中国组织工程研究 ›› 2026, Vol. 30 ›› Issue (29): 7619-7631.doi: 10.12307/2026.398
• 组织构建综述 tissue construction review • 上一篇 下一篇
信号通路调控细胞铁死亡防治骨科疾病的新策略
赫 龙1,高 爽1,陈 超2,覃国忠1,冉庆森1,王政春1,杨亚锋1,任 航1,邱云开1,杨 洋1,李 伟1
- 1深圳平乐骨伤科医院,广东省深圳市 518000;2南方医科大学附属南方医院,广东省广州市 510000
-
收稿日期:2025-08-08修回日期:2025-10-18出版日期:2026-10-18发布日期:2026-03-05 -
通讯作者:李伟,硕士,副主任医师,深圳平乐骨伤科医院,广东省深圳市 518000 -
作者简介:赫龙,男,黑龙江中医药大学毕业,硕士,主要从事中西医结合治疗骨病方面的研究。 -
基金资助:广东省中医药局项目(20241199),项目负责人:陈超;深圳市医疗卫生“三名工程”项目资助(SZZYSM202108013),项目负责人:李伟
A new strategy for preventing and treating orthopedic diseases by regulating ferroptosis through signaling pathways
He Long1, Gao Shuang1, Chen Chao2, Qin Guozhong1, Ran Qingsen1, Wang Zhengchun1, Yang Yafeng1, Ren Hang1, Qiu Yunkai1, Yang Yang1, Li Wei1
- 1Shenzhen Pingle Orthopaedic Hospital, Shenzhen 518000, Guangdong Province, China; 2Nanfang Hospital, Southern Medical University, Guangzhou 510000, Guangdong Province, China
-
Received:2025-08-08Revised:2025-10-18Online:2026-10-18Published:2026-03-05 -
Contact:Li Wei, MS, Associate chief physician, Shenzhen Pingle Orthopaedic Hospital, Shenzhen 518000, Guangdong Province, China -
About author:He Long, MS, Shenzhen Pingle Orthopaedic Hospital, Shenzhen 518000, Guangdong Province, China -
Supported by:Project of Guangdong Provincial Bureau of Traditional Chinese Medicine, No. 20241199 (to CC); Shenzhen Medical "Sanming Projects," No. SZZYSM202108013 (to LW)
摘要:
文题释义:
铁死亡:作为一种新型的铁依赖性非凋亡细胞死亡形式,在形态学上呈现出线粒体形态变小、膜密度增加、外膜破损、嵴减少甚至消失的特征。铁死亡的发生主要与胞质内活性氧或脂质过氧化物产生过多、堆积以及铁代谢的异常有关,使得细胞膜发生破损。
纳米药物:其核心是药物的纳米化技术,包括药物的直接纳米化和纳米载药系统。前者通过纳米沉淀技术或超细粉碎技术直接制备纳米药物颗粒;后者则通过将药物溶解、分散、包裹、吸附、偶联等方式与载体结合成为纳米分散体。药物经纳米化后,其物理化学性质以及生物学特性等发生改变,从而影响药物的吸收、分布、代谢和排泄,最终实现增强药物疗效、降低药物不良反应、提高药物效果等目的。
背景:实验证实铁死亡与多种骨科疾病有着密切联系,但调控铁死亡引起骨科疾病的具体机制尚不清晰,目前证据表明,信号通路可能是调控铁死亡发生的重要途径。
目的:概述参与调控骨科疾病(骨关节炎、脊髓损伤、骨质疏松、椎间盘退化、类风湿关节炎、骨肉瘤、激素性股骨头坏死)中铁死亡的相关信号通路,并描述通过调节信号通路的传导,靶向骨科疾病铁死亡途径的关键调节因子。深入研究骨科疾病中铁死亡的调控机制,为此类疾病的防治提供理论依据。
方法:检索PubMed和中国知网等数据库建库至2025年2月发表的铁死亡与骨科疾病的相关文献,英文检索词为“ferroptosis,osteoarthritis,osteoporosis,spinal cord injury,intervertebral disc degeneration,osteosarcomas,rheumatoid arthritis,steroid-induced osteonecrosis of the femoral head”,中文检索词为“铁死亡,骨关节炎,脊髓损伤,骨质疏松,椎间盘退化,类风湿关节炎,骨肉瘤,激素性股骨头坏死”,最终共纳入138篇文献进行综述分析。
结果与结论:①在多种信号通路的调控下,可以诱发细胞内铁离子、活性氧等物质堆积,引起成骨细胞、软骨细胞、骨肉瘤细胞等发生铁死亡,导致微环境发生变化,继而促进或抑制相关骨科疾病的发生;②研究证实信号通路调控铁死亡在骨科疾病的发生机制中具有重要意义;③但是目前信号通路、铁死亡和骨科疾病相互之间的作用机制仍处于初步阶段,需要进行更深入的研究,为治疗骨科疾病提供更多的策略。
https://orcid.org/0009-0009-3522-0074 (赫龙)
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
中图分类号:
引用本文
赫 龙, 高 爽, 陈 超, 覃国忠, 冉庆森, 王政春, 杨亚锋, 任 航, 邱云开, 杨 洋, 李 伟. 信号通路调控细胞铁死亡防治骨科疾病的新策略[J]. 中国组织工程研究, 2026, 30(29): 7619-7631.
He Long, Gao Shuang, Chen Chao, Qin Guozhong, Ran Qingsen, Wang Zhengchun, Yang Yafeng, Ren Hang, Qiu Yunkai, Yang Yang, Li Wei. A new strategy for preventing and treating orthopedic diseases by regulating ferroptosis through signaling pathways[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(29): 7619-7631.
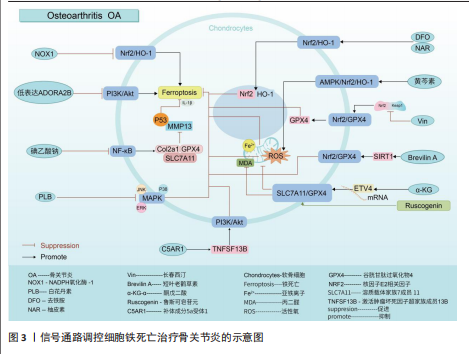
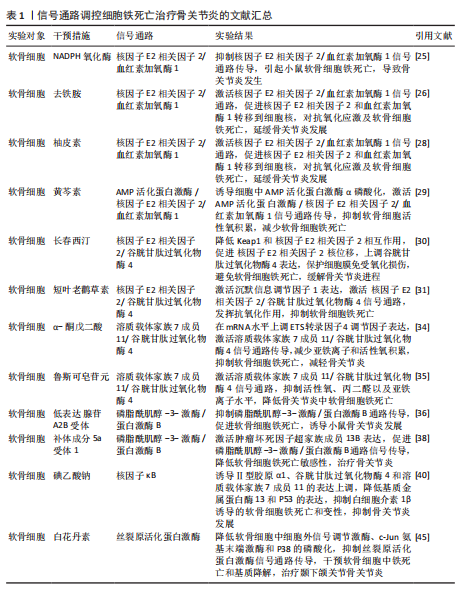
核因子E2相关因子2信号通路:核因子E2相关因子2作为一种抗氧化转录因子中的关键因子,可通过诱导血红素加氧酶1激活达到抑制氧化应激反应以及铁死亡的目的[23-24]。动物实验报道,NADPH氧化酶1是一种细胞内氧化反应的激活剂,当NADPH氧化酶1水平过高时会抑制核因子E2相关因子2/血红素加氧酶1信号通路的传导,致使氧化还原平衡被打破,继而引起小鼠软骨细胞的铁死亡,导致骨关节炎疾病的发生[25]。GUO等[26]通过对小鼠的关节腔内注射去铁胺(一种铁螯合剂,可用于治疗铁超负荷类疾病)发现,去铁胺通过激活核因子E2相关因子2/血红素加氧酶1信号通路[27],促进核因子E2相关因子2和血红素加氧酶1转移到细胞核中,以对抗氧化应激反应以及软骨细胞的铁死亡,延缓骨关节炎的发展。PAN等[28]通过从中药骨碎补中提取的柚皮素同样通过上调核因子E2相关因子2/血红素加氧酶1信号通路表达,减少软骨细胞中活性氧和脂质过氧化物的积累,减少氧化应激,减轻铁过载下的软骨损伤,达到治疗骨关节炎的目的。针对骨关节炎小鼠的实验发现,黄芩素通过诱导细胞中AMP活化蛋白激酶α磷酸化,激活AMP活化蛋白激酶/核因子E2相关因子2/血红素加氧酶1信号通路的传导,抑制软骨细胞内活性氧积累,减少软骨细胞的铁死亡[29]。长春西汀通过降低Keap1和核因子E2相关因子2的相互作用,减缓核因子E2相关因子2的降解以及促进核因子E2相关因子2的核位移,使得谷胱甘肽过氧化物酶4表达上调,将有害的磷脂过氧化物转化为无害的磷脂醇来保护细胞膜免受氧化损伤,避免软骨细胞发生铁死亡,缓解骨关节炎的进程[30]。在小鼠骨关节炎模型中,短叶老鹳草素通过激活沉默信息调节因子1表达,使得核因子E2相关因子2/谷胱甘肽过氧化物酶4信号通路激活,发挥抗氧化作用,抑制软骨细胞铁死亡,使得小鼠免受骨关节炎的侵害[31]。
溶质载体家族7成员11/谷胱甘肽过氧化物酶4信号通路:系统Xc-中溶质载体家族7成员11通过调控谷胱甘肽过氧化物酶4表达干预细胞铁死亡[32-33]。α-酮戊二酸因具有强大抗氧化、抗炎特性而闻名。目前有证据证实,α-酮戊二酸在mRNA水平上调ETS转录因子4调节因子表达,进而激活溶质载体家族7成员11/谷胱甘肽过氧化物酶4信号通路传导,减少亚铁离子和活性氧积累,抑制软骨细胞铁死亡,减轻骨关节炎[34]。鲁斯可皂苷元治疗骨关节炎疾病,同样是通过激活溶质载体家族7成员11/谷胱甘肽过氧化物酶4信号通络,抑制活性氧、丙二醛以及亚铁离子水平,达到降低骨关节炎中软骨细胞铁死亡的目的[35]。
磷脂酰肌醇-3-激酶/蛋白激酶B信号通路:磷脂酰肌醇-3-激酶/蛋白激酶B是丝氨酸/苏氨酸蛋白激酶家族成员之一,该信号通路具备促进代谢、增殖、生长和血管生成等多种功能。LI等[36]通过对引起软骨细胞铁死亡的基因筛选发现,低表达的腺苷A2B受体通过抑制磷脂酰肌醇-3-激酶/蛋白激酶B通路传导促进软骨细胞铁死亡,诱导小鼠骨关节炎的发展。另外,补体成分5a受体1被证实为骨关节炎发生发展过程中的关键基因[37]。通过骨关节炎大鼠的实验发现,补体成分5a受体1通过激活肿瘤坏死因子超家族成员13B表达,促进磷脂酰肌醇-3-激酶/蛋白激酶B通路信号传导,进而降低软骨细胞铁死亡的敏感性,达到治疗骨关节炎疾病的目的[38]。
核因子κB信号通路:核因子κB信号通路通过对骨关节炎中软骨代谢的影响促进或者抑制骨关节炎的发生发展[39]。当核因子κB信号通路传导被抑制时,诱导Ⅱ型胶原α1、谷胱甘肽过氧化物酶4和溶质载体家族7成员11的表达上调,同时降低基质金属蛋白酶13和P53的表达,从而抑制白细胞介素1β诱导的软骨细胞铁死亡和变性,抑制骨关节炎的发展[40]。不仅如此,骨关节炎早期软骨下骨的病理改变与破骨细胞异常激活密切相关,而核因子κB信号通路是激活破骨细胞形成的重要通路之一[41]。那是否可以得出碘乙酸钠通过核因子κB信号通路控制骨关节炎疾病中两种细胞的存活,这为未来骨关节炎的治疗提供了新的思路。
丝裂原活化蛋白激酶信号通路:丝裂原活化蛋白激酶信号通路由细胞外信号调节激酶、c-Jun氨基末端激酶和p38共同组成,当它们接受信号刺激时,会将信号从细胞膜传递至细胞核,以达到调控细胞的增殖分化、炎症以先天免疫等[42]。有关研究报道,人骨关节炎软骨中磷酸化的细胞外信号调节激酶、c-Jun氨基末端激酶以及p38丰富程度均高于正常软骨中的水平[43-44]。CUI等[45]通过白花丹素治疗大鼠颞下颌关节骨关节炎发现,白花丹素主要通过降低软骨细胞中 细胞外信号调节激酶、c-Jun氨基末端激酶和P38的磷酸化,抑制丝裂原活化蛋白激酶信号通路的传导,进而干预软骨细胞中铁死亡和基质降解,达到治疗颞下颌关节骨关节炎的目的。因此骨关节炎的发展可能与丝裂原活化蛋白激酶通路的激活有着密切相关性。
通过上述文献的总结可知,在多种信号通路的调控下可抑制软骨细胞的铁死亡,干预骨关节炎的发生。然而骨关节炎的发生并不仅仅局限于软骨细胞,滑膜细胞等同样是引起骨关节炎发生的重要因素。在骨关节炎疾病相关文献的收集中并未有关于信号通路调控滑膜细胞铁死亡的报道,而在类风湿关节炎的相关文献中,可见大量关于信号通路调控滑膜细胞铁死亡的报道,那么,同样的信号通路调控滑膜细胞铁死亡是否也可以干预骨关节炎的发生?这为未来的实验研究寻找了一个新的方向。见图3及表1。
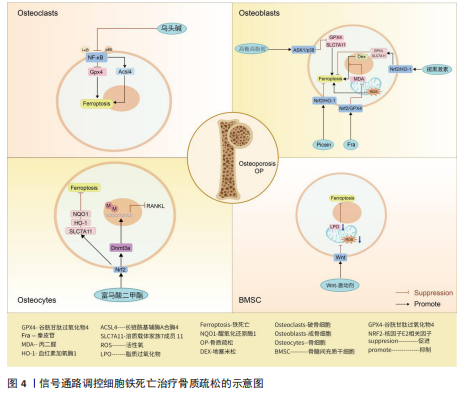
2.2 骨质疏松 骨质疏松是以骨密度减低、骨小梁等细微结构破坏而导致骨脆性增加、易发生骨折为特征的全身性代谢性骨病[46],其发生机制主要与骨代谢平衡被打破密切相关。机体内成骨细胞数量降低、骨基质合成减少等因素抑制骨形成,破骨细胞数量增加、骨吸收因子释放增加等因素上调骨吸收,当骨形成小于骨吸收,则引起骨流失、骨密度降低[47-48]。因此,具备促成骨细胞生成、存活或者抑制破骨细胞生成、凋亡的药物可能是治疗骨质疏松的新途径。根据报道显示,骨细胞、成骨细胞、破骨细胞以及骨髓间充质干细胞在铁死亡的调节下影响骨质疏松的发生发展[49-51]。
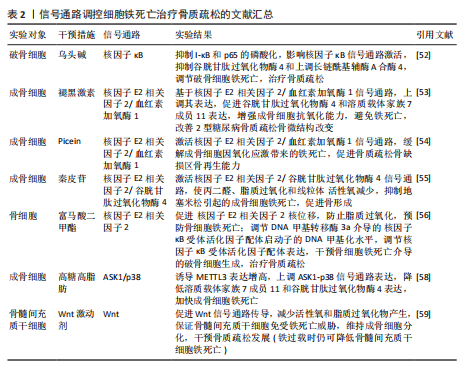
