中国组织工程研究 ›› 2026, Vol. 30 ›› Issue (32): 8544-8554.doi: 10.12307/2026.428
• 生物材料综述 biomaterial review • 上一篇 下一篇
搭载生物材料细胞外泌体修复脊髓损伤的作用机制
郑 颖1,李梦瑶1,郑帆帆2,何 昭1,张 宁1,邹嘉伦1,李优磊1,高 枫1
- 1延安大学延安医学院,陕西省延安市 716000;2晋城市人民医院中医科,山西省晋城市 048000
-
接受日期:2026-01-01出版日期:2026-11-18发布日期:2026-04-29 -
通讯作者:高枫,硕士,硕士生导师,教授,延安大学延安医学院,陕西省延安市 716000 -
作者简介:郑颖,女,1999年生,甘肃省酒泉市人,汉族,硕士在读,主要从事神经损伤的修复与再生研究。 -
基金资助:陕西省自然科学基础研究计划面上项目(2025JC-YBMS-1027),项目负责人:高枫;陕西省秦创原“科学家+工程师”队伍建设项目(2025QCY-KXJ-185),项目负责人:高枫
Mechanism of action of extracellular vesicles loaded with biomaterials in repairing spinal cord injury
Zheng Ying1, Li Mengyao1, Zheng Fanfan2, He Zhao1, Zhang Ning1, Zou Jialun1, Li Youlei1, Gao Feng1
- 1Yan’an Medical College, Yan’an University, Yan’an 716000, Shaanxi Province, China; 2Department of Traditional Chinese Medicine, Jin Cheng People’s Hospital, Jincheng 048000, Shanxi Province, China
-
Accepted:2026-01-01Online:2026-11-18Published:2026-04-29 -
Contact:Gao Feng, MS, Master’s supervisor, Professor, Yan’an Medical College, Yan’an University, Yan’an 716000, Shaanxi Province, China -
About author:Zheng Ying, MS candidate, Yan’an Medical College, Yan’an University, Yan’an 716000, Shaanxi Province, China -
Supported by:Natural Science Foundation Project of Shaanxi Province, No. 2025JC-YBMS-1027 (to GF); Shaanxi Province’s “Scientist+Engineer” Team Construction Project of Qinchuangyuan, No. 2025QCY-KXJ-185 (to GF)
摘要:
文题释义:
继发性脊髓损伤:是指在原发性脊髓损伤的基础上,由于多种因素导致的脊髓组织进一步损害。脊髓组织损伤后会发生一系列复杂的生化反应,如炎症递质释放、自由基生成等,这些物质会进一步损伤神经细胞和神经纤维,导致脊髓功能进行性恶化,尤其是感觉、运动功能障碍明显,还可能伴有括约肌功能障碍等其他并发症。
外泌体:是一种由细胞分泌的微小囊泡,直径在30-150 nm之间。外泌体的膜结构富含磷脂和胆固醇,内部包含多种蛋白质、mRNA、微小RNA等,可以携带信号分子、实现细胞间信息传递,从而调节受体细胞的功能和行为,并且能在细胞之间传递营养物质和代谢产物,促进细胞的增殖、分化和迁移,有助于组织的修复和再生。
背景:通过将外泌体与水凝胶、生物支架等生物材料相结合可实现靶向递送功能,不仅为受损组织提供有效支撑,还显著提升了外泌体的治疗效率,从而对脊髓损伤的修复产生积极影响。
目的:综述外泌体与生物材料结合在脊髓损伤治疗中的作用机制及其研究进展。
方法:检索中国知网、PuMed数据库、Web of Science数据库,英文检索词为“spinal cord injury,exosomes,hydrogel,biological scaffold,neuroinflammation,oxidative stress,axonal regeneration,angiogenesis ”,中文检索词为“脊髓损伤,外泌体,水凝胶,生物支架,神经炎症,氧化应激,轴突再生,血管再生”,根据入选标准,最终纳入106篇文献进行综述。
结果与结论:目前的研究聚焦于脊髓损伤后继发性损伤的修复,如何更好地缓解继发性损伤带来的伤害是目前研究的重点问题。外泌体结合生物材料治疗脊髓损伤主要通过调节神经炎症、促进轴突再生、缓解氧化应激等途径发挥治疗效果,既能避免发生免疫排斥反应、解决外泌体生物利用率低等问题,又能为损伤部位提供有效组织支撑,有效缓解脊髓损伤后的继发性症状。负载外泌体生物材料治疗脊髓损伤的研究多数仅限于细胞及动物实验,缺少临床试验数据,未来仍需要进行更多的机制研究、安全性评价和相关临床试验。
https://orcid.org/0009-0002-1727-3366 (郑颖)
中国组织工程研究杂志出版内容重点:生物材料;骨生物材料;口腔生物材料;纳米材料;缓释材料;材料相容性;组织工程
中图分类号:
引用本文
郑 颖, 李梦瑶, 郑帆帆, 何 昭, 张 宁, 邹嘉伦, 李优磊, 高 枫. 搭载生物材料细胞外泌体修复脊髓损伤的作用机制[J]. 中国组织工程研究, 2026, 30(32): 8544-8554.
Zheng Ying, Li Mengyao, Zheng Fanfan, He Zhao, Zhang Ning, Zou Jialun, Li Youlei, Gao Feng. Mechanism of action of extracellular vesicles loaded with biomaterials in repairing spinal cord injury[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(32): 8544-8554.
