Chinese Journal of Tissue Engineering Research ›› 2022, Vol. 26 ›› Issue (10): 1610-1614.doi: 10.12307/2022.211
Previous Articles Next Articles
Application advantages of hydroxyapatite surface modification as a bone scaffold for tissue engineering
Hou Jianfei, Wang Fuke, Yang Guiran, Liao Xinyu, Jia Di, Liu Dejian
- Department of Sports Medicine, First Affiliated Hospital of Kunming Medical University, Kunming 650032, Yunnan Province, China
-
Received:2020-12-02Revised:2020-12-04Accepted:2021-01-07Online:2022-04-08Published:2021-10-27 -
Contact:Wang Fuke, MD, Professor, Associate chief physician, Master’s supervisor, Department of Sports Medicine, First Affiliated Hospital of Kunming Medical University, Kunming 650032, Yunnan Province, China -
About author:Hou Jianfei, Master candidate, Department of Sports Medicine, First Affiliated Hospital of Kunming Medical University, Kunming 650032, Yunnan Province, China -
Supported by:the Joint Special Fund for Applied Basic Research of Kunming Medical University, No. 201701UH00095 (to WFK); Clinical Medical Center Construction Statement of Yunnan Provincial Health Commission, No. 2X2019-03-04 (to WFK)
CLC Number:
Cite this article
Hou Jianfei, Wang Fuke, Yang Guiran, Liao Xinyu, Jia Di, Liu Dejian. Application advantages of hydroxyapatite surface modification as a bone scaffold for tissue engineering[J]. Chinese Journal of Tissue Engineering Research, 2022, 26(10): 1610-1614.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
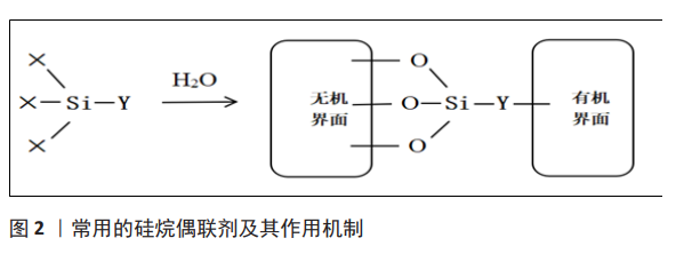
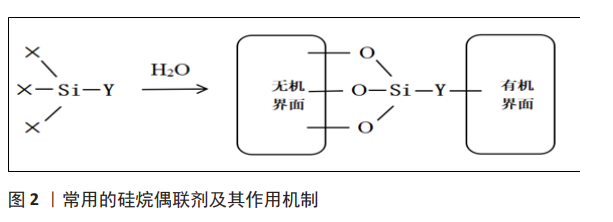
程利等[14]使用硬脂酸对纳米羟基磷灰石表面改性后与聚乳酸复合,硬脂酸的羧基与羟基磷灰石表面的 Ca2+之间发生静电相互作用,可以提高羟基磷灰石纳米粒子与聚乳酸之间界面的结合程度,扫描电镜下观察发现表面改性后的羟基磷灰石与复合材料的界面更模糊,在基体中的分散更均匀。添加适宜的表面活性剂可以使羟基磷灰石颗粒聚集现象显著改善,而材料的性能很大程度是由其微观结构决定,因此改性后羟基磷灰石复合材料的弹性模量、弯曲强度和抗冲击性能等力学性能及生物学性能都有所提高。DING等[15]采用12-羟基硬脂酸表面改性纳米羟基磷灰石并制备羟基磷灰石/聚乳酸-羟基乙酸共聚物复合材料,12-羟基硬脂酸的疏水烃链可改善羟基磷灰石在聚乳酸-羟基乙酸共聚物基体中的分散,结果表明羟基磷灰石表现出较好的分散性,复合材料的拉伸强度等力学性能显著提高,特别是添加了质量分数5%纳米羟基磷灰石后其抗拉强度比纯聚乳酸-羟基乙酸共聚物提高了23.81%,体外细胞培养实验结果表明复合材料具有较好的细胞生物相容性。GHEYSARI等[16]采用十二烷基硫酸钠对羟基磷灰石纳米结构进行改性,并制备了明胶/羧甲基纤维素/羟基磷灰石的高孔纳米复合材料,结果显示羟基磷灰石的尺寸为15-25 nm(直径)和100-150 nm(长度),结晶度为27%,并且发现细胞的附着和黏附得到有效改善。 由于溴化十六烷三甲基铵的CTA+基团能够与羟基磷灰石的PO43-形成化学键,故李沃等[17]采用溴化十六烷三甲基铵、聚乙二醇和人血清对羟基磷灰石纳米颗粒进行表面改性,结果显示,溴化十六烷三甲基铵表面改性的羟基磷灰石纳米颗粒具有更好的分散性和悬浮稳定性,与带负电荷的物质结合能力更强。