Chinese Journal of Tissue Engineering Research ›› 2021, Vol. 25 ›› Issue (26): 4230-4235.doi: 10.12307/2021.126
Previous Articles Next Articles
The role of signal pathways and receptors in various layers of mucosa in overactive bladder
Li Wen, Chen Yuelai
- Shanghai University of Traditional Chinese Medicine, Shanghai 201203, China
-
Received:2020-06-17Revised:2020-06-24Accepted:2020-07-23Online:2021-09-18Published:2021-05-13 -
Contact:Chen Yuelai, MD, Chief physician, Shanghai University of Traditional Chinese Medicine, Shanghai 201203, China E-mail:chenyuelai@163.com -
About author:Li Wen, Master, Shanghai University of Traditional Chinese Medicine, Shanghai 201203, China -
Supported by:the National Natural Science Foundation of China, No. 81674090 (to CYL)
CLC Number:
Cite this article
Li Wen, Chen Yuelai. The role of signal pathways and receptors in various layers of mucosa in overactive bladder[J]. Chinese Journal of Tissue Engineering Research, 2021, 25(26): 4230-4235.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks

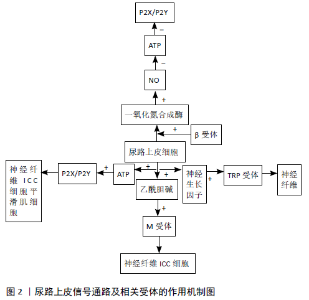
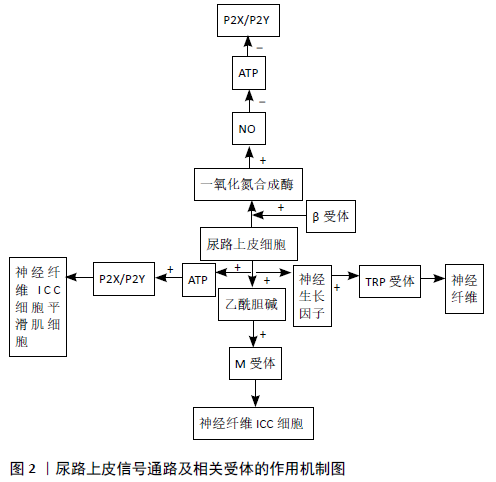
2.1.1 ATP通路 ATP是最早被学者发现从尿路上皮释放的神经递质,是由化学刺激或与膀胱扩张程度成比例的拉伸引起的。ATP作为膀胱内重要的传入信号分子在激活排尿反射中发挥重要作用。研究发现,膀胱在受到扩张等机械拉伸刺激时尿路上皮作出反应并释放ATP,拉伸扩张后50%-75%的膀胱传入神经由尿路上皮释放的ATP所激活,尿路上皮释放ATP的量与膀胱扩张程度呈正相关[6]。70%的ATP释放来源于尿路上皮,通过P2X2、P2X3或P2X2/3等嘌呤能受体的介导进而激活膀胱传入神经的兴奋性,触发膀胱充盈感觉[7]。 ATP的释放和嘌呤能受体表达的异常在许多人类膀胱疾病和膀胱病理的动物模型中被重视。在正常大鼠膀胱内灌注ATP后发现膀胱尿路上皮的传入神经兴奋性增强,大鼠出现膀胱过度活动现象[8],而将ATP水解酶灌注于膀胱腔内会降低膀胱活动[9]。此外,在P2X3基因敲除的小鼠和传入神经末梢缺失P2X3、P2X2/3受体的小鼠中出现传入神经放电减少、膀胱反射减退的现象[7 ,10]。在膀胱过度活动症患者或环磷酰胺诱导的膀胱过度活动大鼠模型尿路上皮中均可以检测到更高水平的P2X3受体[11-12]。有学者在探究臭氧膀胱灌注疗法对膀胱过度活动症大鼠膀胱黏膜P3X3的影响时发现,尿动力显示臭氧灌注后大鼠膀胱功能改善、P2X3受体表达下降[13]。P2Y6受体免疫反应活性在大鼠膀胱尿路上皮中表达最丰富,P2Y6受体的激活可以导致尿路上皮ATP的释放和排尿频率的增加[14]。膀胱灌注P2Y6选择性激动剂诱导大鼠出现膀胱过度活动症,P2Y6受体拮抗剂MRS2578可降低大鼠膀胱过度活动症[15]。SILVA等[16]发现P2X6受体介导的嘌呤能自分泌作用是通过Pannexin通道而非囊泡释放发生的,且膀胱过度活动症患者尿路上皮细胞P2Y6受体激活增加了黏膜ATP的释放。 根据以上结果可知,ATP和嘌呤能受体在膀胱排尿功能调节中发挥作用,可为日后临床治疗膀胱过度活动症打下坚实基础,考虑其特异性拮抗剂为药物研发提供新的思路。 2.1.2 胆碱能通路 由乙酰胆碱介导控制的副交感神经系统是调节排尿的主要途径,不仅直接作用于逼尿肌层面,还通过尿路上皮M受体的激活、增强传入信号的传递影响传入神经兴奋性[17],从而激活诱导膀胱功能障碍的传入途径,导致膀胱过度活动症和间质性膀胱炎/膀胱疼痛综合征相关症状[18]。MCLATCHIE等[19]发现,应用M受体激动剂Carbachol显著增加了ATP释放,M3选择性拮抗剂4-DAMP或M2选择性拮抗剂Methoctramine明显减少ATP释放。临床发现在治疗剂量下抗胆碱能药物主要在充盈期起作用,在排空时对逼尿肌收缩作用不大,这支持了尿路上皮M受体参与传入冲动的产生[20]。