2.1.1 外泌体的生物学特性 细胞外囊泡是一类由细胞内释放到细胞外基质的膜性小囊泡,它们在细胞间的信息传递和物质运输中起着重要作用,根据直径大小分为三大类,分别是凋亡小体、微粒和外泌体[12]。其中,外泌体的直径最小,为30-150 nm,通常呈球形或椭圆形。外泌体起源于内体系统,其生物发生过程是由细胞内吞小分子物质入胞启动的,早期内体成熟后,产生了包裹货物的腔内囊泡,内体进一步成熟为晚期内体,融合出现出现多囊泡,被称为多囊泡体,当多囊泡体与细胞质膜融合,腔内囊泡将释放到胞质外,成为外泌体。外泌体一经分泌,便具有多种生物活性,可以调节细胞间通讯与信号传递[13]。目前外泌体也被用作药物载体定向递送至目标组织或细胞,能极大提高疾病的治愈率。
2.1.2 生物材料的生物学特性 生物材料根据功能的特性主要分为两大类:具有多功能特性的水凝胶材料,能在药物递送系统和组织工程支架构建中展现出双重应用价值;用于组织结构支撑的生物支架。生物材料的构成材料为天然或合成产物,如胶原蛋白基质、海藻酸盐凝胶、硫酸化透明质酸复合物以及自组装超分子纳米材料等[14],这些材料通过模拟细胞外基质结构和功能化修饰发挥生物相容性和组织整合特性,为组织提供高效有利支撑,为组织提供良好的修复环境。
2.1.3 外泌体与生物材料结合发挥治疗最大优势 随着研究的不断进行,已有大量学者发现,单纯的外泌体治疗或生物材料治疗均不能发挥治疗的最大优势,外泌体在治疗过程中存在生物利用率低、靶向作用不准确等问题,而采用水凝胶或生物支架负载药物治疗可能存在药物难以透过血脊髓屏障或仅仅发挥组织支撑作用。将外泌体与生物材料结合,既能实现定向递送关键损伤部位,又能最大程度地发挥外泌体的生物活性效果与生物材料的生物相容性,使组织与支架更好地相互作用,促进细胞黏附和增殖,为损伤组织提供最接近体内稳态的的修复环境[15]。外泌体的发展及其与生物材料联合应用历史脉络,见表1。
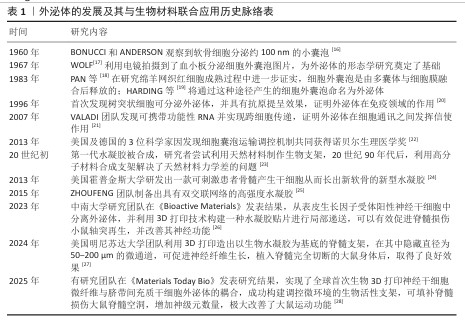
2.2 细胞来源的外泌体 脊髓损伤发生后,由于复杂的病理机制,单一的治疗手段并不能发挥较好的治疗效果,而外泌体治疗在脊髓损伤中表现出显著的治疗效果。外泌体的细胞来源包括间充质干细胞、神经干细胞、小胶质细胞、施万细胞等多种细胞,外泌体对于脊髓损伤后的神经炎症、轴突和血管再生障碍、血-脊髓屏障修复均有治疗作用[29]。外泌体的来源和作用机制见表2。外泌体的分类见图4。
2.2.1 间充质干细胞来源外泌体 间充质干细胞是具有多向分化潜能和自我更新能力的多能干细胞,具有再生成各种组织、器官和细胞的潜力,最常见的类型有骨髓间充质干细胞、脂肪间充质干细胞、脐带间充质干细胞、牙髓间充质干细胞等[42-43]。间充质干细胞来源外泌体可以抑制炎症并减轻细胞凋亡,促进轴突再生和血管生成,改善损伤部位微环境,减轻神经炎症和神经胶质增生,促进脊髓损伤后的运动改善和感觉恢复。主要发挥调节作用的是间充质干细胞来源外泌体中携带的miRNA,它们能有效调节炎症信号通路,XUE等[31]研究正好印证这一结论,他们发现骨髓间充质干细胞来源外泌体中所含的 miR-216a-5p,能够通过抑制Toll样受体4/核因子κB信号通路减轻脊髓损伤后产生的神经炎症。
2.2.2 神经干细胞来源外泌体 神经干细胞来源外泌体是神经干细胞自身所分泌的小分子复合物,通过递送生物分子调节邻近神经元或胶质细胞的功能,对神经炎症也具有抑制作用[44]。神经干细胞来源外泌体可以抑制小胶质细胞向M1型转化,同时抑制星形胶质细胞向A1型活化,例如,ZHAO等[45]验证了神经干细胞来源外泌体可抑制M1极化、促进M2极化,减轻小胶质细胞介导的神经炎症;LI等[46]的研究结果也可印证上述结论,神经干细胞来源外泌体通过肥胖相关基因抑制核因子E2相关因子2 甲基化修饰,将小胶质细胞从M1促炎表型转化为M2抗炎表型,减轻了小胶质细胞炎症反应对神经元的损伤,从而产生神经保护作用。
2.2.3 小胶质细胞来源外泌体 在生理状态下,作为中枢神经系统的固有免疫细胞,小胶质细胞通过动态监测微环境维持稳态在损伤或感染时迅速激活,分化为促炎(M1型)或抗炎(M2型)表型,参与炎症反应和组织修复[47]。小胶质细胞来源外泌体可以通过抑制神经元凋亡来促进轴突再生,其中发挥重要作用的是小胶质细胞外泌体所包含的miRNA,是治疗脊髓损伤的关键分子。例如,miR-672-5p是与M2型小胶质细胞来源外泌体相关的最关键miRNA,通过抑制黑色素瘤缺乏因子2/凋亡相关斑点样蛋白/caspase-1信号通路抑制神经元焦亡,最终促进轴突再生和降低脊髓神经元焦亡水平,促进脊髓损伤小鼠功能的恢复[48]。M2小胶质细胞来源外泌体能抑制星形胶质细胞的过度活化、减少胶质瘢痕形成,为轴突再生提供微环境支持。
2.2.4 施万细胞来源外泌体 施万细胞是周围神经系统的髓鞘胶质细胞,具有髓鞘再生能力,能够减少受伤部位的炎症,最终促进轴突的产生来缓解脊髓损伤[49]。施万细胞来源外泌体可通过降低腺苷酸活化蛋白激酶磷酸化水平改善脊髓损伤后线粒体形态及功能,进一步促进线粒体自噬,减少由于脊髓损伤导致的氧化应激和炎症反应,有效抑制细胞坏死,减少细胞凋亡,促进轴突再生和运动功能恢复[35]。施万细胞来源外泌体可以作用于损伤部位的内皮细胞,促进内皮细胞增殖,刺激损伤部位新生毛细血管生成,新生毛细血管可以减少损伤区域的脊髓空洞,丰富周围血液供应。施万细胞来源外泌体通过递送整合素β1促进血管内皮细胞迁移和再生,增强损伤部位血管生成,能较好地促进脊髓损伤后的功能恢复[50]。
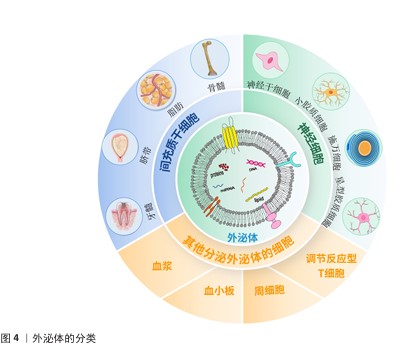
