020;259:118218. [49] CHO BS, KIM JO, HA DH, et al. Exosomes derived from human adipose tissue-derived mesenchymal stem cells alleviate atopic dermatitis. Stem Cell Res Ther. 2018;9(1):187. [50] QU Y, ZHANG Q, CAI X, et al. Exosomes derived from miR-181-5p-modified adipose-derived mesenchymal stem cells prevent liver fibrosis via autophagy activation. J Cell Mol Med. 2017;21(10):2491-2502. [51] 李洪超,王皙,李莉,等.人脂肪干细胞来源外泌体对四氯化碳诱导肝纤维化模型大鼠的治疗作用[J].中国组织工程研究,2020, 24(13):1996-2004. [52] ZHU LL, HUANG X, YU W, et al. Transplantation of adipose tissue-derived stem cell-derived exosomes ameliorates erectile function in diabetic rats. Andrologia. 2018;50(2):e12871. [53] CHEN F, ZHANG H, WANG Z, et al. Adipose-Derived Stem Cell-Derived Exosomes Ameliorate Erectile Dysfunction in a Rat Model of Type 2 Diabetes. J Sex Med. 2017; 14(9):1084-1094. [54] LIU X, WANG S, WU S, et al. Exosomes secreted by adipose-derived mesenchymal stem cells regulate type I collagen metabolism in fibroblasts from women with stress urinary incontinence. Stem Cell Res Ther. 2018;9(1):159. [55] 肖向阳,郑德义,王宝云,等.过表达miR-34a脂肪干细胞外泌体调控增生性瘢痕成纤维细胞增殖及凋亡的研究[J].安徽医科大学学报,2020,55(6):887-893. [56] 钟琼慧,黄波,卢婉,等.脂肪干细胞来源外泌体携带miR-27抑制白色脂肪的棕色化[J].天津医药,2020,48(3):165-170. [57] KIM YS, KIM JY, CHO R, et al. Adipose stem cell-derived nanovesicles inhibit emphysema primarily via an FGF2-dependent pathway. Exp Mol Med. 2017;49(1):e284. [58] WANG C, SONG W, CHEN B, et al. Exosomes Isolated From Adipose-Derived Stem Cells: A New Cell-Free Approach to Prevent the Muscle Degeneration Associated With Torn Rotator Cuffs. Am J Sports Med. 2019;47(13):3247-3255. [59] HUANG B, LU J, DING C, et al. Exosomes derived from human adipose mesenchymal stem cells improve ovary function of premature ovarian insufficiency by targeting SMAD. Stem Cell Res Ther. 2018;9(1):216. [60] ZHAO S, QI W, ZHENG J, et al. Exosomes Derived from Adipose Mesenchymal Stem Cells Restore Functional Endometrium in a Rat Model of Intrauterine Adhesions. Reprod Sci. 2020;27(6):1266-1275. [61] AN Y, ZHAO J, NIE F, et al. Exosomes from Adipose-Derived Stem Cells (ADSCs) Overexpressing miR-21 Promote Vascularization of Endothelial Cells. Sci Rep. 2019;9(1):12861. [62] WANG X, ZHANG H, YANG H, et al. Cell-derived Exosomes as Promising Carriers for Drug Delivery and Targeted Therapy. Curr Cancer Drug Targets. 2018;18(4):347-354. [63] LOU G, SONG X, YANG F, et al. Exosomes derived from miR-122-modified adipose tissue-derived MSCs increase chemosensitivity of hepatocellular carcinoma. J Hematol Oncol. 2015;8:122. [64] LIANG X, ZHANG L, WANG S, et al. Exosomes secreted by mesenchymal stem cells promote endothelial cell angiogenesis by transferring miR-125a. J Cell Sci. 2016;129(11):2182-2189. [65] HE C, ZHENG S, LUO Y, et al. Exosome Theranostics: Biology and Translational Medicine. Theranostics. 2018;8(1):237-255. [66] CHAROENVIRIYAKUL C, TAKAHASHI Y, NISHIKAWA M, et al. Preservation of exosomes at room temperature using lyophilization. Int J Pharm. 2018;553(1-2):1-7. [67] KIM H, WANG SY, KWAK G, et al. Exosome-Guided Phenotypic Switch of M1 to M2 Macrophages for Cutaneous Wound Healing. Adv Sci (Weinh). 2019;6(20):1900513. [68] TAKAHASHI Y, NISHIKAWA M, SHINOTSUKA H, et al. Visualization and in vivo tracking of the exosomes of murine melanoma B16-BL6 cells in mice after intravenous injection. J Biotechnol. 2013;165(2):77-84. [69] LIN R, WANG S, ZHAO RC. Exosomes from human adipose-derived mesenchymal stem cells promote migration through Wnt signaling pathway in a breast cancer cell model. Mol Cell Biochem. 2013;383(1-2):13-20. [70] 王明霜,龙洋,吴剑,等.脂肪干细胞外泌体对子宫内膜癌HEC-251细胞增殖和凋亡的影响[J].基因组学与应用生物学,2019,38(7): 3336-3340. |
