Chinese Journal of Tissue Engineering Research ›› 2022, Vol. 26 ›› Issue (30): 4780-4786.doi: 10.12307/2022.728
Previous Articles Next Articles
Integrin-targeted peptide promotes proliferation of bone marrow mesenchymal stem cells in SD rats
Jia Qiyu1, Huang Xiaoxia1, Guo Jian1, Huang Jinyong2, Guo Xiaobin3, Abdussalam·Alimujiang1, Wu Tong1, Ma Chuang1
- 1Department of Microprosthetic Surgery, 2Department of Traumatology and Orthopedics, 3Department of Joint Surgery, Orthopedic Center, First Affiliated Hospital of Xinjiang Medical University, Urumqi 830054, Xinjiang Uygur Autonomous Region, China
-
Received:2021-10-25Accepted:2021-12-10Online:2022-10-28Published:2022-03-29 -
Contact:Ma Chuang, PhD, Chief physician, Master’s supervisor, Department of Microprosthetic Surgery, Orthopedic Center, First Affiliated Hospital of Xinjiang Medical University, Urumqi 830054, Xinjiang Uygur Autonomous Region, China -
About author:Jia Qiyu, Master candidate, Department of Microprosthetic Surgery, Orthopedic Center, First Affiliated Hospital of Xinjiang Medical University, Urumqi 830054, Xinjiang Uygur Autonomous Region, China -
Supported by:Natural Science Foundation of Xinjiang Uygur Autonomous Region, No. 2020D01C263 (to GXB); National Natural Science Foundation of China, No. 81760397 (to MC)
CLC Number:
Cite this article
Jia Qiyu, Huang Xiaoxia, Guo Jian, Huang Jinyong, Guo Xiaobin, Abdussalam·Alimujiang, Wu Tong, Ma Chuang. Integrin-targeted peptide promotes proliferation of bone marrow mesenchymal stem cells in SD rats[J]. Chinese Journal of Tissue Engineering Research, 2022, 26(30): 4780-4786.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks

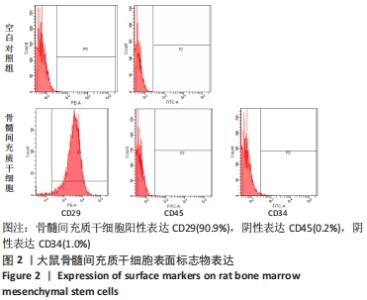
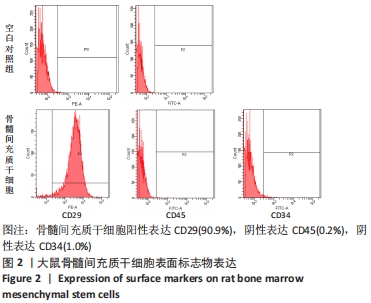
2.1 大鼠骨髓间充质干细胞形态学变化 原代细胞体外分离培养的第3天,镜下可见大量的贴壁细胞,总体呈散状分布,大体形态不统一,部分单侧伪足伸出,部分细胞整体呈梭形,两端均探出伪足,部分细胞有触角探出,另有部分呈多角形,见图 1A-C;轻轻晃动瓶身可见视野中存在漂浮的大量死细胞,对其进行清洗换液。首次换液时,镜下可见细胞密度较低,且分布不均,部分细胞呈聚集态成簇分布,少部分呈团块状,大概率为骨髓小粒未吹散,细胞整体饱满,折光性佳。生长达10 d时,细胞即可增殖至约瓶底面积的90%。随之进入快速增殖阶段,并且随着代数的增加,生长排列愈发整齐,异型细胞逐渐减少,细胞大体形态趋向均一化,部分呈漩涡状生长,见图1D-F。"


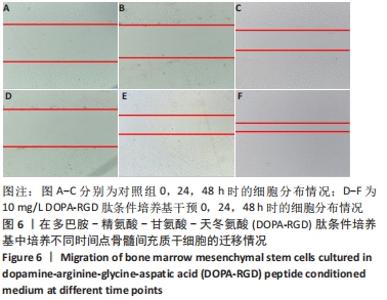
2.3 DOPA-RGD肽作用下骨髓间充质干细胞的形态变化 DOPA-RGD肽作用第 1 天,质量浓度> 20 mg/L时,出现了细胞生长抑制现象,且随着DOPA-RGD肽质量浓度的升高,瓶中漂浮的细胞愈发增多,凋亡现象较明显,细胞形态发生极大改变,部分仍为与之前一致的片状多极化生长,大部分细胞呈现两极化生长趋势,伪足呈细丝状改变,当质量浓度≤20 mg/L 时无显著变化,细胞仍呈密集化聚集生长,整体排布较为整齐,镜下可见均存在少量的漂浮细胞。DOPA-RGD肽作用第3天,质量浓度> 20 mg/L时,细胞形态与第1天观察所见趋势相同,瓶底出现愈发增多的漂浮细胞,80 mg/L组细胞大量凋亡,镜下细胞量显著减少;10 mg/L组镜下可见仍存在少部分细胞漂浮于视野中,但细胞形态也已发生显著改变,形态呈多极化菱形趋势、聚集态生长,细胞密度显著高于其他组,且立体感更强,100倍镜下细胞伸出的细丝状伪足互相交联呈网状。DOPA-RGD肽作用第5天和第7 天时各组细胞形态变化趋势均同前一致,无显著改变,见图3。"

