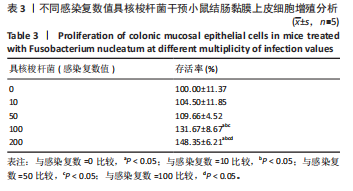
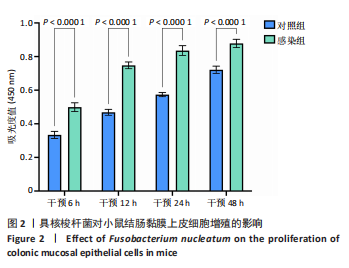
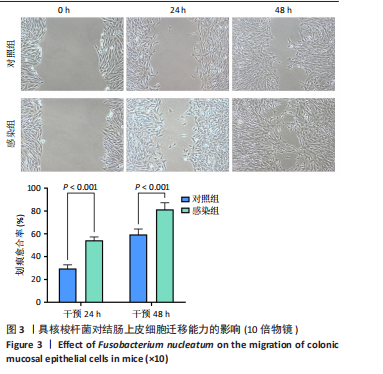
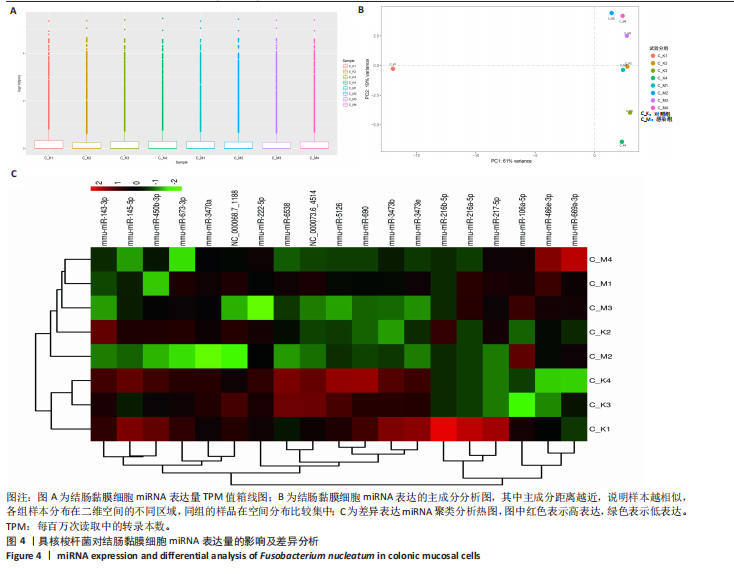
[1] 王佳佳,刘杰,王敏.构建具核梭杆菌诱导的实验性根尖周炎小鼠模型[J].中国组织工程研究,2022,26(2):176-181.
[2] MONDAL T, CHATTOPADHYAY D, SAHA MONDAL P, et al. Fusobacterium nucleatum modulates the Wnt/β-catenin pathway in colorectal cancer development. Int J Biol Macromol. 2025;299:140196.
[3] 郑文香,丛立春,韩斌.沉默信息调节因子1在具核梭杆菌诱导肠上皮细胞炎症和凋亡中的作用[J].中国病原生物学杂志,2019,14(6):643-649+655.
[4] HUH JW, ROH TY. Opportunistic detection of Fusobacterium nucleatum as a marker for the early gut microbial dysbiosis. BMC Microbiol. 20203;20(1):208.
[5] 王子奇,谭诗云.溃疡性结肠炎患者结直肠息肉复发的相关危险因素分析[J].胃肠病学和肝病学杂志,2025,34(3):373-376.
[6] SHI L, LI H, LI S, et al. Pseudoinvasion and squamous metaplasia/morules in colorectal adenomatous polyp: a case report and literature review. Diagn Pathol. 2024;19(1):126.
[7] LIU Y, LIN XX, HU SS, et al. The microbiota comparative analysis of the characteristics between colorectal adenomatous polyps and normal mucosal intestinal. Eur J Gastroenterol Hepatol. 2024;36(11):1305-1313.