YAHIA等[18]采用两种表面活性剂溴化十六烷三甲基铵和十二烷基硫酸钠对微波法制备的羟基磷灰石进行表面改性,扫描电镜结果表明羟基磷灰石纳米棒的尺寸在 5-10 nm范围内,并且使用表面活性剂合成的羟基磷灰石具有良好的与蛋白质结合的能力。 不同形状的羟基磷灰石颗粒取决于阳离子和阴离子表面活性剂的浓度,阳离子表面活性剂浓度较高时颗粒呈片状排列,而阴离子表面活性剂浓度较高时颗粒呈棒状,但这种方法制备的纳米颗粒表面积小,孔径不均匀。为了克服这些问题,研究者们尝试使用非离子表面活性剂,取得了较好效果。非离子型表面活性剂在水中不电离,但是具有可与羟基磷灰石表面 Ca2+络合的基团,如氧乙烯基(-CH2CH2O-)、醚基(-O-)、羟基(-OH)或酰胺基(-CONH2),可与羟基磷灰石发生相互作用,加强羟基磷灰石与基体的联系,从而增进二者之间的相容性。周尧等[19]利用十二烷基醇改性纳米羟基磷灰石制备了羟基磷灰石/聚乳酸复合材料,结果发现羟基磷灰石在复合材料中的聚集程度更低,分散也更均匀,而且复合材料的弯曲强度、拉伸强度和冲击强度均高于未经改性的羟基磷灰石/聚乳酸复合材料。李刚等[20]采用非离子表面活性剂聚乙二醇、聚乙烯醇对羟基磷灰石进行表面改性,结果表明聚乙烯醇对羟基磷灰石的分散性优于聚乙二醇,且聚乙烯醇的添加量为3%时羟基磷灰石的结晶程度趋于完善,羟基磷灰石粉末的团聚程度最小,比表面积最大,为607.4 m2/kg。ELBASUNEY等[21]以聚乙烯-丙烯酸共聚物表面改性羟基磷灰石纳米片,研究表明表面改性剂使纳米羟基磷灰石的表面性质从亲水性完全变为疏水性,提高了纳米颗粒在壳聚糖、胶原、聚乙二醇等生物相容性聚合物中的相容性和分散性,其力学性能也得到提高。 2.2 硅烷偶联剂 硅烷偶联剂具有特殊的两亲性,它的一些分子与有机物有亲和力,而另一些分子与无机物有亲和力,羟基磷灰石的表面是亲水的,而有机材料的表面是疏水的,因此硅烷偶联剂可以充当羟基磷灰石和有机材料之间的“分子桥”,将羟基磷灰石颗粒的亲水性表面转变为疏水性有机活性表面,有助于改善复合材料之间的界面附着力,还促进了颗粒在有机基质中的均匀分布,从而制备出性能优良的复合材料[22]。硅烷偶联剂的通式可以表示为Y-R-Si-X3,其中Y代表能够与聚合物分子有亲和性的活性基团,如氨基、烷烃、乙烯基等;X代表能够水解的基团,如甲氧基、乙氧基、氯基等[23],当这些基团被水解时,它们可以产生硅醇与无机相结合并形成硅氧烷,见图2。硅烷偶联剂按可水解基团的不同分为甲氧基硅烷和乙氧基硅烷,根据有机基团的不同还可分为乙烯基、氨基、环氧树脂等[24]。"
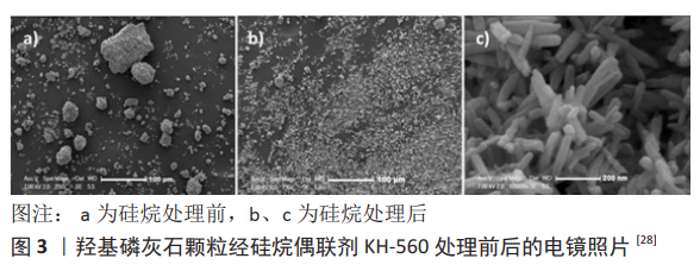
WANG等[25]采用γ-(2,3-环氧丙氧基)丙基三甲氧基硅烷(KH-560)对羟基磷灰石纳米粒子进行改性,KH560可以通过甲氧基与羟基磷灰石的羟基水解缩合对羟基磷灰石表面进行改性,通过注射成型法制备了纳米羟基磷灰石/聚醚醚酮复合材料,扫描电镜观察表明纳米颗粒在基体中分布良好,复合材料的柔韧性、抗冲击性能和机械强度得到明显提高。胡雅菲等[26]采用共沉淀法制备纳米羟基磷灰石,并用KH560对其进行表面改性,结果表明,改性羟基磷灰石在聚醚醚酮基体中分散均匀,与纯聚醚醚酮相比,10%改性羟基磷灰石/聚醚醚酮复合材料的储能模量增加了55.56%,玻璃化温度增加了3.6 ℃,磨痕深度降低了31.1%,拉伸强度为68.33 MPa,能够满足人骨的强度要求。MA等[27]也采用KH560改性纳米羟基磷灰石,通过热压成型法制备羟基磷灰石/聚醚醚酮复合材料,结果表明,复合材料的抗拉强度随羟基磷灰石含量的增加呈现先增大后减小的趋势,当羟基磷灰石含量为5%时复合材料的抗拉强度达到最大值,比纯聚醚醚酮试样的抗拉强度高23%,并且复合材料周围骨组织的生长情况优于其他羟基磷灰石含量的样品。?NER等[28]使用KH-560作为桥梁连接羟基磷灰石和聚羟基丁酸戊酯,电镜观察结果表明,经硅烷处理的羟基磷灰石纳米颗粒具有良好的纳米分散性能,见图3,并且硅烷化可以改善聚羟基丁酸戊 酯/羟基磷灰石的结合,提高其机械强度。 YADAV等[29]分别制备了γ-巯丙基三甲氧基硅烷和γ-氨丙基三乙氧基硅烷处理的纳米羟基磷灰石复合材料,研究发现,两种偶联剂对羟基磷灰石良好的分散性使纳米羟基磷灰石的表面积体积比增加、聚集减少,孔隙含量也随之增加,并且经硅烷偶联剂处理的复合材料显示出更好的硬度。MICHAEL等[30]采用γ-氨丙基三乙氧基硅烷、十二烷基硫酸钠和聚乙烯亚胺3种不同的表面改性剂对纳米羟基磷灰石进行改性,通过熔融混合制备了纳米羟基磷灰石和聚乳酸复合材料,结果表明γ-氨丙基三乙氧基硅烷表面改性制备的复合材料拉伸性能最佳;此外,当改性后的羟基磷灰石添加量增加到30%时,由于聚集作用拉伸强度降低。 经硅烷偶联剂改性的羟基磷灰石与基质之间的界面附着力增强,在一定程度上减少了羟基磷灰石的聚集,促进了颗粒在有机基质中的均匀分布,对复合材料的力学性能特别是机械强度和拉伸性能有显著影响。KARADI等[31]采用γ-甲基丙烯酰氧基丙基三甲氧基硅烷表面改性羟基磷灰石纳米粒子,然后与甲基丙烯酸甲酯混合制备聚合物,研究表明,γ-甲基丙烯酰氧基丙基三甲氧基硅烷处理增强了纳米粒子的分散性和与甲基丙烯酸甲酯基体的界面结合,提高了复合材料的抗冲击强度、表面硬度,并对其吸水性和溶解性有积极的影响。JARAMILLO等[32]采用γ-氨丙基三乙氧基硅烷表面改性羟基磷灰石,然后与环氧树脂混合制备聚合物,结果表明,改性羟基磷灰石实现了在环氧树脂基体中的稳定分散,提高了复合材料的力学性能。 