核因子κB信号通路:XUE等[52]发现乌头碱通过抑制I-κB和p65的磷酸化,进而影响核因子κB信号通路的激活,抑制谷胱甘肽过氧化物酶4和上调长链酰基辅酶A合酶4来调节破骨细胞铁死亡,达到治疗骨质疏松的目的。
核因子E2相关因子2信号通路:学者MA等[53]基于核因子E2相关因子2/血红素加氧酶1信号通路强效的抗氧化能力证实,褪黑激素可以影响核因子E2相关因子2/血红素加氧酶1信号通路表达上调,继而促进谷胱甘肽过氧化物酶4和溶质载体家族7成员11的表达,成骨细胞的抗氧化能力增强,避免细胞遭受氧化应激损伤引起的铁死亡,改善了2型糖尿病骨质疏松骨微结构的改变。HUANG等[54]根据实验发现,Picein治疗骨质疏松主要依赖于激活核因子E2相关因子2/血红素加氧酶1信号通路缓解成骨细胞因氧化应激反应带来的铁死亡,促进骨质疏松骨缺损区的骨再生能力。ZHENG等[55]发现,传统中草药秦皮的关键成分秦皮苷依赖于激活核因子E2相关因子2/谷胱甘肽过氧化物酶4信号通路,致使丙二醛、脂质过氧化和线粒体活性氧减少,有效抑制地塞米松引起的成骨细胞铁死亡,促进骨形成。另外,核因子E2相关因子2信号通路激活不仅通过促进 醌氧化还原酶1、溶质载体家族7成员11和血红素加氧酶1的表达,达到防止脂质过氧化,预防骨细胞铁死亡的目的[56];同时,核因子E2相关因子2可以调节DNA 甲基转移酶3a介导的核因子κB受体活化因子配体启动子的DNA甲基化水平来调节核因子κB受体活化因子配体(诱导破骨细胞生成)的表达[57],干预骨细胞铁死亡介导的破骨细胞生成,调节骨代谢,治疗骨质疏松[56]。
ASK1/p38信号通路:高糖高脂肪状态下会诱导过表达的甲基化转移酶3表达增高,进而上调ASK1/p38 信号通路表达,引起溶质载体家族7成员11和谷胱甘肽过氧化物酶4的表达降低,加快成骨细胞铁死亡,合理解释了2型糖尿病骨质疏松发病过程[58]。
Wnt信号通路:Wnt信号通路的激活可以减少活性氧和脂质过氧化物的产生,从而保证骨髓间充质干细胞免受铁死亡的威胁,维持成骨细胞分化,干预骨质疏松等疾病的发展[59]。但是,若细胞本身处于铁过载的情况,Wnt信
号通路的激活并不能逆转铁过载这一现象,当然,这并不妨碍其具备降低骨髓间充质干细胞铁死亡的能力[59]。由此可以得出,Wnt信号通路虽然在铁过载情况下不具备减少亚铁离子堆积的能力,但是可以是通过减少骨髓间充质干细胞氧化应激损伤而提高铁死亡的阈值,避免铁死亡的发生。
核因子E2相关因子2信号通路作为调控铁死亡发生的重要途径之一,在骨质疏松疾病中不仅可以干预成骨细胞的铁死亡,还可以通过干预核因子κB受体活化因子配体表达,影响骨细胞的破骨分化。众所周知,核因子κB受体活化因子配体是核因子κB的激活剂,两者相互结合诱导破骨细胞生成。由此可以得出推论核因子E2相关因子2信号通路干预骨细胞铁死亡以及破骨分化过程中,可能伴随着核因子κB信号通路的身影。见图4及表2。
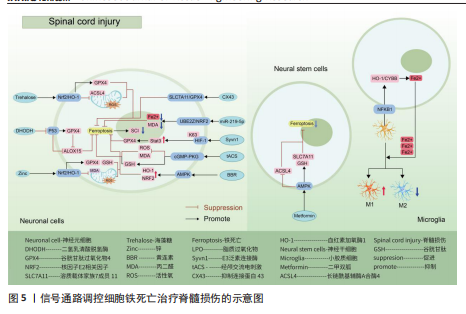
2.3 脊髓损伤 脊髓损伤是由交通意外等机械力引起脊柱骨折或脱位造成原发性损伤,随后引起局部出血、缺血、缺氧、离子失衡、自由基应激等继发性损伤,造成严重的中枢神经损伤,临床表现为截瘫、感觉丧失和其他脊髓异常[60-61]。脊髓损伤之所以具有高致残以及致死率,与其损伤后神经无法再生有着密切联系[62-63]。目前研究成果表明,在脊髓损伤继发性损伤时期,活性氧的过度积累引起的氧化应激反应可能是阻碍神经再生的关键因素[5,60,62]。
这与神经元磷脂膜中丰富的多不饱和脂肪酸残基和清除活性氧的能力差有关,使得神经元本质上容易受到氧化应激的影响有关[64-66]。而活性氧的过度积累引起的氧化应激反应是引起铁死亡的重要途径,因此,干预神经元铁死亡的治疗思路可能是提高神经元存活率的理想方法[67-69]。
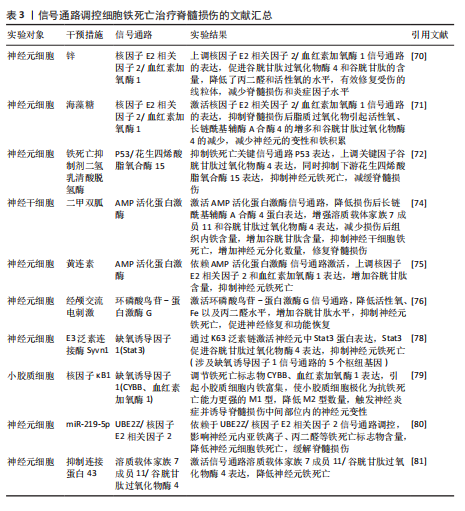
核因子E2相关因子2信号通路:GE等[70]通过脊髓损伤小鼠模型发现,锌通过上调核因子E2相关因子2/血红素加氧酶1信号通路的表达,增加谷胱甘肽过氧化物酶4和谷胱甘肽的含量,降低了丙二醛和活性氧的水平,有效修复受伤的线粒体,减少脊髓损伤和炎症因子水平。GONG等[71]同样运用核因子E2相关因子2/血红素加氧酶1信号通路强效抗氧化能力,运用海藻糖激活核因子E2相关因子2/血红素加氧酶1信号通路的表达,抑制脊髓损伤后脂质过氧化物引起活性氧、长链酰基辅酶A合酶4的增多和谷胱甘肽过氧化物酶4的减少,减少神经元的变性和铁积累,从而促进神经元的存活并改善运动功能的恢复。
P53/花生四烯酸脂氧合酶15信号通路:铁死亡抑制剂二氢乳清酸脱氢酶可以通过抑制P53/花生四烯酸脂氧合酶15信号通路的传导,抑制神经元铁死亡的发生,减缓脊髓损伤[72]。该实验一方面证实,二氢乳清酸脱氢酶是通过抑制铁死亡关键信号通路P53的表达,进而上调关键因子谷胱甘肽过氧化物酶4表达,干预神经元铁死亡;另一方面,二氢乳清酸脱氢酶与P53均可以通过抑制下游花生四烯酸脂氧合酶15的表达,达到抑制神经元铁死亡的目的。P53与花生四烯酸脂氧合酶15之间的相互作
用,可能是通过亚精胺/精胺N1-乙酰转移酶1作为中间介质来完成[73]。
AMP活化蛋白激酶信号通路:二甲双胍被证实可以促进脊髓损伤后的内源性神经干细胞增殖、神经元分化以及抑制铁死亡的发生,其原理主要是依赖激活AMP活化蛋白激酶信号通路,使得损伤后长链酰基辅酶A合酶4蛋白表达降低,溶质载体家族7成员11和谷胱甘肽过氧化物酶4的表达增强,有效减少损伤后组织内的铁含量,增加谷胱甘肽含量,有效改善了神经干细胞的铁死亡,并增加神经元分化数量,达到修复脊髓损伤的目的[74]。另外,黄连素同样是依赖于AMP活化蛋白激酶信号通路的激活,进而上调核因子E2相关因子2和血红素加氧酶1的表达,增加谷胱甘肽的含量,抑制脊髓损伤中神经元铁死亡[75]。
环磷酸鸟苷-蛋白激酶G信号通路:经颅交流电刺激是一种治疗脊髓损伤有效的康复手段,研究表明,其依靠激活环磷酸鸟苷-蛋白激酶G信号通路,从而降低活性氧、亚铁离子以及丙二醛的水平,增加谷胱甘肽水平,抑制神经元铁死亡,促进脊髓损伤患者神经修复和功能恢复[76]。
缺氧诱导因子1信号通路:通过GEO数据库分析发现,缺氧诱导因子1信号通路在脊髓损伤中发挥重要作用,这与缺氧诱导因子1 信号通路的5个枢纽基因(Stat3、Tlr4、血红素加氧酶1、Hif1a和CYBB)有着密切联系[77]。其中,E3泛素连接酶 Syvn1正是通过K63 泛素链激活神经元中Stat3蛋白表达,继而Stat3 促进谷胱甘肽过氧化物酶4表达,抑制神经元铁死亡的发生[78]。另外,转录因子核因子κB1通过调节铁死亡标志物CYBB、血红素加氧酶1表达,引起脊髓损伤中小胶质细胞内铁富集,使得小胶质细胞更易极化为抗铁死亡能力更强的M1型,造成对铁死亡更敏感的M2型数量的降低,触发神经炎症并诱导脊髓损伤中间部位内的神经元变性[79]。通过缺氧诱导因子1 信号通路上的枢纽基因Stat3、血红素加氧酶1可知,在调控铁死亡过程中信号通路存在相互干预的影响,并非完全平行的关系。
UBE2Z/核因子E2相关因子2信号通路:骨髓间充质干细胞来源的外泌体 miR-219-5p依赖于UBE2Z/核因子E2相关因子2信号通路的调控,影响神经元内亚铁离子、丙二醛等铁死亡标志物含量,降低神经元细胞铁死亡,达到缓解脊髓损伤的作用[80]。
溶质载体家族7成员 11/谷胱甘肽过氧化物酶4信号通路:抑制连接蛋白43治疗脊髓损伤,其原理是依赖激活信号通路溶质载体家族7成员 11/谷胱甘肽过氧化物酶4表达,降低神经元铁死亡实现的[81]。
神经炎症反应是创伤后脊髓损伤的重要反应。最早参与炎症反应的细胞是小胶质细胞和中性粒细胞。铁死亡的过程通常伴有炎症反应。目前研究表明,海藻糖或者锌处理后,在保护神经元避免铁死亡的同时,脑脊液和脊髓组织中白细胞介素1β、白细胞介素6和肿瘤坏死因子α 的水平降低,但其具体的作用机制以及是否与铁死亡有关并不清晰,可能与小胶质细胞铁离子富集诱导M1型分化增多有关,具体还需要进一步实验证实。见图5及表3。
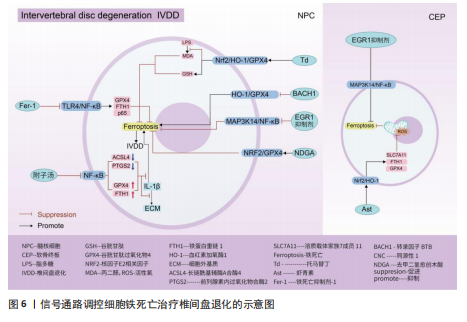
2.4 椎间盘退化 椎间盘退化是连接2个相邻椎骨的椎间盘疾病,是引起临床常见疾病腰部、背部、颈部疼痛发生的主要原因之一[82-83]。从解剖角度出发,椎间盘位于2个椎体之间,由髓核、纤维环和软骨终板组成。从病理生理角度出发,髓核细胞死亡、纤维环裂、软骨终板和细胞外基质代谢失衡均是引起椎间盘退化的重要机制[84]。目前研究表明,铁死亡可通过干预髓核细胞死亡、软骨终板细胞死亡和细胞外基质降解等形式,影响椎间盘退化的进程[85-87]。
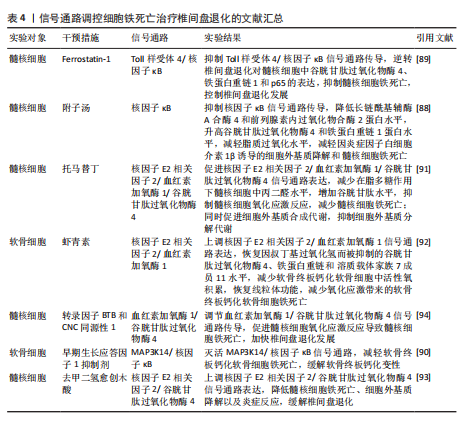
核因子κB信号通路:中药附子汤治疗椎间盘退化是依赖于抑制核因子κB信号通路传导,进而降低长链酰基辅酶A合酶4和前列腺素内过氧化物合酶2蛋白水平,升高谷胱甘肽过氧化物酶4 和铁蛋白重链1蛋白水平,减轻脂质过氧化水平,从而减轻因炎症因子白细胞介素1β诱导的细胞外基质降解和髓核细胞铁死亡[88]。WANG等[89]通过实验表明,经典铁
死亡抑制剂Ferrostatin-1通过抑制Toll样受体4/核因子κB信号通路的传导,可以逆转椎间盘退化对髓核细胞中谷胱甘肽过氧化物酶4、铁蛋白重链1和p65的表达,达到抑制髓核细胞铁死亡的目的,控制椎间盘退化的发展。早期生长应答因子1抑制剂通过灭活MAP3K14/核因子κB信号通路,从而减轻软骨细胞铁死亡,同时缓解软骨终板钙化变性[90]。
核因子E2相关因子2信号通路:LI等[91]发现托马替丁不仅通过促进细胞外基质合成代谢,抑制细胞外基质分解代谢的作用影响椎间盘退化的发生;托马替丁还可以通过促进核因子E2相关因子2/血红素加氧酶1/谷胱甘肽过氧化物酶4信号通路的表达,减少在脂多糖作用下髓核细胞中丙二醛的水平,增加谷胱甘肽的水平,抑制髓核细胞氧化应激反应,减少髓核细胞铁死亡。核因子E2相关因子2/血红素加氧酶1/谷胱甘肽过氧化物酶4信号通路不仅可以调控髓核细胞铁死亡,还具备调控软骨终板钙化软骨细胞铁死亡的能力。虾青素便是依赖上调核因子E2相关因子2/血红素加氧酶1信号通路表达,恢复因叔丁基过氧化氢(具备模拟椎间盘退化环境的能力)而被抑制的谷胱甘肽过氧化物酶4、铁蛋白重链1和溶质载体家族7成员11水平,减少软骨终板钙化软骨细胞中活性氧积累,恢复线粒体功能,减少氧化应激反应带来的软骨终板钙化软骨细胞铁死亡[92]。去甲二氢愈创木酸通过上调核因子E2相关因子2/谷胱甘肽过氧化物酶4信号通路表达,可降低髓核细胞铁死亡、细胞外基质降解以及炎症反应,达到缓解椎间盘退化的目的[93]。
血红素加氧酶1/谷胱甘肽过氧化物酶4信号通路:转录因子BTB和CNC同源性1通过抑制血红素加氧酶1/谷胱甘肽过氧化物酶4信号通路的传导,可促进髓核细胞氧化应激反应导致髓核细胞铁死亡,加快椎间盘退化的发展[94]。在这里转录因子BTB和CNC同源性1直接靶向血红素加氧酶1/谷胱甘肽过氧化物酶4信号通路,而不是通过核因子E2相关因子2信号通路影响血红素加氧酶1表达,说明药物直接靶向下游信号通路同样可以调控铁死亡的发生。
椎间盘退化并不是常规意义的骨科疾病,但它是脊柱退行性疾病发生的源头,因此了解椎间盘退化的全过程对治疗脊柱退行性疾病具有深远的意义。基于上述通过药物调控信号通路干预相关细胞铁死亡的发生治疗椎间盘退化,有望通过药物治疗的手段去缓解或逆转椎间盘退化的发生。但是,药物治疗需要通过血管传输,但鉴于椎间盘特殊的微环境,而其自身无血管结构可能会影响治疗药物的有效传输,在下一步研究中希望得到解决。见图6及表4。
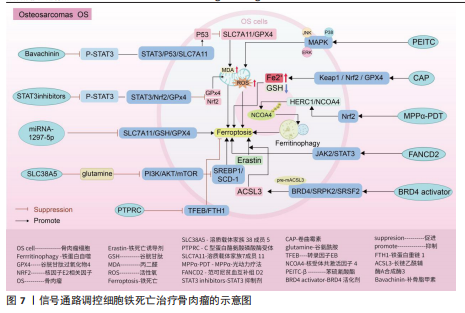
2.5 骨肉瘤 骨肉瘤是一种由间充质干细胞所引发的原发性恶性肿瘤,好发部位为长骨两端的生长板处,在颅骨、下颌骨和骨盆等扁骨中较少出现,多见于15-19岁的青少年人群[95-96]。目前,对于该病常用的治疗方法包括手术治疗、基因治疗、免疫治疗、化疗和分子靶向治疗。新辅助化疗方案的出现使患者的5年生存率提升至70%,但对转移或复发患者,5年生存率仍然只有20%-30%,并且伴随着严重的不良反应及全身系统性的毒性反应[97-99]。近年来研究表明铁死亡与肿瘤的病理发展过程密切相关,针对肿瘤细胞铁死亡的靶向诱导成为治疗肿瘤疾病的新的方向,其中包含骨肉瘤[100-101]。
丝裂原活化蛋白激酶信号通路:经典的肿瘤化学药物β-苯硫氰酸酯通过对丝裂原活化蛋白激酶信号通路中细胞外信号调节激酶、p38和c-Jun氨基末端激酶表达激活,促进骨肉瘤细胞中活性氧过度积累,逆转N-乙酰基-L-半胱氨酸作用下骨肉瘤细胞对铁死亡产生的抗性作用,加快骨肉瘤细胞铁死亡[102]。
Keap1/核因子E2相关因子2 /谷胱甘肽过氧化物酶4信号通路:随着纳米颗粒的发展,通过携带传统抗结核药物卷曲霉素的骨靶向外泌体具备直接靶向骨肉瘤细胞的能力,其通过激活骨肉瘤细胞中Keap1/核因子E2相关因子2/谷胱甘肽过氧化物酶4信号通路表达,诱导亚铁离子聚集、脂质过氧化和谷胱甘肽减少,促进骨肉瘤细胞发生铁死亡,在对机体无明显不良反应的情况下提高患者的生存率[103]。
