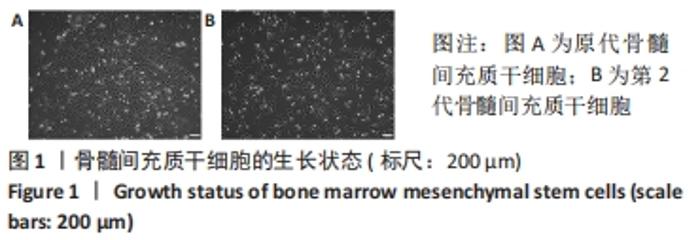
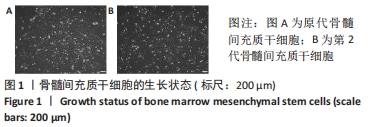
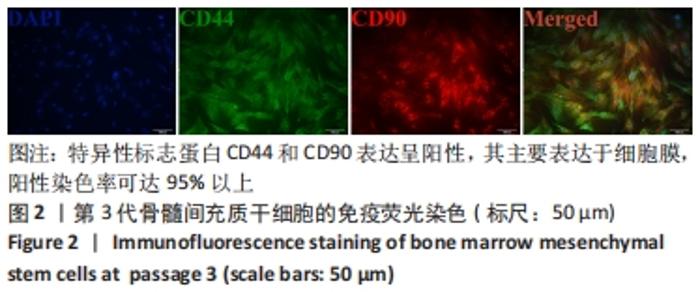
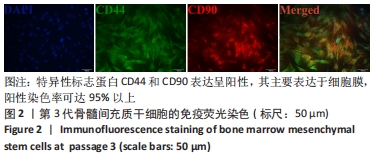
Chinese Journal of Tissue Engineering Research ›› 2022, Vol. 26 ›› Issue (30): 4787-4792.doi: 10.12307/2022.757
Previous Articles Next Articles
Possibility of forskolin and 3-isobutyl-1-methylxanthine in inducing bone marrow mesenchymal stem cells to differentiate into Schwann cells-like phenotype
Wu Xiaosong1, Gong Chao1, Lou Pan1, Wang Wei2, 3, 4
- 1Department of Orthopedics, Jingmen No.1 People’s Hospital, Jingmen 448000, Hubei Province, China; 2Department of Orthopedics, First Affiliated Hospital, Jinzhou Medical University, Jinzhou 121000, Liaoning Province, China; 3Institute of Extra-orbital Sciences, Jinzhou Medical University, Jinzhou 121000, Liaoning Province, China; 4Liaoning Provincial Key Laboratory of Medical Tissue Engineering, Jinzhou 121000, Liaoning Province, China
-
Received:2021-09-16Accepted:2021-10-22Online:2022-10-28Published:2022-03-29 -
Contact:Wang Wei, MD, Chief physician, Professor, Department of Orthopedics, First Affiliated Hospital, Jinzhou Medical University, Jinzhou 121000, Liaoning Province, China; Institute of Extra-orbital Sciences, Jinzhou Medical University, Jinzhou 121000, Liaoning Province, China; Liaoning Provincial Key Laboratory of Medical Tissue Engineering, Jinzhou 121000, Liaoning Province, China -
About author:Wu Xiaosong, Associate chief physician, Department of Orthopedics, Jingmen No.1 People’s Hospital, Jingmen 448000, Hubei Province, China Gong Chao, Master, Physician, Department of Orthopedics, Jingmen No.1 People’s Hospital, Jingmen 448000, Hubei Province, China -
Supported by:School-Enterprise Cooperation Fund Project of Jinzhou Medical University, No. 2020002 (to WW); General Science and Technology Project of Jingmen, No. 2021YFYB072 (to LP)
CLC Number:
Cite this article
Wu Xiaosong, Gong Chao, Lou Pan, Wang Wei. Possibility of forskolin and 3-isobutyl-1-methylxanthine in inducing bone marrow mesenchymal stem cells to differentiate into Schwann cells-like phenotype[J]. Chinese Journal of Tissue Engineering Research, 2022, 26(30): 4787-4792.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
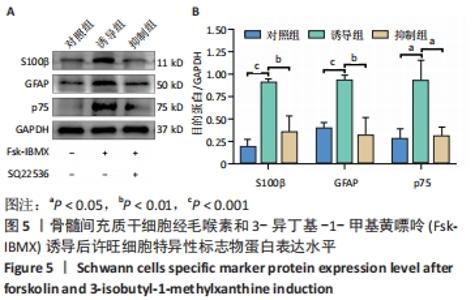
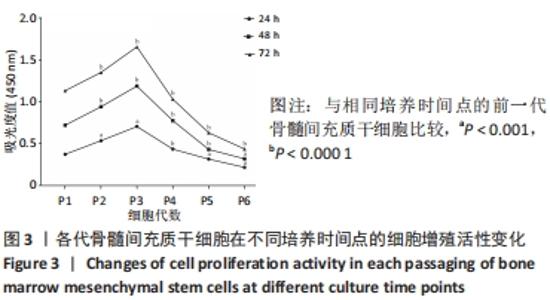
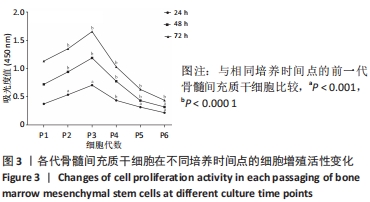
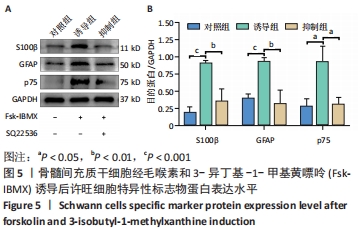
2.5 Fsk-IBMX对许旺细胞特异性标志物表达的影响 Fsk-IBMX诱导后,Western blot检测许旺细胞特异性标志物S100β、GFAP和p75的表达水平,结果显示,诱导组细胞S100β、GFAP和p75的表达水平(0.911±0.038,0.938±0.050,0.932±0.224)与对照组(0.187±0.086,0.394±0.066,0.278±0.113)相比均明显增加(P < 0.001,P < 0.001,P < 0.05),而抑制组细胞S100β、GFAP和p75的表达水平(0.353±0.181,0.315±0.202,0.304±0.106)较对照组无明显变化,与诱导组相比则均明显降低(P < 0.01,P < 0.01,P < 0.05),见图5。"
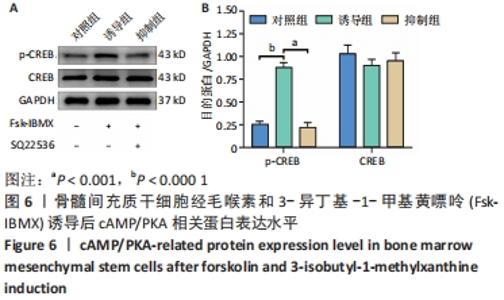
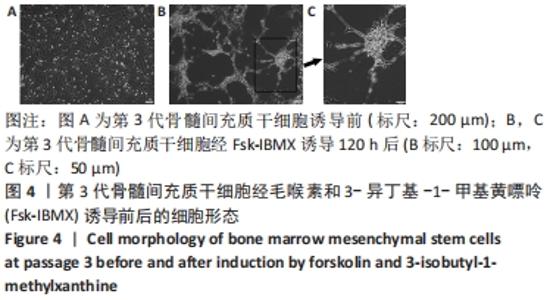
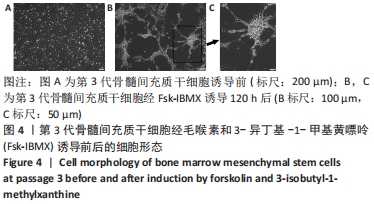
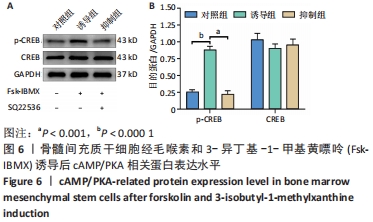