2.3 接枝聚合物改性 接枝是通过共价键将2个单体连接起来的方法,羟基磷灰石表面具有与自由基发生反应的羟基,在适当的条件下聚合物单体可在这些羟基上反应并接枝于表面,可以使羟基磷灰石微粒聚集程度显著降低、分散稳定性增加,并且可以赋予羟基磷灰石更多新的特性[33-34]。常用于表面接枝的聚合物包括甲基丙烯酸、乙烯基膦酸、聚乙烯和聚氨酯等,在羟基磷灰石/聚合物复合材料有着广泛的应用[35]。 羟基磷灰石表面的活性羟基可以直接与聚合物单体进行接枝作用,如TIMPU等[36]分别使用精氨酸和聚乙烯亚胺接枝处理纳米羟基磷灰石,研究结果发现,经10%聚乙烯亚胺处理的纳米羟基磷灰石类似于天然骨成分(30-50 nm长, 15-30 nm宽),将处理后的羟基磷灰石引入胶原-透明质酸复合材料中,压缩模量较与未加填充物的聚合物基质相比提高了6倍。WANG等[37]将乳酸低聚物接枝到纳米羟基磷灰石,将接枝的纳米羟基磷灰石与聚乳酸共混制备得到纳米羟基磷灰石/聚乳酸支架,研究结果表明,羟基磷灰石粒子在复合材料中的分散更均匀,复合材料的界面相容性、力学性能和生物相容性均有改善。KUMAR等[38]采用乙烯基膦酸对纳米羟基磷灰石颗粒进行表面接枝改性,并用发泡法制备了多孔改性纳米羟基磷灰石/聚氨酯复合骨工程材料,结果表明,改性后的羟基磷灰石颗粒表面完全由颗粒结构转变为平板结构,粒径为40 nm;随着羟基磷灰石的浓度从0%增加到30%,复合材料的抗压强度从0.094 MPa增加到22.4 MPa;细胞培养研究表明,纳米羟基磷灰石/聚氨酯复合支架非常适合在骨组织工程中应用。LIUYUN等[39]使用L-赖氨酸接枝纳米羟基磷灰石,并制备了纳米羟基磷灰石/聚乳酸-羟基乙酸复合材料,结果表明,随着L-赖氨酸含量的增加,纳米羟基磷灰石的分散性进一步改善,纳米羟基磷灰石与聚乳酸-羟基乙酸复合材料具有更好的生物相容性、力学性能和体外生物活性。 羟基磷灰石表面的活性羟基数量和反应性有限,因此常通过偶联剂改性在羟基磷灰石上带上不同的反应性官能团,通过这些官能团进一步接枝不同的聚合物。蒋立新等[40]利用γ-氨丙基三乙氧基硅烷和L-丙交酯联合接枝处理纳米羟基磷灰石,然后将其与聚乳酸-羟基乙酸以不同比例混合得到复合材料,γ-氨丙基三乙氧基硅烷的-NH2引发丙交酯开环聚合,结果表明,接枝处理后的纳米粒子在基体中分散均匀,提高了复合材料结晶能力和力学性能,当羟基磷灰石添加量为10%时,复合材料的抗弯强度和拉伸强度分别比未接枝处理的复合材料提高14.4%和11.3%。REHMAN等[41]用γ-氨丙基三乙氧基硅烷接枝纳米羟基磷灰石,然后与琥珀酸酐进一步功能化得到复合材料,结果表明,功能化后的纳米羟基磷灰石已经转化为高度分散的球形颗粒,并且细胞活力测试表明复合材料具有良好的生物相容性。李瑞端等[42]在羟基磷灰石表面引入活性醛基,通过活性醛基接枝到羟基磷灰石表面,研究表明制备的聚乳酸/羟基磷灰石复合材料的界面相容性得到改善,再将经聚乳酸接枝的羟基磷灰石与羟基乙酸共混制备复合材料,该材料的拉伸强度、弯曲强度和吸水率分别为52.62 MPa、87.348 MPa和2.9%。"
| [1] BEURIAT PA, LOHKAMP LN, SZATHMARI A, et al. Repair of Cranial Bone Defects in Children Using Synthetic Hydroxyapatite Cranioplasty (CustomBone). World Neurosurg. 2019;129(9):e104-e113. [2] 陈涛,付海洋,李岩,等.羟基磷灰石基复合骨修复材料研究进展[J].中国药事,2019,33(3):302-309. [3] JEONG J, KIM JH, SHIM JH, et al. Bioactive calcium phosphate materials and applications in bone regeneration. Biomater Res. 2019;23(1):1-11. [4] LI N, WU G, YAO H, et al. Size effect of nano-hydroxyapatite on proliferation of odontoblast-like MDPC-23 cells. Dent Mater J. 2019;38(4):534-539. [5] LU HT, LU TW, CHEN CH, et al. Development of genipin-crosslinked and fucoidan-adsorbed nano-hydroxyapatite/hydroxypropyl chitosan composite scaffolds for bone tissue engineering. Int J Biol Macromol. 2019;128(5):973-984. [6] 余和东,张丽,夏凌云,等.3D打印成型纳米羟基磷灰石/壳聚糖/聚己内酯三元复合支架材料的构建及表征[J]. 