2.2.5 其他细胞来源外泌体 除了上述细胞来源外泌体外,还有一些其他来源外泌体在脊髓损伤中发挥积极的治疗作用。外泌体中所包含的miRNA在治疗过程中的作用较为显著。WU团队发现,脊髓损伤后细胞焦亡主要出现在小胶质细胞中,而Treg细胞分泌的外泌体能够抑制小胶质细胞焦亡,外泌体中的miR-709可抑制脊髓损伤后炎症反应并改善运动恢复[37]。脊髓损伤后,血-脊髓屏障的完整性被破坏,而周细胞在维持屏障稳固方面发挥重要作用,GAO等[38]研究发现,周细胞来源外泌体能促进血-脊髓屏障中内皮细胞的修复,外泌体中的miR-210-5p 能有效改善血-脊髓屏障功能并增强内皮细胞完整性;此外,将血管内皮细胞分泌的外泌体与间充质干细胞共同注射进脊髓损伤大鼠体内后,能有效促进脊髓损伤区域血管再生,维持血-脊髓屏障完整,进而改善大鼠后肢运动功能[39]。除血管内皮细胞分泌的外泌体外,富含血小板的血浆来源外泌体,在促进血-脊髓屏障修复以及抑制炎症反应方面发挥显著作用[40]。外泌体修复脊髓损伤的作用机制见图5。

2.3 可结合外泌体的生物材料
2.3.1 水凝胶 水凝胶在组织修复和重建领域一直发挥着突出作用,尽管水凝胶本身相对缺乏生物活性,但使用水凝胶作为载体可以提高外泌体的稳定性,并有助于将外泌体输送到缺陷部位进行持续原位释放[51]。水凝胶按功能不同分为4种类型,分别是微凝胶和纳米凝胶、刺激响应性水凝胶、黏附性水凝胶以及用于神经损伤修复的导电水凝胶[52]。
微凝胶:微凝胶包含水凝胶微球和纳米凝胶两大类,其中凝胶微球是粒径为微米范围内的颗粒水凝胶,它们的相对表面积更大,更易穿透组织屏障[53]。纳米凝胶能较好地清除脊髓损伤后产生的炎症因子,有效缓解神经炎症。纳米凝胶与外泌体及其分泌的生长因子结合可以有效抑制损伤部位炎症反应,促进血管和髓鞘再生,对脊髓损伤后的运动功能恢复有极大助益[54]。例如,FENG团队构建了一种透明质酸偶联聚多巴胺纳米颗粒水凝胶[55],两者在脊髓损伤中协同发挥作用,水凝胶可以为神经组织提供支撑,促进干细胞的黏附和生长,而纳米颗粒可以缓解脊髓损伤后带来的氧化应激,抑制损伤部位炎症反应,该水凝胶尾静脉注射后可以有效促进损伤部位轴突再生,改善脊髓损伤损伤大鼠的运动功能。
刺激响应性水凝胶:刺激响应性水凝胶是根据光、磁场、温度、机体氧含量、pH值、酶水平和温度等因素的变化,设计出的一种可以通过外部刺激来控制生物活性物质释放的水凝胶,包括化学敏感(如葡萄糖、氧化还原和 pH值)水凝胶、生物敏感(如酶或蛋白分子)水凝胶和物理敏感(如温度、磁性、光和电)水凝胶。刺激响应性水凝胶能够精准控制外泌体的释放,并具有显著的靶向性[56]。脊髓损伤后,体内会产生大量活性氧以及炎症因子,同时机体内活化的免疫细胞会表达基质金属蛋白酶,基质金属蛋白酶在机体内消耗生长因子和周围基质。基于以上因素,CHEN团队[57]制备出一种能够响应活性氧和基质金属蛋白酶的双响应水凝胶,在脊髓损伤后注射机体内会快速清除体内的活性氧以及炎症因子,并释放血管内皮生长因子,促进损伤后期功能的恢复。
黏附性水凝胶:黏附性水凝胶能够使药物或外泌体在损伤部位缓慢释放,增加药物利用率,增长治疗时间。脊髓损伤后,损伤组织末端与健康组织很难重建联系,传统水凝胶只能模拟脊髓组织结构,无法促进损伤部位与健康组织的连接。采用修饰后的具有黏附作用的水凝胶,能有效改善神功能和结构,例如,CHEN团队[58]制备出一种由胶原蛋白和纤维蛋白修饰的具有黏附性且机械性能优良的水凝胶,用于修复大鼠脊髓断裂区,发现在大鼠运动状态下,该水凝胶能够牢固地与脊髓残端结合,增加神经连接,同时募集内源性神经干细胞到达损伤区域并促进神经干细胞向神经元的分化,显著改善大鼠后肢运动功能。
导电水凝胶:生物电信号对于调节细胞行为至关重要,这些细胞行为可以促进细胞因子分泌并改善受损组织的微环境。导电水凝胶可以增强神经干细胞的神经元分化,并为外泌体构建一个稳定微环境,同时控制神经突触伸提供机械和电信号[59]。FAN等[60]发现植入材料的柔软程度会影响神经干细胞的分化,他们开发出一款可以模拟正常脊髓组织结构的导电水凝胶,并将其结合外泌体,发现该复合系统能促进M2型小胶质细胞极化、改善体内神经炎症,并通过激活磷脂酰肌醇-3激酶/蛋白激酶B和丝裂原活化蛋白激酶/细胞外信号调节激酶通路促进内源性神经干细胞迁移到损伤部位,刺激轴突延伸和髓鞘形成,改善脊髓损伤小鼠损伤后的运动功能。
2.3.2 生物支架 生物支架能够起到为细胞提供支撑、黏附的作用,并且可以模拟细胞外基质,为细胞提供更加贴近机体的生存环境[61];此外,支架能够支持充足的气体运输、细胞所需的营养物质,以促进细胞增殖、存活和分化。生物支架按其功能分为三类:细胞培养支架、组织工程支架以及药物缓释支架[62]。
细胞培养支架:细胞培养支架是细胞培养过程中用于支持细胞生长、增殖和分化的三维结构材料,有助于维持细胞的正常形态和功能。具有生物活性的细胞支架可诱导多能干细胞向特定细胞类型分化,促进组织再生。SINGH团队[63]利用一种称为 X-DN宏结构(XDM)的分支四向连接 DNA 宏观结构来研究自组装DNA宏观结构与胶原蛋白之间的相互作用,设计出一款DNA-胶原蛋白活性支架及 XDM/Ⅰ型胶原支架,人神经母细胞瘤细胞系置于 XDM/Ⅰ型胶原支架中培养,能够在支架中分化为成熟神经元。同时另一团队证明,将载有神经干细胞的胶原支架移植到脊髓损伤大鼠中,在损伤部位可诱导神经干细胞分化,有效刺激和引导神经元的生长和轴突再生[64]。
组织工程支架:组织工程支架植入机体后可以暂时替代缺损组织,维持损伤部位形态结构,防止组织塌陷导致的损伤加剧。脊髓损伤后,轴突断裂且神经连接中断,此时利用生物支架能够促进轴突重新建立目标连接。开发可以促进轴突生长的高对齐和高密度以修复受损轴突束的神经导管支架,是脊髓损伤修复的策略之一[65]。YOU团队[66]利用从蚕丝中提取的丝素蛋白生成多通道生物支架导管,与以往的单通道支架不同,多通道支架能更好地促进和引导细胞迁移和轴突再生,并且允许轴突通过并到达损伤部位;粘连蛋白是神经组织中基底膜的主要糖蛋白,在与轴突导向相关的神经突生长中起重要作用;通过将PC12细胞接种于导管上,发现层粘连蛋白功能化显著促进了神经突生长,表明层粘连蛋白在丝素蛋白纳米纤维上的化学偶联可以为神经突生长提供生物活性信号;将该支架种植于脊髓损伤大鼠体内,明显促进了损伤部位血管和轴突再生,并且脊髓损伤大鼠BBB评分以及各项运动功能皆优于对照组。
