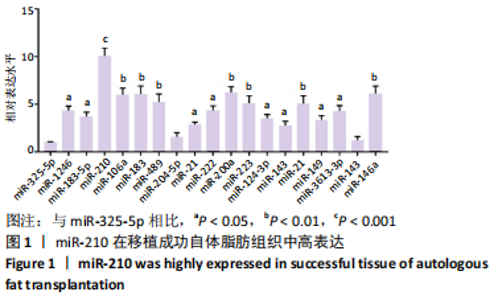
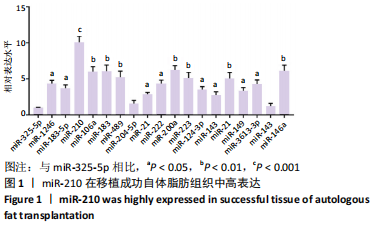
Chinese Journal of Tissue Engineering Research ›› 2022, Vol. 26 ›› Issue (1): 59-63.doi: 10.12307/2022.010
Previous Articles Next Articles
miR-210/YWHAG axis with adipocyte proliferation and angiogenesis after autologous fat transplantation
Yao Yanyi, Chen Xiaoling, Zhang Min, Wang Hong, Hao Liying, Shi Chao, Zhao Bingfeng, Jin Yaqiao, Dong Wei
- Department of Medical Aesthetics, Baotou Eighth Hospital, Baotou 014000, Inner Mongolia Autonomous Region, China
-
Received:2020-07-16Revised:2020-07-18Accepted:2020-09-19Online:2022-01-08Published:2021-10-23 -
Contact:Dong Wei, Master, Physician, Department of Medical Aesthetics, Baotou Eighth Hospital, Baotou 014000, Inner Mongolia Autonomous Region, China -
About author:Yao Yanyi, Master, Chief physician, Department of Medical Aesthetics, Baotou Eighth Hospital, Baotou 014000, Inner Mongolia Autonomous Region, China
CLC Number:
Cite this article
Yao Yanyi, Chen Xiaoling, Zhang Min, Wang Hong, Hao Liying, Shi Chao, Zhao Bingfeng, Jin Yaqiao, Dong Wei. miR-210/YWHAG axis with adipocyte proliferation and angiogenesis after autologous fat transplantation[J]. Chinese Journal of Tissue Engineering Research, 2022, 26(1): 59-63.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
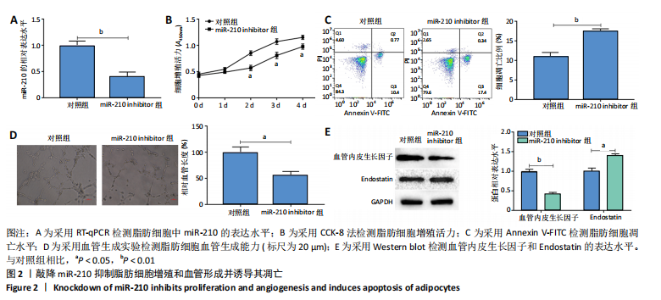
2.2 敲降miR-210抑制脂肪细胞增殖和血管形成并诱导其凋亡 RT-qPCR结果显示,敲降miR-210显著下调miR-210的表达水平(P < 0.01),见图2A;CCK-8结果显示,与对照组相比,敲降miR-210显著下调脂肪细胞的增殖活力(P < 0.05),见图2B;Annexin V-FITC/PI结果显示,敲降miR-210后,脂肪细胞凋亡水平显著高于对照组(P < 0.01),见图2C;血管生成实验结果显示,与对照组相比,敲降miR-210显著抑制脂肪细胞血管生成(P < 0.05),见图2D;Western blot结果显示,与对照组相比,敲降miR-210显著抑制血管内皮生长因子的表达水平(P < 0.01),促进血管生成抑制因子Endostatin的表达水平(P < 0.05),见图2E。综上,敲降miR-210抑制脂肪细胞增殖和血管形成并诱导其凋亡。"
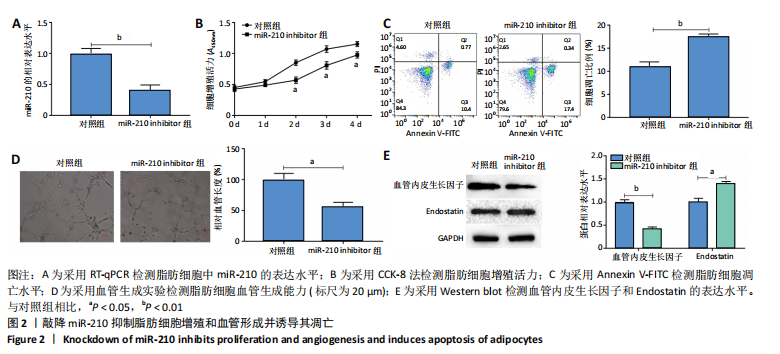
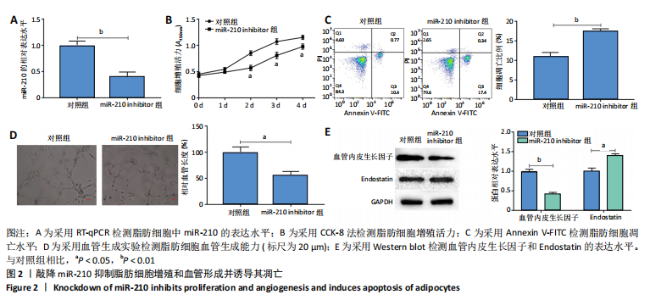
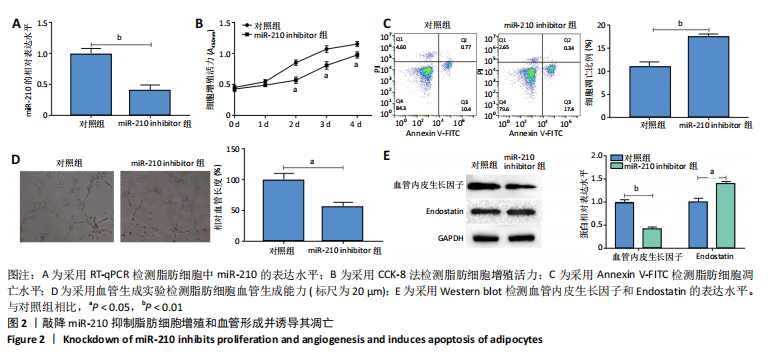
2.2 敲降miR-210抑制脂肪细胞增殖和血管形成并诱导其凋亡 RT-qPCR结果显示,敲降miR-210显著下调miR-210的表达水平(P < 0.01),见图2A;CCK-8结果显示,与对照组相比,敲降miR-210显著下调脂肪细胞的增殖活力(P < 0.05),见图2B;Annexin V-FITC/PI结果显示,敲降miR-210后,脂肪细胞凋亡水平显著高于对照组(P < 0.01),见图2C;血管生成实验结果显示,与对照组相比,敲降miR-210显著抑制脂肪细胞血管生成(P < 0.05),见图2D;Western blot结果显示,与对照组相比,敲降miR-210显著抑制血管内皮生长因子的表达水平(P < 0.01),促进血管生成抑制因子Endostatin的表达水平(P < 0.05),见图2E。综上,敲降miR-210抑制脂肪细胞增殖和血管形成并诱导其凋亡。"
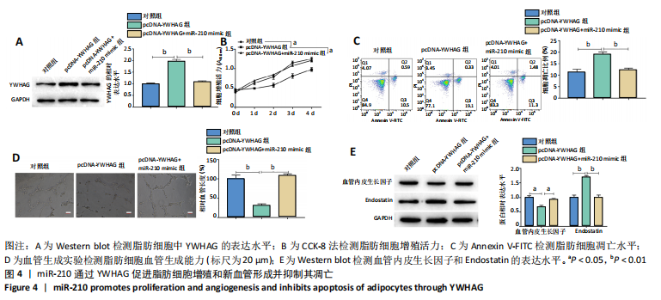
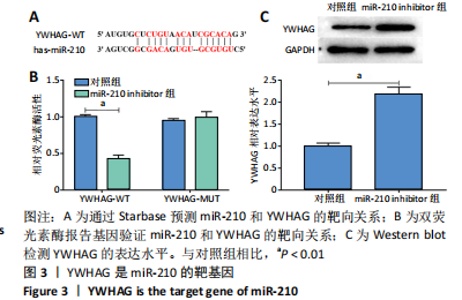
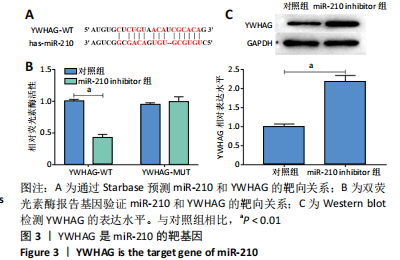
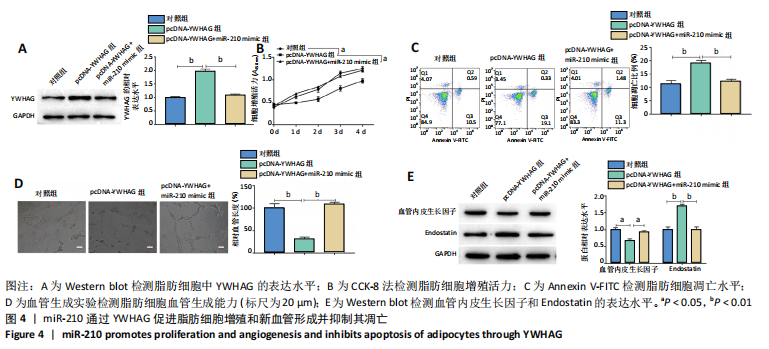
