Chinese Journal of Tissue Engineering Research ›› 2022, Vol. 26 ›› Issue (10): 1631-1640.doi: 10.12307/2022.215
Application advantages of concentrated growth factors in the field of tissue repair and regeneration
Zhou Yi, Liu Xiaoyan, Xiang Bingyan
- Department of Joint Surgery, Third Affiliated Hospital of Zunyi Medical University (Zunyi First People’s Hospital), Zunyi 563000, Guizhou Province, China
-
Received:2021-04-23Revised:2021-04-24Accepted:2021-05-21Online:2022-04-08Published:2021-10-27 -
Contact:Xiang Bingyan, MD, Professor, Chief physician, Master’s supervisor, Department of Joint Surgery, Third Affiliated Hospital of Zunyi Medical University (Zunyi First People’s Hospital), Zunyi 563000, Guizhou Province, China -
About author:Zhou Yi, Master candidate, Department of Joint Surgery, Third Affiliated Hospital of Zunyi Medical University (Zunyi First People’s Hospital), Zunyi 563000, Guizhou Province, China -
Supported by:the Science and Technology Fund Project of Guizhou Provincial Health Commission, No. gzwjkj-2020-1-129 (to XBY)
CLC Number:
Cite this article
Zhou Yi, Liu Xiaoyan, Xiang Bingyan. Application advantages of concentrated growth factors in the field of tissue repair and regeneration[J]. Chinese Journal of Tissue Engineering Research, 2022, 26(10): 1631-1640.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks

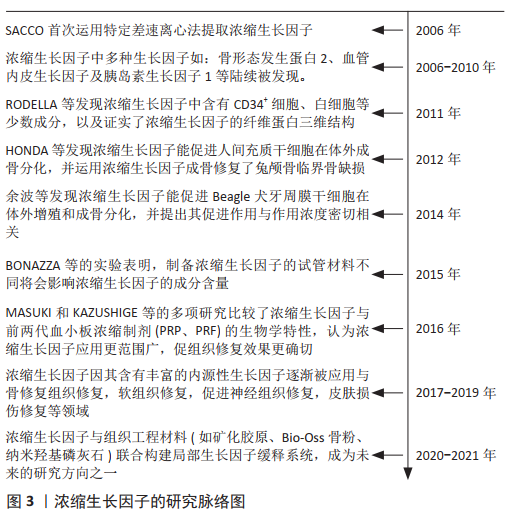
2.1 浓缩生长因子的研究脉 见图3。 血小板浓缩物发展至今,主要分为以下3类:第1代为富血小板血浆,第2代为富血小板纤维蛋白,第3代为浓缩生长因子。20世纪80年代以来,富血小板血浆作为第1代血小板浓缩产物,逐渐应用于软硬组织的修复和再生等领域[2-3]。富血小板血浆的制备需要加入凝血酶等生物制剂,对人体健康有潜在威胁,且制备的富血小板血浆有效成分含量较少,生长因子作用时间短,富血小板血浆的实际疗效和安全性存在一定争议。富血小板纤维蛋白在富血小板血浆的基础上进一步改进和简化,通过模拟生理性的凝血过程,仅对全血进行匀速离心而制备,制备过程中无需添加任何生物制剂[4-5]。有研究表明富血小板纤维蛋白较富血小板血浆释放的生长因子量更大且持续时间更长,富血小板纤维蛋白更利于组织缺损的修复与再生[6-7]。因此富血小板纤维蛋白更多的被广泛应用于颌面外科,整形外科等领域[8-9]。2006年,SACCO首次运用特定的差速离心方法制备出第3代血小板浓缩物浓缩生长因子。与富血小板血浆和富血小板纤维蛋白不同,浓缩生长因子制备采用特定的变速离心,形成了更具韧性的三维纤维蛋白网络,为细胞的黏附、增殖提供了三维空间,变速离心也能够释放出更高浓度的生长因子[10-11],并将白细胞、血小板更为稳定地聚合在浓缩生长因子内[12],使浓缩生长因子的有效成分提取和分离更加完全。此外,浓缩生长因子具有良好的可塑性和兼容性,能被制备成提取液、冻干材料和浓缩生长因子膜等,也可加入培养基中,还能与组织工程的材料结合应用。近年来越来越多的研究对浓缩生长因子的应用价值进行探索。但目前大部分综述仅着重关注浓缩生长因子在促进骨组织修复方面的研究进展,尤其是在口腔种植体修复领域[13],颌面骨修复领域[14-15],然而浓缩生长因子在促进其他组织的修复与再生中仍具有相当的潜力,最近的研究也开始关注浓缩生长因子除促进骨修复以外的作用。"
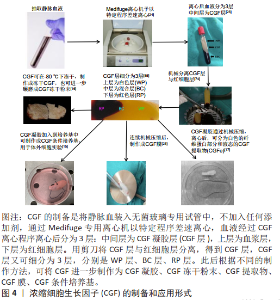
2.2 浓缩生长因子的制备和应用形式 浓缩生长因子的制备和应用方法如图4所示。 浓缩生长因子的制备是将静脉血装入无菌玻璃专用试管中,不加入任何添加剂,通过Medifuge专用离心机离心,其离心程序:加速30 s,以2 700 r/min离心2 min,2 400 r/min 离心4 min,2 700 r/min 离心4 min, 3 000 r/min 离心3 min,减速36 s 至停止。血液经过浓缩生长因子离心程序离心后分为3 层:中间层为浓缩生长因子凝胶层,上层为血浆层,下层为红细胞层[16]。用注射器将管顶部的去血小板血浆抽出,并将浓缩生长因子层和红细胞层从试管上取下。然后用剪刀将两层分离,得到浓缩生长因子层,过程中保留浓缩生长因子层和红细胞层的交界部分,因为这个部分富含血小板、白细胞及各种生长因子。"
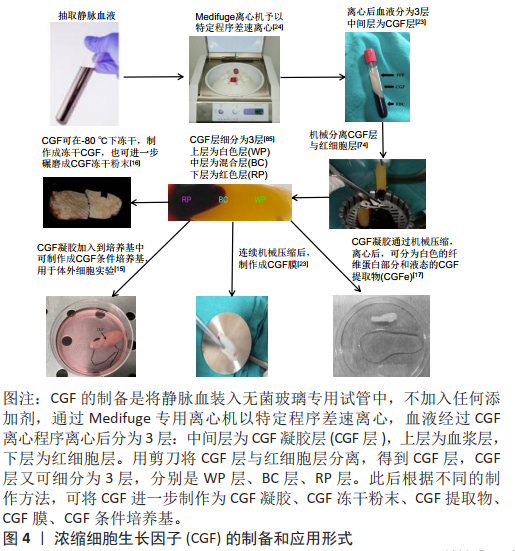
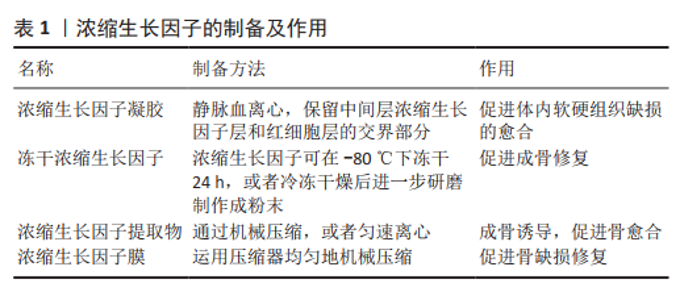
2.3 浓缩生长因子的应用形式 主要包括4种,见表1。 2.3.1 浓缩生长因子凝胶 通过上述方法制备的浓缩生长因子凝胶切成碎片后可以直接植入体内,用于促进体内软硬组织缺损的愈合,效果良好[17-18]。在体外也可以将浓缩生长因子放入条件培养基中,例如直接将浓缩生长因子凝胶加入培养基,再加入体积分数1%胎牛血清,50 μmol/L抗坏血酸盐, 100 nmol/L地塞米松和10 mmol/L β-甘油磷酸酯制作成成骨条件诱导培养基,在体外已被证明有良好的诱导成骨作用。 2.3.2 冻干浓缩生长因子 浓缩生长因子可在-80 ℃下冻干24 h,制作成冻干浓缩生长因子,或者冷冻干燥后进一步研磨,制作成浓缩生长因子粉末直接使用。浓缩生长因子经过冻干后,方便使用和保存,有研究表明冻干的浓缩生长因子粉末,生长因子在浓缩生长因子中处于自由扩散状态,可持续释放胰岛素样生长因子1、血管内皮生长因子、转化生长因子β1和血小板衍生生长因子AB等生长因子[19]。 2.3.3 浓缩生长因子提取物 浓缩生长因子提取物,是浓缩生长因子的另一种应用形式,制作原理是通过将完整的浓缩生长因子机械压缩,或者匀速离心,将浓缩生长因子富含纤维蛋白的部分和富含生长因子的液态浓缩生长因子提取物分离。因浓缩生长因子提取物多成液态或者半凝胶状,在体外加入培养基后,相比直接放入整体的浓缩生长因子,不仅能充分释放各种生长因子,还可以做到定量加入浓缩生长因子提取物,根据浓缩生长因子提取物的浓度制作成不同浓缩生长因子提取物浓度的条件培养基,目前一些研究支持体积分数10%的浓缩生长因子提取物培养基对成骨诱导作用最 大[20-21],高浓度的浓缩生长因子提取物反而抑制细胞的增殖和分化[22]。同样直接植入体内损伤部位可以促进愈合作用,但由于浓缩生长因子提取物失去了纤维蛋白三维结构的缓释作用,可能导致各种生长因子在损伤局部原位过快释放[23]。目前对于浓缩生长因子提取物的最佳浓度和生长因子释放动力学相关研究较少,还需要进一步探究。 2.3.4 浓缩生长因子膜 由于浓缩生长因子富含纤维蛋白成分,运用压缩器均匀地机械压缩后,可以制备成浓缩生长因子膜。有研究表明经过压缩后的浓缩生长因子膜,原有的纤维蛋白三维结构在一定程度上被改变,释放生长因子相较浓缩生长因子凝胶缓慢[24-25],这可能为延长生长因子缓释时间提供了参考。由于薄膜浓缩生长因子植入更加便捷,且浓缩生长因子膜增大了和损伤部位的接触面积,使各种生长因子能大面积释放到损伤局部的各个部位。一些研究借鉴了Masqulet技术生成诱导膜的原理,将浓缩生长因子膜原位覆盖于骨缺损部位的表面,取得了良好的骨缺损修复效果[26-27]。浓缩生长因子膜的发明,确实为临床治疗提供了很大便利,对于慢性皮肤溃疡缺损,浓缩生长因子膜直接覆盖于创面表面,也有良好的愈合效果[28]。 2.4 浓缩生长因子的生物学特性 浓缩生长因子具备生物学活性的两大核心结构是天然的纤维蛋白支架和各种内源性生长因子,此外还有含有少量的白细胞、CD34+免疫细胞、白细胞介素6细胞因子等,使浓缩生长因子还具备一定的抗炎和免疫调节作用[16]。 天然的纤维蛋白支架是在浓缩生长因子特定变速离心过程中不断交联形成的,由于为包含加速和减速过程,大幅度增加了纤维蛋白互相重叠交联的程度,更有利于纤维蛋白原转化为纤维蛋白,相比富血小板纤维蛋白,浓缩生长因子的纤维蛋白网络更加致密和坚固[29]。