核因子E2相关因子2信号通路:光动力疗法作为一种无创、低毒性的新型治疗方法,已被证实是治疗骨肉瘤的有效方法[104-107]。研究表明,MPPα-光动力疗法通过激活核因子E2相关因子2信号通路,进而影响下游靶基因HERC1激活,HERC1作为核受体共激活因子 4的E3泛素连接酶,靶向HERC1,成功抑制了核受体共激活因子4蛋白的泛素化和降解,导致铁蛋白自噬和骨肉瘤细胞铁死亡的激活[108]。
信号转导和转录激活因子3信号通路:研究表明,类黄酮补骨脂甲素可以抑制信号转导和转录激活因子3磷酸化,继而基于信号转导和转录激活因子3/p53/溶质载体家族7成员11信号通路的传导,上调p53表达,抑制溶质载体家族7成员 11、谷胱甘肽过氧化物酶4表达,促进活性氧和丙二醛积累,加快骨肉瘤细胞铁死亡速度[109]。不仅如此,抑制信号转导和转录激活因子3磷酸化还可影响信号转导和转录激活因子3/核因子E2相关因子2/谷胱甘肽过氧化物酶4信号通路的传导,导致核因子E2相关因子2和谷胱甘肽过氧化物酶4蛋白水平降低,导致细胞内丙二醛、活性氧大量堆积,诱导骨肉瘤细胞铁死亡并增加骨肉瘤细胞对顺铂的敏感性[110]。另外,沉默的范可尼贫血互补组 D2通过依赖激活 JAK2/信号转导和转录激活因子3 信号通路表达诱导骨肉瘤细胞铁死亡,抑制骨肉瘤的生长[111]。
溶质载体家族7成员 11/谷胱甘肽/谷胱甘肽过氧化物酶4 信号通路:柳氮磺吡啶、替拉帕嗪和 miRNA-1297-5p可通过抑制 溶质载体家族7成员 11/谷胱甘肽/谷胱甘肽过氧化物酶4 信号通路传导促进骨肉瘤细胞铁死
亡[112-114]。
磷脂酰肌醇-3-激酶/蛋白激酶B/哺乳动物雷帕霉素靶蛋白信号通路:沉默的溶质载体家族38成员5在转运谷氨酰胺介导下抑制磷脂酰肌醇-3-激酶/蛋白激酶B/哺乳动物雷帕霉素靶蛋白信号通路传导,继而抑制调节元件结合蛋白1/硬脂酰辅酶A去饱和酶1信号通路的传导,从而促进骨肉瘤细胞的铁死亡[115]。
转录因子EB/铁蛋白重链 1 信号通路:C型蛋白酪氨酸磷酸酶受体通过阻断转录因子EB/铁蛋白重链1信号传导抑制骨肉瘤细胞的铁死亡,促进骨肉瘤的增殖、迁移和侵袭[116]。
溴结构域蛋白4/丝氨酸精氨酸的蛋白激酶2/丝氨酸精氨酸剪接因子2信号通路:溴结构域蛋白4/丝氨酸精氨酸的蛋白激酶2/丝氨酸精氨酸剪接因子2信号通路通过调控剪切长链乙酰辅酶A合成酶3的RNA前体的效率,确保骨肉瘤细胞中长链乙酰辅酶A合成酶3的丰度,而长链乙酰辅酶A合成酶3的AMP结合结构域是花生四烯酸合成代谢所必需的,该物质的合成有助于增加骨肉瘤细胞对铁死亡的易感性,因此溴结构域蛋白 4/丝氨酸精氨酸的蛋白激酶2/丝氨酸精氨酸剪接因子2信号通路可能是治疗骨肉瘤的潜在靶标[117]。
癌细胞通常具有较高的活性氧水平以维持其恶性增殖,而较高的内源性活性氧水平同样使它们更容易受到氧化应激伤害。因此氧化还原系统的改变和抗氧化系统的功能障碍可能有助于改善耐药性和抗癌治疗的重要途径。而通过上述综述可知,多种药物通过靶向信号通路可以加快骨肉瘤细胞中活性氧的堆积,诱发骨肉瘤细胞的铁死亡,明确了信号通路、铁死亡与骨肉瘤细胞三者之间的密切联系,为骨肉瘤的治疗提供新的方向。但是活性氧的堆积引起的氧化应激反应不仅触发癌细胞的铁死亡,其还引起癌细胞的自噬以及细胞凋亡等程序性死亡的发生。关于骨肉瘤细胞中不同程序性死亡之间是否发生串扰以及同一条信号通路是否可以控制多种程序性死亡的发生,需要进一步通过实验明确。见图7及表5。
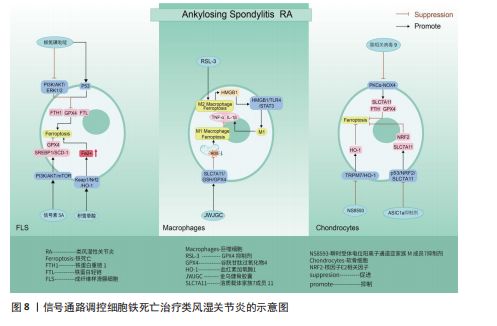
2.6 类风湿关节炎 类风湿关节炎是一种由多因素引起的自身免疫性疾病,其特征主要包含滑膜炎症、关节软骨破坏以及骨侵蚀,在临床

上表现为关节疼痛、肿胀、畸形,甚至残疾[118]。滑膜炎症反应主要是指滑膜内膜层发生的炎症发应,与成纤维样滑膜细胞、滑膜巨噬细胞通过分泌软骨基质酶和细胞因子,促进免疫细胞的浸润有关[119-120]。关节软骨作为关节的重要结构,软骨的退化与损伤是引起类风湿关节炎患者功能障碍的另一重要原因[121-122]。而软骨细胞作为唯一构成软骨的细胞类型,其通过调节软骨基质蛋白(如Ⅱ型胶原蛋白和蛋白多糖)的产生、更新和降解[123],从而维持软骨完整性[124]。因此,通过信号通路的调节干预成纤维样滑膜细胞、滑膜巨噬细胞以及软骨细胞铁死亡的发生,可能会成为类风湿关节炎治疗的新策略。
磷脂酰肌醇-3-激酶/蛋白激酶B、P53信号通路:ZHAO等[125]通过柳氮磺吡啶治疗类风湿关节炎大鼠发现,柳氮磺吡啶主要通过抑制磷脂酰肌醇-3-激酶/蛋白激酶B/细胞外信号调节激酶1/2和促进P53信号通路表达,达到干预成纤维样滑膜细胞中谷胱甘肽过氧化物酶4、铁蛋白重链1、铁蛋白轻链等相关蛋白的表达,促进成纤维样滑膜细胞发生铁死亡,抑制成纤维样滑膜细胞迁移至软骨并对软骨产生侵袭及破坏作用。但在基于柳氮磺吡啶处理下添加磷脂酰肌醇-3-激酶/蛋白激酶B/细胞外信号调节激酶1/2信号通路激活剂以及P53信号通路抑制剂时发现,P53抑制剂的添加会使得成纤维样滑膜细胞活力恢复更佳,侧面说明P53通路在干预成纤维样滑膜细胞铁死亡治疗类风湿关节炎过程中起着更关键的作用。抑制酸敏感离子通道1a 的表达可以抑制p53/核因子E2相关因子2/溶质载体家族7成员11通路中p53的表达,促进软骨细胞中核因子E2相关因子2和溶质载体家族7成员11的表达,减轻类风湿关节炎关节软骨细胞因酸化诱导的铁死亡发生,并抑制因细胞外酸化而发生的细胞外钙内流,抑制关节软骨细胞的退化[126]。信号素5A依赖激活磷脂酰肌醇-3-激酶/蛋白激酶B/哺乳动物雷帕霉素靶蛋白信号通路上调谷胱甘肽过氧化物酶4表达以及调节元件结合蛋白1/硬脂酰辅酶A去饱和酶1信号通路的传导,使得类风湿关节炎-成纤维样滑膜细胞对铁死亡产生抗性[127]。其中磷脂酰肌醇-3-激酶/蛋白激酶B/哺乳动物雷帕霉素靶蛋白信号通路上调谷胱甘肽过氧化物酶4表达,可能与抑制下游分子4E-BP1有关[128]。
Keap1/核因子E2相关因子2/血红素加氧酶1信号通路:SUN等[129]通过积雪草酸治疗类风湿关节炎发现,积雪草酸通过调节Keap1/核因子E2相关因子2/血红素加氧酶1信号通路表达促进亚铁离子的积累,加快类风湿关节炎-成纤维样滑膜细胞铁死亡,有效缓解类风湿关节炎大鼠模型关节炎症反应。
溶质载体家族7成员11/谷胱甘肽/谷胱甘肽过氧化物酶4信号通路:金乌健骨胶囊依赖于调控经典铁死亡信号通路溶质载体家族7成员 11/谷胱甘肽/谷胱甘肽过氧化物酶4的表达,降低活性氧积累,抑制M1巨噬细胞铁死亡,降低肿瘤坏死因子α、白细胞介素1β等相关炎性因子水平,缓解类风湿关节炎的临床表现[130]。但是,FENG等[131]发现,在铁含量高的类风湿关节炎滑膜环境中,M2发生铁死亡的概率高于M1,M2发生铁死亡后会产生高迁移率族蛋白1(内源性危险信号),继而引起高迁移率族蛋白1/Toll样受体4/信号转导和转录激活因子3信号通路的激活,诱导M1巨噬细胞释放肿瘤坏死因子α、白细胞介素1β等炎性细胞因子,促进了类风湿关节炎滑膜中炎症微环境的形成。众所周知,M1/M2平衡是维持关节内炎性状态的关键。通过文献可知M2铁死亡状态下通过信号通路促进M1炎性因子的释放。但是金乌健骨胶囊在抑制M1铁死亡的状态下,却得出抑制相关炎性因子水平的结论,这两种文献给出相悖的结论,可能需要进一步实验。
瞬时受体电位阳离子通道亚家族 M成员7/血红素加氧酶1、PKCα-NADPH氧化酶4信号通路:HAO等[132]通过NS8593对瞬时受体电位阳离子通道亚家族M成员7/血红素加氧酶1信号通路中瞬时受体电位阳离子通道亚家族M成员7表达的抑制,上调血红素加氧酶1的表达,有效减轻软骨细胞铁死亡,缓解类风湿关节炎患者关节软骨的损伤。腺相关病毒9抑制瞬时受体电位阳离子通道亚家族M成员7的表达可以阻断PKCα-NADPH氧化酶4信号通路的传导,增加溶质载体家族7成员11、铁蛋白重链和谷胱甘肽过氧化物酶4表达,减轻类风湿关节炎软骨细胞的铁死亡[133]。
通过上述文献总结可知,信号通路的调控对软骨细胞、成纤维样滑膜细胞以及巨噬细胞的铁死亡具有重要的意义。而类风湿关节炎作为一种慢性自身免疫性炎症疾病,其发生发展与免疫T细胞息息相关,但是,目前尚未有关于信号通路调控T细胞铁死亡治疗类风湿关节炎的相关报道,未来可以加大相关研究的探索。见图8及表6。
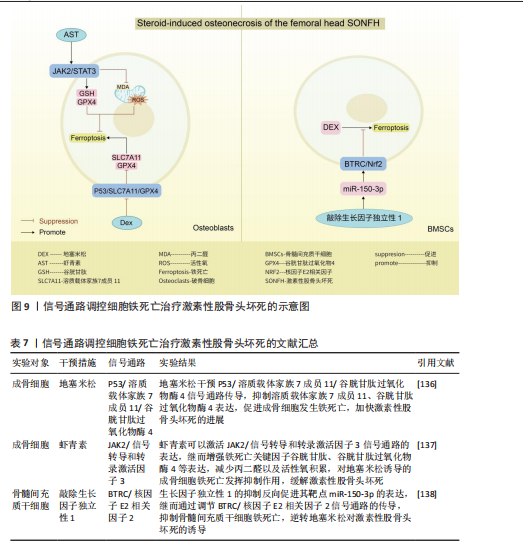
2.7 激素性股骨头坏死 激素性股骨头坏死是指类固醇的滥用引起骨细胞缺血、坏死、骨小梁断裂,临床表现为髋部疼痛、股骨头塌陷和关节功能障碍等,严重者可导致残疾[134]。由于多数患者缺少对激素性股骨头坏死的认知,加之坏死组织修复困难、致残率高,如果不进行早期临床干预,大多数股骨头坏死周围塌陷的患者最终需要进行髋关节置换手术[135]。手术治疗是最后手段,通过药物缓解或逆转激素性股骨头坏死的发展也许是未来治疗该病的更佳方案。目前已知铁死亡可以通过调控相关骨细胞死亡的进程,可能是治疗激素性股骨头坏死的新方向。
P53/溶质载体家族7成员 11/谷胱甘肽过氧化物酶4信号通路:SUN等[136]通过实验证实,地塞米松通过干预P53/溶质载体家族7成员11/谷胱甘肽过氧化物酶4信号通路传导,抑制溶质载体家族7成员11、谷胱甘肽过氧化物酶4表达,促进成骨细胞发生铁死亡,加快激素性股骨头坏死的进展。因此通过干预相关细胞铁死亡发生也许可以逆转类固醇诱发激素性股骨头坏死的结果。
JAK2/信号转导和转录激活因子3信号通路:LIN等[137]通过对激素性股骨头坏死大

鼠骨坏死区注射虾青素发现,虾青素可以激活 JAK2/信号转导和转录激活因子3信号通路的表达,继而增强铁死亡关键因子谷胱甘肽、谷胱甘肽过氧化物酶4等表达,减少丙二醛以及活性氧积累,对地塞米松诱导的成骨细胞铁死亡发挥抑制作用,缓解激素性股骨头坏死。
BTRC/核因子E2相关因子2信号通路:ZHENG等[138]通过沉默生长因子独立性1发现,生长因子独立性1的抑制反向促进其靶点miR-150-3p的表达,继而通过调节BTRC/核因子E2相关因子2信号通路的传导,抑制骨髓间充质干细胞铁死亡,逆转地塞米松对激素性股骨头坏死的诱导。
关于信号通路调控铁死亡治疗激素性股骨头坏死的研究较少,目前有很大可待挖掘的领域,例如,信号通路是否能干预内皮细胞、脂肪细胞铁死亡,从而加快血管损伤的修复、降低髓内压,达到改善股骨头内微循环的目的。见图9及表7。
| [1] KARSENTY G, FERRON M. The contribution of bone to whole-organism physiology. Nature. 2012;481(7381):314-320. [2] MAFFULLI N. Osteoarthritis and the Middle Aged Athlete: The Present and Future. Sports Med Arthrosc Rev. 2022;30(2):77. [3] NAGY G, ROODENRIJS NMT, WELSING PMJ, et al. EULAR points to consider for the management of difficult-to-treat rheumatoid arthritis. Ann Rheum Dis. 2022;81(1):20-33. [4] CHAPARRO-SANABRIA JA, BAUTISTA-MOLANO W, BELLO-GUALTERO JM, et al. Association of adipokines with rheumatic disease activity indexes and periodontal disease in patients with early rheumatoid arthritis and their first-degree relatives. Int J Rheum Dis. 2019;22(11):1990-2000. [5] ANJUM A, YAZID MD, FAUZI DAUD M, et al. Spinal Cord Injury: Pathophysiology, Multimolecular Interactions, and Underlying Recovery Mechanisms. Int J Mol Sci. 2020; 21(20):7533. [6] KERR JF, WYLLIE AH, CURRIE AR. Apoptosis: a basic biological phenomenon with wide-ranging implications in tissue kinetics. Br J Cancer. 1972;26(4):239-257. [7] CHEN H, HAN Z, WANG Y, et al. Targeting Ferroptosis in Bone-Related Diseases: Facts and Perspectives. J Inflamm Res. 2023;16: 4661-4677. [8] DIXON SJ, WINTER GE, MUSAVI LS, et al. Human Haploid Cell Genetics Reveals Roles for Lipid Metabolism Genes in Nonapoptotic Cell Death. ACS Chem Biol. 2015;10(7):1604-1609. [9] LI Y, MAHER P, SCHUBERT D. A role for 12-lipoxygenase in nerve cell death caused by glutathione depletion. Neuron. 