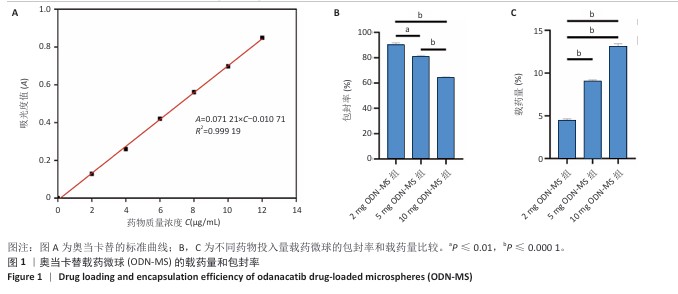
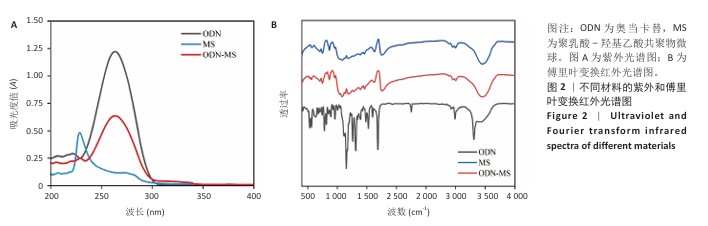
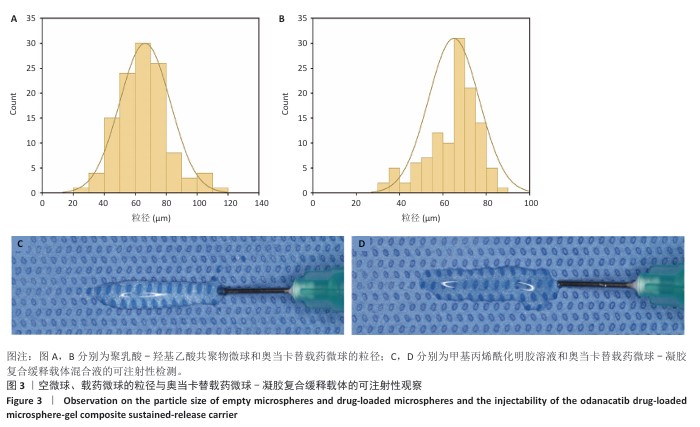
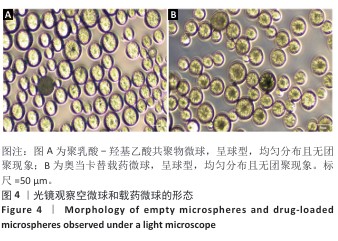
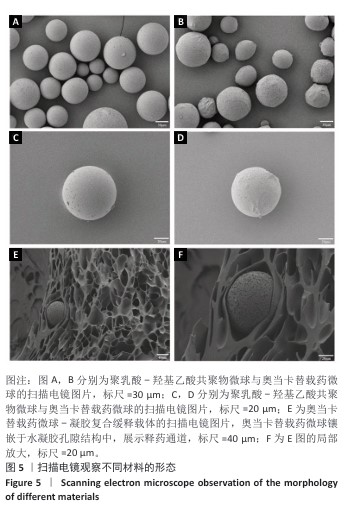
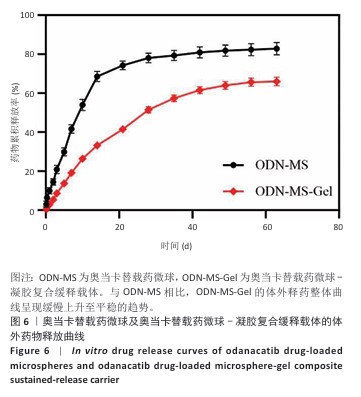
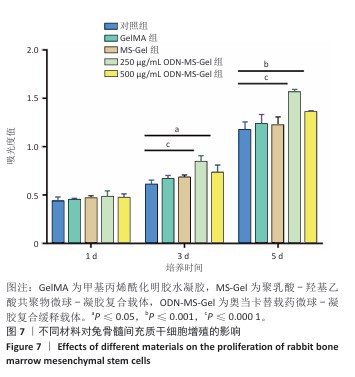
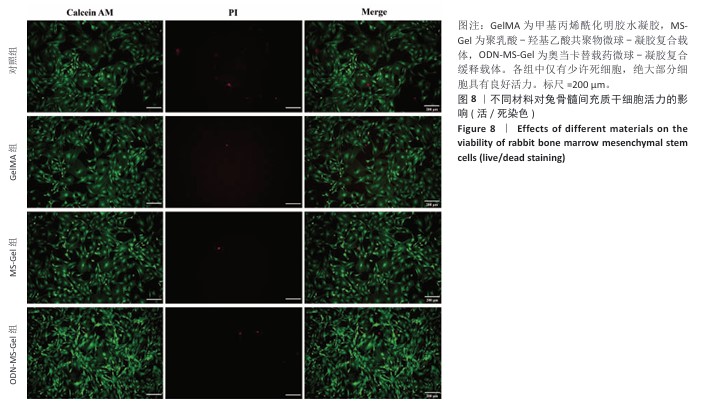
[1] RAZA M, ABUD DG, WANG J, et al. Ease and practicability of the 2017 classification of periodontal diseases and conditions: a study of dental electronic health records. BMC oral Health. 2024;24(1):621.