变速离心过程相比单一匀速离心,能将各沉降系数不同的成分更加完全地分离,因此浓缩生长因子相比富血小板纤维蛋白有更多种类的生长因子和细胞因子[30]。在交替离心力的作用下,各种生长因子和血小板在纤维蛋白网络中嵌入的概率显著增加,这可能有助于生长因子的缓释时间的延长[31]。纤维蛋白支架天然的三维结构本身具有一定的机械支撑作用,同时可与纳米羟基磷灰石等组织工程材料联合形成更为稳定的三维支架结构[32]。这种空间结构为细胞聚集、黏附、分化及增殖提供了适宜的环境,在一定程度上充当了细胞外基质的作用[33]。致密的纤维蛋白网络为浓缩生长因子中的生长因子提供保护,避免被纤溶酶溶解[10]。具备一定孔隙率和孔径的三维纤维蛋白网络支架,避免了各种内源性生长因子爆发性释放,具有一定的缓释作用,更符合生理性释放过程[30]。不同于人工合成的支架材料,天然的纤维蛋白网络自行降解,并且在降解过程中释放出更多的生长因子和血小板,进一步促进损伤组织愈合[34]。 浓缩生长因子在离心过程中因血小板从各个方向相互碰撞破裂,释放出a颗粒,a颗粒中蕴含有大量生长因子,其中主要包括血小板衍生生长因子、转化生长因子β1、血管内皮生长因子、胰岛素样生长因子1、表皮生长因子及骨形成发生蛋白。血小板衍生生长因子作为一种生物趋化因子,也能促进细胞的有丝分裂。 有研究显示,血小板衍生生长因子是启动细胞分化级联反应的关键因子[35-36],能促进口腔黏膜成纤维细胞有丝分裂进而转化成肌成纤维细胞,并促进细胞增殖、趋化和胶原合成[37];骨髓间充质干细胞的趋化,以及向成骨分化过程中,血小板衍生生长因子表达也明显增加[38];因血小板衍生生长因子的趋化作用,在干细胞生物疗法中,可诱导间充质干细胞定向迁移,实现靶向治疗[39]。血小板衍生生长因子促进血管内皮细胞有丝分裂,调控细胞增殖和凋亡,减少血管内皮损伤,可能是动脉粥样硬化治疗的新靶点[40]。转化生长因子β1是属于一组调节细胞生长和分化的转化生长因子β超家族成员,主要作用是促进成纤维细胞的增殖和分化以及调节细胞免疫表型[41-42],目前有研究表明转化生长因子β1还能通过促进间充质细胞的转化加速伤口愈合[43]。血管内皮生长因子一种高度特异性的促血管内皮细胞生长因子,其主要作用是促进血管生长和调节血管通透性。浓缩生长因子中的血管内皮生长因子在感染性骨缺损模型中表达增加,局部血管化作用增强,在骨组织愈合中有关键作用[44]。血管内皮生长因子可联合间充质干细胞促进血管生成和重建,实现早期修复创面[45]。胰岛素样生长因子1是一种多功能细胞增殖调控因子。有研究证实,浓缩生长因子中释放的胰岛素样生长因子1通过胰岛素样生长因子1R/PI3K/AKT信号通路促进耳郭软骨细胞增殖。此外,胰岛素样生长因子1是一种神经营养因子,能保护运动神经元,抑制神经炎症反应,可能对神经损伤的再生有积极作用[46]。表皮生长因子的主要作用是调控上皮细胞增殖和迁移,促进各种表皮组织生长[47]。骨形成发生蛋白能刺激DNA的合成和细胞的复制,促进间充质细胞定向分化为成骨细胞,是骨缺损修复和骨代谢调控的重要因子[48]。在骨组织工程中各种生物材料联合骨形态发生蛋白,通过促进成骨细胞的增殖和分化,能明显增强各种骨缺损模型的修复效果,加速骨组织重建和再生[49-50]。骨形态发生蛋白对软骨的再生也有促进作用,有研究将骨形态发生蛋白用于关节软骨的修复,治疗骨性关节 炎[51],此外骨形态发生蛋白在未成熟软骨发育阶段中有重要调控作用,骨形态发生蛋白2和6在生长板软骨中表达梯度明显增加[52]。"


目前的一些研究致力于比较富血小板血浆、富血小板纤维蛋白、浓缩生长因子中有效生长因子的浓度和释放。LEE等[33]的研究发现浓缩生长因子中血小板衍生生长因子和表皮生长因子的浓度和含量均高于富血小板纤维蛋白,且浓缩生长因子在体外成骨作用大于富血小板纤维蛋白。LEI等[34]的研究中表明,富血小板纤维蛋白和浓缩生长因子 14 d持续稳定释放血小板衍生的生长因子、血管内皮生长因子、骨形态发生蛋白2和转化生长因子β1,生长因子浓度基本相似。MASUKI等[10]的研究发现,与富血小板血浆制剂相比,富血小板纤维蛋白和浓缩生长因子提取液中血小板和血小板衍生生长因子含量均较高,而富血小板纤维蛋白中转化生长因子β1、血小板衍生生长因子BB、血管内皮生长因子浓度大于浓缩生长因子。大多数研究认为富血小板纤维蛋白和浓缩生长因子中内源性生长因子浓度明显高于富血小板血浆,但对于富血小板纤维蛋白和浓缩生长因子中生长因子浓度的差异目前无统一定论。大量内源性生长因子的释放和活化,多信号通路激活,多个作用靶点相互调节,互相协同,是浓缩生长因子促进组织修复和重塑的主要机制,相比单一的某一种生长因子,浓缩生长因子形成综合的生长因子缓释储存库,是其主要优势之一。浓缩生长因子中主要生长因子的生物学作用,见表2。"

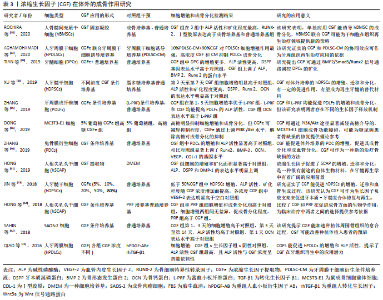
2.5 浓缩生长因子在体外对细胞的作用 由于含有大量促进成骨的活性细胞因子,浓缩生长因子在体外的成骨作用被广泛研究。多项研究评价了浓缩生长因子在体外对细胞的成骨作用。浓缩生长因子体外诱导干细胞成骨的研究6项,包括人牙髓干细胞(hDPSCs)、人骨髓间充质干细胞(hBMSCs)、人牙周膜干细胞(hPDLSCs)及人根尖乳头干细胞(hSCAP);非干细胞研究6项,包括牙髓细胞(DPCs)、牙周膜成纤维细胞(PDLFs)、MC3T3-E1细胞、兔骨膜衍生细胞(PDCs)、SAOS-2成骨样细胞及人牙周膜细胞(hPDLCs)。几乎所有研究都集中于浓缩生长因子体外对细胞的增殖、迁移和成骨分化,成骨分化检测包括碱性磷酸酶活性,茜素红染色评估矿化面积,以及Runx2、骨形态发生蛋白2及BSP等成骨相关基因表达水平,见表3。有3项研究设置了不同的浓缩生长因子提取物浓度[22-23,53],JIN等[23]的研究表明,低于50%浓缩生长因子促进人牙髓干细胞增殖、迁移及成骨分化,高于50%浓度则表现为抑制作用。但AGHAMOHAMADI等[22]的研究却表明:90%浓缩生长因子对牙周膜干细胞增殖仍有显著促进作用,这说明浓缩生长因子对于不同干细胞的增殖分化可能存在不同的最适浓度。有5项研究设置了阳性对照组,以比较浓缩生长因子、LPF、A-LPF、成骨诱导培养基、以及基因修饰后的生长因子对细胞的成骨分化作用。有研究认为富血小板纤维蛋白体外成骨作用强于浓缩生长因子[54],有研究认为富血小板纤维蛋白和浓缩生长因子体外成骨作用无明显差异[55],有研究表明浓缩生长因子体外诱导成骨分化强于单纯成骨诱导培养基[18]。有研究认为浓缩生长因子体外诱导成骨分化强于人重组转化生长因子β1联合人重组血小板衍生生长因子AB[24]。有2项研究涉及浓缩生长因子对PI3K/Akt信号通路和Wnt3a信号通路的影响[24,56]。"
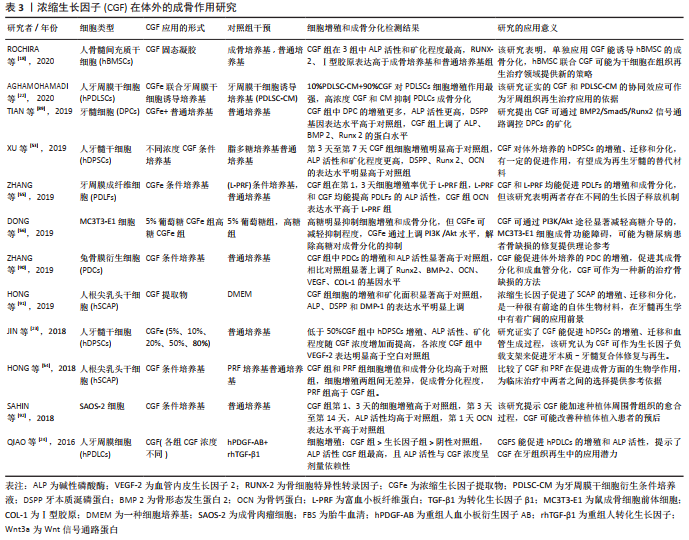
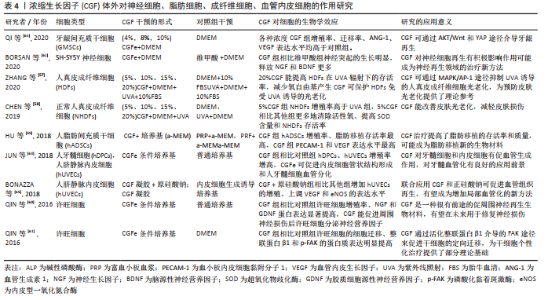
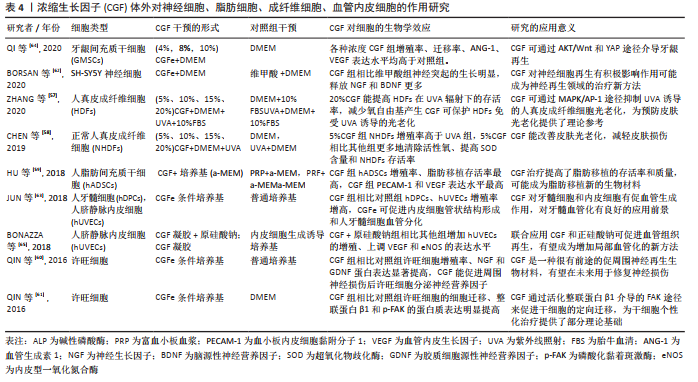
多项研究探讨了浓缩生长因子体外对神经细胞、脂肪细胞、成纤维细胞、血管内皮细胞的影响。其中2项研究表明浓缩生长因子能减轻紫外线照射对人皮肤成纤维细胞的损伤,并减少氧自由基的生成,这可能对皮肤抗老化作用产生积极影响[57-58];此外另一项研究表明浓缩生长因子能提高脂肪移植存活率[59],这表明浓缩生长因子在皮肤美容领域的一定潜力。有3项研究对神经细胞的研究中发现,浓缩生长因子能分泌神经营养因子以及促进神经细胞部分再生,这可能对神经损伤后再修复有积极作用[60-62]。有4项研究表明浓缩生长因子在体外促进了血管内皮生长因子的表 达[59,63-65],这表明浓缩生长因子体外确切的促血管生成作用,局部促血管化作用是组织修复再生过程中的关键之 一,如表4所示。"
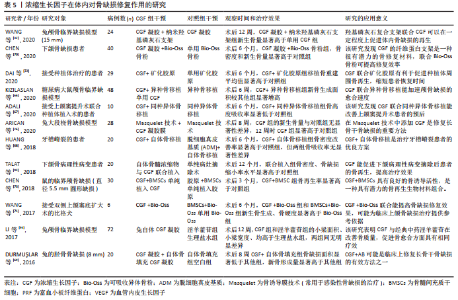
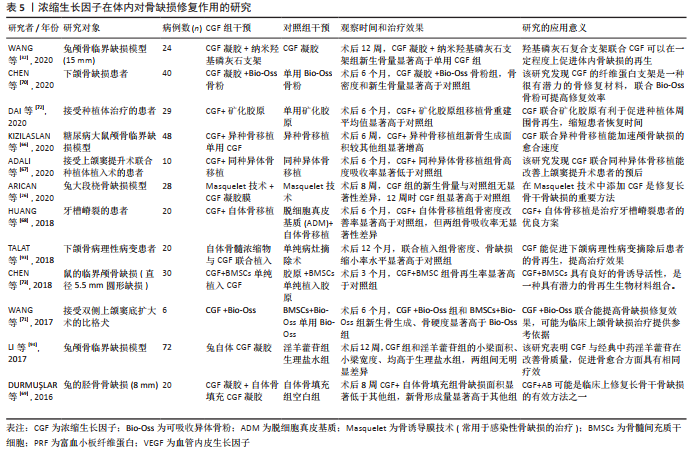
2.6 浓缩生长因子在体内对组织修复和再生的研究 浓缩生长因子在体外的成骨作用已经在大量研究中获得肯定,所以近年有许多研究致力于评估浓缩生长因子在体内对骨缺损修复的作用。在文章的报告中,包括了多项将浓缩生长因子应用于体内骨缺损修复的基础研究和临床研究。骨缺损的类型包括颅骨临界骨缺损、上下颌骨、牙种植体周围骨缺损,以及四肢长骨骨缺损(包括桡骨、股骨和胫骨)。植入的方式多采用浓缩生长因子凝胶或浓缩生长因子凝胶膜。几乎所有研究都肯定了浓缩生长因子在体内对于骨缺损修复的积极作用,仅有1项研究否定浓缩生长因子的效果[17],此研究将浓缩生长因子凝胶植入药物相关性颌骨坏死骨质疏松患者的骨缺损部位,术后6个月的随访结果显示,浓缩生长因子治疗手术组与单纯手术组在骨缺损愈合率上无显著性差异。有4项研究将浓缩生长因子与自体或同种异体骨联合植入体内[66-69],用于修复骨缺损,均表明联合组在骨缺损面积的缩小和骨密度改善方面的显著优势。此外,有4项研究将浓缩生长因子与骨组织工程材料联合用于骨的修复和重建,包括浓缩生长因子联合纳米羟基磷灰石、Bio-Oss骨粉和矿化胶原[32,70-72],这几种联合方式均表现出浓缩生长因子与材料良好的兼容性,以及确切的骨缺损修复效果。 有研究将浓缩生长因子作为支架,搭载骨髓间充质干细胞共同植入[73],在鼠的临界颅骨缺损相比单纯植入浓缩生长因子或胶原,取得更好的骨缺损修复效果,以及观察到这种联合方式可能促进缺损局部血管形成的现象。