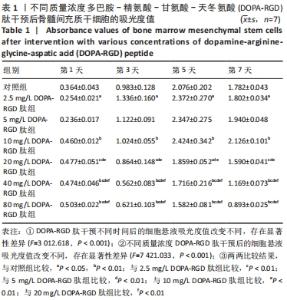
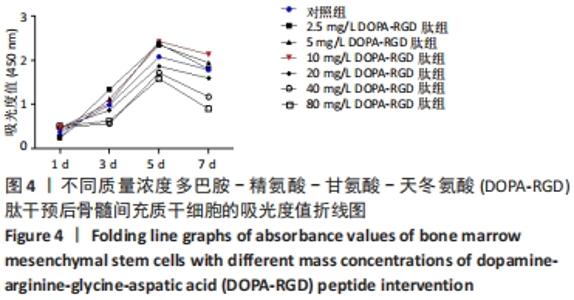
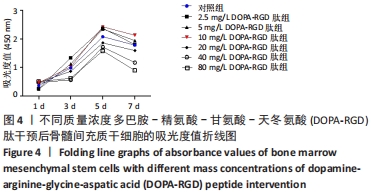
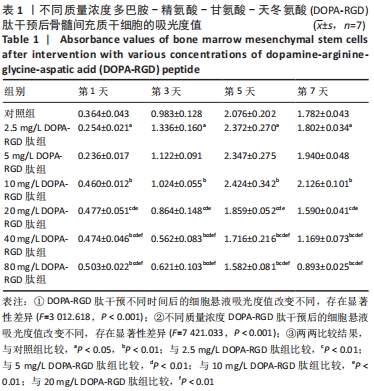
2.4 不同质量浓度DOPA-RGD肽对骨髓间充质干细胞增殖的影响 对所得数据行重复测量方差分析,结果表明经DOPA-RGD肽作用不同时长的骨髓间充质干细胞吸光度值改变不同,存在显著性差异(F=3 012.618,P < 0.001),说明随着DOPA-RGD肽作用时长增加,细胞增殖情况也随之发生明显改变。不同质量浓度DOPA-RGD肽干预骨髓间充质干细胞的吸光度值改变不同,存在显著性差异 (F=7 421.033,P < 0.001),说明经不同质量浓度DOPA-RGD肽干预后,各组细胞增殖情况发生明显变化。 通过 LSD 法进一步对各组进行两两比较,结果显示: 2.5 mg/L DOPA-RGD肽组与质量浓度≥20 mg/L的3个DOPA-RGD肽组及对照组比较均存在显著性差异(P < 0.05);5 mg/L DOPA-RGD肽组与质量浓度≥20 mg/L的3个DOPA-RGD肽组均存在显著性差异(P < 0.05);10 mg/L DOPA-RGD肽组与质量浓度≥20 mg/L的3个DOPA-RGD肽组及对照组比较差异均存在非常显著性差异(P < 0.01);20 mg/L DOPA-RGD肽组与除对照组外的其他各组比较均存在显著性差异(P < 0.01);40 mg/L DOPA-RGD肽组与80 mg/L DOPA-RGD肽组之间无显著差异(P > 0.05),40 mg/L DOPA-RGD肽组与除80 mg/L DOPA-RGD肽组外的其他各组比较均存在显著性差异(P < 0.01);80 mg/L组与质量浓度≤20 mg/L组的4个DOPA-RGD肽组及对照组比较差异均存在显著性差异(P < 0.001);对照组与除5,20 mg/L DOPA-RGD肽组外的其他组均存在显著性差异(P < 0.05)。从各组吸光度值汇总数据中可以看出,2.5-10 mg/L DOPA-RGD肽组于第 5 天的吸光度值明显高于其余组,随后整体呈下降趋势,其中10 mg/L DOPA-RGD肽组在第5天和第7天吸光度值均最高。由此可见,当质量浓度≤10 mg/L 的DOPA-RGD肽在干预 5 天时可促进细胞的增殖。DOPA-RGD肽质量浓度≥20 mg/L会促进细胞凋亡,见表1,图4。"
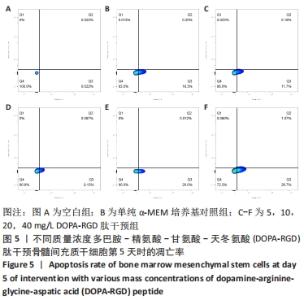
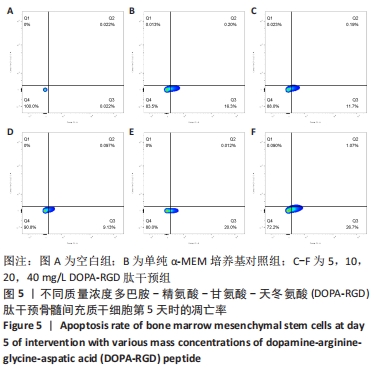
2.5 不同质量浓度DOPA-RGD肽对骨髓间充质干细胞凋亡的影响 在经CCK-8法确定的DOPA-RGD肽最佳干预时间,即干预培养的第5天时,单纯α-MEM培养基组骨髓间充质干细胞凋亡率为(16.24±0.86)%,5,10,20,40 mg/L DOPA-RGD肽干预组细胞凋亡率为(12.00±0.39)%,(9.22±0.54)%,(19.96±0.50)%,(27.88±0.24)%,各组间方差分析结果表明均存在显著性差异(P < 0.001),该结果表明,10 mg/L DOPA-RGD肽干预第5天时可显著降低骨髓间充质干细胞的凋亡率,当DOPA-RGD肽质量浓度≥20 mg/L会促进细胞凋亡,见图5。"
| [1] GIANNOUDIS PV, CALORI GM, BÉGUÉ T, et al. Tissue loss and bone repair: Time to develop an international strategy? Injury. 2015;46 Suppl 8:S1-2. [2] PAPAKOSTIDIS C, BHANDARI M, GIANNOUDIS PV. Distraction osteogenesis in the treatment of long bone defects of the lower limbs: effectiveness, complications and clinical results; a systematic review and meta-analysis. Bone Joint J. 2013;95-B(12):1673-1680. [3] VERLINDEN CR, VAN DE VIJFEIJKEN SE, TUINZING DB, et al. Complications of mandibular distraction osteogenesis for developmental deformities: a systematic review of the literature. Int J Oral Maxillofac Surg. 2015;44(1):44-49. [4] 张进,张永红,王钞琦,等.髓内延长钉技术的研究进展[J].中国修复重建外科杂志,2021,35(5):642-647. [5] PALEY D. Problems, obstacles, and complications of limb lengthening by the Ilizarov technique. Clin Orthop Relat Res. 1990;(250):81-104. [6] YANAGISAWA Y, ITO A, HARA Y, et al. Initial clinical trial of pins coated with fibroblast growth factor-2-apatite composite layer in external fixation of distal radius fractures. J Orthop. 