此外,抗胆碱能药物对无逼尿肌过度活动的膀胱过度活动症患者的急迫症状也有显著治疗作用[21],表明M受体在传入信号传递中的重要性。因此,乙酰胆碱的释放是传感器系统的第一步,它本身调节ATP的进一步释放,从而产生下游效应。 此外,烟碱型胆碱受体也在大鼠尿路上皮细胞中表达,并且通过受体的激活改变排尿反射行为。BECKEL等[22]通过实验发现α7受体对膀胱反射起抑制作用,α3受体则恰恰相反,可能是ATP的释放或抑制相关系[23]。此外,他们还对使用硫酸鱼精蛋白破坏尿路上皮屏障前后膀胱内的烟碱型受体含量进行比较,发现在硫酸鱼精蛋白膀胱灌注之前烟碱型受体起到抑制作用,延长排尿收缩的间隔时间,而当硫酸鱼精蛋白膀胱灌注后烟碱能受体具有相反的效果,刺激膀胱反射,缩短排尿间隔。提示烟碱型受体作用于屏障受损后的尿路上皮下靶点,刺激传入神经末梢,而不经硫酸鱼精蛋白膀胱灌注处理的烟碱能受体则激活尿路上皮中的受体起抑制作用。 综上,乙酰胆碱及其受体的信号作用仍不明确,可能与结合的受体、浓度和膀胱黏膜是否受损等因素相关,可见尿路上皮功能的复杂性,任何环节的改变均可能是膀胱过度活动症致病机制之一。 2.1.3 一氧化氮(NO)通路 尿道上皮细胞含有合成NO的酶机制,并且有证据表明NO可以抑制膀胱传入神经的信号[24]。PANDITA等[25]将NO清除剂(氧合血红蛋白)进行膀胱灌注,显示尿路上皮中NO水平的降低会导致膀胱过度活动;相反,当使用非选择性一氧化氮合成酶抑制剂(L-NAME)治疗环磷酰胺诱导的膀胱过度活动大鼠,可进一步加重膀胱过度活动表现[26]。 尿路上皮释放NO可由膀胱壁扩张、pH值或尿液成分的改变或存在于尿路上皮上的一些受体激活而诱导,其中值得一提的是β肾上腺素受体。β-A受体刺激激活膀胱上皮细胞中的环磷酸腺苷途径,并启动细胞内钙离子的增加,从而触发NO的产生和释放[27]。在NO松弛逼尿肌的同时释放一种抑制收缩的尿路上皮衍生抑制因子(UDIF),共同介导膀胱松弛、增加储尿期膀胱容量[28]。目前众多研究表明,人类尿路上皮细胞释放NO及产生NO是由β肾上腺素受体刺激调节[29],因此β3受体激动剂米拉贝格隆在膀胱过度活动症的临床使用中具有良好疗效。 此外在膀胱活动低下患者的尿路上皮中发现,β3受体和一氧化氮合成酶的表达均明显低于正常人[30],从反面证实NO水平的提高在尿路上皮信号的调节中起抑制作用,也为膀胱活动低下症患者提供有效的治疗手段。 2.1.4 神经生长因子通路 神经生长因子是一种神经营养因子,对周围神经系统神经元的生长、分化、再生、神经递质功能、发育和表型维持起着至关重要的作用。据报道,神经生长因子在小鼠尿路上皮中的过度表达会诱发小鼠骨盆区超敏反应和膀胱过度活动,并导致小鼠膀胱中钙素基因相关肽、P物质和阳性C纤维的密度明显增加[31]。推断神经生长因子可能通过激活正常Aδ纤维和C纤维兴奋传入通路而引起膀胱内的伤害性感觉及膀胱过度活动,提示神经生长因子可能作为膀胱过度活动症诊断治疗的一个潜在的生物学标志物。 已有实验证实,尿路上皮中神经生长因子的增加可以激活TRP通道,进而增强膀胱反射活动,其中TRPV1和TRPV4是研究重点,TRPV1对化学和热刺激敏感,TRPV4则介导机械感觉的传导[32]。最近的证据显示,从膀胱过度活动症患者分离的膀胱尿路上皮细胞中检测到的TRPV1表达和功能增加[33];而在TRPV1基因敲除小鼠模型中则检测到小鼠膀胱容量增加、ATP排泄减少[34]。TRPV1通道是C纤维的特异性通道且对辣椒素敏感,因此临床上常使用膀胱灌注TRPV1激动剂的治疗手段,使C纤维脱敏,以减轻膀胱过度活动症的症状。 TRPV4被发现是尿路上皮中最丰富的TRP通道。最新实验研究发现,TRPV4拮抗剂可降低尿路上皮神经生长因子过表达小鼠的排尿频率、ATP释放和盆腔敏感[35]。在大鼠尿路上皮细胞中,机械牵拉导致TRPV4激活,诱导细胞外Ca2+内流,从而有助于ATP释放,降低大鼠膀胱容量,引起排尿;而TRPV4的阻断或敲除都会导致牵拉诱发的Ca2+内流降低[36],因此TRPV4可以作为治疗膀胱功能障碍的一个有效作用靶点。 膀胱上皮中神经生长因子和TRPV通道的相互作用对膀胱过度活动症研究具有很大的应用价值。此外,对抗胆碱治疗无效的膀胱过度活动症患者进行膀胱灌注辣椒辣素类似物治疗可取得良好的疗效,提示选择降低膀胱敏感性的治疗方案,可能会对膀胱过度活动症患者有良好的治疗效果。 2.2 固有层 固有层是基底膜和逼尿肌之间的一层,是由胶原和弹性蛋白纤维组成的细胞外基质,包含血管网络、淋巴通道、感觉和自主神经末梢,以及几种类型的细胞如肌成纤维细胞、脂肪细胞和间质细胞等。当前下尿路研究的重点是传入机制及传入信息如何在排尿控制中生成并传递到中枢神经系统的过程,尿路上皮是目前研究的重点,但固有层同样是传入信号所必需的功能活性结构。尿路上皮与间质细胞和其下传入神经一起才能够启动膀胱收缩的功能,被称为黏膜信号传导途径[37]。 2.2.1 肌成纤维细胞 肌成纤维细胞是位于固有层的一类细胞,在尿路上皮基底细胞下方,也被称为上皮下肌成纤维细胞或上皮下间质细胞,这些上皮下肌成纤维细胞首先在人类膀胱中被描述[38]。上皮下肌成纤维细胞间的通讯信号通路参与膀胱感觉反应,肌成纤维细胞的表达增加,可能是诱导膀胱过度活动症发病的原因之一[39-40]。HOMMA等[41]提出尿频等相关症状与膀胱超敏反应相关,可能涉及由缝隙连接和局部肾素-血管紧张素系统调节的传入神经和上皮下层肌成纤维细胞之间的相互作用。 