Chinese Journal of Tissue Engineering Research ›› 2022, Vol. 26 ›› Issue (33): 5384-5392.doi: 10.12307/2022.779
Previous Articles Next Articles
Molecular mechanism of magnesium ion promoting bone regeneration
Zhang Jingxin1, Liu Linfeng1, Zhang Shiwen2, Lin Jie2
- 1State Key Laboratory of Oral Diseases/National Clinical Research Center for Oral Diseases/West China Hospital of Stomatology, Sichuan University, Chengdu 610041, Sichuan Province, China; 2State Key Laboratory of Oral Diseases/National Clinical Research Center for Oral Diseases/Department of Oral Implantology, West China Hospital of Stomatology, Sichuan University, Chengdu 610041, Sichuan Province, China
-
Received:2021-12-04Accepted:2021-12-31Online:2022-11-28Published:2022-03-31 -
Contact:Lin Jie, Master, Associate chief nurse, State Key Laboratory of Oral Diseases/National Clinical Research Center for Oral Diseases/Department of Oral Implantology, West China Hospital of Stomatology, Sichuan University, Chengdu 610041, Sichuan Province, China -
About author:Zhang Jingxin, State Key Laboratory of Oral Diseases/National Clinical Research Center for Oral Diseases/West China Hospital of Stomatology, Sichuan University, Chengdu 610041, Sichuan Province, China -
Supported by:Horizontal Project of Sichuan University, No. 21H0441 (to LJ)
CLC Number:
Cite this article
Zhang Jingxin, Liu Linfeng, Zhang Shiwen, Lin Jie. Molecular mechanism of magnesium ion promoting bone regeneration[J]. Chinese Journal of Tissue Engineering Research, 2022, 26(33): 5384-5392.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
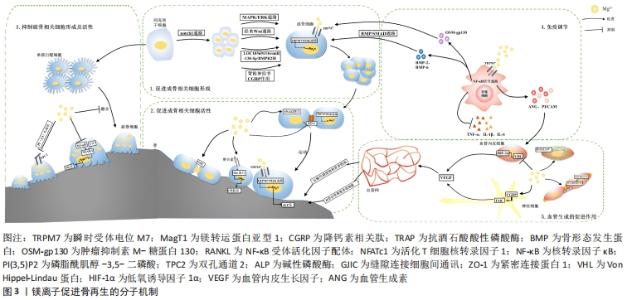
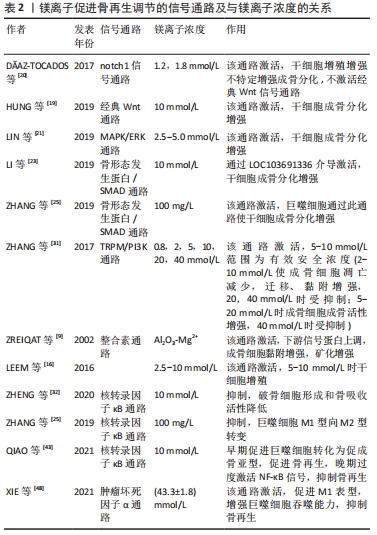
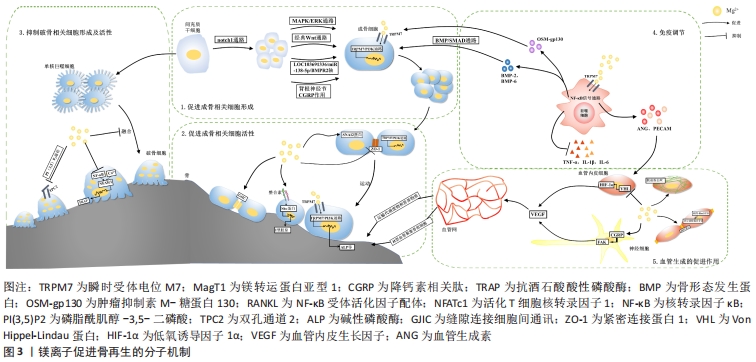
2.1 镁离子对骨相关细胞的影响 2.1.1 间充质干细胞 镁是细胞代谢活动的必要元素,如镁离子与ATP螯合成Mg-ATP,改变酶的结构,参与能量代谢和糖、脂肪等物质的合成与代谢,因此镁离子对代谢循环至关重要[3,10]。间充质干细胞存在于骨髓中,具有强大的增殖能力和分化能力,在体内受多种调控因素,分化形成骨祖细胞,进而形成成骨细胞。 瞬时受体电位M7(transient receptor potential melastatin 7,TRPM7)和镁转运蛋白亚型1(magnesium transporter subtype 1,MagT1)是骨髓间充质干细胞上转运镁离子的主要跨膜离子通道,能感受细胞外镁离子变化,并促使镁离子内流,参与骨再生进程。大量研究表明镁离子对骨髓间充质干细胞的作用离不开TRPM7和MagT1的作用[11-13],镁离子进入细胞后,对增殖、迁移、黏附、成骨分化等多个细胞活动过程起到调节作用。首先镁离子可促进骨髓间充质干细胞增殖[14-16]。LEEM等[16]研究表明镁离子是通过增强整合素α2和α3的表达来促进骨髓间充质干细胞增殖的。同时镁离子能够显著促进骨髓干细胞的迁移与黏附[14-15,17-18],细胞迁移对镁离子的浓度有很强的依赖性,较高的镁离子浓度可更显著促进细胞迁移[15],但 镁离子促进骨髓间充质干细胞迁移与黏附的作用暂未有明确分子机制研究。 骨髓间充质干细胞的成骨分化是镁离子调节骨再生的主要途径,镁离子可通过直接激活骨髓间充质干细胞的成骨信号通路,上调成骨相关基因,促进骨髓间充质干细胞成骨分化,同时镁离子可作用于微环境中的其他细胞,如免疫细胞、神经节细胞等,分泌细胞因子间接作用于干细胞促进成骨分化。MAPK/ERK信号通路和经典Wnt信号通路是调控间充质干细胞成骨分化的重要信号通路,其激活可能与浓度密切相关。HUNG等[19]首次阐明镁离子激活经典Wnt信号通路从而促进骨髓间充质干细胞分化,发现10 mmol/L 镁离子使骨髓间充质干细胞内活性β-连环蛋白的蛋白表达显著增加,其下游靶基因LEF1和Dkk1激活,诱导其成骨分化。而D?-AZ-TOCADOS等[20]发现MgCl2未激活经典Wnt通路,而是通过激活Notch1信号促进其增殖以此促进成骨分化,而不是直接促进骨髓间充质干细胞的成骨分化。这可能与其实验选用的镁离子浓度不同,低于HUNG等实验选用的镁离子浓度,这提示镁离子对骨髓间充质干细胞分化的作用机制可能有浓度依赖性,需进一步探究不同镁离子浓度下促进骨髓间充质干细胞成骨分化的主要分子机制。LIN等[21]发现(2.5-5.0)×10-3 mol/L 是镁离子成骨诱导的最佳浓度,通过激活MAPK/ERK信号通路,显著增强了骨髓间充质干细胞的增殖和成骨分化。 除了以上信号通路激活的成骨分化,近年来,编码RNA,例如长链非编码RNA(lncRNA)和微小RNA(miRNA)在包括成骨分化在内的生物过程中的作用已得到广泛认可[22],LI等[23]用镁离子处理骨髓间充质干细胞后,细胞内lncRNA LOC103691336上调,竞争性与miR-138-5p结合,以减弱其对下游BMPR2的抑制作用(BMPR2是骨形态发生蛋白的激酶受体),其后证明了LOC103691336/miR-138-5p/BMPR2轴通过SMADs和p38信号参与镁离子介导的成骨分化。这是首次研究镁离子对IncRNA的作用促进成骨分化,为镁离子促进骨再生的分子机制研究提供新思路。 另一方面,镁离子通过间充质干细胞与微环境中其他细胞的相互作用,促进间充质干细胞的成骨分化。ZHANG等[24]首次发现神经细胞及其分泌的降钙素相关肽对干细胞成骨分化的作用,镁离子通过背根神经节使骨膜干细胞成骨分化能力增强,使背根神经节分泌降钙素相关肽,降钙素相关肽作用于骨膜干细胞上受体使成骨分化基因表达上调,成骨分化增强。巨噬细胞作为骨免疫微环境中重要的分泌细胞,分泌多种细胞因子促进成骨分化,ZHANG等[25]研究发现镁离子通过巨噬细胞激活骨形态发生蛋白/SMAD信号通路促进成骨分化,QIAN等[26]证实镁离子通过此通路促进成骨分化,同时发现巨噬细胞可分泌肿瘤抑制素M(oncostatin,OSM)-糖蛋白130(glycoprotein130,gp130)信号复合物促进成骨分化。 综上所述,镁离子增强骨髓间充质干细胞增殖、迁移与黏附及成骨分化能力,尤其是成骨分化方面目前分子机制研究较为丰富,但不同镁离子浓度激活的信号通路可能不同,同时目前机制研究主要集中在对骨髓间充质干细胞的直接作用,旁路机制研究较少。 