中国组织工程研究,2020, 24(10):1496-1501. [7] MONTALBANO G, MOLINO G, FIORILLI S, et al. Synthesis and incorporation of rod-like nano-hydroxyapatite into type I collagen matrix: A hybrid formulation for 3D printing of bone scaffolds. J Eur Ceram Soc. 2020;40(11): 3689-3697. [8] SALEEM M, RASHEED S, YOUGEN C. Silk fibroin/hydroxyapatite scaffold: a highly compatible material for bone regeneration. Sci Technol Adv Mater. 2020;21(1):242-266. [9] FENG P, HE J, PENG S, et al. Characterizations and interfacial reinforcement mechanisms of multicomponent biopolymer based scaffold. Mater Sci Eng C Mater Biol Appl. 2019;100(7):809-825. [10] DIAO H, SI Y, ZHU A, et al. Surface modified nano-hydroxyapatite/poly (lactide acid) composite and its osteocyte compatibility. Mater Sci Eng C Mater Biol Appl. 2012;32(7):1796-1801. [11] TURON P, DEL VALLE LJ, ALEMÁN C, et al. Grafting of Hydroxyapatite for Biomedical Applications. Biopolym Graft. 2018:45-80. [12] YANG X. Experimental study on removal of Cu2+ in water by nano-modified hydroxyapatite. IOP Conf Ser: Earth Environ Sci. 2019;227(6):028-062. [13] MOLINO G, PALMIERI MC, MONTALBANO G, et al. Biomimetic and mesoporous nano-hydroxyapatite for bone tissue application: a short review. Biomed Mater. 2020;15(2):001-022. [14] 程利,王鑫,赵雄燕.功能化纳米羟基磷灰石改性聚乳酸复合材料的研究[J].应用化工,2019,48(7):1595-1597. [15] DING HJ, TANG S, JIANG LY, et al. Study on the surface-modification for nano-hydroxyapatite with 12-hydroxystearic acid and the reinforcing effect for poly (lactic-o-glycolide). Express Polym Lett. 2020;14(8):741-756. [16] GHEYSARI H, MOHANDES F, MAZAHERI M, et al. Extraction of Hydroxyapatite Nanostructures from Marine Wastes for the Fabrication of Biopolymer-Based Porous Scaffolds. Mar Drugs. 2019;18(1):26. [17] 李沃,黄东,胡霞,等.三种不同表面活性剂对羟基磷灰石纳米颗粒的表面修饰的比较[J].现代生物医学进展,2016,16(11):2013-2017+2053. [18] YAHIA IS, KESHK SMAS, SHKIR M, et al. An Effect of Cationic CTAB and Anionic SDS Surfactants on the Physicochemical Properties of Hydroxyapatite Nanostructures for Bone Tissue Engineering. J Nanoelectron Optoe. 2020; 15(3):316-324. [19] 周尧,赵雄燕,孙占英,等.改性纳米羟基磷灰石增强聚乳酸复合材料的研究[J].应用化工,2017,46(12):2350-2353. [20] 李刚,梁彦会,郭玉洁,等.非离子表面活性剂改性羟基磷灰石粉体的制备[J].硅酸盐通报,2019,38(2):559-562+579. [21] ELBASUNEY S. Green Synthesis of Hydroxyapatite Nanoparticles with Controlled Morphologies and Surface Properties Toward Biomedical Applications. J Inorg Organomet Polym Mater. 2020;30(3):899-906. [22] 王雅珍,张雪泽,狄语韬.近年硅烷偶联剂在聚合物改性中的研究进展及应用[J].