2018;16(1):69-73. [7] GATHEN M, PLOEGER MM, JAENISCH M, et al. Outcome evaluation of new calcium titanate schanz-screws for external fixators. First clinical results and cadaver studies. J Mater Sci Mater Med. 2019;30(11):124. [8] BAKHSH K, ZIMRI FK, ATIQ-UR-REHMAN, et al. Outcome of complex non-unions of femoral fractures managed with Ilizarov method of distraction osteogenesis. Pak J Med Sci. 2019;35(4):1055-1059. [9] GUICHET JM, DEROMEDIS B, DONNAN LT, et al. Gradual femoral lengthening with the Albizzia intramedullary nail. J Bone Joint Surg Am. 2003;85(5):838-848. [10] GARCÍA-CIMBRELO E, CURTO DE LA MANO A, GARCÍA-REY E, et al. The intramedullary elongation nail for femoral lengthening. J Bone Joint Surg Br. 2002;84(7):971-977. [11] LEE DH, RYU KJ, SONG HR, et al. Complications of the Intramedullary Skeletal Kinetic Distractor (ISKD) in distraction osteogenesis. Clin Orthop Relat Res. 2014;472(12):3852-3859. [12] KRIEG AH, LENZE U, SPETH BM, et al. Intramedullary leg lengthening with a motorized nail. Acta Orthop. 2011;82(3):344-350. [13] COLE JD, JUSTIN D, KASPARIS T, et al. The intramedullary skeletal kinetic distractor (ISKD): first clinical results of a new intramedullary nail for lengthening of the femur and tibia. Injury. 2001;32 Suppl 4:SD129-139. [14] WANG K, EDWARDS E. Intramedullary skeletal kinetic distractor in the treatment of leg length discrepancy--a review of 16 cases and analysis of complications. J Orthop Trauma. 2012;26(9):e138-144. [15] GUICHET JM, SPIVAK JM, TROUILLOUD P, et al. Lower limb-length discrepancy. An epidemiologic study. Clin Orthop Relat Res. 1991;(272):235-241. [16] QI MC, ZOU SJ, HAN LC, et al. Expression of bone-related genes in bone marrow MSCs after cyclic mechanical strain: implications for distraction osteogenesis. Int J Oral Sci. 2009;1(3):143-150. [17] QI MC, HU J, ZOU SJ, et al. Mechanical strain induces osteogenic differentiation: Cbfa1 and Ets-1 expression in stretched rat mesenchymal stem cells. Int J Oral Maxillofac Surg. 2008;37(5):453-458. [18] 姜涛,吴硕,李志强,等.血小板衍生生长因子BB促进SD大鼠骨髓间充质干细胞的增殖[J].中国组织工程研究,2021,25(13):1976-1981. [19] PEREZ JR, KOUROUPIS D, LI DJ, et al. Tissue Engineering and Cell-Based Therapies for Fractures and Bone Defects. Front Bioeng Biotechnol. 