2.1.2 成骨细胞 在经历了骨髓间充质干细胞成骨分化后,骨再生的最终形成还需要经历成骨细胞增殖、细胞凋亡、细胞迁移与黏附、细胞外基质成熟及细胞外基质矿化等阶段。镁离子作为重要的调控因子,可调节细胞各阶段,促进成骨细胞形成。 大量研究表明镁离子通过MagT1和TRPM7离子通道进入成骨细胞促进成骨细胞增殖[27-30]。同时镁离子能减少成骨细胞凋亡,镁及其合金植入材料在体内与体液反应产生OH-,从而造成碱性环境。ZHANG等[31]研究表明镁离子激活成骨细胞内TRPM7/PI3K信号通路,促进细胞增殖,同时在碱性环境下提高镁离子浓度,增强了成骨细胞对碱性环境的抵抗能力,减少了细胞凋亡。ZHENG等[32]证实在镁离子的影响下,成骨细胞对脂多糖等多种细胞毒素的抵抗能力增强,减少细胞凋亡,增加细胞存活率。因此,镁离子调节了成骨细胞的增殖和凋亡,最终使成骨细胞数量增加,为成骨细胞发挥生理功能,促进骨再生打下了良好的基础。 成骨细胞的运动和黏附能力对骨骼生理至关重要,只有成骨细胞到达骨沉积位置并且稳固附着才能实现骨修复。镁离子可促进成骨细胞的细胞骨架排列和形态发生变化,细胞更加细长,片状足突出,细胞表面积增大,均有利于细胞黏附[33]。同时整合素作为成骨细胞上的跨膜蛋白,介导成骨细胞黏附,镁离子可提高其亚型表达,促进细胞黏附[9,33],整合素还可传导信号,激活关键信号蛋白Shc、黏着斑激酶和细胞外基质蛋白Ⅰ型胶原的表达,促进细胞外基质成熟[9]。TRPM7/PI3K信号通路也在镁离子增强成骨细胞运动和黏附能力方面发挥重要作用,使成骨细胞运动和黏附相关基因上调[31],然而CHOI等[34]则发现镁离子并未影响运动相关转录基因表达,而是在蛋白表达水平上上调SNAI2蛋白的表达,促进细胞迁移,同时成骨细胞间紧密连接蛋白(zonula occludens 1,ZO-1)的连接部位定位被破坏并转移到细胞质,可能使细胞获得更高的迁移性。因此成骨细胞的运动和黏附能力的增强是通过何种机制需进一步深入研究。 除此之外,镁离子显著增强成骨细胞的成骨活性,碱性磷酸酶是成骨细胞分泌的标志性酶,在骨形成过程中起重要作用,在一定程度上可反应成骨细胞的成骨能力。大量研究表明镁离子使成骨细胞活性增强,碱性磷酸酶活性增强[30-31,35-37],同时促进细胞外基质Ⅰ型胶原增多[38],细胞外基质成熟矿化[39]。除碱性磷酸酶外,细胞内多种成骨相关基因表达上调,如骨桥蛋白、骨钙素、核转录因子κB受体活化因子配体(receptor activator of nuclear factor-κ B ligand,RANKL)、RUNX2(一种成骨分化转录因子)等,均反映了骨愈合过程中成骨活性的增强。 而值得注意的是,HE等[40]首次研究了镁离子对成骨细胞细胞缝隙连接的影响,其显著促进成骨细胞的缝隙连接细胞间通讯(gap junction intercellular communication,GJIC),调节成骨细胞活性,可能是由于镁离子增强成骨细胞间缝隙连接细胞间通讯,成骨细胞同步功能,改善成骨细胞对刺激信号的反应。因此镁离子现已证实从成骨细胞增殖、凋亡、黏附、运动、成骨活性等多方面促进骨再生,但分子信号通路的研究仍较少,同时镁离子除了对受体的直接结合和激活作用,还可能存在如通过细胞间连接和信号传递协调骨重建等多种分子机制,值得进一步研究,丰富对骨再生的分子机制研究。 2.1.3 破骨细胞 破骨细胞参与骨吸收功能,在骨的发育和形成中与骨形成功能相互协调,在骨再生过程中发挥重要作用。破骨细胞由多个单核巨噬细胞融合而成,抗酒石酸酸性磷酸酶(tartrate resistant acid phosphatase,TRAP)是破骨细胞形成和功能的标志性酶。镁离子使TRAP阳性多核破骨细胞数量减少,多数学者认为是由于镁离子抑制破骨细胞增殖分化,破骨细胞特异性基因表达下调[41],而MARADZE等[27]发现在镁离子作用下TRAP阳性多核破骨细胞数量减少而特异性基因表达未下调,这可能是镁离子抑制单核巨噬细胞融合形成破骨细胞,因此镁离子作用于破骨细胞形成过程中的何种阶段及其分子机制需进一步研究。同时镁离子的浓度不同可能对破骨细胞产生不同效应,WU等[8]研究细胞外镁离子对破骨细胞分化和功能的影响,发现氯化镁随着浓度的提高先促进破骨细胞形成后抑制其形成,因此需控制调节镁离子的浓度以此调节骨吸收功能,与骨形成功能协调,形成更好地骨愈合。 镁离子对破骨细胞形成和活性影响的主要分子机制的关键集中在活化T细胞核转录因子1(nuclear factor-activated T cell 1,NFATc1),NFATc1是破骨细胞生成和功能的重要调节因子,NFATc1受到两条通路的影响,破骨细胞表面RANKL-RANK结合,激活核转录因子κB信号通路,诱导NFATc1表达;而另一方面钙调神经磷酸酶能够促进NFATc1自动扩增,其活性受细胞内钙离子浓度影响,从而钙离子浓度降低将使NFATc1表达降低。NFATc1表达增多能够激活下游破骨相关基因,促进细胞形成,增强骨吸收活性。镁离子抑制核转录因子κB通路,直接抑制NFATc1的表达,抑制破骨细胞相关基因的表达,如TRAP、降钙素受体、基质金属蛋白酶9、NFATc1和组织蛋白酶k[32],抑制破骨细胞形成和骨吸收活性。同时细胞内钙离子浓度受破骨细胞离子通道的调节,镁离子抑制钙离子通道,使细胞内Ca2+浓度降低,最终也使NFATc1表达降低,破骨细胞骨吸收活性降低[32]。除了NFATc1的分子机制调节外,镁离子抑制破骨细胞分化的分子机制也有其他研究,TAKUYA等[42]发现TPC2(Two-pore channel 2)是破骨细胞上的一种钙离子通道和钠离子通道,在1 mmol/L 镁离子条件下由PI(3,5)P2 (phosphatidylinositol-3,5-bisphosphate)途径介导,其活性降低,钠离子内流减少,细胞膜膜去极化,抑制破骨细胞分化。 综上所述,目前镁离子对于破骨细胞的分子机制研究较少,尤其是破骨细胞形成的分子机制,且破骨细胞形成过程涉及到干细胞、单核细胞等多种细胞的增殖、分化、融合、凋亡等代谢过程,镁离子具体主要作用于哪一阶段需要进一步研究,尤其是对融合和凋亡阶段的影响。 2.2 免疫调节 免疫系统中的巨噬细胞可分化为M1型和M2型巨噬细胞,M1型分泌炎症因子,启动早期炎症反应,其后M1型巨噬细胞需及时向M2型巨噬细胞转化,抑制炎症,促进后续成骨能力,若炎症早期将巨噬细胞完全清除,镁离子的促成骨作用消失[43]。镁离子可以促进M1向M2型巨噬细胞转化,调节细胞因子的分泌,抑制促炎因子肿瘤坏死因子α、白细胞介素1β及白细胞介素6的分泌,促进骨形态发生蛋白2和转化生长因子β的表达,最终促进骨再生[44-46]。进一步研究镁离子调节巨噬细胞的分子机制,镁离子的存在促进了单核细胞向成熟巨噬细胞的募集和激活,其后镁离子通过TRPM7离子通道进入巨噬细胞内,调节TLR-核转录因子κB信号通路,诱导巨噬细胞表型改变,在巨噬细胞细胞因子的分泌中起到关键作用[25,43,47]。 然而镁离子的免疫调节作用具有剂量依赖性和时间依赖性,浓度过高和长时间作用可能抑制骨再生作用。XIE等[48]发现高镁离子环境[(43.3±1.8) mmol/L]显著上调肿瘤坏死因子α信号通路,促进巨噬细胞的M1表型,刺激肿瘤坏死因子α和一氧化氮合成酶(iNOS)的表达,下调M2表型标记,并增强巨噬细胞的吞噬能力,高镁浓度引起的过度炎症反应对骨修复产生负面影响。QIAO等[43]则发现早期镁离子暴露,以TRPM7依赖的方式触发巨噬细胞组蛋白H3磷酸化,促进成骨免疫微环境的形成,而镁离子的持续暴露导致巨噬细胞中核转录因子κB信号的过度激活和破骨样细胞数量的增加,而且还通过抑制羟基磷灰石沉淀来减缓骨成熟。除此之外,掺入镁离子后局部微环境蛋白质吸附模式发生改变,材料对抗炎、细胞黏附和组织再生等蛋白质吸附能力增强,有利于骨再生[33]。免疫调节作为镁离子调节骨再生机制的研究热点,免疫调节涉及多细胞在骨微环境的相互作用,镁离子影响巨噬细胞表型转化已得到广泛共识,然而其浓度和时间的对巨噬细胞转化和分泌的影响尚不完全明晰,同时巨噬细胞与成骨、破骨的作用机制可进一步明晰。 2.3 血管生成的促进作用 血管的生成对骨的生长修复至关重要,在骨骼系统中血管不仅能够运输营养物质和代谢废物,利于骨相关细胞的生理代谢过程,还能够为骨形成提供钙和磷酸盐等无机离子,为细胞外基质矿化提供基础[49]。血管内壁由血管内皮细胞组成,镁离子通过TRPM7和MagT1离子通道增强血管内皮细胞增殖、黏附、运动[50-54],同时可使血管内皮细胞纽蛋白和肌动蛋白丝含量增加[30],细胞骨架蛋白产生增多,细胞呈现扩散形态[55],其相关基因表达上升,如血管生成素、血管细胞黏附因子1、膜细胞骨架连接蛋白等[53]。