[2] PINTO GR, CARVALHO FILHO PC, CARVALHO RDO, et al. Subgingival biofilm microbiome in individuals with asthma and periodontitis: metagenomic analysis. Oral Dis. 2024;30(7):4721-4730.
[3] RELVAS M, MEDES-FRIAS A, GONÇALVES M, et al. Salivary il-1β, il-6, and il-10 are key biomarkers of periodontitis severity. Int J Mol Sci. 2024;25(15):8401.
[4] BADAHDAH A,BAHANAN L, BARNAWI RF, et al. Outcomes of nonsurgical periodontal therapy in gingivitis and periodontitis patients treated by senior dental students. Int J Dent Hyg. 2025;23(2):374-383.
[5] 张武阳,李登科,陈楷文,等.我国牙槽骨缺损人工植骨材料的发展现状与趋势[J].中国实用口腔科杂志,2022,15(6):644-647.
[6] BEKLEN A, AI-SAMADI A, KONTTINE YT. Expression of cathepsin k in periodontitis and in gingival fibroblasts. Oral Dis. 2015;21(2):163-169.
[7] ZHANG J, LI D, ZHENG X, et al. TMT-labelled quantitative proteomic analysis to predict the target promoting human odontogenic inflammatory granulation tissue transform into reparative granulation tissue. Acta Odontol Scand. 2021;79(6): 458-465.
[8] ZHANG W, DONG Z, LI D, et al. Cathepsin k deficiency promotes alveolar bone regeneration by promoting jaw bone marrow mesenchymal stem cells proliferation and differentiation via glycolysis pathway. Cell Prolif. 2021;54(7): e13058.
[9] RIZZOLI R, BENHAMOU CL, HALSE J, et al. Continuous treatment with odanacatib for up to 8 years in postmenopausal women with low bone mineral density: a phase 2 study. Osteoporos Int. 2016;27(6):2099-2107.
[10] DAI R, WU Z, CHU H , et al. Cathepsin k: the action in and beyond bone. Front Cell Dev Biol. 2020;8:433.
[11] LI D, ZHANG W, YE W, et al. A multifunctional drug consisting of tetracycline conjugated with odanacatib for efficient periodontitis therapy. Front Pharmacol. 2022;13:1046451.
[12] 郭作娟,梁荣财.PLGA微球用于蛋白/多肽类药物缓释递送的优化策略[J].中国医药工业杂志,2023,54(3):312-319.
[13] WU Y, WEI G, CAO X, et al. Stimuli-responsive dual-drug loaded microspheres with differential drug release for antibacterial and wound repair promotion. Colloids Surf B Biointerfaces. 2025;248:114455.
[14] LI Z, QIANG Y, CHEN D, et al. Dual-modal super-resolution ultrasound and nir-ii fluorescence imaging of ischemic stroke with icg-doped porous plga microspheres. Mater Today Bio. 2025;31:101513.
[15] LI X, WEI Y, WEN K, et al. Novel insights on the encapsulation mechanism of plga terminal groups on ropivacaine. Eur J Pharm Biopharm. 2021;160:143-151.
[16] ALLISON SD. Analysis of initial burst in plga microparticles. Expert Opin Drug Deliv. 2008;5(6):615-628.
[17] LIU L, ZHENG M, LIANG R. Improvement of liraglutide release from plga microspheres by a porous microsphere-gel composite system. Pharm Dev Technol. 2024;29(4):291-299.