2.4 miR-210通过YWHAG促进脂肪细胞增殖和新血管形成并抑制其凋亡 Western blot结果显示,与对照组相比,过表达YWHAG显著上调YWHAG的表达水平(P < 0.01),见图4A,且同时过表达miR-210和YWHAG后,YWHAG的表达水平与对照组差异无显著性意义。CCK-8结果显示,与对照组相比,过表达YWHAG显著抑制脂肪细胞增殖(P < 0.05),见图4B,且同时过表达miR-210和YWHAG的脂肪细胞增殖活力与对照组差异无显著性意义;Annexin V-FITC/PI结果显示,过表达YWHAG后,脂肪细胞凋亡水平显著高于对照组(P < 0.01),见图4C,且同时过表达miR-210和YWHAG的脂肪细胞凋亡水平与对照组差异无显著性意义;血管生成实验及Western blot结果显示,与对照组相比,过表达YWHAG显著抑制脂肪细胞血管生成(P < 0.01),下调血管内皮生长因子的表达水平(P < 0.05),上调血管生成抑制因子 Endostatin的表达水平(P < 0.01),见图4D和4E,且同时过表达miR-210和YWHAG的脂肪细胞血管生成能力及血管内皮生长因子和Endostatin的表达水平与对照组差异无显著性意义。综上,miR-210通过靶向下调YWHAG的表达水平,从而促进脂肪细胞增殖和新血管形成并抑制其凋亡。"
| [1] RENNEKAMPFF HO, REIMERS K, GABKA CJ, et al. Current perspective and limitations of autologous fat transplantation--”consensus meeting” of the German Society of Plastic, Reconstructive and Aesthetic Surgeons at Hannover; September 2009. Handchir Mikrochir Plast Chir. 2010;42(2):137-142. [2] KAKAGIA D, PALLUA N. Autologous fat grafting: in search of the optimal technique. Surg Innov. 2014;21(3):327-336. [3] LAM SM. Fat transfer for the management of soft tissue trauma: the do’s and the don’ts. Facial Plast Surg. 2010;26(6):488-493. [4] DUCIC Y. Fat grafting in trauma and reconstructive surgery. Facial Plast Surg Clin North Am. 2008;16(4):409-416. [5] PU LL. Mechanisms of Fat Graft Survival. Ann Plast Surg. 2016;77 Suppl 1:S84-86. [6] BUCKY LP, PERCEC I. The science of autologous fat grafting: views on current and future approaches to neoadipogenesis. Aesthet Surg J. 2008;28(3):313-321; quiz 322-324. [7] MASHIKO T, YOSHIMURA K. How does fat survive and remodel after grafting? Clin Plast Surg. 2015;42(2):181-190. [8] ZHOU S, CHANG Q, LU F, et al. Injectable Mussel-Inspired Immobilization of Platelet-Rich Plasma on Microspheres Bridging Adipose Micro-Tissues to Improve Autologous Fat Transplantation by Controlling Release of PDGF and VEGF, Angiogenesis, Stem Cell Migration. Adv Healthc Mater. 2017;6(22). doi: 10.1002/adhm.201700131. [9] JIANG A, LI M, DUAN W, et al. Improvement of the survival of human autologous fat transplantation by adipose-derived stem-cells-assisted lipotransfer combined with bFGF. ScientificWorldJournal. 2015;2015:968057. [10] TEMIZ G, SIRINOGLU H, YESILOGLU N, et al. Effects of Deferoxamine on Fat Graft Survival. Facial Plast Surg. 2016;32(4):438-443. [11] CHEN X, YAN L, GUO Z, et al. Adipose-derived mesenchymal stem cells promote the survival of fat grafts via crosstalk between the Nrf2 and TLR4 pathways. Cell Death Dis. 2016;7(9):e2369. [12] YATES LA, NORBURY CJ, GILBERT RJ. The long and short of microRNA. Cell. 2013; 153(3):516-519. [13] CREUGNY A, FENDER A, PFEFFER S. Regulation of primary microRNA processing. FEBS Lett. 2018;592(12):1980-1996. [14] ZHANG B, WANG Q, PAN X. MicroRNAs and their regulatory roles in animals and plants. J Cell Physiol. 