药物缓释支架:药物缓释支架是在一般生物支架的基础上,将具有治疗作用的药物负载于支架表面,利用药物与支架材料之间的相互作用以及支架的物理结构控制药物的释放速度和时间,使药物和外泌体缓慢释放,在病变部位持续发挥作用,达到治疗疾病的目的[67]。基于此,XIAO等[68]开发出一款搭载紫杉醇的纳米纤维支架,紫杉醇可以有效促进脊髓损伤后的轴突再生,并在组织抑制因子存在下还可促进轴突生长电位的恢复,但由于该药不能透过血脊髓屏障,因此设计出可以搭载紫杉醇的纳米纤维支架,该支架可以较好地控制紫杉醇在体内的释放,能够缓解脊髓损伤大鼠脱髓鞘发生、减少神经瘢痕增生、促进大鼠后肢运动功能的恢复。 2.4 外泌体与生物材料结合发挥显著治疗优势 尽管外泌体的治疗作用效果显著,但仍不可避免出现靶向性差、生物利用率低、半衰期短等情况,生物材料缺乏生物活性,仅能发挥支撑和组织保护作用。但生物材料具有稳定的物理支撑结构和生物相容性,并且不同类型的生物材料在组织修复、药物递送方面能发挥巨大优势,这正好弥补了外泌体在体内作用时效短、生物利用率低以及靶向性不明确等问题。目前,外泌体与生物材料相结合在骨组织再生和重建、神经修复、血管再生等领域发挥积极治疗作用。生物材料对外泌体的作用见表3。

2.5 脊髓损伤的病理机制 脊髓损伤分为原发性损伤和继发性损伤,前者是损伤当时的直接破坏,导致脊髓组织撕裂、出血、水肿甚至断裂,后者将持续在数小时至数天内直接发生。继发性损伤是导致病情加剧和神经功能恶化的主要原因,导致继发性损伤的原因包括缺血缺氧、炎症反应、氧化应激、细胞凋亡等,这会加剧损伤部位神经细胞死亡,加快神经瘢痕的形
成[76]。目前,如何干预继发性脊髓损伤带来的进一步损害是临床干预的靶点,当前研究的重点便是减轻继发性损伤带来的神经炎症,同时抑制细胞凋亡,促进损伤区域轴突和血管再生。
2.6 外泌体与水凝胶结合治疗脊髓损伤的作用机制 目前已有大量研究证明,将外泌体与水凝胶结合可以促进脊髓损伤后功能的恢复,既能发挥外泌体的最大效力,又能维持损伤区域的微环境。不同细胞来源外泌体对脊髓损伤的治疗机制主要分为以下三类:抑制神经炎症,缓解细胞凋亡;促进轴突和髓鞘再生,促进运动功能恢复;促进血管再生,恢复损伤区域血供。
2.6.1 抑制神经炎症,缓解细胞凋亡 原发性损伤的严重程度影响了神经炎症的发展,神经炎症的产生源于免疫细胞在组织内的长期浸润,同时会加重组织损伤并进一步破坏正常组织,这一过程是通过多种细胞类型和炎性细胞因子实现的,这些细胞因子和其他趋化因子由损伤部位的小胶质细胞、星形胶质细胞和免疫细胞释放,炎症因子的长期浸润会导致神经元受损;此外,小胶质细胞和星形胶质细胞极易分化为促炎表型,会加重神经炎症,严重影响脊髓损伤预后[77]。外泌体与水凝胶结合后,主要通过抑制神经细胞凋亡、调节小胶质细胞和星形胶质细胞极化缓解炎症因子的释放来抑制继发性损伤造成的神经炎症。
抑制细胞凋亡:在脊髓损伤数小时至数天内,神经细胞在缺血缺氧等情况下通过凋亡途径死亡,这加重了脊髓组织的进一步萎缩,外泌体结合水凝胶可通过多条信号通路来抑制细胞凋亡。YU等[78]制备一种由谷胱甘肽修饰的封装M2型小胶质细胞来源外泌体的二甲双胍纳米凝胶,谷胱甘肽能够穿过血-脊髓屏障,并且具有较高的生物安全性,结合二甲双胍能抑制氧化应激、稳定微管和减少神经元细胞凋亡来促进脊髓损伤运动功能的恢复。WU等[79]将血小板衍生生长因子模拟肽与水凝胶微球结合,可通过调节Bcl-2/Caspase-3/Bax信号通路减少神经细胞调亡,改善损伤区域的炎症反应,增强轴突再生和髓鞘形成,重建损伤部位血管结构,加强突触再生,改善脊髓损伤大鼠运动功能。除此以外,外泌体结合水凝胶也可调节其他凋亡相关信号通路,韩悦[80]将脂肪间充质干细胞来源外泌体与多肽水凝胶结合,发现该复合系统通过调节磷脂酰肌醇-3激酶/蛋白激酶B信号通路抑制脊髓损伤大鼠神经细胞凋亡,促进神经修复,显著提升脊髓损伤大鼠BBB评分,同时下调白细胞介素1β、白细胞介素6、肿瘤坏死因子α等炎症因子的表达。
调节小胶质细胞极化:脊髓损伤后,小胶质细胞会分化为M1促炎型和M2抗炎型,抑制M1型小胶质细胞极化也可较好地缓解炎症因子的释放。GUAN等[81]研究发现,将M2小胶质细胞来源外泌体与单宁酸和聚吡咯组成的导电水凝胶结合后,通过在损伤部位持续释M2型小胶质细胞外泌体刺激磷脂酰肌醇-3激酶/蛋白激酶B信号通路,诱导 M1型小胶质细胞转变为 M2型,从而显著减轻炎症反应。LIU团队[82]开发出一种整合了Fe?O?和钛酸钡的核壳纳米颗粒与人脐带间充质干细胞来源外泌体的仿生磁电水凝胶,在磁场刺激作用下,该水凝胶通过调节磷脂酰肌醇-3激酶/蛋白激酶B信号通路可有效缓解经脂多糖诱导的小胶质细胞炎症反应,促进小胶质细胞向M2型抗炎表型极化,体内实验也能证明该水凝胶通过调节磷脂酰肌醇-3激酶/蛋白激酶B信号通路诱导神级干细胞分化为神经元,阻止细胞向小胶质细胞和星形胶质细胞分化。
上述研究证明了外泌体结合水凝胶在抑制脊髓损伤神经炎症方面发挥着显著作用,外泌体结合水凝胶通过调节相关炎症信号通路发挥治疗作用,在这些炎症通路中,磷脂酰肌醇-3激酶/蛋白激酶B信号通路和Bcl-2/Caspase-3/Bax信号通路的调控作用尤为显著,这提示若能将具有调节上述信号通路上下游蛋白的外泌体进行组合,是否能发挥更好的治疗作用?