[18] LIN SJ, CHAN YC, SU ZC, et al. Growth factor-loaded microspheres in mpeg-polypeptide hydrogel system for articular cartilage repair. J Biomed Mater Res A. 2021;109(12):2516-2526.
[19] ZHOU T, XIONG H, YAO SY, et al. Hypoxia and matrix metalloproteinase 13-responsive hydrogel microspheres alleviate osteoarthritis progression in vivo. Small. 2024;20(19):e2308599.
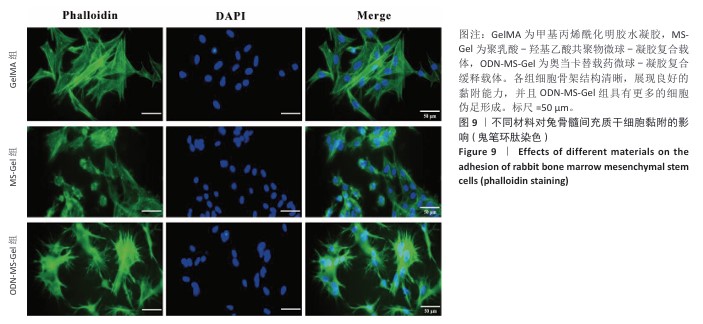
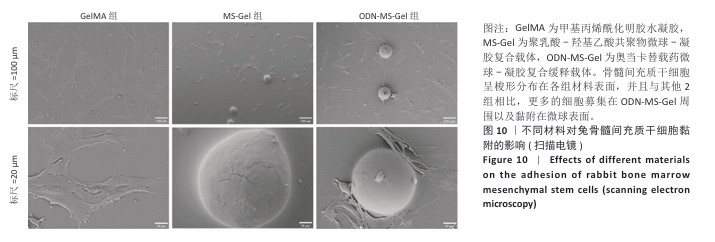
[20] 郭水香,张格格,胡锦玥,等.甲基丙烯酰化明胶水凝胶治疗牙周病的研究进展[J].中国实用口腔科杂志,2025,18(1):99-103.
[21] 国家药典委员会编.中华人民共和国药典.2010年版.二部[M].北京:中国医药科技出版社,2010.
[22] 曲萌菲,王艺璇,张杭,等.羟丙基壳聚糖/氧化甲基纤维素自愈合水凝胶的制备及其载药性能[J].精细化工,2023,40(8):1734-1741.
[23] 国家药典委员会编.中华人民共和国药典 2020版[M].北京:中国医药科技出版社,2020.
[24] 方毅承.多奈哌齐-plga微球的制备与释药机理研究[D].杭州:浙江工业大学,2019.
[25] 闫秋霖,吴恒乾,高岩,等.用于治疗牙周炎的抗菌药局部缓释递送系统的研究进展[J].中国医药工业杂志,2024,55(9):1186-1192.
[26] KORNMAN KS, PAPAPANOU PN. Clinical application of the new classification of periodontal diseases: ground rules, clarifications and “gray zones”. J Periodontol. 2020;91(3):352-360.
[27] MOGI M, OTOGOTO J. Expression of cathepsin-k in gingival crevicular fluid of patients with periodontitis. Arch Oral Biol. 2007;52(9):894-898.
[28] TANG Y, LI X, CAI Y, et al. Hypoxia-induced factor 1α promotes osteotomy-induced regional acceleratory phenomenon via dc-stamp mediated membrane fusion. Oral Dis. 2023;29(5):2139-2153.
[29] ZHOU H, ZHANG Y, ZHANG Q, et al. Cathepsin k inhibition alleviates periodontal bone resorption by promoting type h vessel formation through pdgf-bb/pdgfr-β axis. Oral Dis. 2024;30(8):5335-5348.
[30] BATISTA-CÀRDENAS D, ARAYA-CASTILLO A, ARIAS-CAMPOS MP, et al. Association of the severity and progression rate of periodontitis with systemic medication intake. Front Oral Health. 2024;5:1447019.
[31] RAN S, XUE L, WEI X, et al. Recent advances in injectable hydrogel therapies for periodontitis. J Mater Chem B. 2024;12(25):6005-6032.
[32] KHAING EM, MAHADLEK J, OKONOGI S, et al. Lime peel oil-incorporated rosin-based antimicrobial in situ forming gel. Gels. 2022;8(3):169.
[33] SENARAT S, PICHAYAKORN W, PHAECHAMUD T, et al. Antisolvent eudragit® polymers based in situ forming gel for periodontal controlled drug delivery. J Drug Deliv Sci Technol. 2023;82:104361.
|