1997;19(2): 453-463. [10] SEILER A, SCHNEIDER M, FÖRSTER H, et al. Glutathione peroxidase 4 senses and translates oxidative stress into 12/15-lipoxygenase dependent- and AIF-mediated cell death. Cell Metab. 2008;8(3):237-248. [11] CONRAD M, PRATT DA. The chemical basis of ferroptosis. Nat Chem Biol. 2019;15(12):1137-1147. [12] GAO L, HUA W, TIAN L, et al. Molecular Mechanism of Ferroptosis in Orthopedic Diseases. Cells. 2022;11(19):2979. [13] LI J, CAO F, YIN HL, et al. Ferroptosis: past, present and future. Cell Death Dis. 2020; 11(2):88. [14] FORCINA GC, DIXON SJ. GPX4 at the Crossroads of Lipid Homeostasis and Ferroptosis. Proteomics. 2019;19(18):e1800311. [15] WANG W, GREEN M, CHOI JE, et al. CD8+ T cells regulate tumour ferroptosis during cancer immunotherapy. Nature. 2019; 569(7755):270-274. [16] WU J, MINIKES AM, GAO M, et al. Intercellular interaction dictates cancer cell ferroptosis via NF2-YAP signalling. Nature. 2019;572(7769):402-406. [17] ABDALKADER M, LAMPINEN R, KANNINEN KM, et al. Targeting Nrf2 to Suppress Ferroptosis and Mitochondrial Dysfunction in Neurodegeneration. Front Neurosci. 2018; 12:466. [18] ARTYUKHOVA MA, TYURINA YY, CHU CT, et al. Interrogating Parkinson’s disease associated redox targets: Potential application of CRISPR editing. Free Radic Biol Med. 2019;144:279-292. [19] SONG X, XIE Y, KANG R, et al. FANCD2 protects against bone marrow injury from ferroptosis. Biochem Biophys Res Commun. 2016;480(3):443-449. [20] HUNTER DJ, BIERMA-ZEINSTRA S. Osteoarthritis. Lancet. 2019;393(10182): 1745-1759. [21] YAO Q, WU X, TAO C, et al. Osteoarthritis: pathogenic signaling pathways and therapeutic targets. Signal Transduct Target Ther. 2023; 8(1):56. [22] ZHANG S, XU J, SI H, et al. The Role Played by Ferroptosis in Osteoarthritis: Evidence Based on Iron Dyshomeostasis and Lipid Peroxidation. Antioxidants (Basel). 2022;11(9):1668. [23] ZHAO L, TAO X, QI Y, et al. Protective effect of dioscin against doxorubicin-induced cardiotoxicity via adjusting microRNA-140-5p-mediated myocardial oxidative stress. Redox Biol. 2018;16:189-198. [24] LIU X, YUAN X, LIANG G, et al. z. Free Radic Biol Med. 2020;160:820-836. [25] TAO L, YANG K, WANG K, et al. NOX1-mediated oxidative stress induces chondrocyte ferroptosis by inhibiting the Nrf2/HO-1 pathway. Sci Rep. 2024;14(1):19877. [26] GUO Z, LIN J, SUN K, et al. Deferoxamine Alleviates Osteoarthritis by Inhibiting Chondrocyte Ferroptosis and Activating the Nrf2 Pathway. Front Pharmacol. 2022;13: 791376. [27] YAO X, ZHANG Y, HAO J, et al. Deferoxamine promotes recovery of traumatic spinal cord injury by inhibiting ferroptosis. Neural Regen Res. 2019;14(3):532-541. [28] PAN Z, HE Q, ZENG J, et al. Naringenin protects against iron overload-induced osteoarthritis by suppressing oxidative stress. Phytomedicine. 2022;105:154330. [29] WAN Y, SHEN K, YU H, et al. Baicalein limits osteoarthritis development by inhibiting chondrocyte ferroptosis. Free Radic Biol Med. 2023;196:108-120. [30] WANG J, YANG J, FANG Y, et al. Vinpocetine protects against osteoarthritis by inhibiting ferroptosis and extracellular matrix degradation via activation of the Nrf2/GPX4 pathway. Phytomedicine. 2024;135:156115. [31] RUAN Q, WANG C, ZHANG Y, et al. Brevilin A attenuates cartilage destruction in osteoarthritis mouse model by inhibiting inflammation and ferroptosis via SIRT1/Nrf2/GPX4 signaling pathway. Int Immunopharmacol. 2023;124(Pt B): 110924. [32] DIXON SJ, PATEL DN, WELSCH M, et al. Pharmacological inhibition of cystine-glutamate exchange induces endoplasmic reticulum stress and ferroptosis. Elife. 2014;3: e02523. [33] CHEN X, KANG R, KROEMER G, et al. Ferroptosis in infection, inflammation, and immunity. J Exp Med. 2021;218(6): e20210518. [34] HE R, WEI Y, PENG Z, et al. α-Ketoglutarate alleviates osteoarthritis by inhibiting ferroptosis via the ETV4/SLC7A11/GPX4 signaling pathway. Cell Mol Biol Lett. 2024; 29(1):88. [35] RUAN Q, WANG C, ZHANG Y, et al. Ruscogenin attenuates cartilage destruction in osteoarthritis through suppressing chondrocyte ferroptosis via Nrf2/SLC7A11/GPX4 signaling pathway. Chem Biol Interact. 2024;388:110835. [36] LI S, HAN J, CAO J, et al. ADORA2B, transcriptionally suppressing by MYC, promotes ferroptosis of chondrocytes via inhibition of the PI3K/Akt pathway in mice with osteoarthritis. Environ Toxicol. 2024;39(5):2487-2501. [37] LIU Y, LU T, LIU Z, et al. Six macrophage-associated genes in synovium constitute a novel diagnostic signature for osteoarthritis. Front Immunol. 2022;13:936606. [38] LV M, CAI Y, HOU W, et al. The C5AR1/TNFSF13B axis alleviates osteoarthritis by activating the PI3K/Akt/GSK3β/Nrf2/HO-1 pathway to inhibit ferroptosis. Exp Cell Res. 2024;441(2):114195. [39] JIMI E, FEI H, NAKATOMI C. NF-κB Signaling Regulates Physiological and Pathological Chondrogenesis. Int J Mol Sci. 2019;20(24):6275. [40] HAN J, ZHAN LN, HUANG, et al. Moderate mechanical stress suppresses chondrocyte ferroptosis in osteoarthritis by regulating NF-κB p65/GPX4 signaling pathway. Sci Rep. 2024;14(1):5078. [41] FANG C, GUO JW, WANG YJ, et al. Diterbutyl phthalate attenuates osteoarthritis in ACLT mice via suppressing ERK/c-fos/NFATc1 pathway, and subsequently inhibiting subchondral osteoclast fusion. Acta Pharmacol Sin. 2022;43(5):1299-1310. [42] KIM EK, CHOI EJ. Compromised MAPK signaling in human diseases: an update. Arch Toxicol. 