2.6.2 促进轴突和髓鞘再生,促进运动功能恢复 轴突在神经元信号传递过程中发挥重要作用,能够维持细胞间通讯,具有传导神经冲动、传递电信号、递送神经营养物质、形成神经网络等功能。脊髓损伤后,髓鞘磷脂产生大量的髓鞘相关糖蛋白、少突胶质细胞髓鞘糖蛋白等轴突生长抑制因子,这些抑制因子导致轴突生长锥崩裂,使轴突再生受限;同时损伤区域星形胶质细胞增生、肥大,并分泌胶质纤维酸性蛋白和硫酸软骨素蛋白多糖参与构成神经胶质瘢痕,导致神经传导通路的废用[83]。外泌体与水凝胶结合在很大程度上减少了神经瘢痕增生,促进轴突和髓鞘再生,对脊髓损伤后的功能恢复产生积极影响。
促进轴突再生:外泌体中的miRNA在促进脊髓损伤轴突再生中能发挥显著优势。SUN团队[84]通过单细胞RNA测序筛选出骨髓间充质干细胞中的一种亚型,即CD271+CD56+骨髓间充质干细胞亚群,该亚群分泌的外泌体有较为显著的促进轴突再生作用,为保证外泌体能在体内稳定长久地释放,将其搭载水凝胶在脊髓损伤部位进行注射,结果显示注射CD271+CD56+骨髓间充质干细胞来源外泌体后的小鼠轴突再生能力、损伤区域恢复情况明显优于对照组;此外,他们对 CD271+CD56+骨髓间充质干细胞进行了 microRNA 测序,发现miR-431-3p在轴突再生中发挥重要调控作用,能增加轴突的延伸距离和分支数量;脊髓损伤后,排斥引导分子家族成员a 高表达,它是抑制轴突再生的潜在靶基因,CD271+CD56+骨髓间充质干细胞来源外泌体能调控miR-431-3p作用于损伤神经元中促进轴突再生,并且抑制分子家族成员a的表达,对脊髓损伤后的小鼠运动功能恢复起到促进作用。神经元利用微管作为其主要结构元素来产生细长的轴突,因此,微管的稳定对于轴突再生至关重要。组蛋白脱乙酰化酶6是位于细胞质中的组蛋白脱乙酰化酶,抑制组蛋白脱乙酰化酶6表达能够激活神经细胞自噬水平、影响神经微管的乙酰化水平,以此促进神经突生长[85]。所以,另一研究团体从表皮生长因子受体阳性神经干细胞中分离出外泌体,将其包埋进3D打印水凝胶贴片中,外泌体能较为轻易地穿过血-脊髓屏障,该外泌体中的miR-34a-5p能够递送至神经元并下调组蛋白脱乙酰化酶6,从而激活自噬过程,促进神经突生长;体外实验可以证明,表皮生长因子受体阳性神经干细胞来源外泌体能够有效缓解硫酸软骨素干预后对轴突生长的抑制,体内实验证明,该水凝胶贴片可促进脊髓损伤小鼠的轴突生长、缩小组织损伤面积、提升BMS评分[86]。以上结果均能证明外泌体中携带的miRNA在促进轴突再生中具有显著效果,并且水凝胶能明显增强外泌体的作用时长,为外泌体的稳定释放提供环境保护。
促进髓鞘修复:将三维培养的间充质干细胞来源外泌体与热敏甲基丙烯明胶杂交,制备成微针阵列贴片,可以降低脊髓损伤后星形胶质细胞中胶质纤维酸性蛋白的表达,避免星形胶质细胞过度积累引起的神经胶质细胞/纤维化瘢痕的形成[87]。此外,骨髓间充质干细胞来源外泌体与导电水凝胶可组成生物敷料,通过核因核因子κB信号通路调节M2巨噬细胞极化,减轻炎症反应,促进施万细胞的附着和迁移,促进轴突再生,从而改善肌肉去神经支配萎缩并进一步促进功能恢复。除调节核因子κB 信号通路外,骨髓间充质干细胞来源外泌体与导电水凝胶结合可以抑制神经炎症,增强中枢神经系统中神经元和少突胶质细胞分化,促进神经元和轴突再生,并通过激活磷脂酰-3激酶/蛋白激酶B/雷帕霉素靶蛋白信号通路促进轴突生长,加快脊髓半切术后小鼠的运动恢复进程[88]。WANG团队[89]利用源自人诱导多能干细胞的皮质神经元分泌的外泌体和人脐带间充质干细胞的脱细胞细胞外基质开发了一种黏附性水凝胶,将其递送至脊髓损伤部位,显著降低了损伤区域的神经瘢痕形成,促进髓鞘再生,极大地改善了脊髓损伤大鼠的运动功能。
以上研究显示,外泌体结合水凝胶在促进轴突再生、促进髓鞘恢复方面的效果显著,但相关文献报道数量较少,后续可继续探究外泌体中的活性成分与水凝胶的相互作用,发掘调控轴突和髓鞘再生的信号通路,拓展对于脊髓损伤治疗思路。
2.6.3 促进血管再生 脊髓损伤后,血-脊髓屏障和微血管破坏是加重继发性损伤的关键因素,受损部位血管破裂后形成大面积血肿,而局部微环境的改变可能导致血管内皮细胞凋亡,加速炎性递质如自由基、一氧化氮的释放,这些递质会进一步影响血管再生[90]。脊髓损伤导致局部血流动力学的改变,如血流速度减慢、血液黏稠度增加等,这些改变可能影响血管再生。在脊髓损伤修复过程中,轴突再生通常沿着血管生长,血管再生的破坏会阻止受伤组织的再生,因此,促进血管内皮再生在修复神经系统中起着至关重要的作用。神经干细胞来源外泌体具有促进血管内皮细胞增殖的作用,使用黏附性透明质酸水凝胶搭神经干细胞来源外泌体注射至脊髓损伤区域,发现该水凝胶可促进血管生成[91]。另外MU团队[92]证明装载间充质干细胞来源外泌体的透明质酸水凝胶,可促进血管内皮增殖、提高损伤部位血供。除细胞来源的外泌体外,富含血小板的血浆来源外泌体在血管再生中也发挥重要作用,例如,YUN团队[93]研发出一种负载血浆来源外泌体的三重网络水凝胶,其中富集神经营养因子、血小板衍生生长因子和胰岛素样生长因子1,能显著促进血管内皮细胞的增殖、黏附和迁移,并通过激活磷脂酰肌-3激酶/蛋白激酶B/雷帕霉素靶蛋白信号通路诱导神经发生和血管生成,促进细胞外基质的产生和血管再生。
综上所述,血管内皮再生对于脊髓损伤后组织的修复至关重要,持续的血供是加速脊髓损伤后期功能恢复的基础。外泌体能够较好地调控血管内皮细胞的增殖,由于不同细胞来源外泌体都具有促进血管内皮增殖的作用,提示未来可将多种外泌体联合应用以促进脊髓损伤后功能的恢复。外泌体结合水凝胶治疗脊髓损伤的作用机制,见表4。

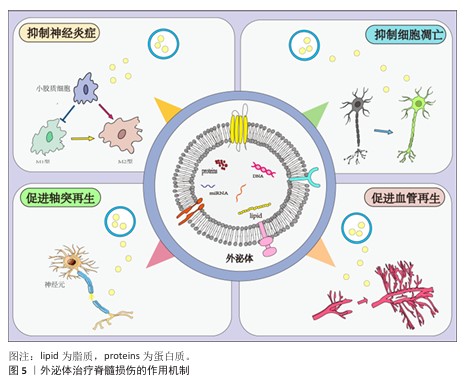
2.7 外泌体与生物支架结合治疗脊髓损伤的作用机制 生物支架能够起到为细胞提供支撑、黏附的作用,并且可以模拟细胞外基质,为细胞提供更加贴近机体的生存环境;此外,支架能够支持充足的气体运输、细胞所需的营养物质,以促进细胞增殖、存活和分化。将外泌体与生物支架结合,既可以支撑损伤区域,为损伤区域提供营养支持,又可以定向给药,极大提高治疗效率。外泌体与生物材料结合治疗脊髓损伤的作用机制主要包括以下两点:抑制神经炎症;促进轴突和髓鞘再生。