2.6 Fsk-IBMX对cAMP/PKA相关蛋白表达的影响 Fsk-IBMX诱导第3代骨髓间充质干细胞120 h后,Western blot检测了cAMP效应元件结合蛋白(cAMP-response element binding protein,CREB)及磷酸化CREB蛋白(phospho-CREB protein,p-CREB)的表达水平,结果显示,诱导组细胞p-CREB表达水平(0.885±0.050)与对照组(0.247±0.041)相比明显增加(P < 0.000 1),而CREB表达水平(0.908±0.062;1.036±0.090)变化则不明显(P > 0.05)。在加入cAMP/PKA抑制剂SQ22536抑制组中,p-CREB的表达水平(0.218±0.056)明显低于诱导组(P < 0.001),CREB的表达水平(0.958±0.084)仍无明显变化(P > 0.05),这表明SQ22536可以减弱Fsk-IBMX对cAMP/PKA通路的影响,见图6。"
| [1] DE LA ROSA MB, KOZIK EM, SAKAGUCHI DS. Adult Stem Cell-Based Strategies for Peripheral Nerve Regeneration. Adv Exp Med Biol. 2018;1119:41-71. [2] MEENA P, KAKKAR A, KUMAR M, et al. Advances and clinical challenges for translating nerve conduit technology from bench to bed side for peripheral nerve repair. Cell Tissue Res. 2021;383(2):617-644. [3] PANAGOPOULOS GN, MEGALOIKONOMOS PD, MAVROGENIS AF. The Present and Future for Peripheral Nerve Regeneration. Orthopedics. 2017;40(1):e141-e156. [4] LI R, LIU Z, PAN Y, et al. Peripheral nerve injuries treatment: a systematic review. Cell Biochem Biophys. 2014;68(3):449-454. [5] SULLIVAN R, DAILEY T, DUNCAN K, et al. Peripheral Nerve Injury: Stem Cell Therapy and Peripheral Nerve Transfer. Int J Mol Sci. 2016;17(12):2101. [6] MIN Q, PARKINSON DB, DUN XP. Migrating Schwann cells direct axon regeneration within the peripheral nerve bridge. Glia. 2021;69(2):235-254. [7] HOPF A, SCHAEFER DJ, KALBERMATTEN DF, et al. Schwann Cell-Like Cells: Origin and Usability for Repair and Regeneration of the Peripheral and Central Nervous System. Cells. 2020;9(9):1990. [8] SAYAD FATHI S, ZAMINY A. Stem cell therapy for nerve injury. World J Stem Cells. 2017;9(9):144-151. [9] YI S, ZHANG Y, GU X, et al. Application of stem cells in peripheral nerve regeneration. Burns Trauma. 2020;8:tkaa002. [10] WAKAO S, HAYASHI T, KITADA M, et al. Long-term observation of auto-cell transplantation in non-human primate reveals safety and efficiency of bone marrow stromal cell-derived Schwann cells in peripheral nerve regeneration. Exp Neurol. 2010;223(2):537-547. [11] MATHOT F, SHIN AY, VAN WIJNEN AJ. Targeted stimulation of MSCs in peripheral nerve repair. Gene. 2019;710:17-23. [12] SOWA Y, KISHIDA T, TOMITA K, et al. Direct Conversion of Human Fibroblasts into Schwann Cells that Facilitate Regeneration of Injured Peripheral Nerve In Vivo. Stem Cells Transl Med. 2017;6(4):1207-1216. [13] ZHENG M, DUAN J, HE Z, et al. Transplantation of bone marrow stromal stem cells overexpressing tropomyosin receptor kinase A for peripheral nerve repair. Cytotherapy. 2017;19(8):916-926. [14] OLIVEIRA JT, MOSTACADA K, DE LIMA S, et al. Bone marrow mesenchymal stem cell transplantation for improving nerve regeneration. Int Rev Neurobiol. 2013;108:59-77. [15] GUO MG, LI DP, WU LX, et al. Bone marrow mesenchymal stem cells repair brachial plexus injury in rabbits through ERK pathway. Eur Rev Med Pharmacol Sci. 2020;24(3):1515-1523. [16] REN Z, WANG Y, PENG J, et al. Role of stem cells in the regeneration and repair of peripheral nerves. Rev Neurosci. 2012;23(2):135-143. [17] REISMAN M, ADAMS KT. Stem cell therapy: a look at current research, regulations, and remaining hurdles. P T. 2014;39(12):846-857. [18] 曹丽芝,冯乃波,王娟,等.间充质干细胞在周围神经损伤修复中的应用现状及前景分析[J].