2015;89(6):867-882. [43] CLANCY R, REDISKE J, KOEHNE C, et al. Activation of stress-activated protein kinase in osteoarthritic cartilage: evidence for nitric oxide dependence. Osteoarthritis Cartilage. 2001;9(4):294-299. [44] FAN Z, SÖDER S, OEHLER S, et al. Activation of interleukin-1 signaling cascades in normal and osteoarthritic articular cartilage. Am J Pathol. 2007;171(3):938-946. [45] CUI T, LAN Y, YU F, et al. Plumbagin alleviates temporomandibular joint osteoarthritis progression by inhibiting chondrocyte ferroptosis via the MAPK signaling pathways. Aging (Albany NY). 2023;15(22):13452-13470. [46] QADIR A, LIANG S, WU Z, et al. Senile Osteoporosis: The Involvement of Differentiation and Senescence of Bone Marrow Stromal Cells. Int J Mol Sci. 2020; 21(1):349. [47] QIN Y, YANG X, NING Z. Causal roles of educational duration in bone mineral density and risk factors for osteoporosis: a Mendelian randomization study. BMC Musculoskelet Disord. 2024;25(1):345. [48] WANG D, YANG Y. The Relationship Between Serum 25-Hydroxyvitamin D Levels and Osteoporosis in Postmenopausal Women. Clin Interv Aging. 2023;18:619-627. [49] YANG Y, JIANG Y, QIAN D, et al. Prevention and treatment of osteoporosis with natural products: Regulatory mechanism based on cell ferroptosis. J Orthop Surg Res. 2023;18(1):951. [50] JING Z, LI Y, ZHANG H, et al. Tobacco toxins induce osteoporosis through ferroptosis. Redox Biol. 2023;67:102922. [51] YIN Y, CHEN GJ, YANG C, et al. Osteocyte ferroptosis induced by ATF3/TFR1 contributes to cortical bone loss during ageing. Cell Prolif. 2024;57(10):e13657. [52] XUE C, LUO H, WANG L, et al. Aconine attenuates osteoclast-mediated bone resorption and ferroptosis to improve osteoporosis via inhibiting NF-κB signaling. Front Endocrinol (Lausanne). 2023;14: 1234563. [53] MA H, WANG X, ZHANG W, et al. Melatonin Suppresses Ferroptosis Induced by High Glucose via Activation of the Nrf2/HO-1 Signaling Pathway in Type 2 Diabetic Osteoporosis. Oxid Med Cell Longev. 2020; 2020:9067610. [54] HUANG L, WANG J, YU J, et al. Picein alleviates oxidative stress and promotes bone regeneration in osteoporotic bone defect by inhibiting ferroptosis via Nrf2/HO-1/GPX4 pathway. Environ Toxicol. 2024;39(7):4066-4085. [55] ZHENG X, YE FC, SUN T, et al. Delay the progression of glucocorticoid-induced osteoporosis: Fraxin targets ferroptosis via the Nrf2/GPX4 pathway. Phytother Res. 2024; 38(11):5203-5224. [56] JIANG Z, QI G, HE X, et al. Ferroptosis in Osteocytes as a Target for Protection Against Postmenopausal Osteoporosis. Adv Sci (Weinh). 2024;11(12):e2307388. [57] TROUVIN AP, GOËB V. Receptor activator of nuclear factor-κB ligand and osteoprotegerin: maintaining the balance to prevent bone loss. Clin Interv Aging. 2010;5:345-354. [58] LIN Y, SHEN X, KE Y, et al. Activation of osteoblast ferroptosis via the METTL3/ASK1-p38 signaling pathway in high glucose and high fat (HGHF)-induced diabetic bone loss. FASEB J. 2022;36(3):e22147. [59] LUO C, XU W, TANG X, et al. Canonical Wnt signaling works downstream of iron overload to prevent ferroptosis from damaging osteoblast differentiation. Free Radic Biol Med. 2022;188:337-350. [60] ALIZADEH A, DYCK SM, KARIMI-ABDOLREZAEE S. Traumatic Spinal Cord Injury: An Overview of Pathophysiology, Models and Acute Injury Mechanisms. Front Neurol. 2019;10:282. [61] ZHENG Q, WANG D, LIN R, et al. Pyroptosis, ferroptosis, and autophagy in spinal cord injury: regulatory mechanisms and therapeutic targets. Neural Regen Res. 2025;20(10): 2787-2806. [62] SOFRONIEW MV. Dissecting spinal cord regeneration. Nature. 2018;557(7705):343-350. [63] TRAN AP, WARREN PM, SILVER J. The Biology of Regeneration Failure and Success After Spinal Cord Injury. Physiol Rev. 2018;98(2):881-917. [64] ZOU Y, HENRY WS, RICQ EL, et al. Plasticity of ether lipids promotes ferroptosis susceptibility and evasion. Nature. 2020;585(7826):603-608. [65] GENG Z, GUO Z, GUO R, et al. Ferroptosis and traumatic brain injury. Brain Res Bull. 2021; 172:212-219. [66] INGÓLFSSON HI, CARPENTER TS, BHATIA H, et al. Computational Lipidomics of the Neuronal Plasma Membrane. Biophys J. 2017; 113(10):2271-2280. [67] KANG Y, LI Q, ZHU R, et al. Identification of Ferroptotic Genes in Spinal Cord Injury at Different Time Points: Bioinformatics and Experimental Validation. Mol Neurobiol. 2022; 59(9):5766-5784. [68] HU X, XU Y, XU H, et al. Progress in Understanding Ferroptosis and Its Targeting for Therapeutic Benefits in Traumatic Brain and Spinal Cord Injuries. Front Cell Dev Biol. 2021;9:705786. [69] SHI Z, YUAN S, SHI L, et al. Programmed cell death in spinal cord injury pathogenesis and therapy. Cell Prolif. 2021;54(3):e12992. [70] GE MH, TIAN H, MAO L, et al. Zinc attenuates ferroptosis and promotes functional recovery in contusion spinal cord injury by activating Nrf2/GPX4 defense pathway. CNS Neurosci Ther. 2021;27(9):1023-1040. [71] GONG F, GE T, LIU J, et al. Trehalose inhibits ferroptosis via NRF2/HO-1 pathway and promotes functional recovery in mice with spinal cord injury. Aging (Albany NY). 2022; 14(7):3216-3232. [72] LI D, LU X, XU G, et al. Dihydroorotate dehydrogenase regulates ferroptosis in neurons after spinal cord injury via the P53-ALOX15 signaling pathway. CNS Neurosci Ther. 2023;29(7):1923-1939. [73] OU Y, WANG SJ, LI D, et al. Activation of SAT1 engages polyamine metabolism with p53-mediated ferroptotic responses. Proc Natl Acad Sci U S A. 2016;113(44):E6806-E6812. [74] XING C, LIU S, WANG L, et al. Metformin enhances endogenous neural stem cells proliferation, neuronal differentiation, and inhibits ferroptosis through activating AMPK pathway after spinal cord injury. J Transl Med. 2024;22(1):723. [75] MA H, XING C, WEI H, et al. Berberine attenuates neuronal ferroptosis via the AMPK-NRF2-HO-1-signaling pathway in spinal cord-injured rats. Int Immunopharmacol. 