血管内皮细胞形成新的血管和血管网形成的能力,不论是数量、分支还是长度在掺入镁离子后也均有显著提升[30,37,53,56]。除此之外,在镁离子作用下调节S1P1-Rac1途径和内皮屏障稳定介质增强血管内皮屏障[54],增强内皮细胞之间的连接蛋白(ZO-1)使屏障更为稳固[32, 54]。因此镁离子改善血管内皮细胞的增殖、黏附、管形成、屏障能力,促进血管生成。 镁离子促血管生成作用对骨再生的作用也得到了研究证实。镁离子能够在体内共促进血管和骨生长[56]。Von Hippel-Lindau蛋白(VHL)与低氧诱导因子1α结合并通过泛素化使其降解,HAMUSHAN等[57]研究发现镁离子通过VHL/低氧诱导因子1α/血管内皮生长因子信号通路,抑制VHL,减少低氧诱导因子1α的降解,促进血管内皮生长因子表达,从而促进骨周围新生血管形成,进一步促进骨形成。此外,YE等[58]研究表明镁离子激活神经细胞内CGRP-FAK-血管内皮生长因子通路,促进血管生成,进而修复骨缺损部位。另外KUSUMBE等[59]发现H型血管为骨骼特有的血管,H型血管内皮细胞强表达CD31和EMCN,骨祖细胞选择性聚集在此型血管周围,与成骨关系密切,有研究表明H型血管可通过增强notch信号通路、低氧诱导因子1α、白细胞介素33分泌等方式[59-61],促进骨再生。同时HAN等[62]研究发现镁离子可促进H型血管的形成,促进骨祖细胞在H型血管周围聚集,使骨形成增加,然而镁离子是通过何种途径增强H型血管形成而进一步促进骨形成的分子机制尚未有研究报道。因此,目前镁离子促进血管形成可促进骨再生的观点已得到了广泛认可,镁离子促进血管再生和骨再生的研究较多,但主要是研究了镁离子促进血管形成的分子机制和最终促进骨再生的作用,然而镁离子促进血管形成从而促进骨再生的分子机制仍需进一步研究,尤其是H型血管内皮细胞,不能仅停留于血管再生对骨再生的促进机制和镁离子对血管和骨再生的作用,而应该将两者结合起来进一步研究证实。 镁离子通过多个途径促进骨再生,分子机制丰富,首先对骨相关细胞的增殖、迁移与黏附、活性、凋亡等代谢过程均有影响。对于间充质干细胞,镁离子显著促进其增殖、迁移与黏附、成骨分化,尤其是既可以通过经典Wnt通路、MAPK/ERK通路等直接促其成骨分化,也可通过促进神经节细胞分泌CGRP、巨噬细胞分泌骨形态发生蛋白等细胞因子间接促进成骨分化,其间接作用在成骨分化方面较为新颖。对于成骨细胞,镁离子主要通过TRPM/PI3K信号通路促进其增殖、迁移与黏附、成骨活性,抑制其凋亡。对于破骨细胞镁离子主要通过核转录因子κB信号通路抑制破骨细胞形成及骨吸收活性。除此之外,镁离子调节骨环境中的免疫反应,转化巨噬细胞表型,促进促成骨因子分泌,降低炎症因子分泌,降低炎症反应,同时促进血管内皮细胞增殖、迁移与黏附、成血管能力,促进血管生成。镁离子通过对免疫调节与血管生成的作用从而调节骨再生微环境的分子机制为近年来研究热点,尤其是巨噬细胞的核转录因子κB通路和H型血管内皮细胞对骨再生的作用,共同为骨再生创造了良好的微环境。见图3,表1。"

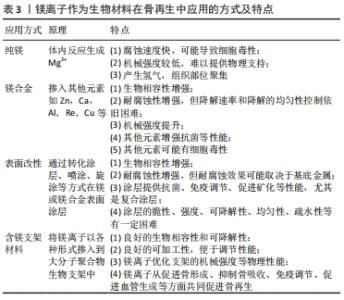
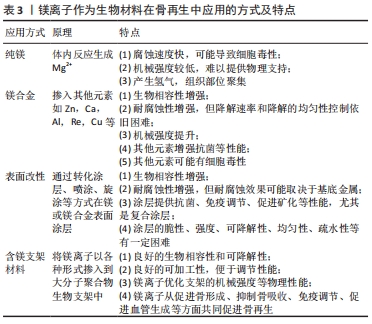
2.4.1 镁及镁合金 镁离子由于其良好的生物相容性和促骨再生作用,有望在骨再生方面得到广泛应用,金属镁在体内发生反应产生镁离子,然而由于强度较低且降解速度较快,镁离子毒性较大,且产生较多氢气,限制了其在骨科的应用,因此镁合金作为一种新型可降解植入物,在骨再生方面有良好的应用前景。镁合金通过掺入其他金属并调整各金属比例的方式,既能够降低降解速度和镁离子释放速度,同时能够改变物理性能,使镁合金植入材料与骨的密度及力学性能相匹配,更适用于骨再生[63]。 镁合金常掺入的金属元素包括锌、钙、铝及稀土元素等,以AZ合金(Mg-Al-Zn)、WE合金(Mg-Y-Re-Zr)、Mg-Zn-Ca合金为代表的镁合金兼具良好的机械性能和耐腐蚀性,证实其良好的骨再生作用。ZHANG等[64]首次在体外和体内对有前景的 Mg-2Zn-0.5Ca合金的表征、机械性能、降解行为、抗菌性能和生物相容性进行与纯镁的比较研究,其降解速度降低,促进骨髓间充质干细胞黏附,还显示出对耐甲氧西林金黄色葡萄球菌的抗菌性能,其临床转化潜力良好。有研究对植入体内的WE合金进行长达一年内的随访,其显示出长期稳定性和生物相容性,并且其周围形成层状骨,骨再生进展顺利[65]。同时Sr,Ag和Cu等微量元素掺入镁合金中,除了促进成骨细胞活化,促进骨再生外,还可提供作为抑菌作用等,从而创造良好的骨再生环境[66-69]。 总之,适当地使用合金元素可以改善镁合金的机械强度和耐腐蚀性等多种性能,但镁合金的降解速率仍是主要问题,最佳的镁合金意味着既控制了材料的分解速率,又保持了其机械强度。理想的生物可降解种植体应在种植初期保持机械完整性,至少4-16周,骨折复位后降解较快,但鲜有镁合金能达到。并且大多数镁合金由于其不均匀的微观结构而容易发生降解速率不均的问题,损害骨再生过程中的完整性。同时合金中其他元素的毒性仍需要注意,因为它们在降解过程中也会溶解在体液中,可能产生细胞毒性。 2.4.2 镁及镁合金的表面改性 镁及镁合金的表面改性可构建降解速度更可控、机械强度更优的骨再生生物植入材料,表面改性主要采用微弧氧化、阳极氧化、化学转化涂层等方式,而在骨再生方面镁合金表面改性的方式主要是无机类陶瓷涂层和聚合物涂层,无机类陶瓷涂层是通过转化涂层的方式,镁及镁合金与环境发生化学或电化学互相作用,从而在表面形成的,而聚合物涂层主要是通过喷涂、旋涂、浸涂等方式制备的[70]。无机类陶瓷涂层中的羟基磷灰石涂层其化学组成与结构与骨相似,可有效提高镁及镁合金耐腐蚀性,增强骨再生,羟基磷灰石表面涂层的形态和结构会影响骨再生[71],但其脆性高、强度低,降解性较差,限制了在骨再生方面的应用。并且有研究证明羟基磷灰石涂层抑制镁或镁合金腐蚀效果取决于腐蚀产物的性质[72],将羟基磷灰石涂层于5种镁或镁合金表面,植入大鼠股骨内观察到于WE43合金表面涂层抑制腐蚀效果最优,羟基磷灰石涂层表面骨再生增强。 磷酸钙涂层(tricalcium phosphate,TCP)也是广泛运用于镁及镁合金表面涂层,除了有良好的生物相容性和增强镁合金耐腐蚀性[73],还显示出良好的免疫调节作用,活化M2巨噬细胞表型,释放骨形态发生蛋白2,加速骨再生[74]。大分子聚合物涂层是近年来镁合金表面改性的研究重点,如聚己内酯、聚乳酸等,尤其是复合涂层的应用可综合多种涂层的性能[75]。有研究在镁螺钉上均匀涂层聚己内酯,其植入体内后涂层稳定,腐蚀更少,周围形成更致密更厚的骨[76]。还有研究在AZ31镁合金上制备聚己内酯和纳米羟基磷灰石双层纳米复合涂层,涂层后植入物促进早期细胞附着和生物矿化,骨形成更多[77]。因此镁及镁合金表面改性的多种涂层及结合应用提高镁及镁合金的耐腐蚀性和生物相容性,并可通过促进细胞增殖、黏附、免疫调节等作用促进骨再生,但仍需考虑涂层的可降解性、均匀性、疏水性、强度等问题,其抗炎性能、自愈性等潜力仍有待开发。 2.4.3 含镁支架材料 得益于优异的生物可降解性和可吸收性,由聚(α-羟基酯)及其共聚物组成的大分子合成聚合物支架已广泛用于骨组织再生,尤其是聚(乳酸-乙醇酸共聚物)[Poly(lactic-co-glycolic acid),PLGA]在骨再生方面的应用,然而其单独使用机械强度较低,可通过其他材料及镁离子增强性能。镁离子可通过各种镁的表现形式加入支架材料中,如镁粉和Mg(OH)2等形式。有研究将镁粉掺入新型多孔PLGA/TCP支架,其微观结构无改变,但机械强度显著提升,为骨修复提供更合适的机械支持,在体内,支架同时显著促进骨和血管的形成,具有协同作用,在修复骨缺损方面有巨大的临床转化潜力[78]。PLGA支架的分解产物可能会在植入部位产酸,引起细胞坏死和炎症,有研究开发含TCP/Mg(OH)2的PLGA纳米材料,由于Mg(OH)2的添加抑制肿瘤坏死因子α炎性细胞因子的表达和破骨细胞活化程度,促进骨缺损部位骨再生[79]。聚己内酯掺入不同比例的Mg,PCL/10%Mg支架表现出最优的骨再生作用[80],PCL/β-TCP/MgO2支架也有良好的机械性能,持续释放镁离子约3周,促进骨髓间充质干细胞存活、增殖、迁移和成骨分化[81]。含镁支架材料有良好的可加工性,不同材料的掺入与比例可调节其强度、延展性、韧性等,并赋予其协同促进骨再生与血管再生、调节炎症反应等作用,在骨再生方面应用前景良好。"