有研究创新性地将Masquelet膜诱导技术与浓缩生长因子凝胶膜结合,并表明在术后12周时,这种创新性地结合治疗方式比对照组有更多的新骨生成[26]。 浓缩生长因子的体内研究不仅仅局限在骨组织等硬组织的修复作用,由于含有多种内源性生长因子以及多种灵活可变的应用形式,浓缩生长因子也被应用于修复多种软组织缺损。CHEN等[28]的Meta分析中,包括578例患者,8项随机对照试验,表明浓缩生长因子对于修复皮肤慢性溃疡创面的积极效果。ZHAO等[74]的研究中,将自体浓缩生长因子膜植入鼻中隔黏膜缺损部位,术后6个月黏膜完全修复,且无并发症发生。有研究将浓缩生长因子凝胶植入干槽症患者牙槽周围软组织缺损中,在术后第7天浓缩生长因子治疗组的新生肉芽组织数量显著高于单纯手术 组[75]。与促进骨修复不同的是,浓缩生长因子修复软组织的效果存在一些争议。在牙龈退缩的修复2项研究中,一项研究认为浓缩生长因子仅能改善牙龈角化宽度和龈厚,修复效果有限[76];另一项研究则认为结缔组织比浓缩生长因子凝胶更有利于治疗牙龈萎缩[77]。此外,有研究在治疗种植体周围炎的研究中也表明,胶原膜植入在治疗牙龈愈合及骨再生方面优于浓缩生长因子膜[78]。 有2项关于浓缩生长因子和拔牙术后并发症的研究中,一项研究表示浓缩生长因子可降低下颌第三磨牙拔除术后牙槽骨炎发生率[79],另一项研究却认为浓缩生长因子和A-富血小板纤维蛋白对下颌第三磨牙拔除术后患者的疼痛、局部肿胀程度的缓解没有作用[80]。另外2项关于皮肤的研究中表明,浓缩生长因子能够改善皮肤光老化损伤程度以及在治疗面部凹陷瘢痕的良好效果[81-82]。此外,有研究发现浓缩生长因子联合米诺地尔能治疗雄激素性脱发,且效果优于单独米诺地尔治疗[83]。还有研究将浓缩生长因子与Masquelet技术结合[27],发现浓缩生长因子治疗组形成的诱导膜厚度、炎症程度、膜血管化程度、膜增殖率、干细胞比例均优于对照组,可见浓缩生长因子可能为优化Masquelet技术提供了一种参考,如表5所示。"
| [1] KANG YH, JEON SH, PARK JY, et al. Platelet-rich fibrin is a Bioscaffold and reservoir of growth factors for tissue regeneration. Tissue Eng Part A. 2011; 17(3-4):349-359. [2] SAMPSON S, REED M, SILVERS H, et al. Injection of platelet-rich plasma in patients with primary and secondary knee osteoarthritis: a pilot study. Am J Phys Med Rehabil. 2010;89(12):961-969. [3] NAGAVENI NB, PRAVEEN RB, UMASHANKAR KV, et al. Efficacy of platelet-rich-plasma (PRP) in bone regeneration after cyst enucleation in pediatric patients--a clinical study. J Clin Pediatr Dent. 2010;35(1):81-87. [4] DOHAN DM, CHOUKROUN J, DISS A, et al. Platelet-rich fibrin (PRF): a second-generation platelet concentrate. Part I: technological concepts and evolution. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2006; 101(3):e37-e44. [5] WANG Z, HAN L, SUN T, et al. Preparation and effect of lyophilized platelet-rich fibrin on the osteogenic potential of bone marrow mesenchymal stem cells in vitro and in vivo. Heliyon. 2019;5(10):e02739. [6] NOLAN GS, SMITH OJ, MOSAHEBI A. Enhancing fat graft survival with autologous growth factors: platelet-rich fibrin (PRF) vs platelet-rich plasma (PRP). Aesthet Surg J. 2021;41(5):NP241. [7] RIZK HM, SALAH AL-DEEN MSM, EMAM AA. Comparative evaluation of Platelet Rich Plasma (PRP) versus Platelet Rich Fibrin (PRF) scaffolds in regenerative endodontic treatment of immature necrotic permanent maxillary central incisors: a double blinded randomized controlled trial. Saudi Dent J. 2020;(5):224-231. [8] FEIGIN K, SHOPE B. Use of platelet-rich plasma and platelet-rich fibrin in dentistry and oral surgery: introduction and review of the literature. J Vet Dent. 2019;36(2):109-123. [9] DENTAL SUPPLEMENT, TESTORI T, PANDA S, et al. Short implants and platelet-rich fibrin for transcrestal sinus floor elevation: a prospective multicenter clinical study. J Biol Regul Homeost Agents. 2019;33(6 Suppl 2):121-135. [10] MASUKI H, OKUDERA T, WATANEBE T, et al. Growth factor and pro-inflammatory cytokine contents in platelet-rich plasma (PRP), plasma rich in growth factors (PRGF), advanced platelet-rich fibrin (A-PRF), and concentrated growth factors (CGF). Int J Implant Dent. 2016;2(1):19-27. [11] AIZAWA H, TSUJINO T, WATANABE T, et al. Quantitative near-infrared imaging of platelets in platelet-rich fibrin (PRF) matrices: comparative analysis of Bio-PRF, leukocyte-rich PRF, advanced-PRF and concentrated growth factors. Int J Mol Sci. 2020;21(12):4426-4433. [12] YAMAGUCHI S, AIZAWA H, SATO A, et al. Concentrated growth factor matrices prepared using silica-coated plastic tubes are distinguishable from those prepared using glass tubes in platelet distribution: application of a novel near-infrared imaging-based, quantitative technique. Front Bioeng Biotechnol. 2020;8:600-609. [13] 李佳,何东宁.CGF在种植中促进成骨的研究探讨及应用进展[J].口腔颌面修复学杂志,2021,22(1):76-80. [14] 宁佳,张玮,柳洪志.浓缩生长因子在骨组织再生和修复上的作用研究进展[J].中国美容医学,2012,21(15):2073-2075. [15] 王瑾,张月,刘克达,等.应用浓缩生长因子促进干细胞增殖及成骨分化修复口腔颌面部缺损的研究进展[J].