化工新型材料,2018,46(11):5-7+12. [23] SHUAI C, YU L, FENG P, et al. Interfacial reinforcement in bioceramic/biopolymer composite bone scaffold: the role of coupling agent. Colloids Surf B Biointerfaces. 2020;193:083-111. [24] 王岩,肖艳,郎美东.羟基磷灰石表面改性的研究进展[J].化学通报, 2011,74(9):783-790. [25] WANG L, WENG L, WU Z, et al. The Properties of Polyetheretherketone Biocomposite Reinforced by Surface-Modified Nano-Hydroxyapatite. Adv Mater Res. 2015;1096:214-218. [26] 胡雅菲,单英杰,邱丽,等.羟基磷灰石的表面改性对羟基磷灰石/聚醚醚酮复合材料力学和摩擦性能的影响[J].复合材料学报,2018,35(10): 2651-2657. [27] MA R, LI Q, WANG L, et al. Mechanical properties and in vivo study of modified-hydroxyapatite/polyetheretherketone biocomposites. Mater Sci Eng C Mater Biol Appl. 2017;73:429-439. [28] Öner M, İlhan B. Fabrication of poly (3-hydroxybutyrate-co-3-hydroxyvalerate) biocomposites with reinforcement by hydroxyapatite using extrusion processing. Mater Sci Eng C Mater Biol Appl. 2016;65:19-26. [29] YADAV S, GANGWAR S. In vitro wear characteristics of a nano-hydroxyapatite filled dental resin composite under two-body wear condition. J Mater Sci. 2020;55:10202-10220. [30] MICHAEL FM, RATNAM CT, KHALID M, et al. Surface modification of nanohydroxyapatite and its loading effect on polylactic acid properties for load bearing implants. Polym Composite. 2018;39(8):2880-2888. [31] KARADI RH, HUSSEIN B. Effect of Modified Nano Hydroxyapatite Fillers Addition on Some Properties of Heat Cured Acrylic Denture Base Materials. J Baghdad Coll Dent. 2017;29(2):49-54 [32] JARAMILLO AF, MEDINA C, FLORES P, et al. Improvement of thermomechanical properties of composite based on hydroxyapatite functionalized with alkylsilanes in epoxy matrix. Ceram Int. 2020;46(6): 8368-8378. [33] MICHAEL F M, KHALID M, WALVEKAR R, et al. Surface modification techniques of biodegradable and biocompatible polymers. Biodegrad Biocompatible Polym Compos. 2018;2:33-54. [34] LEE HJ, CHOI HW, KIM KJ, et al. Modification of hydroxyapatite nanosurfaces for enhanced colloidal stability and improved interfacial adhesion in nanocomposites. Chem Mater. 