2.7.1 抑制神经炎症 原发性脊髓损伤后,组织破损导致免疫细胞、血管活性肽和炎症因子流入脊髓,完全破坏脊髓组织微环境,加重了神经细胞凋亡,并刺激小胶质细胞极化,进一步加重了继发性损伤严重程度,因此,抑制神经炎症是治疗脊髓损伤的重中之重[94]。
调节小胶质细胞极化:LI等[95]利用3D打印技术研发出一种神经干细胞微纤维支架,将其与脐带间充质干细胞衍生外泌体结合,研究显示,装载外泌体的水凝胶支架能改善脂多糖诱导的BV2细胞炎症反应,促进小胶质细胞向M2型转化,同时将支架植入损伤区域后可抑制炎症因子的释放、促进神经突再生,发挥神经保护作用。XIAO等[96]通过测序验证脐带间充质干细胞来源外泌体中的主要作用成分,发现发挥治疗作用的主要成分是miR-138-5p,基于此,分别制备了包裹脐带间充质干细胞来源外泌体的支架和只包裹miR-138-5p的水凝胶支架,研究显示miR-138-5p在体外能抑制脂多糖诱导的BV2细胞向M1型极化,miR-138-5p显著降低了NLRP3/caspase-1的蛋白表达,有效抑制了神经炎症因子的表达;对脊髓损伤大鼠尾静脉注射包裹外泌体的水凝胶支架后,大鼠后肢拖拽明显改善且后肢摇摆幅度减小;步态分析结果显示,注射只包裹miR-138-5p水凝胶支架组小鼠步幅和步宽均优于注射脐带间充质干细胞的外泌体支架组,并且炎症因子水平如白细胞介素6、肿瘤坏死因子α等水平下降更明显,证明外泌体中miR-138-5p在调节神经炎症、改善运动功能恢复方面的重要作用。
抑制经胞细神凋亡: 甲泼尼龙是一类糖皮质激素,能够调节神经炎症,显著改善脊髓损伤后微环境,促进神经元的存活,缓解脊髓损伤诱导的炎症微环境[97]。ZHU等[98]将搭载甲泼尼龙的施万细胞来源外泌体与纳米纤维支架以及透明质酸水凝胶组合,制备复合贴片,放置于受损区域以实现药物的定向递送,发现该复合贴片有效抑制了Cleaved-caspase-3蛋白的表达、缓解细胞凋亡、促进损伤神经元的存活,进而改善脊髓损伤大鼠的运动功能。
通过以上文献可以发现,外泌体与生物支架结合在抑制神经炎症方面的效果显著,生物支架不仅发挥组织支撑作用,同时能够调控外泌体在组织内的释放,但相关文献表述较少且上述文献中主要提及的外泌体多为间充质干细胞,对其他细胞来源外泌体提及较少。未来研究可探索其余细胞来源外泌体与生物支架结合后对脊髓损伤的治疗作用,拓展治疗
思路。
2.7.2 促进轴突和髓鞘再生 促凋亡信号通路的激活和缺血性损伤引发许多功能性神经元的丢失以及轴突髓鞘的断裂,导致细胞之间的信号传递中断。生物支架能够发挥支撑和保护作用,防止组织损伤进一步加重,生物支架与外泌体结合能促进神经元轴突再生,加强脊髓损伤后细胞间的信息传递以及物质运输,促进功能恢复[99]。
促进轴突再生:RAN等[100]采用新型组合肽将组合肽与自体血浆来源外泌体结合,生成一种可以治疗脊髓损伤的生物支架,该支架的可塑性高且与周围组织融合性好,能靶向于脊髓损伤区域的受损神经元,促进轴突再生,促进脊髓损伤小鼠运动功能的恢复。
促进髓鞘再生:已有实验证明miR-21在脊髓损伤后以时间依赖性方式上调,miR-21具有调节星形胶质细胞肥大和神经胶质瘢痕的作用,但单一治疗无法保证miR-21的有效递送。LIU等[101]设计将胶原结合结构域融合溶酶体相关膜糖蛋白2b蛋白和 miR-21在宿主HEK293T(293T)细胞中过度表达,用于产生工程化的 miR-21负载外泌体,将负载miR21的工程外泌体通过浓缩和富集植入Ⅰ型胶原蛋白支架中,该生物支架能够有效抑制神经元凋亡并增加神经元的活性和存活率,减少神经瘢痕增生,缓解脊髓损伤。神经肽前体被证明与髓鞘形成和少突胶质细胞的发育密切相关,通过调控神经肽前体的表达水平可以有效促进轴突再生,减少神经瘢痕的形成。HE等[102]将间充质干细胞来源外泌体与纤维蛋白结合形成纤维蛋白凝胶支架,注射进脊髓损伤小鼠体内,结果显示纤维蛋白凝胶能显著上调神经肽前体表达水平、促进少突胶质细胞生成、加快髓鞘从头生成,改善脊髓损伤后的组织结构。 此外,另一团队利用间充质干细胞来源外泌体作为药物递送载体,通过新型双重生物特异性肽设计了一种胶原支架,通过间充质干细胞来源外泌体提高紫杉醇的递送效率,这种多功能支架通过促进神经细胞迁移增强神经再生和减少瘢痕沉积,在促进大鼠脊髓损伤后的运动功能恢复方面表现出优异的性能[103]。
通过总结上述研究发现,外泌体结合生物支架促进轴突与髓鞘再生主要是通过调节轴突或髓鞘相关蛋白或miRNA发挥治疗作用的,在未来的研究中可以更为直接和更有针对性地干预轴突和髓鞘相关调控蛋白,达到更好的治疗效果。此外,文献中通过制备工程化外泌体使得外泌体批量化获取具有可行性,为后期临床应用提供了新的思路。外泌体结合生物支架治疗脊髓损伤的作用机制,见表5,图6。

| 1] STERNER RC, STERNER RM. Immune response following traumatic spinal cord injury: Pathophysiology and therapies. Front Immunol. 2023;13:1084101. [2] HU X, XU W, REN Y, et al. Spinal cord injury: molecular mechanisms and therapeutic interventions. Signal Transduct Target Ther. 2023;8(1):245. [3] 何光侣,储婉玉,李妍,等.Orexin-A通过调节促离子型谷氨酸受体促进脊髓损伤大鼠运动功能恢复[J]. 