中国实用口腔科杂志,2020,13(1): 47-50, 54. [16] RODELLA LF, FAVERO G, BONINSEGNA R, et al. Growth factors, CD34 positive cells, and fibrin network analysis in concentrated growth factors fraction. Microsc Res Tech. 2011;74(8):772-783. [17] YÜCE MO, ADALı E, IŞıK G. The effect of concentrated growth factor (CGF) in the surgical treatment of medication-related osteonecrosis of the jaw (MRONJ) in osteoporosis patients: a randomized controlled study. Clin Oral Investig. 2021. doi: 10.1007/s00784-020-03766-8. [18] ROCHIRA A, SICULELLA L, DAMIANO F, et al. Concentrated Growth Factors (CGF) Induce osteogenic differentiation in human bone marrow stem cells. Biology (Basel). 2020;9(11):370-381. [19] WANG L, WAN M, LI Z, et al. A comparative study of the effects of concentrated growth factors in two different forms on osteogenesis in vitro. Mol Med Rep. 2019;20(2):1039-1048. [20] HONDA H, TAMAI N, NAKA N,et al. Bone tissue engineering with bone marrow-derived stromal cells integrated with concentrated growth factor in Rattus norvegicus calvaria defect model. J Artif Organs. 2013;16(3):305-315. [21] CHEN X, CHEN Y, HOU Y, et al. Modulation of proliferation and differentiation of gingiva‑derived mesenchymal stem cells by concentrated growth factors: potential implications in tissue engineering for dental regeneration and repair. Int J Mol Med. 2019;44(1):37-46. [22] AGHAMOHAMADI Z, KADKHODAZADEH M, TORSHABI M, et al. A compound of concentrated growth factor and periodontal ligament stem cell-derived conditioned medium. Tissue Cell. 2020;65:101373. [23] JIN R, SONG G, CHAI J, et al. Effects of concentrated growth factor on proliferation, migration, and differentiation of human dental pulp stem cells in vitro. J Tissue Eng. 2018;21;9:2041731418817505. [24] QIAO J, AN N. Effect of concentrated growth factors on function and Wnt3a expression of human periodontal ligament cells in vitro. Platelets. 2017;28(3):281-286. [25] TONE T, SHIMIZU Y, SAITO H, et al. In vivo behavior of untreated and compressed concentrated growth factors as biomaterials in rabbits. Dent Mater J. 2021;40(1):8-15. [26] ARıCAN G, ÖZMERIÇ A, FIRAT A, et al. Micro-ct findings of concentrated growth factors (cgf) on bone healing in masquelet’s technique-an experimental study in rabbits. Arch Orthop Trauma Surg. 2020. doi: 10.1007/s00402-020-03596-z. [27] YıLMAZ O, ÖZMERIÇ A, ALEMDAROĞLU KB, et al. Effects of concentrated growth factors (CGF) on the quality of the induced membrane in Masquelet’s technique - An experimental study in rabbits. Injury. 2018;49(8): 1497-1503. [28] CHEN J, WAN Y, LIN Y, et al. Platelet-rich fibrin and concentrated growth factors as novel platelet concentrates for chronic hard-to-heal skin ulcers: a systematic review and Meta-analysis of randomized controlled trials. J Dermatolog Treat. 2020;1:1-9. [29] ISOBE K, WATANEBE T, KAWABATA H, et al. Mechanical and degradation properties of advanced platelet-rich fibrin (A-PRF), concentrated growth factors (CGF), and platelet-poor plasma-derived fibrin (PPTF). Int J Implant Dent. 2017;3(1):17-26. [30] YU M, WANG X, LIU Y, et al. Cytokine Release Kinetics of Concentrated Growth Factors in Different Scaffolds. Clin Oral Invest. 2019;23(4):1663-1671. [31] HE L, LIN Y, HU X, et al. A comparative study of platelet-rich fibrin (PRF) and platelet-rich plasma (PRP) on the effect of proliferation and differentiation of rat osteoblasts in vitro. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2009;108(5):707-713. [32] WANG X, TONG S, HUANG S, et al. Application of a new type of natural calcined bone repair material combined with concentrated growth factors in bone regeneration in rabbit critical-sized calvarial defect. Biomed Res Int. 2020;2020:8810747. [33] LEE HM, SHEN EC, SHEN JT, et al. Tensile strength, growth factor content and proliferation activities for two platelet concentrates of platelet-rich fibrin and concentrated growth factor. J Dent Sci. 2020;15(2):141-146. [34] LEI L, YU Y, HAN J, et al. Quantification of growth factors in advanced platelet-rich fibrin and concentrated growth factors and their clinical efficacy as adjunctive to the GTR procedure in periodontal intrabony defects. J Periodontol. 