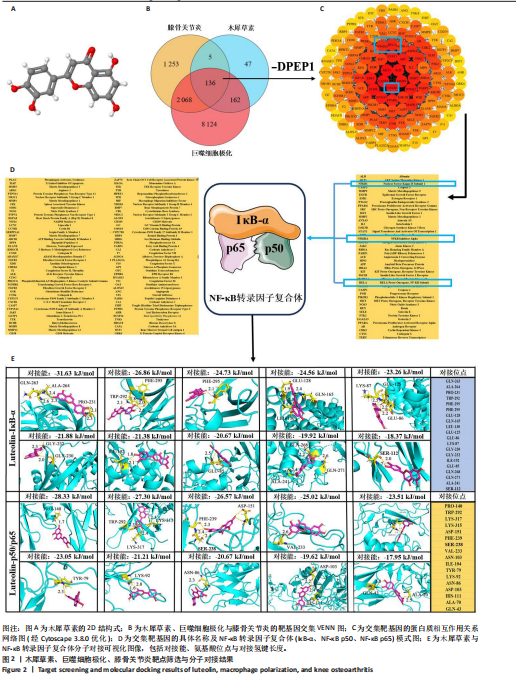
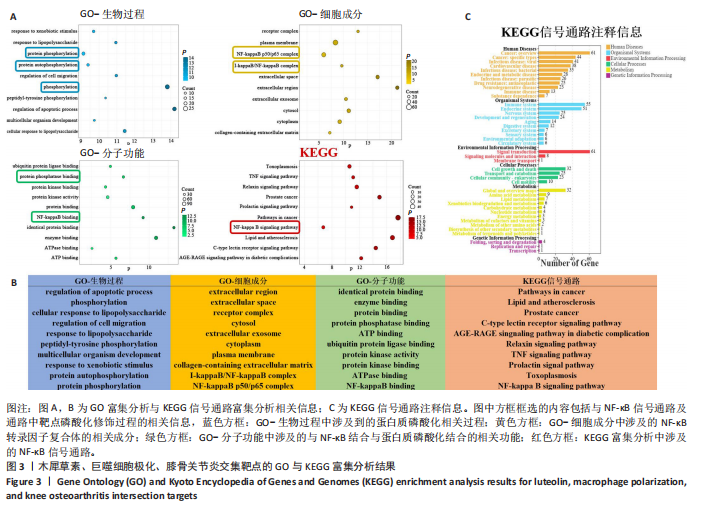
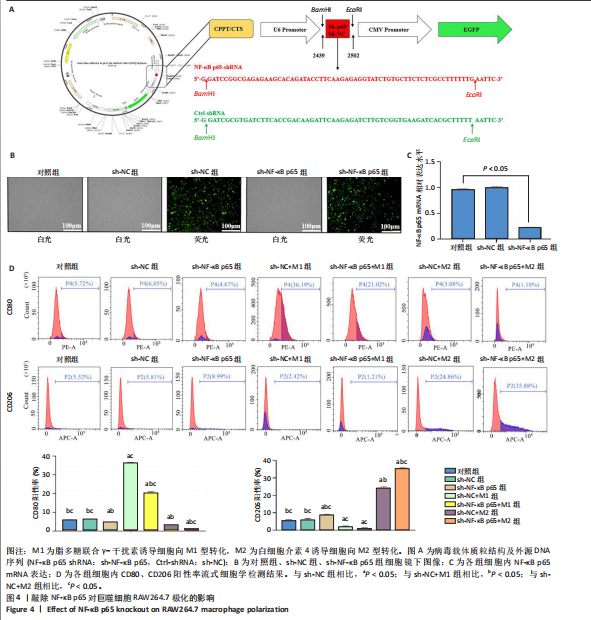
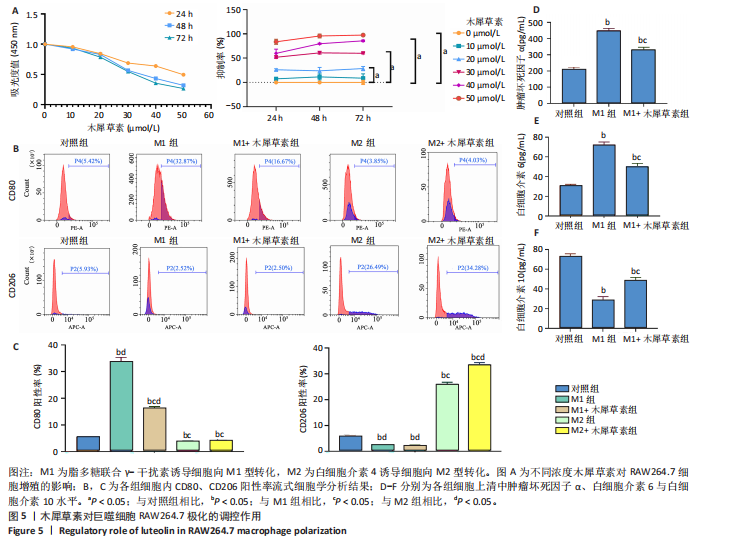
[1] MAGNUSSON K, TURKIEWICZ A, SNOEKER B, et al. The heritability of doctor-diagnosed traumatic and degenerative meniscus tears. Osteoarthritis Cartilage. 2021;29(7):979-985.