缝隙连接是存在于大多数细胞类型的质膜中的特殊结构,其形成通道以连接相邻细胞的细胞质,以便促进细胞质成分的直接交换,而不受细胞外元素的干扰[41]。Cx43是缝隙连接的主要蛋白质成分,负责缝隙连接通道的功能产生。Cx43的上调使相邻细胞(特别是膀胱平滑肌细胞)之间的兴奋性通讯增加,可能是膀胱过度活动症肌源性改变的主要原因[42]。但现在越来越多的观点认为,上皮下肌成纤维细胞诱导自发的电活动和细胞内Ca2+反应,这些细胞通过缝隙连接相互连接,并通过缝隙连接与逼尿肌平滑肌细胞连接,缝隙连接是调节协调和同步逼尿肌平滑肌收缩的功能连接[43],可能为膀胱感觉障碍提供新的治疗靶点。 2.2.2 传入神经纤维 传入神经检测来自周围的感觉信息,如膀胱充盈和疼痛,并将这些信息传递给脊髓。传入神经纤维在固有层形成神经丛,部分延伸到逼尿肌层,少数神经末梢终止于上皮的基底细胞层,包含有髓鞘的Aδ纤维和无髓鞘的C纤维。 在啮齿类动物中,Aδ纤维和C类纤维的传入在刺激方式的基础上是不可区分的;这两种类型的传入都由机械敏感和化学敏感的群体组成,可以根据背根神经节神经元的电生理和化学特性来区分。C纤维群体的特征是对辣椒素的敏感性,与TRPV1受体的表达相关,具有高的激活阈值和长时间的动作电位。 有髓鞘Aδ纤维和无髓鞘C纤维的传入神经是调节排尿的重要环节之一[44],C纤维主要是对膀胱黏膜化学性和冷刺激敏感,具有高机械性阈值特征。C纤维对机械、热和化学刺激后产生反应,释放多种神经递质如NO、ATP、乙酰胆碱和P物质等,这些递质作用于周围的感觉神经和细胞等,输入信号从外周传递到中枢神经系统,对其产生兴奋或抑制作用[45]。研究表明,虽然C纤维在正常膀胱中是“沉默的”,但C纤维的激活被认为是膀胱过度活动症发生的主要原因之一。膀胱灌注利多卡因是膀胱过度活动症的治疗手段之一。有研究将利多卡因灌注入膀胱过度活动症大鼠膀胱,使用c-Fos标记C纤维,发现处理后膀胱过度活动症大鼠尿动力学明显改善,C纤维的传入活动减弱,改善了膀胱过度活动症状[46]。 以上结果提示,C纤维的激活和兴奋性增强可直接影响膀胱传入功能,引起膀胱过度活动症。 2.2.3 黏膜血管学 血管系统是一个潜在的、重要的、未被深入研究的黏膜成分。在临床上进行膀胱镜检查时,最显著的可视化特征是透明尿路上皮下广泛的血管网络[47]。使用血管腐蚀铸造技术对小鼠膀胱血管进行解剖学研究发现,固有层内有丰富的毛细血管丛,由小动脉供应,由小静脉引流,并与尿路上皮细胞密切接触。 研究人员将黏膜单独剥离并去除尿路上皮,通过对电场刺激和各种激动剂/拮抗剂的反应研究黏膜中微静脉的收缩性。该实验得出结论,小静脉对交感神经活动的反应是收缩和增加收缩频率,因此通常被认为在膀胱平滑肌上起作用的药剂(如α阻滞剂、β3-激动剂和抗胆碱能药)也可能与黏膜血管相互作用导致黏膜信号的变化[48]。此外,相邻的血管与周围间质细胞相互作用对小静脉的自发性血管收缩中产生影响,有利于膀胱充盈阶段维持血流,维持正常的排尿行为[49]。 因此,膀胱灌注不足和由此产生的局部缺血容易影响尿路上皮和固有层及其相关细胞,导致黏膜信号传导功能改变,推测可能是造成膀胱过度活动症的因素之一。"

| [1] GORMLEY EA, LIGHTNER DJ, FARADAY M, et al. Diagnosis and treatment of overactive bladder (non-neurogenic) in adults:AUA /SUFU guideline amendment. J Urol. 2015;193(5):1572-1580. [2] CORCOS J, PRZYDACZ M, CAMPEAU L, et al. CUA guideline on adult overactive bladder. Can Urol Assoc J. 2017;11(5):E142-E173. [3] CHUANG YC, KAUFMANN JH, CHANCELLOR DD, et al. Bladder instillation of liposome encapsulated onabotulinumtoxina improves overactive bladder symptoms:a prospective,multicenter,double-blind,randomized trial. J Urol. 2014;192(6):1743-1749. [4] KRHUT J, NAVRATILOVA M, SYKORA R, et al. Intravesical instillation of onabotulinum toxin A embedded in inert hydrogel in the treatment of idiopathic overactive bladder: A double-blind randomized pilot study. Scand J Urol. 2016;50(3):200-205. [5] ANDERSSON K, ARNER A. Urinary bladder contraction and relaxation: physiology and pathophysiology. Physiol Rev. 2004;84(3):935-986. [6] LEE S, ROSE’MEYER R, MCDERMOTT C, et al. Diabetes-induced alterations in urothelium function: Enhanced ATP release and nerve-evoked contractions in the streptozotocin rat bladder. Clin Exp Pharmacol Physiol. 