2020;91(4):462-472. [35] ROCKER AJ, LEE DJ, SHANDAS R, et al. Injectable polymeric delivery system for spatiotemporal and sequential release of therapeutic proteins to promote therapeutic angiogenesis and reduce inflammation. ACS Biomater Sci Eng. 2020;6(2):1217-1227. [36] LI Y, LI X, YANG F, et al. Target-induced activation of polymerase activity for recycling signal amplification cascades for sensitive aptamer-based detection of biomarkers. Analyst. 2021;146(5):1590-1595. [37] WANG J, YOU J, GONG D, et al. PDGF-BB induces conversion, proliferation, migration, and collagen synthesis of oral mucosal fibroblasts through PDGFR-β/PI3K/ AKT signaling pathway. Cancer Biomark. 2021;30(4):407-415. [38] ZHANG N, LO CW, UTSUNOMIYA T, et al. PDGF-BB and IL-4 co-overexpression is a potential strategy to enhance mesenchymal stem cell-based bone regeneration. Stem Cell Res Ther. 2021;12(1):40-47. [39] OH J, SON YS, KIM WH, et al. Mesenchymal stem cells genetically engineered to express platelet-derived growth factor and heme oxygenase-1 ameliorate osteoarthritis in a canine model. J Orthop Surg Res. 2021;16(1):43-51. [40] JIAO Y, ZHAO D, GAO F, et al. MicroRNA-520c-3p suppresses vascular endothelium dysfunction by targeting RELA and regulating the AKT and NF-κB signaling pathways. J Physiol Biochem. 2021;77(1):47-61. [41] MISHIMA Y, OKA A, ISHIHARA S. Detection and quantification of transforming growth factor-β1 produced by murine B cells: pros and cons of different techniques. Methods Mol Biol. 2021;2270:113-124. [42] MORA-NAVARRO C, BADILEANU A, GRACIOSO MARTINS AM, et al. Porcine vocal fold lamina propria-derived biomaterials modulate tgf-β1-mediated fibroblast activation in vitro. ACS Biomater Sci Eng. 2020;6(3):1690-1703. [43] EL-BAZ LMF, SHOUKRY NM, HAFEZ HS, et al. Fibroblast growth factor 2 augments transforming growth factor beta 1 induced epithelial-mesenchymal transition in lung cell culture model. Iran J Allergy Asthma Immunol. 2020;19(4):348-361. [44] WEI P, JING W, YUAN Z, et al. Vancomycin- and strontium-loaded microspheres with multifunctional activities against bacteria, in angiogenesis, and in osteogenesis for enhancing infected bone regeneration. ACS Appl Mater Interfaces. 2019;11(34):30596-30609. [45] SHUKLA A, CHOUDHURY S, CHAUDHARY G, et al. Chitosan and gelatin biopolymer supplemented with mesenchymal stem cells (Velgraft®) enhanced wound healing in goats (Capra hircus): involvement of VEGF, TGF and CD31. J Tissue Viability. 2021;30(1):59-66. [46] ZHAO L, ZHANG B, HUANG S, et al. Insulin-like growth factor-1 enhances motoneuron survival and inhibits neuroinflammation after spinal cord transection in Zebrafish. Cell Mol Neurobiol. 2021. doi: 10.1007/s10571-020-01022-x. [47] ZENG F, HARRIS RC. Epidermal growth factor, from gene organization to bedside. Semin Cell Dev Biol. 2014;28:2-11. [48] BABOOTA RK, BLÜHER M, SMITH U. Emerging role of bone morphogenetic protein 4 in metabolic disorders. Diabetes. 2021;70(2):303-312. [49] CARREIRA AC, ZAMBUZZI WF, ROSSI MC, et al. Bone Morphogenetic Proteins: Promising Molecules for Bone Healing, Bioengineering, and Regenerative Medicine. Vitam Horm. 2015;99:293-322. [50] MUZIO G, MARTINASSO G, BAINO F, et al. Key role of the expression of bone morphogenetic proteins in increasing the osteogenic activity of osteoblast-like cells exposed to shock waves and seeded on bioactive glass-ceramic scaffolds for bone tissue engineering. J Biomater Appl. 2014;29(5):728-736. [51] IIJIMA H, ITO A, NAGAI M, et al. Physiological exercise loading suppresses post-traumatic osteoarthritis progression via an increase in bone morphogenetic proteins expression in an experimental rat knee model. Osteoarthritis Cartilage. 2017;25(6):964-975. [52] GARRISON P, YUE S, HANSON J, et al. Spatial regulation of bone morphogenetic proteins (BMPs) in postnatal articular and growth plate cartilage. PLoS One. 2017;12(5):e0176752. [53] XU F, QIAO L, ZHAO Y, et al. The potential application of concentrated growth factor in pulp regeneration: an in vitro and in vivo study. Stem Cell Res Ther. 2019;10(1):134-142. [54] HONG S, CHEN W, JIANG B. A Comparative evaluation of concentrated growth factor and platelet-rich fibrin on the proliferation, migration, and differentiation of human stem cells of the apical papilla. J Endod. 