2018;6: 105. [20] DUCHAMP DE LAGENESTE O, JULIEN A, Abou-Khalil R, et al. Periosteum contains skeletal stem cells with high bone regenerative potential controlled by Periostin. Nat Commun. 2018;9(1):773. [21] GUO X, LIU Y, BAI J, et al. Efficient Inhibition of Wear-Debris-Induced Osteolysis by Surface Biomimetic Engineering of Titanium Implant with a Mussel-Derived Integrin-Targeting Peptide. Adv Biosyst. 2019;3(2):e1800253. [22] PAN G, SUN S, ZHANG W, et al. Biomimetic Design of Mussel-Derived Bioactive Peptides for Dual-Functionalization of Titanium-Based Biomaterials. J Am Chem Soc. 2016;138(45):15078-15086. [23] VERSTAPPEN JFM, JIN J, KOÇER G, et al. RGD-functionalized supported lipid bilayers modulate pre-osteoblast adherence and promote osteogenic differentiation. J Biomed Mater Res A. 2020;108(4):923-937. [24] HORI N, UENO T, MINAMIKAWA H, et al. Electrostatic control of protein adsorption on UV-photofunctionalized titanium. Acta Biomater. 2010;6(10): 4175-4180. [25] AITA H, HORI N, TAKEUCHI M, et al. The effect of ultraviolet functionalization of titanium on integration with bone. Biomaterials. 2009;30(6):1015-1025. [26] GUTIÉRREZ-SÁNCHEZ M, ESCOBAR-BARRIOS VA, POZOS-GUILLÉN A, et al. RGD-functionalization of PLA/starch scaffolds obtained by electrospinning and evaluated in vitro for potential bone regeneration. Mater Sci Eng C Mater Biol Appl. 2019;96:798-806. [27] LUO F, HONG G, MATSUI H, et al. Initial osteoblast adhesion and subsequent differentiation on zirconia surfaces are regulated by integrins and heparin-sensitive molecule. Int J Nanomedicine. 2018;13:7657-7667. [28] CHEN X, SEVILLA P, APARICIO C. Surface biofunctionalization by covalent co-immobilization of oligopeptides. Colloids Surf B Biointerfaces. 2013;107: 189-197. [29] KANTLEHNER M, SCHAFFNER P, FINSINGER D, et al. Surface coating with cyclic RGD peptides stimulates osteoblast adhesion and proliferation as well as bone formation. Chembiochem. 2000;1(2):107-114. [30] MAS-MORUNO C, FRAIOLI R, ALBERICIO F, et al. Novel peptide-based platform for the dual presentation of biologically active peptide motifs on biomaterials. ACS Appl Mater Interfaces. 2014;6(9):6525-6536. [31] WAITE JH. The DOPA Ephemera: A Recurrent Motif in Invertebrates. Biol Bull. 1992;183(1):178-184. [32] WAITE JH. The phylogeny and chemical diversity of quinone-tanned glues and varnishes. Comp Biochem Physiol B. 1990;97(1):19-29. [33] 李娜,章非敏.多巴胺与Ⅰ型胶原对MC3T3-E1细胞初期粘附形态的影响[J].