2018;45(11):1161-1169. [7] COCKAYNE DA, DUNN PM, ZHONG Y, et al. P2X2 knockout mice and P2X2/P2X3 double knockout mice reveal a role for the P2X2 receptor subunit in mediating multiple sensory effects of ATP. J Physiol. 2005; 567(2):621-639. [8] UEDA N, KONDO M, TAKEZAWA K, et al. Intravesical ATP instillation induces urinary frequency because of activation of bladder afferent nerves without inflammatory changes in mice: A promising model for overactive bladder. Biochem Biophys Res Commun. 2018;506(3):498-503. [9] TAKEZAWA K, KONDO M, NONOMURA N, et al. Urothelial ATP signaling: what is its role in bladder sensation? Neurourol Urodyn. 2017;36(4): 966-972. [10] VLASKOVSKA M, KASAKOV L, RONG W, et al. P2X3 knock-out mice reveal a major sensory role for urothelially released ATP. J Neurosci. 2001;21(15):5670-5677. [11] NORTH RA. P2X receptors. Philos Trans R Soc Lond B Biol Sci. 2016; 371(1700):20150427. [12] LEE WC, WU CC, CHUANG YC, et al. Ba-Wei-Die-Huang-Wan (Hachimi-jio-gan) can ameliorate cyclophosphamide-induced ongoing bladder overactivity and acidic adenosine triphosphate solution-induced hyperactivity on rats prestimulated bladder. J Ethnopharmacol. 2016; 184:1-9. [13] 陈从波,王黎,黄力,等.臭氧膀胱灌注对膀胱过度活动症大鼠膀胱黏膜 P2X3 的影响[J].湖北医药学院学报,2018,37(6):526-530. [14] TIMÓTEO MA, CARNEIRO I, SILVA I, et al. ATP released via pannexin-1 hemichannels mediates bladder overactivity triggered by urothelial P2Y6 receptors. Biochem Pharmacol. 2014;87(2):371-379. [15] CARNEIRO I, TIMÓTEO MA, SILVA I, et al. Activation of P2Y6 receptors increases the voiding frequency in anaesthetized rats by releasing ATP from the bladder urothelium. Br J Pharmacol. 2014;171(14):3404-3419. [16] SILVA I, FERREIRINHA F, MAGALHÃES-CARDOSO MT, et al. Activation of P2Y6 Receptors Facilitates Nonneuronal Adenosine Triphosphate and Acetylcholine Release from Urothelium with the Lamina Propria of Men with Bladder Outlet Obstruction. J Urol. 2015;194(4):1146-1154. [17] ARRIGHI N, BODEI S, LUCENTE A, et al. Muscarinic receptors stimulate cell proliferation in the human urothelium-derived cell line UROtsa. Pharmacol Res. 2011;64(4):420-425. [18] NEUHAUS