2007;210(2):279-289. [15] NAN Y, GUO H, GUO L, et al. MiRNA-451 Inhibits Glioma Cell Proliferation and Invasion Through the mTOR/HIF-1α/VEGF Signaling Pathway by Targeting CAB39. Hum Gene Ther Clin Dev. 2018;29(3):156-166. [16] LIANG D, LI J, WU Y, et al. miRNA-204 drives cardiomyocyte proliferation via targeting Jarid2. Int J Cardiol. 2015;201:38-48. [17] ZHAO K, ZHANG Y, KANG L, et al. Epigenetic silencing of miRNA-143 regulates apoptosis by targeting BCL2 in human intervertebral disc degeneration. Gene. 2017;628:259-266. [18] TIWARI A, MUKHERJEE B, DIXIT M. MicroRNA Key to Angiogenesis Regulation: MiRNA Biology and Therapy. Curr Cancer Drug Targets. 2018;18(3):266-277. [19] JING R, ZHONG QQ, LONG TY, et al. Downregulated miRNA-26a-5p induces the apoptosis of endothelial cells in coronary heart disease by inhibiting PI3K/AKT pathway. Eur Rev Med Pharmacol Sci. 2019;23(11):4940-4947. [20] TU Y, LIU L, ZHAO D, et al. Overexpression of miRNA-497 inhibits tumor angiogenesis by targeting VEGFR2. Sci Rep. 2015;5:13827. [21] YU NZ, HUANG JZ, ZHANG H, et al. A systemic review of autologous fat grafting survival rate and related severe complications. Chin Med J (Engl). 2015;128(9): 1245-1251. [22] HAMED S, EGOZI D, KRUCHEVSKY D, et al. Erythropoietin improves the survival of fat tissue after its transplantation in nude mice. PLoS One. 2010;5(11):e13986. [23] CAI J, FENG J, LIU K, et al. Early Macrophage Infiltration Improves Fat Graft Survival by Inducing Angiogenesis and Hematopoietic Stem Cell Recruitment. Plast Reconstr Surg. 2018;141(2):376-386. [24] WANG Y, SUN B, SUN H, et al. Regulation of proliferation, angiogenesis and apoptosis in hepatocellular carcinoma by miR-26b-5p. Tumour Biol. 2016;37(8): 10965-10979. [25] ZHOU Y, LI S, LI J, et al. Effect of microRNA-135a on Cell Proliferation, Migration, Invasion, Apoptosis and Tumor Angiogenesis Through the IGF-1/PI3K/Akt Signaling Pathway in Non-Small Cell Lung Cancer. Cell Physiol Biochem. 2017;42(4):1431-1446. [26] LIU XL, WANG G, SONG W, et al. microRNA-137 promotes endothelial progenitor cell proliferation and angiogenesis in cerebral ischemic stroke mice by targeting NR4A2 through the Notch pathway. J Cell Physiol. 