| [1] FILIP S, GüNTER F. Vascularization strategies in bone tissue engineering. Cells. 2021;10(7):1749. [2] 王子瑞,朱金亮,何志敏,等.人工合成骨修复材料的临床应用及展望[J].生物骨科材料与临床研究,2021,18(4):8-17. [3] DE BAAIJ JH, HOENDEROP JG, BINDELS RJ. Magnesium in man:implications for health and disease. Physiol Rev. 2015;95(1):1-46. [4] ZOFKOVA I, DAVIS M, BLAHOS J. Trace elements have beneficial, as well as detrimental effects on bone homeostasis. Physiol Res. 2017;66(3):391-402. [5] 崔云霞,李保胜,韩春雨,等.生物可降解医用镁合金应用于骨缺损修复中的研究展望 [J]. 现代口腔医学杂志,2019,33(1):46-49. [6] 刘印,马敏先.镁离子在骨再生应用的研究进展[J].生物医学工程与临床, 2021,25(4):522-526. [7] 施泽文,刘辰,陈先军,等.表面改性可降解镁的生物性能及其在骨修复领域的应用[J].国际骨科学杂志,2021,42(4):226-230. [8] WU L, LUTHRINGER BJ, FEYERABEND F, et al. Effects of extracellular magnesium on the differentiation and function of human osteoclasts. Acta Biomater. 2014; 10(6):2843-2854. [9] ZREIQAT H, HOWLETT CR, ZANNETTINO A, et al. Mechanisms of magnesium-stimulated adhesion of osteoblastic cells to commonly used orthopaedic implants. J Biomed Mater Res. 2002;62(2):175-184. [10] AL ALAWI AM, MAJONI SW, FALHAMMAR H. Magnesium and human health: perspectives and research directions. Int J Endocrinol. 2018;2018:9041694. [11] CASTIGLIONI S, ROMEO V, LOCATELLI L, et al. TRPM7 and MagT1 in the osteogenic differentiation of human mesenchymal stem cells in vitro. Sci Rep. 2018;8(1):16195. [12] ZHENG J, MAO X, LING J, et al. Role of magnesium transporter subtype 1 (magt1) in the osteogenic differentiation of rat bone marrow stem cells. Biol Trace Elem Res. 2016;171(1):131-137. [13] ZHENG JM, KONG YY, LI YY, et al. MagT1 regulated the odontogenic differentiation of BMMSCs induced byTGC-CM via ERK signaling pathway. Stem Cell Res Ther. 2019;10(1):48. [14] CHEN Y, LU X, ZHAO F, et al. A study on the biocompatibility of mgo coating prepared by anodic oxidation method on magnesium metal. J Bionic Eng. 2020; 17(1):76-91. [15] YUAN Z, WEI P, HUANG Y, et al. Injectable PLGA microspheres with tunable magnesium ion release for promoting bone regeneration. Acta Biomater. 2019; 85:294-309. [16] LEEM YH, LEE KS, KIM JH, et al. Magnesium ions facilitate integrin alpha 2- and alpha 3-mediated proliferation and enhance alkaline phosphatase expression and activity in hBMSCs. J Tissue Eng Regen Med. 2016;10(10):E527-E536. [17] KUNYU Z, SIEN L, QIAN F, et al. Nanocomposite hydrogels stabilized by self-assembled multivalent bisphosphonate-magnesium nanoparticles mediate sustained release of magnesium ion and promote in-situ bone regeneration. Acta Biomater. 2017;64:389-400. [18] WANG J, XU J, SONG B, et al. Magnesium (Mg) based interference screws developed for promoting tendon graft incorporation in bone tunnel in rabbits. Acta Biomater. 2017;63:393-410. [19] HUNG CC, CHAYA A, LIU K, et al. The role of magnesium ions in bone regeneration involves the canonical Wnt signaling pathway. Acta Biomater. 2019;98:246-255. [20] DÃAZ-TOCADOS JM, HERENCIA C, MARTÃNEZ-MORENO JM, et al. Magnesium chloride promotes osteogenesis through notch signaling activation and expansion of mesenchymal stem cells. Sci Rep. 2017;7(1):7839. [21] LIN S, YANG G, JIANG F, et al. A magnesium-enriched 3D culture system that mimics the bone development microenvironment for vascularized bone regeneration. Adv Sci (Weinh). 2019;6(12):1900209. [22] WANG L, WANG Y, LI Z, et al. Differential expression of long noncoding ribonucleic acids during osteogenic differentiation of human bone marrow mesenchymal stem cells. Int Orthop. 2015;39(5):1013-1019. [23] LI D, YU K, XIAO T, et al. LOC103691336/miR-138-5p/BMPR2 axis modulates Mg-mediated osteogenic differentiation in rat femoral fracture model and rat primary bone marrow stromal cells. J Cell Physiol. 