J, SCHULTE-BAUKLOH H, STOLZENBURG JU, et al. Individual receptor profling as anovel tool to support diagnosis of bladder pain syndrome/interstitial cystitis (BPS/IC). World J Urol. 2012;30(5):693-700. [19] MCLATCHIE LM, YOUNG JS, FRY CH. Regulation of ACh release from guinea pig bladder urothelial cells: potential role in bladder filling sensations. Br J Pharmacol. 2014;171(14):3394-3403. [20] FINNEY SM, ANDERSSON KE, GILLESPIE JI, et al. Antimuscarinic drugs in detrusor overactivity and the overactive bladder syndrome: motor or sensory actions? BJU Int. 2006;98(3):503-507. [21] ROVNER E, KENNELLY M, SCHULTE BH, et al. Urodynamic results and clinical outcomes with intradetrusor injections of onabotulinumtoxinA in a randomized, placebo-controlled dose-finding study in idiopathic overactive bladder. Neurourol Urodyn. 2011;30(4):556-562. [22] BECKEL JM, KANAI AJ, LEE SJ, et al. Expression of functional nicotinic acetylcholine receptors in rat urinary bladder epithelial cells. Am J Physiol Renal Physiol. 2006;290(1):F103-F110. [23] BECKEL JM, BIRDER LA. Differential expression and function of nicotinic acetylcholine receptors in the urinary bladder epithelium of the rat. J Physiol PMID.2012;590(6):1465-1480. [24] AIZAWA N, IGAWA Y, NISHIZAWA O, et al. Effects of nitric oxide on the primary bladder afferent activities of the rat with and without intravesical acrolein treatment. Eur Urol. 2011;59(2):264-271. [25] PANDITA RK, MIZUSAWA H, ANDERSSON K. Intravesical Oxyhemoglobin initiates bladder overactivity in conscious,normal rats. J Urol. 2000; 164(2):545-550. [26] ANDERSSON M, ARONSSON K, GIGLIO DA, et al. Pharmacological modulation of the micturition pattern in normal and cyclophosphamide pretreated conscious rats. Auton Neurosci. 2011;159(1-2):77-83. [27] TYAGI PD, THOMAS CA, YOSHIMURA NK, et al. Investigations into the presence of functional Beta1, Beta2 and Beta3-adrenoceptors in urothelium and detrusor of human bladder. Int Braz J Urol. 2009;35(1): 76-83. [28] IMRAN M, NAJMI AK, TABREZ S. Mirabegron for overactive bladder:a novel, first-in-class β3-agonist therapy. Urol J. 2013;10(3):935-940. [29] NOGUCHI K, SUGAYA K, NISHIJIMA S, et al. Evaluation of a rat model of functional urinary bladder outlet obstruction produced by chronic inhibition of nitric oxide synthase. Life