2006;18(21):5111-5118. [35] ANWAR A, REHMAN IU, DARR JA. Low-temperature synthesis and surface modification of high surface area calcium hydroxyapatite nanorods incorporating organofunctionalized surfaces. J Phys Chem C. 2016;120(51):29069-29076. [36] TIMPU D, SACARESCU L, VASILIU T, et al. Surface cationic functionalized nano-hydroxyapatite–preparation, characterization, effect of coverage on properties and related applications. Eur Polym J. 2020;132(5):109759. [37] WANG Z, XU Y, WANG Y, et al. Enhanced in vitro mineralization and in vivo osteogenesis of composite scaffolds through controlled surface grafting of L-lactic acid oligomer on nanohydroxyapatite. Biomacromolecules. 2016; 17(3):818-829. [38] KUMAR L, AHUJA D. Preparation and characterization of aliphatic polyurethane and modified hydroxyapatite composites for bone tissue engineering. Polym Bull. 2019;77:1-14. [39] LIUYUN J, LIXIN J, CHENGDONG X, et al. Effect of l-lysine-assisted surface grafting for nano-hydroxyapatite on mechanical properties and in vitro bioactivity of poly (lactic acid-co-glycolic acid). J Biomater Appl. 2016;30(6): 750-758. [40] 蒋立新,蒋柳云,马驰,等.新型改性的n-HA与PLGA复合材料的制备及性能研究[J].无机材料学报,2013,28(7):751-756. [41] REHMAN S, KHAN K, MUJAHID M, et al. Synthesis of nano-hydroxyapatite and its rapid mediated surface functionalization by silane coupling agent. Mater Sci Eng C Mater Biol Appl. 2016;58:675-681. [42] 李瑞端,张洪彬,戴传波,等.功能聚乳酸改性羟基磷灰石有机无机复合材料的制备[J].科学技术与工程,2017,17(19):99-102. |
| [1] | Li Rui, Liu Zhen, Guo Zige, Lu Ruijie, Wang Chen. Aspirin-loaded chitosan nanoparticles and polydopamine modified titanium sheets improve osteogenic differentiation [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(3): 374-379. |
| [2] | Jin Tao, Liu Lin, Zhu Xiaoyan, Shi Yucong, Niu Jianxiong, Zhang Tongtong, Wu Shujin, Yang Qingshan. Osteoarthritis and mitochondrial abnormalities [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1452-1458. |
| [3] | Zhang Lichuang, Xu Hao, Ma Yinghui, Xiong Mengting, Han Haihui, Bao Jiamin, Zhai Weitao, Liang Qianqian. Mechanism and prospects of regulating lymphatic reflux function in the treatment of rheumatoid arthritis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1459-1466. |
| [4] | Yao Xiaoling, Peng Jiancheng, Xu Yuerong, Yang Zhidong, Zhang Shuncong. Variable-angle zero-notch anterior interbody fusion system in the treatment of cervical spondylotic myelopathy: 30-month follow-up [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1377-1382. |