2018; 44(6):977-983. [55] ZHANG Z, LI X, ZHAO J, et al. Effect of autogenous growth factors released from platelet concentrates on the osteogenic differentiation of periodontal ligament fibroblasts: a comparative study. PeerJ. 2019;7:e7984. [56] DONG K, HAO P, ZHOU W, et al. Concentrate growth factors regulate osteogenic dysfunction of MC3T3-E1 cells induced by high glucose through PI3K/Akt signaling pathway. Implant Dent. 2019;28(5):478-483. [57] ZHANG M, ZHANG T, TANG Y, et al. Concentrated growth factor inhibits UVA-induced photoaging in human dermal fibroblasts via the MAPK/AP-1 pathway. Biosci Rep. 2020;40(7):BSR20193566. [58] CHEN J, JIAO D, ZHANG M, et al. Concentrated growth factors can inhibit photoaging damage induced by ultraviolet A (UVA) on the human dermal fibroblasts in vitro. Med Sci Monit. 2019;25:3739-3749. [59] HU Y, JIANG Y, WANG M, et al. Concentrated growth factor enhanced fat graft survival: a comparative study. Dermatol Surg. 2018;44(7):976-984. [60] QIN J, WANG L, SUN Y, et al. Concentrated growth factor increases Schwann cell proliferation and neurotrophic factor secretion and promotes functional nerve recovery in vivo. Int J Mol Med. 2016;37(2):493-500. [61] QIN J, WANG L, ZHENG L, et al. Concentrated growth factor promotes Schwann cell migration partly through the integrin β1-mediated activation of the focal adhesion kinase pathway. Int J Mol Med. 2016;37(5):1363-1370. [62] BORSANI E, BUFFOLI B, BONAZZA V, et al. In vitro effects of concentrated growth factors (CGF) on human SH-SY5Y neuronal cells. Eur Rev Med Pharmacol Sci. 2020;24(1):304-314. [63] JUN H, LEI D, QIFANG Y, et al. Effects of concentrated growth factors on the angiogenic properties of dental pulp cells and endothelial cells: an in vitro study. Braz Oral Res. 2018;32:e48. [64] QI L, LIU L, HU Y, et al. Concentrated growth factor promotes gingival regeneration through the AKT/Wnt/β-catenin and YAP signaling pathways. Artif Cells Nanomed Biotechnol. 2020;48(1):920-932. [65] BONAZZA V, BORSANI E, BUFFOLI B, et al. In vitro treatment with concentrated growth factors (CGF) and sodium orthosilicate positively affects cell renewal in three different human cell lines. Cell Biol Int. 2018; 42(3):353-364. [66] KIZILASLAN S, KARABUDA ZC, OLGAC V. The effect of concentrated growth factor on calvarial bone in diabetic healing. J Craniofac Surg. 2020;31(1): 158-161. [67] ADALı E, YÜCE MO, GÜNBAY T, et al. Does concentrated growth factor used with allografts in maxillary sinus lifting have adjunctive benefits? J Oral Maxillofac Surg. 2021;79(1):98-108. [68] HUANG L, ZOU R, HE J, et al.Comparing osteogenic effects between concentrated growth factors and the acellular dermal matrix. Braz Oral Res. 2018;32:e29. [69] DURMUŞLAR MC, BALLI U, DEDE FÖ, et al. Histological evaluation of the effect of concentrated growth factor on bone healing. J Craniofac Surg. 2016;27(6):1494-1507. [70] CHEN J, JIANG H. Clinical application of concentrated growth factor fibrin combined with bone repair materials in jaw defects. J Oral Maxillofac Surg. 2020;78(7):1041-1049. [71] WANG F, LI Q, WANG Z. A comparative study of the effect of Bio-Oss® in combination with concentrated growth factors or bone marrow-derived mesenchymal stem cells in canine sinus grafting. J Oral Pathol Med. 2017; 46(7):528-536. [72] DAI Y, HAN XH, HU LH, et al. Efficacy of concentrated growth factors combined with mineralized collagen on quality of life and bone reconstruction of guided bone regeneration. Regen Biomater. 2020;7(3): 313-320. [73] CHEN X, WANG J, YU L, et al. Effect of concentrated growth factor (CGF) on the promotion of osteogenesis in bone marrow stromal cells (BMSC) in vivo. Sci Rep. 2018;8(1):5876-5887. [74] ZHAO QM, GAO J, HUANG XX, et al. Concentrated growth factors extracted from blood plasma used to repair nasal septal mucosal defect after rhinoplasty. Aesthetic Plast Surg. 2020;44(2):511-516. [75] KAMAL A, SALMAN B, ABDUL RAZAK NH, et al. The efficacy of concentrated growth factor in the healing of alveolar osteitis: a clinical study. Int J Dent. 2020;2020:9038629. [76] BOZKURT DOĞAN Ş, ÖNGÖZ DEDE F, BALLı U, et al. Concentrated growth factor in the treatment of adjacent multiple gingival recessions: a split-mouth randomized clinical trial. J Clin Periodontol. 