2019;234(11):21316-21330. [24] ZHANG Y, XU J, RUAN YC, et al. Implant-derived magnesium induces local neuronal production of CGRP to improve bone-fracture healing in rats. Nat Med. 2016;22(10):1160-1169. [25] ZHANG X, CHEN Q, MAO X, et al. Magnesium Enhances Osteogenesis of BMSCs by Tuning Osteoimmunomodulation. Biomed Res Int. 2019;2019:7908205. [26] QIAN W, LEI X, REGINE W, et al. Macrophage-derived oncostatin M/bone morphogenetic protein 6 in response to Mg-based materials influences pro-osteogenic activity of human umbilical cord perivascular cells. Acta Biomater. 2020;133:268-279. [27] MARADZE D, MUSSON D, ZHENG Y, et al. High magnesium corrosion rate has an effect on osteoclast and mesenchymal stem cell role during bone remodelling. Sci Rep. 2018;8(1):10003. [28] SUN Y, LIU H, SUN X, et al. In vitro and in vivo study on the osseointegration of magnesium and strontium ion with two different proportions of mineralized collagen and its mechanism. J Biomater App. 2021;36(3):528-540. [29] BURMESTER A, WILLUMEIT-ROMER R, FEYERABEND F. Behavior of bone cells in contact with magnesium implant material. J Biomed Mater Res B Appl Biomater. 2017;105(1):165-179. [30] GAO P, FAN B, YU X, et al. Biofunctional magnesium coated Ti6Al4V scaffold enhances osteogenesis and angiogenesis in vitro and in vivo for orthopedic application. Bioact Mater. 2020;5(3):680-693. [31] ZHANG X, ZU H, ZHAO D, et al. Ion channel functional protein kinase TRPM7 regulates Mg ions to promote the osteoinduction of human osteoblast via PI3K pathway:In vitro simulation of the bone-repairing effect of Mg-based alloy implant. Acta Biomater. 2017;63:369-382. [32] ZHENG LZ, WANG JL, XU JK, et al. Magnesium and vitamin C supplementation attenuates steroid-associated osteonecrosis in a rat model. Biomaterials. 2020; 238:119828. [33] ANDREIA C, FRANCISCO R-G, IñAKI G-A, et al. Characterization of magnesium doped sol-gel biomaterial for bone tissue regeneration:The effect of Mg ion in protein adsorption. Mater Sci Eng C Mater Biol Appl. 2021;125:112114. [34] CHOI S, KIM KJ, CHEON S, et al. Biochemical activity of magnesium ions on human osteoblast migration. Biochem Biophys Res Commun. 2020;531(4):588-594. [35] LIU C, YANG G, ZHOU M, et al. Magnesium ammonium phosphate composite cell-laden hydrogel promotes osteogenesis and angiogenesis in vitro. ACS Omega. 2021;6(14):9449-9459. [36] YANG Y, WANG H, YANG H, et al. Magnesium-based whitlockite bone mineral promotes neural and osteogenic activities. ACS Biomater Sci Eng. 2020;6(10): 5785-5796. [37] ZHANG X, HUANG P, JIANG G, et al. A novel magnesium ion-incorporating dual-crosslinked hydrogel to improve bone scaffold-mediated osteogenesis and angiogenesis. Mater Sci Eng C Mater Biol Appl. 2021;121:111868. [38] TIAN L, SHENG Y, HUANG L, et al. An innovative Mg/Ti hybrid fixation system developed for fracture fixation and healing enhancement at load-bearing skeletal site. Biomaterials. 2018;180:173-183. [39] BELéN RRG, MANUEL DLJ, MICHELE I, et al. Biomimetic mineralization of recombinant collagen type I derived protein to obtain hybrid matrices for bone regeneration. J Struct Biol. 2016;196(2):138-146. [40] HE LY, ZHANG XM, LIU B, et al. Effect of magnesium ion on human osteoblast activity. Braz J Med Biol Res. 2016;49(7):e5257. [41] JäHN K, SAITO H, TAIPALEENMäKI H, et al. Intramedullary Mg2Ag nails augment callus formation during fracture healing in mice. Acta Biomater. 