Sci. 2019;234:116772. [30] JIANG YH, LEE CL, KUO HC. Urothelial Dysfunction, Suburothelial Inflammation and Altered Sensory Protein Expression in Men with Bladder Outlet Obstruction and Various Bladder Dysfunctions: Correlation with Urodynamics. J Urol. 2016;196(3):831-837. [31] SCHNEGELSBERG B, SUN TT, CAIN G, et al. Overexpression of NGF in mouse urothelium leads to neuronal hyperinnervation, pelvic sensitivity, and changes in urinary bladder function. Am J Physiol Regul Integr Comp Physiol. 2010;298(3):R534-R547. [32] ANDERSSON KE. TRP Channels as Lower Urinary Tract Sensory Targets.Med Sci (Basel). 2019;7(5):1-15. [33] BIRDER LA, KANAI A J, GROAT WC, et al. Vanilloid receptor expression suggests a sensory role for urinary bladder epithelial cells. Proc Natl Acad Sci USA. 2001;98(23):13396-13401. [34] BIRDER L, NAKAMURA Y, KISS S, et al. Altered urinary bladder function in mice lacking the vanilloid receptor. Nat Neurosci. 2002;5(9):856-860. [35] GIRARD BM, CAMPBELL SE, PERKINS M, et al. TRPV4 blockade reduces voiding frequency, ATP release, and pelvic sensitivity in mice with chronic urothelial overexpression of NGF. Am J Physiol Renal Physiol. 2019;317(6):F1695-F1706. [36] JANSSEN DAW, HOENDEROP JG, HEESAKKERS JPFA, et al. TRPV4 mediates afferent pathways in the urinary bladder. A spinal c-fos study showing TRPV1 related adaptations in the TRPV4 knockout mouse. Pflugers Arch. 2016;468(10):1741-1749. [37] KANAI A, ANDERSSON KE. Bladder afferent signalling:Recent findings. J Urol. 2010;183(4):1288-1295. [38] ANDERSSON KE, MARTIN N, NITTI V. Selective β3- adrenoceptor agonists for the treatment of overactive bladder. J Urol. 2013;190(4): 1173-1180. [39] SUI GP, ROTHERY S, DUPONT E, et al. Gap junctions and connexin expression in human suburothelial interstitial cells. BJU Int. 2002; 90(1):118-129. [40] McCloskey KD. Interstitial cells in the urinary bladder localization and function. Neurourol Urodyn. 2010;29(1):82-87. [41] HOMMA Y, UEDA T, TOMOE H, et al. Clinical guidelines for interstitial cystitis and hypersensitive bladder syndrome. Int J Urol. 2009;16(7): 597-615. [42] BABAOGLU M, ZUMRUTBAS AE, ACAR IC, et al. Gap junction expression and the effects of gap junction inhibitors in overactive bladder models:Does ovariectomy have a role? Int Urol Nephrol. 