[8] BHATTACHARJEE R, PRABHAKAR N, KUMAR L, et al. Crosstalk between long noncoding RNA and microRNA in Cancer. Cell Oncol (Dordr). 2023;46(4):885-908.
[9] 张林,高林波.miR-143和miR-145与肿瘤的研究进展[J].西安交通大学学报(医学版),2013,34(1):1-6.
[10] 刘久莲,刘童,高善语.microRNA在结直肠癌耐药及治疗中作用的研究进展[J].中国肿瘤生物治疗杂志,2023,30(5):438-444.
[11] 叶伟标,徐咏强,李妤玲,等.MicroRNA-200c在结直肠癌中的表达及对侵袭和转移的影响[J].中山大学学报(医学版),2018,39(3):335-340.
[12] GHOFRANI-SHAHPAR M, PAKRAVAN K, RAZMARA E, et al. Cancer-associated fibroblasts drive colorectal cancer cell progression through exosomal miR-20a-5p-mediated targeting of PTEN and stimulating interleukin-6 production. BMC Cancer. 2024;24(1):400.
[13] GUO S, CHEN J, CHEN F, et al. Exosomes derived from Fusobacterium nucleatum-infected colorectal cancer cells facilitate tumour metastasis by selectively carrying miR-1246/92b-3p/27a-3p and CXCL16. Gut. 2020. doi: 10.1136/gutjnl-2020-321187.
[14] AKTAN Ç, TEKIN F, ORUÇ N, et al. CHRM3-Associated miRNAs May Play a Role in Bile Acid-Induced Proliferation of H508 Colon Cancer Cells. Turk J Gastroenterol. 2023;34(3):298-307.
[15] 周锋,沙德胜,卢瑗瑗,等.基于Wnt/β-catenin通路探究迷迭香酸对结直肠癌上皮-间质转化的抑制作用[J].西安交通大学学报(医学版),2024,45(5): 726-733.
[16] WANG N, FANG JY. Fusobacterium nucleatum, a key pathogenic factor and microbial biomarker for colorectal cancer. Trends Microbiol. 2023;31(2):159-172.
[17] 邵雨琪,洪洁.肠道菌群和结直肠癌发生发展及治疗的研究进展[J].中国肿瘤临床,2024,51(17):881-887.
[18] ZEPEDA-RIVERA M, MINOT SS, BOUZEK H, et al. A distinct Fusobacterium nucleatum clade dominates the colorectal cancer niche. Nature. 2024;628(8007): 424-432.
[19] ELLAKWA DE, MUSHTAQ N, KHAN S, et al. Molecular functions of microRNAs in colorectal cancer: recent roles in proliferation, angiogenesis, apoptosis, and chemoresistance. Naunyn Schmiedebergs Arch Pharmacol. 2024;397(8):5617-5630.
[20] 张晓云,王立峰.miR-143在结肠癌组织中的差异表达及其对结肠癌细胞凋亡和迁移的影响[J].上海交通大学学报(医学版),2017,37(3):325-329.
[21] BAUER KM, HUMMON AB. Effects of the miR-143/-145 microRNA cluster on the colon cancer proteome and transcriptome. J Proteome Res. 2012;11(9):4744-4754.
[22] XU C, FAN L, LIN Y, et al. Fusobacterium nucleatum promotes colorectal cancer metastasis through miR-1322/CCL20 axis and M2 polarization. Gut Microbes. 2021;13(1):1980347.
[23] ZHANG L, LENG XX, QI J, et al. The adhesin RadD enhances Fusobacterium nucleatum tumour colonization and colorectal carcinogenesis. Nat Microbiol. 2024;9(9):2292-2307.