2018;233(7):5255-5266. [27] HORIE M, SUZUKI M, TAKAHASHI E, et al. Cloning, expression, and chromosomal mapping of the human 14-3-3gamma gene (YWHAG) to 7q11.23.Genomics. 1999; 60(2):241-243. [28] WANG H, ZHI H, MA D, et al. MiR-217 promoted the proliferation and invasion of glioblastoma by repressing YWHAG. Cytokine. 2017;92:93-102. [29] AI Q, YU J, LI Y, et al. MicroRNA-182 promoted esophageal squamous cell carcinoma cell growth and metastasis via targeting YWHAG. J BUON. 2018;23(5):1439-1447. [30] YOO JO, KWAK SY, AN HJ, et al. miR-181b-3p promotes epithelial-mesenchymal transition in breast cancer cells through Snail stabilization by directly targeting YWHAG. Biochim Biophys Acta. 2016;1863(7 Pt A):1601-1611. [31] SATHE G, NA CH, RENUSE S, et al. Quantitative Proteomic Profiling of Cerebrospinal Fluid to Identify Candidate Biomarkers for Alzheimer’s Disease. Proteomics Clin Appl. 2019;13(4):e1800105. |
| [1] | Xiang Xinjian, Liu Fang, Wu Liangliang, Jia Daping, Tao Yue, Zhao Zhengnan, Zhao Yu. High-dose vitamin C promotes the survival of autologous fat transplantation in rats [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1298-1302. |
| [2] | Wen Dandan, Li Qiang, Shen Caiqi, Ji Zhe, Jin Peisheng. Nocardia rubra cell wall skeleton for extemal use improves the viability of adipogenic mesenchymal stem cells and promotes diabetes wound repair [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1086-1092. |
| [3] | Zhang Yujie, Yang Jiandong, Cai Jun, Zhu Shoulei, Tian Yuan. Mechanism by which allicin inhibits proliferation and promotes apoptosis of rat vascular endothelial cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1128-1132. |
| [4] | Zhang Jinglin, Leng Min, Zhu Boheng, Wang Hong. Mechanism and application of stem cell-derived exosomes in promoting diabetic wound healing [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1163-1169. |
| [5] | Hou Jingying, Guo Tianzhu, Yu Menglei, Long Huibao, Wu Hao. Hypoxia preconditioning targets and downregulates miR-195 and promotes bone marrow mesenchymal stem cell survival and pro-angiogenic potential by activating MALAT1 [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1053-1059. |
| [6] | Deng Shuang, Pu Rui, Chen Ziyang, Zhang Jianchao, Yuan Lingyan . Effects of exercise preconditioning on myocardial protection and apoptosis in a mouse model of myocardial remodeling due to early stress overload [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(5): 717-723. |