南方医科大学学报, 2025,45(5): 1023-1030. [4] ANJUM A, YAZID MD, FAUZI DAUD M, et al. Spinal Cord Injury: Pathophysiology, Multimolecular Interactions, and Underlying Recovery Mechanisms. Int J Mol Sci. 2020; 21(20):7533. [5] LIU WZ, MA ZJ, LI JR, et al. Mesenchymal stem cell-derived exosomes: therapeutic opportunities and challenges for spinal cord injury. Stem Cell Res Ther. 2021;12(1):102. [6] XIE Y, SUN Y, LIU Y, et al. Targeted Delivery of RGD-CD146(+)CD271(+) Human Umbilical Cord Mesenchymal Stem Cell-Derived Exosomes Promotes Blood-Spinal Cord Barrier Repair after Spinal Cord Injury. ACS Nano. 2023;17(18):18008-18024. [7] FENG J, ZHANG Y, ZHU Z, et al. Emerging Exosomes and Exosomal MiRNAs in Spinal Cord Injury. Front Cell Dev Biol. 2021;9: 703989. [8] ZHANG X, JIANG W, LU Y, et al. Exosomes combined with biomaterials in the treatment of spinal cord injury. Front Bioeng Biotechnol. 2023;11:1077825. [9] GUIJARRO-BELMAR A, VARONE A, BALTZER MR, et al. Effectiveness of biomaterial-based combination strategies for spinal cord repair - a systematic review and meta-analysis of preclinical literature. Spinal Cord. 2022;60(12):1041-1049. [10] JEON J, PARK SH, CHOI J, et al. Association between neural stem/progenitor cells and biomaterials in spinal cord injury therapies: A systematic review and network meta-analysis. Acta Biomater. 2024;183:50-60. [11] VERSTAPPEN K, AQUARIUS R, KLYMOV A, et al. Systematic Evaluation of Spinal Cord Injury Animal Models in the Field of Biomaterials. Tissue Eng Part B Rev. 2022;28(6):1169-1179. [12] KALLURI R. The biology and function of extracellular vesicles in immune response and immunity. Immunity. 2024;57(8):1752-1768. [13] KIMIZ-GEBOLOGLU I, ONCEL SS. Exosomes: Large-scale production, isolation, drug loading efficiency, and biodistribution and uptake. J Control Release. 2022;347:533-543. [14] FENG C, DENG L, YONG Y, et al. The Application of Biomaterials in Spinal Cord Injury. Int J Mol Sci. 2023;24(1):816. [15] HOU Y, LIU X, GUO Y, et al. Strategies for Effective Neural Circuit Reconstruction After Spinal Cord Injury: Use of Stem Cells and Biomaterials. World Neurosurg. 2022;161:82-89. [16] BONUCCI E. Fine structure of early cartilage calcification. J Ultrastruct Res. 1967;20(1): 33-50. [17] WOLF P. The nature and significance of platelet products in human plasma. Br J Haematol. 