[24] XIANG Z, LI X, WANG X, et al. Fusobacterium nucleatum exacerbates colitis via STAT3 activation induced by Acetyl-CoA accumulation. Gut Microbes. 2025; 17(1):2489070.
[25] SAMENI F, ELKHICHI PA, DADASHI A, et al. Global prevalence of Fusobacterium nucleatum and Bacteroides fragilis in patients with colorectal cancer: an overview of case reports/case series and meta-analysis of prevalence studies. BMC Gastroenterol. 2025;25(1):71.
[26] SUN L, ZHANG Y, CAI J, et al. Bile salt hydrolase in non-enterotoxigenic Bacteroides potentiates colorectal cancer. Nat Commun. 2023;14(1):755.
[27] PROENÇA MA, BISELLI JM, SUCCI M, et al. Relationship between Fusobacterium nucleatum, inflammatory mediators and microRNAs in colorectal carcinogenesis. World J Gastroenterol. 2018;24(47):5351-5365.
[28] 冯志平,陈富坤,杨传周,等.miR-143-3p靶向KRAS阻滞PI3K/Akt信号通路抑制分化型甲状腺癌细胞增殖、侵袭和迁移[J].现代肿瘤医学,2021,29(14): 2387-2393.
[29] ZHANG L, SHAO W. MicroRNA-143-3p Inhibits Wilms’ Tumor Cell Growth By Targeting the Ras/Raf/MEK/ERK Pathway. Altern Ther Health Med. 2023;29(2): 140-147.
[30] ABD ELHAFEEZ AS, GHANEM HM, SWELLAM M, et al. Involvement of FAM170B-AS1, hsa-miR-1202, and hsa-miR-146a-5p in breast cancer. Cancer Biomark. 2024;39(4):313-333.
[31] 李震,姚弘虞,王世豪,等.WNT信号通路在KRAS基因突变型结直肠癌中的作用[J].郑州大学学报(医学版),2021,56(3):417-422.
[32] CHENG X, SHEN T, LIU P, et al. mir-145-5p is a suppressor of colorectal cancer at early stage, while promotes colorectal cancer metastasis at late stage through regulating AKT signaling evoked EMT-mediated anoikis. BMC Cancer. 2022;22(1):1151.
[33] YIN Y, YAN ZP, LU NN, et al. Downregulation of miR-145 associated with cancer progression and VEGF transcriptional activation by targeting N-RAS and IRS1. Biochim Biophys Acta. 2013;1829(2):239-247.
[34] YOON JH, BAE E, NAGAFUCHI Y, et al. Repression of SMAD3 by STAT3 and c-Ski induces conventional dendritic cell differentiation. Life Sci Alliance. 2024;7(9): e201900581.
[35] SHAN H, TIAN G, ZHANG Y, et al. Exploring the molecular mechanisms and therapeutic potential of SMAD4 in colorectal cancer. Cancer Biol Ther. 2024; 25(1):2392341.
[36] LIU L, WANG Y, YU S, et al. Transforming Growth Factor Beta Promotes Inflammation and Tumorigenesis in Smad4-Deficient Intestinal Epithelium in a YAP-Dependent Manner. Adv Sci(Weinh). 2023;10(23):e2300708.
[37] ROSHANI ASL E, RASMI Y, BARADARAN B. MicroRNA-124-3p suppresses PD-L1 expression and inhibits tumorigenesis of colorectal cancer cells via modulating STAT3 signaling. J Cell Physiol. 2021;236(10):7071-7087.
[38] NIKOLAIEVA N, SEVCIKOVA A, OMELKA R, et al. Gut Microbiota-MicroRNA Interactions in Intestinal Homeostasis and Cancer Development. Microorganisms. 2022;11(1):107.
[39] SHI H, LIANG GF, LI Y, et al. Preparation and Evaluation of Upconversion Nanoparticles Based miRNA Delivery Carrier in Colon Cancer Mice Model. J Biomed Nanotechnol. 2019;15(11):2240-2250.
|