2013;45(4): 1001-1008. [43] ZHOU F, LI H, ZHOU C, et al. Structural and functional changes in gap junctional intercellular communication in a rat model of overactive bladder syndrome induced by partial bladder outlet obstruction. Exp Ther Med. 2016;11(6):2139-2146. [44] BIRDER LA, KULLMANN FA. Role of neurogenic inflammation in local communication in the visceral mucosa. Semin Immunopathol. 2018;40(3):261-279. [45] LERON E, WEINTRAUB AY, MASTROLIA SA, et al. Overactive bladder syndrome:evaluation and management. Curr Urol. 2018;11(3):117-125. [46] JUSZCZAK K, ZIOMBER A, WYCZOLKOWSKI M, et al. Urodynamic effects of the bladder C-fiber afferent activity modulation in chronic model of overactive bladder in rats. J Physiol Pharmacol. 2009;60(4):85-91. [47] KANG HJ, KIM SW, LEE YS, et al. Effects of 1% Lidocaine Instillation on Overactive Bladder Induced by Bladder Outlet Obstruction in Rats. Urol J. 2019;157(5):210-229. [48] HOSSLER FE, LAMETSCHWANDTNER A, KAO R, et al. Microvascular architecture of mouse urinary bladder described with vascular corrosion casting,light microscopy, SEM, and TEM. Microsc. Microanal. 2013;19(6):1428-1435. [49] SHIMIZU Y, MOCHIZUKI S, MITSUI R, et al. Neurohumoral regulation of spontaneous constrictions in suburothelial venules of the rat urinary bladder. Vasc Pharmacol. 2014;60(2):84-94. |
| [1] | An Yang, Liao Yinan, Xie Chengxin, Li Qinglong, Huang Ge, Jin Xin, Yin Dong. Mechanism of Inulae flos in the treatment of osteoporosis: an analysis based on network pharmacology [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-8. |
| [2] | Pu Rui, Chen Ziyang, Yuan Lingyan. Characteristics and effects of exosomes from different cell sources in cardioprotection [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-. |
| [3] | Zhang Chao, Lü Xin. Heterotopic ossification after acetabular fracture fixation: risk factors, prevention and treatment progress [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1434-1439. |
| [4] | Zhou Jihui, Li Xinzhi, Zhou You, Huang Wei, Chen Wenyao. Multiple problems in the selection of implants for patellar fracture [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1440-1445. |
| [5] | Wang Debin, Bi Zhenggang. Related problems in anatomy mechanics, injury characteristics, fixed repair and three-dimensional technology application for olecranon fracture-dislocations [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1446-1451. |