中国组织工程研究,2019,23(33):5385-5391. [19] WALSH S, MIDHA R. Use of stem cells to augment nerve injury repair. Neurosurgery. 2009;65(4 Suppl):A80-86. [20] LAVORATO A, RAIMONDO S, BOIDO M, et al. Mesenchymal Stem Cell Treatment Perspectives in Peripheral Nerve Regeneration: Systematic Review. Int J Mol Sci. 2021;22(2):572. [21] MUSAVI L, BRANDACHER G, HOKE A, et al. Muscle-derived stem cells: important players in peripheral nerve repair. Expert Opin Ther Targets. 2018;22(12):1009-1016. [22] COONEY DS, WIMMERS EG, IBRAHIM Z, et al. Mesenchymal Stem Cells Enhance Nerve Regeneration in a Rat Sciatic Nerve Repair and Hindlimb Transplant Model. Sci Rep. 2016;6:31306. [23] ZHANG L, SEITZ LC, ABRAMCZYK AM, et al. cAMP initiates early phase neuron-like morphology changes and late phase neural differentiation in mesenchymal stem cells. Cell Mol Life Sci. 2011;68(5):863-876. [24] SHAHBAZI A, SAFA M, ALIKARAMI F, et al. Rapid Induction of Neural Differentiation in Human Umbilical Cord Matrix Mesenchymal Stem Cells by cAMP-elevating Agents. Int J Mol Cell Med. 2016;5(3):167-177. [25] THOMPSON R, CASALI C, CHAN C. Forskolin and IBMX Induce Neural Transdifferentiation of MSCs Through Downregulation of the NRSF. Sci Rep. 2019; 9(1):2969. [26] LV P, WANG W, CAO Z, et al. Fsk and IBMX inhibit proliferation and proapoptotic of glioma stem cells via activation of cAMP signaling pathway. J Cell Biochem. 2019;120(1):321-331. [27] CALEBIRO D, DI DALMAZI G, BATHON K, et al. cAMP signaling in cortisol-producing adrenal adenoma. Eur J Endocrinol. 2015;173(4):M99-106. [28] STRATTON JA, KUMAR R, SINHA S, et al. Purification and Characterization of Schwann Cells from Adult Human Skin and Nerve. eNeuro. 2017;4(3): ENEURO.0307-16.2017. [29] KINGHAM PJ, KOLAR MK, NOVIKOVA LN, et al. Stimulating the neurotrophic and angiogenic properties of human adipose-derived stem cells enhances nerve repair. Stem Cells Dev. 2014;23(7):741-754. [30] GEORGIOU M, GOLDING JP, LOUGHLIN AJ, et al. Engineered neural tissue with aligned, differentiated adipose-derived stem cells promotes peripheral nerve regeneration across a critical sized defect in rat sciatic nerve. Biomaterials. 2015; 37:242-251. [31] TOMITA K, MADURA T, SAKAI Y, et al. Glial differentiation of human adipose-derived stem cells: implications for cell-based transplantation therapy. Neuroscience. 2013;236:55-65. [32] LI WY, ZHU GY, YUE WJ, et al. KLF7 overexpression in bone marrow stromal stem cells graft transplantation promotes sciatic nerve regeneration. J Neural Eng. 2019;16(5):056011. [33] KEILHOFF G, STANG F, GOIHL A, et al. Transdifferentiated mesenchymal stem cells as alternative therapy in supporting nerve regeneration and myelination. Cell Mol Neurobiol. 2006;26(7-8):1235-1252. [34] TOHILL M, MANTOVANI C, WIBERG M, et al. Rat bone marrow mesenchymal stem cells express glial markers and stimulate nerve regeneration. Neurosci Lett. 2004; 362(3):200-203. [35] CHEN CJ, OU YC, LIAO SL, et al. Transplantation of bone marrow stromal cells for peripheral nerve repair. Exp Neurol. 