2015;42(9):868-875. [77] AKCAN SK, ÜNSAL B. Gingival recession treatment with concentrated growth factor membrane: a comparative clinical trial. J Appl Oral Sci. 2020; 28:e20190236. [78] ISLER SC, SOYSAL F, CEYHANLı T, et al. Regenerative surgical treatment of peri-implantitis using either a collagen membrane or concentrated growth factor: a 12-month randomized clinical trial. Clin Implant Dent Relat Res. 2018;20(5):703-712. [79] ÖZVERI KOYUNCU B, IŞıK G, ÖZDEN YÜCE M, et al. Effect of concentrated growth factors on frequency of alveolar Osteitis following partially-erupted mandibular third molar surgery: a randomized controlled clinical study. BMC Oral Health. 2020;20(1):222-231. [80] TORUL D, OMEZLI MM, KAHVECI K. Evaluation of the effects of concentrated growth factors or advanced platelet rich-fibrin on postoperative pain, edema, and trismus following lower third molar removal: a randomized controlled clinical trial. J Stomatol Oral Maxillofac Surg. 2020;121(6):646-651. [81] ZHOU R, WANG M, ZHANG X, et al. Therapeutic effect of concentrated growth factor preparation on skin photoaging in a mouse model. J Int Med Res. 2020;48(10):300060520962946. [82] 孙佳琳,王军杰,崔正军,等.浓缩生长因子联合血浆蛋白凝胶治疗面部凹陷瘢痕的临床效果[J].中华烧伤杂志,2020,36(3):210-218. [83] TAN PC, ZHANG PQ, XIE Y, et al. Autologous concentrated growth factors combined with topical minoxidil for the treatment of male androgenetic alopecia: a randomized controlled clinical trial. Facial Plast Surg Aesthet Med. 2020. doi:10.1089/fpsam.2020.0288. [84] BONAZZA V, BORSANI E, BUFFOLI B, et al. How the different material and shape of the blood collection tube influences the Concentrated Growth Factors production. Microsc Res Tech. 2016;79(12):1173-1178. [85] TAKAGI N, KAWAKAMI K, KANNO E, et al. IL-17A promotes neutrophilic inflammation and disturbs acute wound healing in skin. Exp Dermatol. 2017;26(2):137-144. [86] FU J, HUANG J, LIN M, et al. Quercetin promotes diabetic wound healing via switching macrophages from M1 to M2 polarization. J Surg Res. 2020;246:213-223. [87] BONAZZA V, HAJISTILLY C, PATEL D, et al. Growth factors release from concentrated growth factors: effect of β-tricalcium phosphate addition. J Craniofac Surg. 2018;29(8):2291-2295. [88] ANTUNES JC, OLIVEIRA JM, REIS RL, et al. Novel poly(L-lactic acid)/hyaluronic acid macroporous hybrid scaffolds: characterization and assessment of cytotoxicity. J Biomed Mater Res A. 2010;94(3):856-869. [89] TIAN S, WANG J, DONG F, et al. Concentrated growth factor promotes dental pulp cells proliferation and mineralization and facilitates recovery of dental pulp tissue. Med Sci Monit. 2019;25:10016-10028. [90] ZHANG L, AI H. Concentrated growth factor promotes proliferation, osteogenic differentiation, and angiogenic potential of rabbit periosteum-derived cells in vitro. J Orthop Surg Res. 2019;14(1):146-157. [91] HONG S, LI L, CAI W, et al. The potential application of concentrated growth factor in regenerative endodontics. Int Endod J. 2019;52(5):646-655. [92] SAHIN IO, GOKMENOGLU C, KARA C. Effect of concentrated growth factor on osteoblast cell response. J Stomatol Oral Maxillofac Surg. 2018;119(6):477-481. [93] TALAAT WM, GHONEIM MM, SALAH O, et al. Autologous bone marrow concentrates and concentrated growth factors accelerate bone regeneration after enucleation of mandibular pathologic lesions. J Craniofac Surg. 2018; 29(4):992-997. [94] LI G, WANG J, REN G, et al. Morphological observation on critical-sized cranial defect repaired by icariin and autologous concentrate growth factors in rabbits. Med Sci Monit. 2017;23:2373-2381. [95] PARK HC, KIM SG, OH JS, et al. Early bone formation at a femur defect using cgf and prf grafts in adult dogs: a comparative study. Implant Dent. 2016;25(3):387-393. [96] TAKEDA Y, KATSUTOSHI K, MATSUZAKA K, et al. The effect of concentrated growth factor on rat bone marrow cells in vitro and on calvarial bone healing in vivo. Int J Oral Maxillofac Implants. 2015;30(5):1187-1196. |