2024;142(Pt B):113227. [76] HUANG K, FANG J, XIAO S, et al. Transcranial alternating current stimulation inhibits ferroptosis and promotes functional recovery in spinal cord injury via the cGMP-PKG signalling pathway. Life Sci. 2025;362: 123341. [77] DONG H, ZHANG C, SHI D, et al. Ferroptosis related genes participate in the pathogenesis of spinal cord injury via HIF-1 signaling pathway. Brain Res Bull. 2023;192:192-202. [78] XIAO S, ZHANG Y, WANG S, et al. The Syvn1 inhibits neuronal cell ferroptosis by activating Stat3/Gpx4 axis in rat with spinal cord injury. Cell Prolif. 2024;57(10):e13658. [79] WANG S, YANG L, WU Z, et al. Ferroptosis-related genes participate in the microglia-induced neuroinflammation of spinal cord injury via NF-κB signaling: evidence from integrated single-cell and spatial transcriptomic analysis. J Transl Med. 2025;23(1):43. [80] DONG J, GONG Z, BI H, et al. BMSC-derived exosomal miR-219-5p alleviates ferroptosis in neuronal cells caused by spinal cord injury via the UBE2Z/NRF2 pathway. Neuroscience. 2024;556:73-85. [81] HUANG Q, SHA W, GU Q, et al. Inhibition of Connexin43 Improves the Recovery of Spinal Cord Injury Against Ferroptosis via the SLC7A11/GPX4 Pathway. Neuroscience. 2023;526:121-134. [82] KOS N, GRADISNIK L, VELNAR T. A Brief Review of the Degenerative Intervertebral Disc Disease. Med Arch. 2019;73(6):421-424. [83] YANG S, ZHANG F, MA J, et al. Intervertebral disc ageing and degeneration: The antiapoptotic effect of oestrogen. Ageing Res Rev. 2020;57:100978. [84] MILLECAMPS M, STONE LS. Delayed onset of persistent discogenic axial and radiating pain after a single-level lumbar intervertebral disc injury in mice. Pain. 2018;159(9):1843-1855. [85] WANG W, JING X, DU T, et al. Iron overload promotes intervertebral disc degeneration via inducing oxidative stress and ferroptosis in endplate chondrocytes. Free Radic Biol Med. 2022;190:234-246. [86] YANG RZ, XU WN, ZHENG HL, et al. Involvement of oxidative stress-induced annulus fibrosus cell and nucleus pulposus cell ferroptosis in intervertebral disc degeneration pathogenesis. J Cell Physiol. 2021;236(4):2725-2739. [87] CUI P, SHENG Y, WU C, et al. Puerarin modulates proliferation, inflammation and ECM metabolism in human nucleus pulposus mesenchymal stem cells via the lncRNA LINC01535. Heliyon. 2024;10(12):e33083. [88] LIU X, PAN F, SHA C, et al. Fuzi decoction ameliorates intervertebral disc degeneration through ferroptosis modulation by suppressing NF-κB pathway. Int Immunopharmacol. 2025; 148:114155. [89] WANG YG, YU XJ, QU YK, et al. Ferrostatin-1 Inhibits Toll-Like Receptor 4/NF-κB Signaling to Alleviate Intervertebral Disc Degeneration in Rats. Am J Pathol. 2023;193(4):430-441. [90] ZHANG J, HE L, LI Q, et al. EGR1 knockdown confers protection against ferroptosis and ameliorates intervertebral disc cartilage degeneration by inactivating the MAP3K14/NF-κB axis. Genomics. 2023;115(5):110683. [91] LI Z, CHENG P, XI H, et al. Tomatidine Alleviates Intervertebral Disc Degeneration by Activating the Nrf2/HO-1/GPX4 Signaling Pathway. Drug Des Devel Ther. 2024;18:6313-6329. [92] YANG G, LIU X, JING X, et al. Astaxanthin suppresses oxidative stress and calcification in vertebral cartilage endplate via activating Nrf-2/HO-1 signaling pathway. Int Immunopharmacol. 2023;119:110159. [93] ZHANG Y, LI H, CHEN Y, et al. Nordihydroguaiaretic acid suppresses ferroptosis and mitigates intervertebral disc degeneration through the NRF2/GPX4 axis. Int Immunopharmacol. 2024;143(Pt 3):113590. [94] YAO B, CAI Y, WAN L, et al. BACH1 promotes intervertebral disc degeneration by regulating HMOX1/GPX4 to mediate oxidative stress, ferroptosis, and lipid metabolism in nucleus pulposus cells. J Gene Med. 2023; 25(6):e3488. [95] AHMAD A, BIERSACK B, LI Y, et al. Targeted regulation of PI3K/Akt/mTOR/NF-κB signaling by indole compounds and their derivatives: mechanistic details and biological implications for cancer therapy. Anticancer Agents Med Chem. 2013;13(7):1002-1013. [96] RITTER J, BIELACK SS. Osteosarcoma. Ann Oncol. 2010;21 Suppl 7:vii320-5. [97] DOBBIN ZC, LANDEN CN. The importance of the PI3K/AKT/MTOR pathway in the progression of ovarian cancer. Int J Mol Sci. 2013;14(4):8213-8227. [98] ROSEN G, TAN C, SANMANEECHAI A, et al. The rationale for multiple drug chemotherapy in the treatment of osteogenic sarcoma. Cancer. 1975;35(3 suppl):936-945. [99] MIALOU V, PHILIP T, KALIFA C, et al. Metastatic osteosarcoma at diagnosis: prognostic factors and long-term outcome--the French pediatric experience. Cancer. 2005;104(5):1100-1109. [100] WEN RJ, DONG X, ZHUANG HW, et al. Baicalin induces ferroptosis in osteosarcomas through a novel Nrf2/xCT/GPX4 regulatory axis. Phytomedicine. 2023;116:154881. [101] LIU J, HONG M, LI Y, et al. Programmed Cell Death Tunes Tumor Immunity. Front Immunol. 2022;13:847345. [102] LV H, ZHEN C, LIU J, et al. β-Phenethyl Isothiocyanate Induces Cell Death in Human Osteosarcoma through Altering Iron Metabolism, Disturbing the Redox Balance, and Activating the MAPK Signaling Pathway. Oxid Med Cell Longev. 2020;2020:5021983. [103] CHEN W, LI Z, YU N, et al. Bone-targeting exosome nanoparticles activate Keap1 / Nrf2 / GPX4 signaling pathway to induce ferroptosis in osteosarcoma cells. J Nanobiotechnology. 2023;21(1):355. [104] WANG Y, ZHANG L, ZHAO G, et al. Homologous targeting nanoparticles for enhanced PDT against osteosarcoma HOS cells and the related molecular mechanisms. J Nanobiotechnology. 