2016;36:350-360. [42] TAKUYA N, MIYUKI K, AKIKO H, et al. Role of lysosomal channel protein TPC2 in osteoclast differentiation and bone remodeling under normal and low-magnesium conditions. J Biol chem. 2017;292(51):20998-1010. [43] QIAO W, WONG KHM, SHEN J, et al. TRPM7 kinase-mediated immunomodulation in macrophage plays a central role in magnesium ion-induced bone regeneration. Nat Commun. 2021;12(1):2885. [44] WANG M, YU Y, DAI K, et al. Improved osteogenesis and angiogenesis of magnesium-doped calcium phosphate cement via macrophage immunomodulation. Biomater Sci. 2016;4(11):1574-1583. [45] COSTANTINO MD, SCHUSTER A, HELMHOLZ H, et al. Inflammatory response to magnesium-based biodegradable implant materials. Acta Biomater. 2020;101: 598-608. [46] BESSA-GONçALVES M, SILVA AM, BRáS JP, et al. Fibrinogen and magnesium combination biomaterials modulate macrophage phenotype, NF-kB signaling and crosstalk with mesenchymal stem/stromal cells. Acta Biomater. 2020;114:471-484. [47] ZHENG Z, CHEN Y, HONG H, et al. The “Yin and Yang” of immunomodulatory magnesium-enriched graphene oxide nanoscrolls decorated biomimetic scaffolds in promoting bone regeneration. Adv Healthc Mater. 2021;10(2):e2000631. [48] XIE K, WANG N, GUO Y, et al. Additively manufactured biodegradable porous magnesium implants for elimination of implant-related infections: an in vitro and in vivo study. Bioact Mater. 2021;8:140-152. [49] 李旭, 陈依民, 王鼎予, 等.骨血管形成机制及功能的研究进展[J].解剖学报, 2019,50(5):703-706. [50] GU Y, ZHANG J, ZHANG X, et al. Three-dimensional printed mg-doped β-TCP bone tissue engineering scaffolds:effects of magnesium ion concentration on osteogenesis and angiogenesis in vitro. Tissue Eng Regen Med. 2019;16(4):415-429. [51] XU L, WILLUMEIT-ROMER R, LUTHRINGER-FEYERABEND B. Hypoxia influences the effects of magnesium degradation products on the interactions between endothelial and mesenchymal stem cells. Acta Biomater. 2020;101:624-636. [52] LIU W, GUO S, TANG Z, et al. Magnesium promotes bone formation and angiogenesis by enhancing MC3T3-E1 secretion of PDGF-BB. Biochem Biophys Res Commun. 2020;528(4):664-670. [53] XU L, WILLUMEIT-ROMER R, LUTHRINGER-FEYERABEND BJC. Effect of magnesium-degradation products and hypoxia on the angiogenesis of human umbilical vein endothelial cells. Acta Biomater. 2019;98:269-283. [54] ZHU D, YOU J, ZHAO N, et al. Magnesium regulates endothelial barrier functions through TRPM7, MagT1, and S1P1. Adv Sci (Weinh). 2019;6(18):1901166. [55] GU Y, ZHANG J, ZHANG X, et al. Three-dimensional printed Mg-doped beta-TCP bone tissue engineering scaffolds: effects of magnesium ion concentration on osteogenesis and angiogenesis in vitro. Tissue Eng Regen Med. 2019;16(4):415-429. [56] MA L, CHENG S, JI X, et al. Immobilizing magnesium ions on 3D printed porous tantalum scaffolds with polydopamine for improved vascularization and osteogenesis. Mater Sci Eng C Mater Biol Appl. 2020;117:111303. [57] HAMUSHAN M, CAI W, ZHANG Y, et al. High-purity magnesium pin enhances bone consolidation in distraction osteogenesis model through activation of the VHL/HIF-1alpha/VEGF signaling. J Biomater Appl. 2020;35(2):224-236. [58] YE L, XU J, MI J, et al. Biodegradable magnesium combined with distraction osteogenesis synergistically stimulates bone tissue regeneration via CGRP-FAK-VEGF signaling axis. Biomaterials. 2021;275:120984. [59] KUSUMBE AP, RAMASAMY SK, ADAMS RH. Coupling of angiogenesis and osteogenesis by a specific vessel subtype in bone. Nature. 2014;507(7492): 323-328. [60] RAMASAMY SK, KUSUMBE AP, WANG L, et al. Endothelial Notch activity promotes angiogenesis and osteogenesis in bone. Nature. 