| [5] | Zhu Chan, Han Xuke, Yao Chengjiao, Zhou Qian, Zhang Qiang, Chen Qiu. Human salivary components and osteoporosis/osteopenia [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1439-1444. |
| [6] | Wang Jing, Xiong Shan, Cao Jin, Feng Linwei, Wang Xin. Role and mechanism of interleukin-3 in bone metabolism [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1260-1265. |
| [7] | Zhu Chan, Han Xuke, Yao Chengjiao, Zhang Qiang, Liu Jing, Shao Ming. Acupuncture for Parkinson’s disease: an insight into the action mechanism in animal experiments [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1272-1277. |
| [8] | Guo Jia, Ding Qionghua, Liu Ze, Lü Siyi, Zhou Quancheng, Gao Yuhua, Bai Chunyu. Biological characteristics and immunoregulation of exosomes derived from mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1093-1101. |
| [9] | Wu Weiyue, Guo Xiaodong, Bao Chongyun. Application of engineered exosomes in bone repair and regeneration [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1102-1106. |
| [10] | Zhou Hongqin, Wu Dandan, Yang Kun, Liu Qi. Exosomes that deliver specific miRNAs can regulate osteogenesis and promote angiogenesis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1107-1112. |
| [11] | Zhang Jinglin, Leng Min, Zhu Boheng, Wang Hong. Mechanism and application of stem cell-derived exosomes in promoting diabetic wound healing [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1113-1118. |
| [12] | Huang Chenwei, Fei Yankang, Zhu Mengmei, Li Penghao, Yu Bing. Important role of glutathione in stemness and regulation of stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1119-1124. |
| [13] | Hui Xiaoshan, Bai Jing, Zhou Siyuan, Wang Jie, Zhang Jinsheng, He Qingyong, Meng Peipei. Theoretical mechanism of traditional Chinese medicine theory on stem cell induced differentiation [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1125-1129. |
| [14] | An Weizheng, He Xiao, Ren Shuai, Liu Jianyu. Potential of muscle-derived stem cells in peripheral nerve regeneration [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1130-1136. |
| [15] | Fan Yiming, Liu Fangyu, Zhang Hongyu, Li Shuai, Wang Yansong. Serial questions about endogenous neural stem cell response in the ependymal zone after spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1137-1142. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||