2022;20(1):83. [105] YU H, ZHANG Y, ZUO Q, et al. Targeting X box-binding protein-1 (XBP1) enhances the sensitivity of HOS osteosarcoma cells to pyropheophorbide- α methyl ester-mediated photodynamic therapy. Photodiagnosis Photodyn Ther. 2022;37:102646. [106] ZHAN F, HE T, CHEN Z, et al. RhoA enhances osteosarcoma resistance to MPPa-PDT via the Hippo/YAP signaling pathway. Cell Biosci. 2021;11(1):179. [107] ZUO Q, OU Y, ZHONG S, et al. Targeting GRP78 enhances the sensitivity of HOS osteosarcoma cells to pyropheophorbide-α methyl ester-mediated photodynamic therapy via the Wnt/β-catenin signaling pathway. Acta Biochim Biophys Sin (Shanghai). 2021;53(10):1387-1397. [108] ZHANG Y, CHEN Y, MOU H, et al. Synergistic induction of ferroptosis by targeting HERC1-NCOA4 axis to enhance the photodynamic sensitivity of osteosarcoma. Redox Biol. 2024; 76:103328. [109] LUO Y, GAO X, ZOU L, et al. Bavachin Induces Ferroptosis through the STAT3/P53/SLC7A11 Axis in Osteosarcoma Cells. Oxid Med Cell Longev. 2021;2021:1783485. [110] LIU Q, WANG K. The induction of ferroptosis by impairing STAT3/Nrf2/GPx4 signaling enhances the sensitivity of osteosarcoma cells to cisplatin. Cell Biol Int. 2019;43(11):1245-1256. [111] LI X, LIU J. FANCD2 inhibits ferroptosis by regulating the JAK2/STAT3 pathway in osteosarcoma. BMC Cancer. 2023;23(1):179. [112] SHI Y, GONG M, DENG Z, et al. Tirapazamine suppress osteosarcoma cells in part through SLC7A11 mediated ferroptosis. Biochem Biophys Res Commun. 2021;567:118-124. [113] LIU J, LOU C, ZHEN C, et al. Iron plays a role in sulfasalazine-induced ferroptosis with autophagic flux blockage in K7M2 osteosarcoma cells. Metallomics. 2022;14(5): mfac027. [114] XU Z, CHEN L, WANG C, et al. MicroRNA-1287-5p promotes ferroptosis of osteosarcoma cells through inhibiting GPX4. Free Radic Res. 2021;55(11-12):1119-1129. [115] HUANG X, XIA K, WEI Z, et al. SLC38A5 suppresses ferroptosis through glutamine-mediated activation of the PI3K/AKT/mTOR signaling in osteosarcoma. J Transl Med. 2024;22(1):1004. [116] SHAO Y, ZUO X. PTPRC Inhibits Ferroptosis of Osteosarcoma Cells via Blocking TFEB/FTH1 Signaling. Mol Biotechnol. 2024;66(10):2985-2994. [117] LUO SH, TIAN JM, CHU Y, et al. The BRD4-SRPK2-SRSF2 signal modulates the splicing efficiency of ACSL3 pre-mRNA and influences erastin-induced ferroptosis in osteosarcoma cells. Cell Death Dis. 2023;14(11):760. [118] DENG C, ZHANG Q, HE P, et al. Targeted apoptosis of macrophages and osteoclasts in arthritic joints is effective against advanced inflammatory arthritis. Nat Commun. 2021; 12(1):2174. [119] NYGAARD G, FIRESTEIN GS. Restoring synovial homeostasis in rheumatoid arthritis by targeting fibroblast-like synoviocytes. Nat Rev Rheumatol. 2020;16(6):316-333. [120] NÉMETH T, NAGY G, PAP T. Synovial fibroblasts as potential drug targets in rheumatoid arthritis, where do we stand and where shall we go? Ann Rheum Dis. 2022;81(8):1055-1064. [121] REN P, NIU H, GONG H, et al. Morphological, biochemical and mechanical properties of articular cartilage and subchondral bone in rat tibial plateau are age related. J Anat. 2018;232(3):457-471. [122] OSTROWSKA M, MAŚLIŃSKI W, PROCHOREC-SOBIESZEK M, et al. Cartilage and bone damage in rheumatoid arthritis. Reumatologia. 2018;56(2):111-120. [123] BUCKWALTER JA, MANKIN HJ. Articular cartilage: degeneration and osteoarthritis, repair, regeneration, and transplantation. Instr Course Lect. 1998;47:487-504. [124] PARK S, BAEK IJ, RYU JH, et al. PPARα-ACOT12 axis is responsible for maintaining cartilage homeostasis through modulating de novo lipogenesis. Nat Commun. 2022; 13(1):3. [125] ZHAO C, YU Y, YIN G, et al. Sulfasalazine promotes ferroptosis through AKT-ERK1/2 and P53-SLC7A11 in rheumatoid arthritis. Inflammopharmacology. 2024;32(2):1277-1294. [126] TANG J, LIU W, LI Z, et al. Inhibition of ASIC1a reduces ferroptosis in rheumatoid arthritis articular chondrocytes via the p53/NRF2/SLC7A11 pathway. FASEB J. 2025;39(1):e70298. [127] CHENG Q, CHEN M, LIU M, et al. Semaphorin 5A suppresses ferroptosis through activation of PI3K-AKT-mTOR signaling in rheumatoid arthritis. Cell Death Dis. 2022;13(7):608. [128] ZHANG Y, SWANDA RV, NIE L, et al. mTORC1 couples cyst(e)ine availability with GPX4 protein synthesis and ferroptosis regulation. Nat Commun. 2021;12(1):1589. [129] SUN M, WANG Q, HUANG J, et al. Asiatic acid induces ferroptosis of RA-FLS via the Nrf2/HMOX1 pathway to relieve inflammation in rheumatoid arthritis. Int Immunopharmacol. 2024;137:112394. [130] LING Y, YANG Y, REN N, et al. Jinwu Jiangu capsule attenuates rheumatoid arthritis via the SLC7A11/GSH/GPX4 pathway in M1 macrophages. Phytomedicine. 2024;135: 156232. [131] FENG Z, MENG F, HUO F, et al. Inhibition of ferroptosis rescues M2 macrophages and alleviates arthritis by suppressing the HMGB1/TLR4/STAT3 axis in M1 macrophages. Redox Biol. 2024;75:103255. [132] HAO W, ZHU R, ZHANG H, et al. NS8593 inhibits chondrocyte ferroptosis and alleviates cartilage injury in rat adjuvant arthritis through TRPM7 / HO-1 pathway. Int J Biochem Cell Biol. 2024;174:106618. [133] ZHOU R, CHEN Y, LI S, et al. TRPM7 channel inhibition attenuates rheumatoid arthritis articular chondrocyte ferroptosis by suppression of the PKCα-NOX4 axis. Redox Biol. 2022;55:102411. [134] WANG W, JIANG H, YU J, et al. Astaxanthin-mediated Nrf2 activation ameliorates glucocorticoid-induced oxidative stress and mitochondrial dysfunction and impaired bone formation of glucocorticoid-induced osteonecrosis of the femoral head in rats. J Orthop Surg Res. 2024;19(1):294. [135] YANG J, SUN P, LIU Z, et al. Mid-term Clinical Outcomes of “Light Bulb” Core Decompression with Arthroscopic Assistance in Peri-collapse Osteonecrosis of the Femoral Head: A Retrospective Comparative Study. Orthop Surg. 2024;16(6):1399-1406. [136] SUN F, ZHOU JL, LIU ZL, et al. Dexamethasone induces ferroptosis via P53/SLC7A11/GPX4 pathway in glucocorticoid-induced osteonecrosis of the femoral head. Biochem Biophys Res Commun. 2022;602:149-155. [137] LIN YZ, CHEN ZH, YANG JF, et al. Astaxanthin Prevents Glucocorticoid-Induced Femoral Head Osteonecrosis by Targeting Ferroptosis through the JAK2/STAT3 Signaling Pathway. J Agric Food Chem. 2025;73(7):4270-4287. [138] ZHENG L, ZHANG C, LIAO L, et al. Knockdown of Gfi1 increases BMSCs exosomal miR-150-3p to inhibit osteoblast ferroptosis in steroid-induced osteonecrosis of the femoral head through BTRC/Nrf2 axis. Endocr J. 2025;72(2):205-219. |