[2] KATANO H, OZEKI N, KOHNO Y, et al. Trends in arthroplasty in Japan by a complete survey, 2014-2017. J Orthop Sci. 2021;26(5):812-822.
[3] MORA JC, PRZKORA R, CRUZ-ALMEIDA Y. Knee osteoarthritis: pathophysiology and current treatment modalities. J Pain Res. 2018;11:2189-2196.
[4] 晏润祺.中西医治疗膝关节骨性关节炎的研究进展[J].临床医学研究与实践,2025,10(2):193-198.
[5] 梅斌,饶毅,蔡毅,等.内热针联合PRP注射治疗对膝关节骨性关节炎患者血清TNF-α、NF-кB水平的影响[J].时珍国医国药,2024,35(7):1664-1666.
[6] 谢筱虎,李虹成.MRI联合MMP-9、IL-7R对膝关节骨性关节炎发生发展的诊断价值研究[J].中国实验诊断学,2024,28(5):563-567.
[7] 缪祎,张德新,魏海强,等.基于CX3CL1探讨不同干预剂量石榴籽油对膝关节骨性关节炎大鼠滑膜细胞及软骨基质的影响[J].中国老年学杂志,2025, 45(11):2704-2708.
[8] 吴志明,李曾艳.黄芩苷对大鼠膝关节骨性关节炎的炎症调节作用研究[J].中国临床药理学杂志,2024,40(12):1808-1812.
[9] 余文英,修忠标,林泽豪,等.基于Notch1/Jagged1/Hes1信号通路探讨针刀调控巨噬细胞极化抑制兔膝关节骨关节炎滑膜炎性反应的作用机制[J]. 针刺研究,2025,50(6):649-657.
[10] LATOURTE A, KLOPPENBURG M, RICHETTE P. Emerging pharmaceutical therapies for osteoarthritis. Nat Rev Rheumatol. 2020;16(12):673-688.
[11] 周琦,孙慧娟,于栋华,等.巨噬细胞M1/M2型极化在不同疾病中的作用机制[J].中国药理学通报,2020,36(11):1502-1506.
[12] GENSEL JC, ZHANG B. Macrophage activation and its role in repair and pathology after spinal cord injury. Brain Res. 2015;1619:1-11.
[13] 张颖,王永福.巨噬细胞极化相关机制的研究进展[J].细胞与分子免疫学杂志,2016,32(11):1570-1573.
[14] JIE L, ZHANG L, FU H, et al. Xibining inhibition of the PI3K-AKT pathway reduces M1 macrophage polarization to ameliorate KOA synovial inflammation and nociceptive sensitization. Phytomedicine. 2025;136:156281.
[15] LI GS, CUI L, WANG GD. miR-155-5p regulates macrophage M1 polarization and apoptosis in the synovial fluid of patients with knee osteoarthritis. Exp Ther Med. 2021;21(1):68.
[16] 扎西达娃.M2-巨噬细胞来源的外泌体通过抑制膝关节骨关节炎大鼠的PI3K/AKT/mTOR通路发挥治疗作用的影响[D].西宁:青海大学,2022.
[17] 杜娟,左非非,魏双艳,等.山楂降脂软胶囊中4种黄酮含量的测定[J].郑州大学学报(医学版),2017,52(4):456-459.
[18] LEYVA-LÓPEZ N, GUTIERREZ-GRIJALVA EP, AMBRIZ-PEREZ DL, et al. Flavonoids as Cytokine Modulators: A Possible Therapy for Inflammation-Related Diseases. Int J Mol Sci. 2016;17(6):921.
[19] 王书侠,姚孝明,葛亮,等.木犀草素对巨噬细胞炎症极化的影响及机制[J].解放军医学杂志,2018,43(12):1013-1018.
[20] 李渊深,索艳晖,谢延平,等.紫草素调节NF-κB/ERK信号通路对膝关节骨性关节炎大鼠炎症反应的影响[J].中国免疫学杂志,2023,39(12):2577-2581.
[21] KUANG S, LIU Z, LIU L, et al. Polygonatum sibiricum polysaccharides protect against knee osteoarthritis by inhibiting the TLR2/NF-κB signaling pathway in vivo and in vitro. Int J Biol Macromol. 2024;274(Pt 1):133137.
[22] JI L, LI W, QIAN W, et al. The Role of Nanoparticles-Mediated Ligustrazine in the Treatment of Knee Osteoarthritis and Its Effect on Matrix Metalloproteinases and Upstream NF-κB Signaling Pathway in Knee Osteoarthritis. J Nanosci Nanotechnol. 2021;21(2):1372-1377.