2014;507(7492):376-380. [61] KENSWIL KJG, JARAMILLO AC, PING Z, et al. Characterization of endothelial cells associated with hematopoietic niche formation in humans identifies IL-33 as an anabolic factor. Cell Rep. 2018;22(3):666-678. [62] HAN HS, JUN I, SEOK HK, et al. Biodegradable magnesium alloys promote angio-osteogenesis to enhance bone repair. Adv Sci (Weinh). 2020;7(15):2000800. [63] STAIGER MP, PIETAK AM, HUADMAI J, et al. Magnesium and its alloys as orthopedic biomaterials:a review. Biomaterials. 2006;27(9):1728-1734. [64] ZHANG C, LIN J, NGUYEN NT, et al. Antimicrobial bioresorbable Mg-Zn-Ca alloy for bone repair in a comparison study with Mg-Zn-Sr alloy and pure Mg. ACS Biomater Sci Eng. 2020;6(1):517-538. [65] OSHIBE N, MARUKAWA E, YODA T, et al. Degradation and interaction with bone of magnesium alloy WE43 implants: a long-term follow-up in vivo rat tibia study. J Biomater Appl. 2019;33(9):1157-1167. [66] HAN J, WAN P, GE Y, et al. Tailoring the degradation and biological response of a magnesium-strontium alloy for potential bone substitute application. Mater Sci Eng C Mater Biol Appl. 2016;58:799-811. [67] LIU C, FU X, PAN H, et al. Biodegradable Mg-Cu alloys with enhanced osteogenesis, angiogenesis, and long-lasting antibacterial effects. Sci Rep. 2016;6:27374. [68] CHEN Y, DOU J, YU H, et al. Degradable magnesium-based alloys for biomedical applications: the role of critical alloying elements. J Biomater Appl. 2019;33(10): 1348-1372. [69] YU K, DAI Y, LUO Z, et al. In vitro and in vivo evaluation of novel biodegradable Mg-Ag-Y alloys for use as resorbable bone fixation implant. J Biomed Mater Res A. 2018;106(7):2059-2069. [70] YANG J, KOONS GL, CHENG G, et al. A review on the exploitation of biodegradable magnesium-based composites for medical applications. Biomed Mater. 2018; 13(2):022001. [71] ROJAEE R, FATHI M, RAEISSI K. Comparing nanostructured hydroxyapatite coating on AZ91 alloy samples via sol-gel and electrophoretic deposition for biomedical applications. IEEE Trans Nanobiosci. 2014;13(4):409-414. [72] HIROMOTO S, NOZOE E, HANADA K, et al. In vivo degradation and bone formation behaviors of hydroxyapatite-coated Mg alloys in rat femur. Mater Sci Eng C Mater Biol Appl. 2021;122:111942. [73] CHAI H, GUO L, WANG X, et al. In vitro and in vivo evaluations on osteogenesis and biodegradability of a β-tricalcium phosphate coated magnesium alloy. J Biomed Mater Res A. 2012;100(2):293-304. [74] CHEN Z, MAO X, TAN L, et al. Osteoimmunomodulatory properties of magnesium scaffolds coated with β-tricalcium phosphate. Biomaterials. 2014;35(30):8553-8565. [75] LI L Y, CUI LY, ZENG RC, et al. Advances in functionalized polymer coatings on biodegradable magnesium alloys - A review. Acta Biomater. 2018;79:23-36. [76] KIM YK, LEE KB, KIM SY, et al. Improvement of osteogenesis by a uniform PCL coating on a magnesium screw for biodegradable applications. Sci Rep. 2018;8(1):13264. [77] PERUMAL G, RAMASAMY B, NANDKUMAR AM, et al. Bilayer nanostructure coated AZ31 magnesium alloy implants: in vivo reconstruction of critical-sized rabbit femoral segmental bone defect. Nanomedicine. 2020;29:102232. [78] LAI Y, LI Y, CAO H, et al. Osteogenic magnesium incorporated into PLGA/TCP porous scaffold by 3D printing for repairing challenging bone defect. Biomaterials. 2019;197:207-219. [79] GO EJ, KANG EY, LEE SK, et al. An osteoconductive PLGA scaffold with bioactive β-TCP and anti-inflammatory Mg(OH)(2) to improve in vivo bone regeneration. Biomater Sci. 2020;8(3):937-948. [80] ZHAO S, XIE K, GUO Y, et al. Fabrication and biological activity of 3D-printed polycaprolactone/magnesium porous scaffolds for critical size bone defect repair. ACS Biomater Sci Eng. 2020;6(9):5120-5131. [81] PENG Z, WANG C, LIU C, et al. 3D printed polycaprolactone/beta-tricalcium phosphate/magnesium peroxide oxygen releasing scaffold enhances osteogenesis and implanted BMSCs survival in repairing the large bone defect. J Mater Chem B. 2021;9(28):5698-5710. |