2007;204(1):443-453. [36] WANG Y, LI ZW, LUO M, et al. Biological conduits combining bone marrow mesenchymal stem cells and extracellular matrix to treat long-segment sciatic nerve defects. Neural Regen Res. 2015;10(6):965-971. [37] FAN L, YU Z, LI J, et al. Schwann-like cells seeded in acellular nerve grafts improve nerve regeneration. BMC Musculoskelet Disord. 2014;15:165. [38] JORI FP, NAPOLITANO MA, MELONE MA, et al. Molecular pathways involved in neural in vitro differentiation of marrow stromal stem cells. J Cell Biochem. 2005; 94(4):645-655. [39] DENG W, OBROCKA M, FISCHER I, et al. In vitro differentiation of human marrow stromal cells into early progenitors of neural cells by conditions that increase intracellular cyclic AMP. Biochem Biophys Res Commun. 2001;282(1): 148-152. [40] KRAMPERA M, MARCONI S, PASINI A, et al. Induction of neural-like differentiation in human mesenchymal stem cells derived from bone marrow, fat, spleen and thymus. Bone. 2007;40(2):382-390. [41] DEZAWA M, KANNO H, HOSHINO M, et al. Specific induction of neuronal cells from bone marrow stromal cells and application for autologous transplantation. J Clin Invest. 2004;113(12):1701-1710. [42] YANG L. Neuronal cAMP/PKA Signaling and Energy Homeostasis. Adv Exp Med Biol. 2018;1090:31-48. [43] WANG L, HAN L, XUE P, et al. Dopamine suppresses osteoclast differentiation via cAMP/PKA/CREB pathway. Cell Signal. 2021;78:109847. [44] JIN P, DENG S, TIAN M, et al. INT-777 prevents cognitive impairment by activating Takeda G protein-coupled receptor 5 (TGR5) and attenuating neuroinflammation via cAMP/ PKA/ CREB signaling axis in a rat model of sepsis. Exp Neurol. 2021;335: 113504. [45] XIE W, LI F, HAN Y, et al. Neuropeptide Y1 receptor antagonist promotes osteoporosis and microdamage repair and enhances osteogenic differentiation of bone marrow stem cells via cAMP/PKA/CREB pathway. Aging (Albany NY). 2020; 12(9):8120-8136. [46] NAKAGAWA S, KIM JE, LEE R, et al. Localization of phosphorylated cAMP response element-binding protein in immature neurons of adult hippocampus. J Neurosci. 2002;22(22):9868-9876. [47] NAKAGAWA S, KIM JE, LEE R, et al. Regulation of neurogenesis in adult mouse hippocampus by cAMP and the cAMP response element-binding protein. J Neurosci. 2002;22(9):3673-3682. |
| [1] | Wang Baojuan, Zheng Shuguang, Zhang Qi, Li Tianyang. Miao medicine fumigation can delay extracellular matrix destruction in a rabbit model of knee osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1180-1186. |
| [2] | Xiao Hao, Liu Jing, Zhou Jun. Research progress of pulsed electromagnetic field in the treatment of postmenopausal osteoporosis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1266-1271. |
| [3] | Gao Yujin, Peng Shuanglin, Ma Zhichao, Lu Shi, Cao Huayue, Wang Lang, Xiao Jingang. Osteogenic ability of adipose stem cells in diabetic osteoporosis mice [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 999-1004. |
| [4] | An Weizheng, He Xiao, Ren Shuai, Liu Jianyu. Potential of muscle-derived stem cells in peripheral nerve regeneration [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1130-1136. |