口腔生物医学,2018,9(4):187-190. [34] HUANG YZ, WANG JJ, HUANG YC, et al. Organic composite-mediated surface coating of human acellular bone matrix with strontium. Mater Sci Eng C Mater Biol Appl. 2018;84:12-20. [35] HUANG S, LIANG N, HU Y, et al. Polydopamine-Assisted Surface Modification for Bone Biosubstitutes. Biomed Res Int. 2016;2016:2389895. [36] DE JONGE LT, LEEUWENBURGH SC, WOLKE JG, et al. Organic-inorganic surface modifications for titanium implant surfaces. Pharm Res. 2008; 25(10):2357-2369. [37] BRUBAKER CE, MESSERSMITH PB. Enzymatically degradable mussel-inspired adhesive hydrogel. Biomacromolecules. 2011;12(12):4326-4334. [38] SECCHI AG, GRIGORIOU V, SHAPIRO IM, et al. RGDS peptides immobilized on titanium alloy stimulate bone cell attachment, differentiation and confer resistance to apoptosis. J Biomed Mater Res A. 2007;83(3):577-584. [39] ELMENGAARD B, BECHTOLD JE, SØBALLE K. In vivo study of the effect of RGD treatment on bone ongrowth on press-fit titanium alloy implants. Biomaterials. 2005;26(17):3521-3526. [40] KROESE-DEUTMAN HC, VAN DEN DOLDER J, SPAUWEN PH, et al. Influence of RGD-loaded titanium implants on bone formation in vivo. Tissue Eng. 2005;11(11-12):1867-1875. |
| [1] | Xiao Hao, Liu Jing, Zhou Jun. Research progress of pulsed electromagnetic field in the treatment of postmenopausal osteoporosis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1266-1271. |
| [2] | Tian Chuan, Zhu Xiangqing, Yang Zailing, Yan Donghai, Li Ye, Wang Yanying, Yang Yukun, He Jie, Lü Guanke, Cai Xuemin, Shu Liping, He Zhixu, Pan Xinghua. Bone marrow mesenchymal stem cells regulate ovarian aging in macaques [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 985-991. |
| [3] | Hou Jingying, Guo Tianzhu, Yu Menglei, Long Huibao, Wu Hao. Hypoxia preconditioning targets and downregulates miR-195 and promotes bone marrow mesenchymal stem cell survival and pro-angiogenic potential by activating MALAT1 [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1005-1011. |
| [4] | Liang Xuezhen, Yang Xi, Li Jiacheng, Luo Di, Xu Bo, Li Gang. Bushen Huoxue capsule regulates osteogenic and adipogenic differentiation of rat bone marrow mesenchymal stem cells via Hedgehog signaling pathway [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1020-1026. |
| [5] | Fang Xiaolei, Leng Jun, Zhang Chen, Liu Huimin, Guo Wen. Systematic evaluation of different therapeutic effects of mesenchymal stem cell transplantation in the treatment of ischemic stroke [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1085-1092. |