| [1] | Yao Xiaoling, Peng Jiancheng, Xu Yuerong, Yang Zhidong, Zhang Shuncong. Variable-angle zero-notch anterior interbody fusion system in the treatment of cervical spondylotic myelopathy: 30-month follow-up [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1377-1382. |
| [2] | Xue Yadong, Zhou Xinshe, Pei Lijia, Meng Fanyu, Li Jian, Wang Jinzi . Reconstruction of Paprosky III type acetabular defect by autogenous iliac bone block combined with titanium plate: providing a strong initial fixation for the prosthesis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1424-1428. |
| [3] | Wu Weiyue, Guo Xiaodong, Bao Chongyun. Application of engineered exosomes in bone repair and regeneration [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1102-1106. |
| [4] | Zhou Hongqin, Wu Dandan, Yang Kun, Liu Qi. Exosomes that deliver specific miRNAs can regulate osteogenesis and promote angiogenesis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1107-1112. |
| [5] | Zhang Jinglin, Leng Min, Zhu Boheng, Wang Hong. Mechanism and application of stem cell-derived exosomes in promoting diabetic wound healing [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1113-1118. |
| [6] | An Weizheng, He Xiao, Ren Shuai, Liu Jianyu. Potential of muscle-derived stem cells in peripheral nerve regeneration [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1130-1136. |
| [7] | Gao Yujin, Peng Shuanglin, Ma Zhichao, Lu Shi, Cao Huayue, Wang Lang, Xiao Jingang. Osteogenic ability of adipose stem cells in diabetic osteoporosis mice [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 999-1004. |
| [8] | Huang Shibo, Xie Hui, Wang Zongpu, Wang Weidan, Qin Kairong, Zhao Dewei. Application of degradable high-purity magnesium screw in the treatment of developmental dysplasia of the hip [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 493-498. |
| [9] | Tan Guozhong, Tu Xinran, Guo Liyang, Zhong Jialin, Zhang Yang, Jiang Qianzhou. Biosafety evaluation of three-dimensional printed gelatin/sodium alginate/58S bioactive glass scaffolds for bone defect repair [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 521-527. |
| [10] | He Yunying, Li Lingjie, Zhang Shuqi, Li Yuzhou, Yang Sheng, Ji Ping. Method of constructing cell spheroids based on agarose and polyacrylic molds [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 553-559. |
| [11] | He Guanyu, Xu Baoshan, Du Lilong, Zhang Tongxing, Huo Zhenxin, Shen Li. Biomimetic orientated microchannel annulus fibrosus scaffold constructed by silk fibroin [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 560-566. |
| [12] | Yang Feng, Zhao Qian, Zhang Shixuan, Zhao Tienan, Feng Bo. Effectiveness and safety of rapamycin combined with CD133 antibody stent in preventing vascular restenosis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 579-584. |
| [13] | Chen Xiaoxu, Luo Yaxin, Bi Haoran, Yang Kun. Preparation and application of acellular scaffold in tissue engineering and regenerative medicine [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 591-596. |
| [14] | Kang Kunlong, Wang Xintao. Research hotspot of biological scaffold materials promoting osteogenic differentiation of bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 597-603. |
| [15] | Shen Jiahua, Fu Yong. Application of graphene-based nanomaterials in stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 604-609. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||