| [6] | Ji Zhixiang, Lan Changgong. Polymorphism of urate transporter in gout and its correlation with gout treatment [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1290-1298. |
| [7] | Yuan Mei, Zhang Xinxin, Guo Yisha, Bi Xia. Diagnostic potential of circulating microRNA in vascular cognitive impairment [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1299-1304. |
| [8] | Wang Xianyao, Guan Yalin, Liu Zhongshan. Strategies for improving the therapeutic efficacy of mesenchymal stem cells in the treatment of nonhealing wounds [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1081-1087. |
| [9] | Wan Ran, Shi Xu, Liu Jingsong, Wang Yansong. Research progress in the treatment of spinal cord injury with mesenchymal stem cell secretome [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1088-1095. |
| [10] | Liao Chengcheng, An Jiaxing, Tan Zhangxue, Wang Qian, Liu Jianguo. Therapeutic target and application prospects of oral squamous cell carcinoma stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1096-1103. |
| [11] | Zhao Min, Feng Liuxiang, Chen Yao, Gu Xia, Wang Pingyi, Li Yimei, Li Wenhua. Exosomes as a disease marker under hypoxic conditions [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1104-1108. |
| [12] | Xie Wenjia, Xia Tianjiao, Zhou Qingyun, Liu Yujia, Gu Xiaoping. Role of microglia-mediated neuronal injury in neurodegenerative diseases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1109-1115. |
| [13] | Li Shanshan, Guo Xiaoxiao, You Ran, Yang Xiufen, Zhao Lu, Chen Xi, Wang Yanling. Photoreceptor cell replacement therapy for retinal degeneration diseases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1116-1121. |
| [14] | Jiao Hui, Zhang Yining, Song Yuqing, Lin Yu, Wang Xiuli. Advances in research and application of breast cancer organoids [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1122-1128. |
| [15] | Wang Shiqi, Zhang Jinsheng. Effects of Chinese medicine on proliferation, differentiation and aging of bone marrow mesenchymal stem cells regulating ischemia-hypoxia microenvironment [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1129-1134. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||