| [1] | 于晨锜, 刘洋, 余建锋, 康康, 邓垚歌, 夏小伟, 张一健, 朱雪松. 仿生黑磷纳米系统调控滑膜巨噬细胞极化治疗骨关节炎[J]. 中国组织工程研究, 2026, 30(在线): 1-13. |
| [2] | 陈秋函, 杨 龙, 袁代柱, 吴展羽, 邹梓豪, 叶 川. 膝关节周围截骨治疗膝骨关节炎:治疗策略的优化[J]. 中国组织工程研究, 2026, 30(9): 2303-2312. |
| [3] | 张子峥, 罗 旺, 刘长路. 膝内侧间室骨关节炎单髁置换中有限元分析的应用价值[J]. 中国组织工程研究, 2026, 30(9): 2313-2322. |
| [4] | 刘文龙, 董 磊, 肖争争, 聂 宇. 骨质疏松患者行固定平台单髁置换后胫骨假体松动的有限元分析[J]. 中国组织工程研究, 2026, 30(9): 2191-2198. |
| [5] | 陈 龙, 王小阵, 席金涛, 鲁齐林. 短节段置钉联合可扩张聚醚醚酮置换体在骨质疏松椎体中的生物力学性能[J]. 中国组织工程研究, 2026, 30(9): 2226-2235. |
| [6] | 张 楠, 孟庆华, 鲍春雨. 踝关节有限元模型的特性及临床应用[J]. 中国组织工程研究, 2026, 30(9): 2343-2349. |
| [7] | 陈惠挺, 曾伟权, 周剑鸿, 王 杰, 庄聪颖, 陈培友, 梁泽乾, 邓伟明. 椎体成形中拖尾锚定治疗伴裂隙征骨质疏松性椎体压缩骨折的有限元分析[J]. 中国组织工程研究, 2026, 30(9): 2145-2152. |
| [8] | 曾 轩, 翁 汭, 叶仕成, 唐佳栋, 莫 凌, 李文超. 两种腰椎旋扳手法治疗腰椎间盘突出症:生物力学差异的有限元分析[J]. 中国组织工程研究, 2026, 30(9): 2153-2161. |
| [9] | 程旗圣, 居来提·买提肉孜, 肖 扬, 张陈伟, 帕尔哈提·热西提. 新型变径螺钉在腰椎改良皮质骨轨迹中的有限元分析[J]. 中国组织工程研究, 2026, 30(9): 2162-2171. |
| [10] | 黎清斌, 林建辉, 黄文杰, 王明爽, 杜间开, 劳永锵. 膝关节周围骨巨细胞瘤病灶扩大刮除后填充骨水泥:软骨下植骨与不植骨的比较[J]. 中国组织工程研究, 2026, 30(8): 1896-1902. |
| [11] | 陈豪杰, 王 黛, 沈 山. 种植体周围炎中的免疫炎症微环境机制[J]. 中国组织工程研究, 2026, 30(8): 2054-2062. |
| [12] | 傅律鹏, 于 鹏, 梁国彦, 昌耘冰. 脊柱外科领域应用的电活性材料[J]. 中国组织工程研究, 2026, 30(8): 2113-2123. |
| [13] | 胡雄科, 刘少华, 谭 谦, 刘 昆, 朱光辉. 紫草素干预骨髓间充质干细胞改善老年小鼠股骨的微结构[J]. 中国组织工程研究, 2026, 30(7): 1609-1615. |
| [14] | 宋浦蓁, 马贺宾, 陈宏广, 章亚东. 骨髓间充质干细胞外泌体联合转化生长因子β1对巨噬细胞的作用[J]. 中国组织工程研究, 2026, 30(7): 1616-1623. |
| [15] | 韩念荣, 黄异飞, 艾克热木·吾斯曼, 刘岩路, 胡 炜. 高糖微环境中程序性细胞死亡受体1抑制大鼠骨髓间充质干细胞的成骨分化[J]. 中国组织工程研究, 2026, 30(7): 1649-1657. |
人作为一个多细胞生物,机体任何一个角落都是通过细胞来维持其稳定性,而细胞是否能正常发生程序性死亡(主动有序的细胞死亡过程[6])可能决定了许多疾病是否发生,包含骨科疾病[7]。目前已知的程序性死亡包含细胞凋亡、坏死、焦亡以及铁死亡等,其中,铁死亡与其他程序性死亡不同,它是一种铁依赖性细胞死亡模式。调控铁死亡的分子机制主要为以下3个方面:①磷脂代谢异常:磷脂中含多不饱和脂肪酸的磷脂更易发生过氧化反应,它们在长链酰基辅酶A合酶4的催化下,转移至细胞膜磷脂层中,加快细胞质膜的过氧化,引起细胞铁死亡[8]。②铁代谢异常:首先,铁可以催化脂氧合酶和植物原叶绿素酸酯氧化还原酶等相关代谢酶,引起细胞磷脂过氧化以及活性氧的生成[9-10];其次,磷脂氢过氧化物作为细胞铁死亡的执行者,磷脂氢过氧化物可以与亚铁离子和3价铁离子反应,分别产生自由基PLO•和 PLOO•。这些自由基与含多不饱和脂肪酸的磷脂反应以进一步传播磷脂氢过氧化物的产生,这是促进细胞铁死亡的标志[11]。③经典谷胱甘肽过氧化物酶4抗氧化失调:谷胱甘肽过氧化物酶4作为一种硒蛋白,一方面可以将谷胱甘肽转化为氧化谷胱甘肽,增强细胞的抗氧化作用,消除脂质过氧化物并保护细胞膜的完整性[12];另一方面,谷胱甘肽过氧化物酶4可将细胞毒性脂质过氧化物还原为相应的醇类,减少脂质过氧化物的积累[13]。相反,当谷胱甘肽过氧化物酶4表达被抑制时,脂质过氧化物积累增多是引起细胞铁死亡的关键标志[14]。
根据现有研究表明,铁死亡正向调节可以被编程以杀死癌细胞,为癌症提供了一种新的治疗方式[15-16]。而铁死亡的负向调节可以抑制相关细胞死亡,用以治疗退行性神经疾病等相关疾病[17-18]。铁死亡作为细胞死亡的新形式,其在骨科疾病的研究中受到重点关注,例如,范可尼贫血互补组D2(一种参与DNA损伤修复的核蛋白)通过干预细胞铁死亡的发生达到治疗脊髓损伤的目的[19]。实验证实,铁死亡干预细胞死亡的进程能有效治疗相关骨科疾病,但是,如何通过最精准的方式调控细胞铁死亡的发生,是治疗相关疾病的重中之重。信号通路作为细胞外向细胞内传递信息的重要方式,参与细胞铁死亡的发生。
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
1.1 资料来源
1.1.1 检索人及检索时间 由第一作者在2018年1月至2025年2月进行文献检索。
1.1.2 检索时限 各数据库建库时间至2025年2月。
1.1.3 检索数据库 中国知网、PubMed数据库。
1.1.4 检索途径 主题词检索、关键词检索、摘要检索等。
1.1.5 检索词 中文检索词为“铁死亡,骨关节炎、脊髓损伤、骨质疏松、类风湿关节炎、骨肉瘤、激素性股骨头坏死”;英文检索词为“ferroptosis,osteoarthritis,osteoporosis,spinal cord injury,intervertebral disc degeneration,osteosarcomas,rheumatoid arthritis,steroid-induced osteonecrosis of the femoral head”。
1.1.6 文献检索策略 以PubMed数据库为例的检索略策略见图1。
1.1.7 检索文献类型 实验研究。
1.1.8 手工检索情况 手工检索引用文献中价值较高的参考文献,阅读评估是否符合纳入标准。
1.1.9 检索文献量 初步检索到1 341篇文献。
1.2 入选标准
纳入标准:①铁死亡分子机制研究的相关文献;②相关骨科疾病的相关研究的文献;③铁死亡与信号通路及骨科疾病相互作用的文献;④以信号通路为靶点调控铁死亡治疗骨科疾病的相关文献。
排除标准:与研究主题无关或重复阐述相同观点、年代久远、无法获得全文的文献。
1.3 质量评估及数据的提取 通过阅读文献标题、摘要、简介及正文等部分,严格按照纳入排除标准,最终纳入138篇文献进行综述,见图2。
铁死亡作为一种新型细胞程序性死亡过程,其在不同层面受到严格调控,干预多种疾病的发生发展。自2012年以后,针对铁死亡的研究逐步深入骨科疾病领域。该篇综述展示了信号通路通过调控细胞铁死亡的发生在骨科疾病的发病机制中起到关键作用。通过实验证实,信号通路通过两种方式调控细胞铁死亡在骨科疾病中发挥重要作用:①信号通路通过负调节抑制细胞内铁离子以及活性氧等物质过分堆积,避免软骨细胞、成骨细胞以及神经元细胞遭受氧化应激刺激而发生铁死亡;②信号通路通过正向调节促进骨肉瘤细胞、M1巨噬细胞以及成纤维样滑膜细胞细胞发生铁死亡。
通过上述文献的总结,发现几个有趣且值得思考的问题。首先,不同的骨科疾病之间看似是相互独立的,但是细胞的铁死亡是共同的。类风湿关节炎、骨关节炎以及椎间盘退化都存在软骨细胞铁死亡,那么既有研究结果是否是互通的,例如,虾青素上调核因子E2相关因子2/血红素加氧酶1信号通路的表达抑制椎间盘退化中软骨细胞铁死亡,其是否也可以治疗因软骨损伤而发病的类风湿关节炎或者骨关节炎。其次,既然有一种信号通路干预多种细胞铁死亡的证据,例如磷脂酰肌醇-3-激酶/蛋白激酶B信号通路通过调节下游细胞外信号调节激酶1/2表达,促进成纤维样滑膜细胞发生铁死亡,抑制成纤维样滑膜细胞迁移至软骨并对软骨产生侵袭及破坏作用,同时,磷脂酰肌醇-3-激酶/蛋白激酶B信号通路还可以影响哺乳动物雷帕霉素靶蛋白的表达,继而抑制调节元件结合蛋白1/硬脂酰辅酶A去饱和酶1信号通路的传导,从而促进骨肉瘤细胞的铁死亡[115,125]。那是否可以认为柳氮磺吡啶可以通过干预磷脂酰肌醇-3-激酶/蛋白激酶B信号通路治疗类风湿关节炎,同样也可以通过磷脂酰肌醇-3-激酶/蛋白激酶B信号通路治疗骨肉瘤。另外,信号通路之间控制铁死亡发展过程并不是相互平行的,它们之间相互交叉、相互影响,形成一个庞大的信号网络,在未来可以通过大量数据收集,找到信号通路之间互相联系的枢纽基因,然后运用药物针对性的干预,对于治疗疾病更加具备针对性。最后,铁死亡与其他程序性死亡(凋亡、自噬等)共同决定细胞的命运,那么同一种信号通路是否对同一种疾病细胞的多种程序性死亡起到干预作用。当然这些假设需要大量的实验研究,但若得到证实,未来可以节省大量的实验成本,并且大大明确未来对疾病治疗的方向以及提高效率,为骨科疾病的治疗提供新的思路。
目前,关于信号通路调控铁死亡的研究仅局限于动物实验研究,因此临床试验转化存在着巨大的挑战。首先,靶向信号通路调控铁死亡不可控,可能引发免疫系统障碍、神经毒性甚至肝肾等器官损伤,未来可以考虑纳米科技进行精准的靶向给药。其次,铁死亡相关生物标志物并未明确,因此在临床诊断、预后评估中缺乏诊断标准。从基础研究到临床应用的转化,面临着巨大的挑战,未来需要投入大量的时间和资源。
总之,信号通路对细胞铁死亡的干预与骨质疏松、椎间盘退化、类风湿关节炎和激素性股骨头坏死等骨科疾病密切相关。深入研究信号通路调控铁死亡的相关机制将为骨相关疾病的诊断和治疗提供新靶点。
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
铁死亡作为一种新型细胞程序性死亡过程,其在不同层面受到严格调控,干预多种疾病的发生发展。自2012年以后,针对铁死亡的研究逐步深入骨科疾病领域。但是关于调控铁死亡的具体机制并不清晰,而信号通路是已知的细胞内外信息传递的重要方式。因此该篇文章首先提出信号通路是干预骨科疾病相关细胞铁死亡的主要途径。然后通过对国内外大量文献的收集,论证了信号通路调控细胞铁死亡干预骨科疾病发生的可能性。该篇论文详细阐明了信号通路调控细胞铁死亡的相关因子,干预铁死亡的发生,梳理出信号通路、铁死亡、骨科疾病三者之间的密切联系。并且文章搜集了大量的实验数据,为文章的奠定了坚实的理论基础。再者,作者在整理资料的同时提出了一些对信号通路干预铁死亡治疗骨科疾病的建议和看法,为未来的实验方向提出了一些新的方向。
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程#br#
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||