| [7] | Yang Sidi, Wang Qian, Xu Nuo, Wang Ronghan, Jin Chuanqi, Lu Ying, Dong Ming. Biodentine enhances the proliferation and differentiation of osteoblasts through upregulating bone morphogenetic protein-2 [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 516-520. |
| [8] | Li Anan, Jiang Tao, Zhan Min, Cai Yuning, Song Min, Li Congcong, Lin Wenzheng, Zhang Jiayuan, Liu Wengang. Pharmacological mechanism of Shenling Baizhu San in the treatment of knee osteoarthritis based on network pharmacology and molecular docking [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(2): 197-204. |
| [9] | Wei Wenyue, Wang Yuyin, Guo Minfang, Zhang Jing, Gu Qingfang, Song Lijuan, Chai Zhi, Yu Jiezhong, Ma Cungen. Fasudil inhibits neuronal apoptosis via regulating mitochondrial dynamics in APP/PS1 mice [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(2): 232-238. |
| [10] | Feng Dongfei, He Hongxu, Xie Qi, Zhang Lili, Zhou Hui, Li Wei. Selection of key genes related to biological functions and regulation pathway in periodontal reconstruction [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(2): 253-259. |
| [11] | Zhou Yi, Liu Xiaoyan, Xiang Bingyan. Application advantages of concentrated growth factors in the field of tissue repair and regeneration [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(10): 1710-1720. |
| [12] | Dong Hongfei, Huang Qilin, Yang Xiong, Li Shuai, Gu Rui, Sun Hongyu, Tang Lijun. Dermal extracellular matrix hydrogel in promoting acute skin wound healing in rats [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(10): 1627-1632. |
| [13] | Cai Shengsheng, Mei Heng, Zhang Xuequan, Deng Jin, Cao Jun, He Bin. Prepared HPe6DF composite nanoparticles enhance the effect of photodynamic therapy [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(10): 1638-1645. |
| [14] | Li Xuan, Sun Yimin, Li Longbiao, Wang Zhenming, Yang Jing, Wang Chenglin, Ye Ling. Manufacturing of nano-modified polycaprolactone microspheres and its biological effects in dental pulp cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(10): 1602-1608. |
| [15] | Li Zhongkang, Zheng Jiahua, Tian Yanpeng, Huang Xianghua. Latest progress and mechanisms of mesenchymal stem cells on premature ovarian failure [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(1): 141-147. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||