[23] FENG X, ZHU S, QIAO J, et al. CX3CL1 promotes M1 macrophage polarization and osteoclast differentiation through NF-κB signaling pathway in ankylosing spondylitis in vitro. J Transl Med. 2023;21(1):573.
[24] PAOLUCCI T, PINO V, ELSALLABI O, et al. Quantum Molecular Resonance Inhibits NLRP3 Inflammasome/Nitrosative Stress and Promotes M1 to M2 Macrophage Polarization: Potential Therapeutic Effect in Osteoarthritis Model In Vitro. Antioxidants (Basel). 2023;12(7):1358.
[25] MO H, WANG Z, HE Z, et al. Decreased Peli1 expression attenuates osteoarthritis by protecting chondrocytes and inhibiting M1-polarization of macrophages. Bone Joint Res. 2023;12(2):121-132.
[26] 赵新然,王玉娜,曾倩.基于NF-κB信号通路探究木犀草素对宫颈癌细胞增殖和凋亡的调控作用[J].中医药导报,2023,29(6):22-27.
[27] 刘克新,龚本新,何飞雪,等.木犀草素通过TLR4/NF-κB途径对哮喘模型幼鼠炎症的影响[J].黑龙江医学,2023,47(2):133-136.
[28] 邹国涛,王英娟,曾毅文,等.基于TLR4/NF-κB信号通路研究木犀草素对幼鼠急性肺损伤的作用机制[J].中药新药与临床药理,2021,32(5):661-666.
[29] 孔学礼,霍桂桃,李佳,等.木犀草素通过抑制p65 NF-κB、促进p85 PI3K调节微血管内皮细胞VCAM-1表达(英文)[J].生物化学与生物物理进展, 2020,47(8):675-684.
[30] 胡涂.镁通过调控巨噬细胞极化影响间充质干细胞软骨分化的研究[D].上海:上海交通大学,2018.
[31] 程锐,周健.宣痹汤加味联合常规西药治疗膝骨关节炎湿热痹阻证34例[J].浙江中医杂志,2022,57(9):653-654.
[32] 党兴,张铎安.关节克痹丸联合西药治疗寒湿痹阻型膝骨关节炎临床研究[J].新中医,2024,56(5):117-122.
[33] KNIGHTS AJ, REDDING SJ, MAERZ T. Inflammation in osteoarthritis: the latest progress and ongoing challenges. Curr Opin Rheumatol. 2023;35(2):128-134.
[34] LOESER RF. Articular cartilage nuclear receptors: an emerging target for treatment of osteoarthritis. Osteoarthritis Cartilage. 2009;17(7):829-831.
[35] KO CY, LIN YY, ACHUDHAN D, et al. Omentin-1 ameliorates the progress of osteoarthritis by promoting IL-4-dependent anti-inflammatory responses and M2 macrophage polarization. Int J Biol Sci. 2023;19(16):5275-5289.
[36] DING D, YAN J, FENG G, et al. Dihydroartemisinin attenuates osteoclast formation and bone resorption via inhibiting the NF‑κB, MAPK and NFATc1 signaling pathways and alleviates osteoarthritis. Int J Mol Med. 2022;49(1):4.
[37] YUNNA C, MENGRU H, LEI W, et al. Macrophage M1/M2 polarization. Eur J Pharmacol. 2020;877:173090.
[38] XU J, CHEN X, ZHANG H, et al. Platelet-rich plasma relieves inflammation and pain by regulating M1/M2 macrophage polarization in knee osteoarthritis rats. Sci Rep. 2025;15(1):12805.
[39] LIU L, GUO H, SONG A, et al. Progranulin inhibits LPS-induced macrophage M1 polarization via NF-кB and MAPK pathways. BMC Immunol. 2020;21(1):32.
[40] YANG K, XU J, FAN M, et al. Lactate Suppresses Macrophage Pro-Inflammatory Response to LPS Stimulation by Inhibition of YAP and NF-κB Activation via GPR81-Mediated Signaling. Front Immunol. 2020;11:587913.
[41] XU Y, HU X, CAI J, et al. Atractylenolide-III alleviates osteoarthritis and chondrocyte senescence by targeting NF-κB signaling. Phytother Res. 2023;37(10):4607-4620.
[42] SUN SC. The non-canonical NF-κB pathway in immunity and inflammation. Nat Rev Immunol. 2017;17(9):545-558.
[43] 冯逸佳,李庆芳,董壮壮,等.黄酮化合物对巨噬细胞中白细胞介素介导的TLR4/MyD88/NF-κB信号转导通路的调节作用[J].医药导报,2024,43(5): 689-695.
[44] XUE L, JIN X, JI T, et al. Luteolin ameliorates DSS-induced colitis in mice via suppressing macrophage activation and chemotaxis. Int Immunopharmacol. 2023;124(Pt B):110996.
[45] XU YL, LI XJ, CAI W, et al. Diosmetin-7-O-β-D-glucopyranoside from Pogostemonis Herba alleviated SARS-CoV-2-induced pneumonia by reshaping macrophage polarization and limiting viral replication. J Ethnopharmacol. 2025;336:118704.
|