| [6] | Zhang Jinglin, Leng Min, Zhu Boheng, Wang Hong. Mechanism and application of stem cell-derived exosomes in promoting diabetic wound healing [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1113-1118. |
| [7] | Feng Jianbo, Li Chencheng, Liu Jinyue, Wang Xiaomin, Peng Jiachen. Implantation of Kirschner wire with Staphylococcus aureus biofilm establishes a traumatic osteomyelitis model in rats [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(5): 700-705. |
| [8] | Kang Kunlong, Wang Xintao. Research hotspot of biological scaffold materials promoting osteogenic differentiation of bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 597-603. |
| [9] | Yang Sidi, Wang Qian, Xu Nuo, Wang Ronghan, Jin Chuanqi, Lu Ying, Dong Ming. Biodentine enhances the proliferation and differentiation of osteoblasts through upregulating bone morphogenetic protein-2 [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 516-520. |
| [10] | Wu Chengcong, Wang Fang, Wan Jianshan, Wu Zheng, Sun Rong, Huang Hefei, Qian Xuankun, Ou Hua, Ren Jing. Adenovirus-mediated bone morphogenetic protein 2 induces osteogenic differentiation of rabbit bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(30): 4757-4761. |
| [11] | Dong Yi, Shan Shuai, Liu Jialin, Han Xiangzhen, He Huiyu. Circular RNA mmu_circ_0001775 knockdown improves the osteogenic ability of mouse bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(30): 4767-4772. |
| [12] | Shen Enpu, Huang Ba, Liu Danping, Qi Hui, Wu Zhiwen, Li Beibei. Exosomes derived from melatonin-modified bone marrow mesenchymal stem cells promote osteogenesis of bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(30): 4800-4805. |
| [13] | Huang Gao, Xu Jun, Chen Wenge. Implantation of bone marrow mesenchymal stem cells-loaded platelet-rich plasma combined with extracorporeal shock wave in the repair of bone defects [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(30): 4812-4818. |
| [14] | Zhan Yuanbo, Liu Xinpeng, Xu Wenxia, Miao Nan, Mu Sen, Zhang Ruimin, Li Ying. Expression of histone deacetylase 9 in bone marrow mesenchymal stem cells during senescence [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(30): 4762-4766. |
| [15] | Wu Xiaosong, Gong Chao, Lou Pan, Wang Wei. Possibility of forskolin and 3-isobutyl-1-methylxanthine in inducing bone marrow mesenchymal stem cells to differentiate into Schwann cells-like phenotype [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(30): 4787-4792. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||