| [5] | Tian Chuan, Zhu Xiangqing, Yang Zailing, Yan Donghai, Li Ye, Wang Yanying, Yang Yukun, He Jie, Lü Guanke, Cai Xuemin, Shu Liping, He Zhixu, Pan Xinghua. Bone marrow mesenchymal stem cells regulate ovarian aging in macaques [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 985-991. |
| [6] | Hou Jingying, Guo Tianzhu, Yu Menglei, Long Huibao, Wu Hao. Hypoxia preconditioning targets and downregulates miR-195 and promotes bone marrow mesenchymal stem cell survival and pro-angiogenic potential by activating MALAT1 [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1005-1011. |
| [7] | Liang Xuezhen, Yang Xi, Li Jiacheng, Luo Di, Xu Bo, Li Gang. Bushen Huoxue capsule regulates osteogenic and adipogenic differentiation of rat bone marrow mesenchymal stem cells via Hedgehog signaling pathway [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1020-1026. |
| [8] | Wang Jifang, Bao Zhen, Qiao Yahong. miR-206 regulates EVI1 gene expression and cell biological behavior in stem cells of small cell lung cancer [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1027-1031. |
| [9] | Fang Xiaolei, Leng Jun, Zhang Chen, Liu Huimin, Guo Wen. Systematic evaluation of different therapeutic effects of mesenchymal stem cell transplantation in the treatment of ischemic stroke [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1085-1092. |
| [10] | Tang Jiping, Zhang Yeting. Exercise regulates adult hippocampal neurogenesis in Alzheimer’s disease: mechanism and role [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(5): 798-803. |
| [11] | Fan Jianchao, Xu Paidi, Han Yongli, Wen Caiyuzhu, Zhang Hongxing, Pan Xiaoli. Effects of electroacupuncture at Zusanli acupoint on visceral hypersensitivity in rats with functional dyspepsia [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(5): 663-668. |
| [12] | Mo Weibin, Huang Tianchang, Zeng Zhiwei, Yan Linbo. Effects of Pueraria lobata flavonoids on expressions of beta-catenin and glycogen synthase kinase 3beta in the brain of rats undergoing exhaustive exercise after long endurance exercise [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(5): 736-741. |
| [13] | Li Jiajun, Xia Tian, Liu Jiamin, Chen Feng, Chen Haote, Zhuo Yinghong, Wu Weifeng. Molecular mechanism by which icariin regulates osteogenic signaling pathways in the treatment of steroid-induced avascular necrosis of the femoral head [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(5): 780-785. |
| [14] | Liu Jin, Li Zhen, Hao Huiqin, Wang Ze, Zhao Caihong, Lu Wenjing. Ermiao san aqueous extract regulates proliferation, migration, and inflammatory factor expression of fibroblast-like synovial cells in collagen-induced arthritis rats [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(5): 688-693. |
| [15] | Li Weiming, Xu Qingwen, Li Yijun, Sun Yanbo, Cui Jin, Xu Pengyuan . Deep seawater promotes wound healing in diabetic mice by activating PI3K/Akt pathway [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(5): 724-729. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||