| [1] | Xue Yadong, Zhou Xinshe, Pei Lijia, Meng Fanyu, Li Jian, Wang Jinzi . Reconstruction of Paprosky III type acetabular defect by autogenous iliac bone block combined with titanium plate: providing a strong initial fixation for the prosthesis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1424-1428. |
| [2] | Wu Weiyue, Guo Xiaodong, Bao Chongyun. Application of engineered exosomes in bone repair and regeneration [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1102-1106. |
| [3] | Zhang Jinglin, Leng Min, Zhu Boheng, Wang Hong. Mechanism and application of stem cell-derived exosomes in promoting diabetic wound healing [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1113-1118. |
| [4] | Yang Sidi, Wang Qian, Xu Nuo, Wang Ronghan, Jin Chuanqi, Lu Ying, Dong Ming. Biodentine enhances the proliferation and differentiation of osteoblasts through upregulating bone morphogenetic protein-2 [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 516-520. |
| [5] | Tan Guozhong, Tu Xinran, Guo Liyang, Zhong Jialin, Zhang Yang, Jiang Qianzhou. Biosafety evaluation of three-dimensional printed gelatin/sodium alginate/58S bioactive glass scaffolds for bone defect repair [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 521-527. |
| [6] | Zhou Liang, Chen Xingzhen, Li Zhenyu, Zhang Zekun, Duan Guoqing. The mechanism of lncRNA HOTAIR in interleukin-1beta-mediated osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(35): 5607-5613. |
| [7] | Zhang Jingxin, Liu Linfeng, Zhang Shiwen, Lin Jie. Molecular mechanism of magnesium ion promoting bone regeneration [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(33): 5384-5392. |
| [8] | Tang Kai, Zhao Wenhua, Shang Qi, Shen Gengyang, Zhang Zhida, He Jiahui, Zhang Peng, Yu Fuyong, Chen Guifeng, Ren Hui, Jiang Xiaobing, Yu Xiang. Effects of concentrated growth factors on the proliferation, osteogenic differentiation and migration of rat bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(31): 4935-4939. |
| [9] | Huang Gao, Xu Jun, Chen Wenge. Implantation of bone marrow mesenchymal stem cells-loaded platelet-rich plasma combined with extracorporeal shock wave in the repair of bone defects [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(30): 4812-4818. |
| [10] | Jia Qiyu, Huang Xiaoxia, Guo Jian, Huang Jinyong, Guo Xiaobin, Abdussalam·Alimujiang, Wu Tong, Ma Chuang. Integrin-targeted peptide promotes proliferation of bone marrow mesenchymal stem cells in SD rats [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(30): 4780-4786. |
| [11] | Zhao Dun, Fang Bin, Yi Chunzhi, He Mincong, Zheng Jiaqian, Li Yue. Effects of total flavonoids of Rhizoma drynariae on bone remodeling and expression of bone morphogenetic protein-2, vascular endothelial growth factor and CD31 [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(29): 4638-4642. |
| [12] | Lu Renpei, Zou Zhichen, Zhao Fengnian, Zheng Changming, Song Keguan. Role and application of polylactic acid-hydroxyacetic acid copolymer scaffolds in bone defect repair and regeneration [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(28): 4525-4531. |
| [13] | Fan Yi, Liu Yadong, Cui Yutao, Liu He, Tian Yuhang, Li Shaorong, Wang Gan, Wu Dankai, Peng Chuangang. Modification of natural and composite alginate hydrogels and repair of bone defect with composite systems [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(28): 4532-4538. |
| [14] | Ci Wentao, Zhang Xinlong, Yan Shi, Wang Zhao. Reducing the recurrence of infection after the application of Masquelet technique for osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(28): 4546-4552. |
| [15] | Lu Yunan, Zhang Xinzhao, Lin Binbin, Xu Gan, Chen Jingdi, Chen Shunyou. Naringin-chitosan/hydroxyapatite composite scaffold in repair of rat skull defect [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(28): 4441-4445. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||