| [1] | Zhu Chan, Han Xuke, Yao Chengjiao, Zhou Qian, Zhang Qiang, Chen Qiu. Human salivary components and osteoporosis/osteopenia [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1439-1444. |
| [2] | Jin Tao, Liu Lin, Zhu Xiaoyan, Shi Yucong, Niu Jianxiong, Zhang Tongtong, Wu Shujin, Yang Qingshan. Osteoarthritis and mitochondrial abnormalities [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1452-1458. |
| [3] | Zhang Lichuang, Xu Hao, Ma Yinghui, Xiong Mengting, Han Haihui, Bao Jiamin, Zhai Weitao, Liang Qianqian. Mechanism and prospects of regulating lymphatic reflux function in the treatment of rheumatoid arthritis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1459-1466. |
| [4] | Xiao Hao, Liu Jing, Zhou Jun. Research progress of pulsed electromagnetic field in the treatment of postmenopausal osteoporosis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1266-1271. |
| [5] | Zhu Chan, Han Xuke, Yao Chengjiao, Zhang Qiang, Liu Jing, Shao Ming. Acupuncture for Parkinson’s disease: an insight into the action mechanism in animal experiments [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1272-1277. |
| [6] | Wang Jing, Xiong Shan, Cao Jin, Feng Linwei, Wang Xin. Role and mechanism of interleukin-3 in bone metabolism [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1260-1265. |
| [7] | An Weizheng, He Xiao, Ren Shuai, Liu Jianyu. Potential of muscle-derived stem cells in peripheral nerve regeneration [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1130-1136. |
| [8] | Fan Yiming, Liu Fangyu, Zhang Hongyu, Li Shuai, Wang Yansong. Serial questions about endogenous neural stem cell response in the ependymal zone after spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1137-1142. |
| [9] | Wu Weiyue, Guo Xiaodong, Bao Chongyun. Application of engineered exosomes in bone repair and regeneration [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1102-1106. |
| [10] | Zhou Hongqin, Wu Dandan, Yang Kun, Liu Qi. Exosomes that deliver specific miRNAs can regulate osteogenesis and promote angiogenesis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1107-1112. |
| [11] | Zhang Jinglin, Leng Min, Zhu Boheng, Wang Hong. Mechanism and application of stem cell-derived exosomes in promoting diabetic wound healing [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1113-1118. |
| [12] | Huang Chenwei, Fei Yankang, Zhu Mengmei, Li Penghao, Yu Bing. Important role of glutathione in stemness and regulation of stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1119-1124. |
| [13] | Hui Xiaoshan, Bai Jing, Zhou Siyuan, Wang Jie, Zhang Jinsheng, He Qingyong, Meng Peipei. Theoretical mechanism of traditional Chinese medicine theory on stem cell induced differentiation [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1125-1129. |
| [14] | Tian Chuan, Zhu Xiangqing, Yang Zailing, Yan Donghai, Li Ye, Wang Yanying, Yang Yukun, He Jie, Lü Guanke, Cai Xuemin, Shu Liping, He Zhixu, Pan Xinghua. Bone marrow mesenchymal stem cells regulate ovarian aging in macaques [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 985-991. |
| [15] | Wen Dandan, Li Qiang, Shen Caiqi, Ji Zhe, Jin Peisheng. Nocardia rubra cell wall skeleton for extemal use improves the viability of adipogenic mesenchymal stem cells and promotes diabetes wound repair [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1038-1044. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||