1967;13(3):269-288. [18] PAN BT, JOHNSTONE RM. Fate of the transferrin receptor during maturation of sheep reticulocytes in vitro: selective externalization of the receptor. Cell. 1983;33(3):967-978. [19] HARDING C, HEUSER J, STAHL P. Receptor-mediated endocytosis of transferrin and recycling of the transferrin receptor in rat reticulocytes. J Cell Biol. 1983;97(2):329-339. [20] BASTOS-AMADOR P, PEREZ-CABEZAS B, IZQUIERDO-USEROS N, et al. Capture of cell-derived microvesicles (exosomes and apoptotic bodies) by human plasmacytoid dendritic cells. J Leukoc Biol. 2012;91(5):751-758. [21] VALADI H, EKSTROM K, BOSSIOS A, et al. Exosome-mediated transfer of mRNAs and microRNAs is a novel mechanism of genetic exchange between cells. Nat Cell Biol. 2007; 9(6):654-659. [22] KAISER CA, SCHEKMAN R. Distinct sets of SEC genes govern transport vesicle formation and fusion early in the secretory pathway. Cell. 1990;61(4):723-733. [23] GUTIERREZ AM, FRAZAR EM, X KLAUS MV, et al. Hydrogels and Hydrogel Nanocomposites: Enhancing Healthcare through Human and Environmental Treatment. Adv Healthc Mater. 2022;11(7):e2101820. [24] SHARMA B, FERMANIAN S, GIBSON M, et al. Human cartilage repair with a photoreactive adhesive-hydrogel composite. Sci Transl Med. 2013;5(167):167ra6. [25] LIN P, MA S, WANG X, et al. Molecularly engineered dual-crosslinked hydrogel with ultrahigh mechanical strength, toughness, and good self-recover. Adv Mater. 2015;27(12): 2054-2059. [26] LOU W, QIU X, QIN Y, et al. 3D-printed advanced scaffold armed with exosomes derived from human skeletal stem cell identified by single-cell RNA sequencing enhances osteochondral regeneration. Bioact Mater. 2025;51:231-256. [27] HAN G, LAVOIE NS, PATIL N, et al. 3D-Printed Scaffolds Promote Enhanced Spinal Organoid Formation for Use in Spinal Cord Injury. Adv Healthc Mater. 2025;14(24):e04817. [28] HU X, LIU Z, ZHOU X, et al. Small extracellular vesicles derived from mesenchymal stem cell facilitate functional recovery in spinal cord injury by activating neural stem cells via the ERK1/2 pathway. Front Cell Neurosci. 2022;16:954597. [29] YANG Z, RAO J, LIN F, et al. The Role of Exosomes and Exosomal Noncoding RNAs From Different Cell Sources in Spinal Cord Injury. Front Cell Neurosci. 2022;16:882306. [30] 郑伊桐,汪永新,刘文,等.神经内镜下人脐带间充质干细胞外泌体鞘内移植修复脊髓损伤的作用机制[J].中国组织工程研究, 2025,29(36):7743-7751. [31] XUE H, RAN B, LI J, et al. Bone marrow mesenchymal stem cell exosomes-derived microRNA-216a-5p on locomotor performance, neuronal injury, and microglia inflammation in spinal cord injury. Front Cell Dev Biol. 2023;11: 1227440. [32] ZHOU Z, LI C, BAO T, et al. Exosome-Shuttled miR-672-5p from Anti-Inflammatory Microglia Repair Traumatic Spinal Cord Injury by Inhibiting AIM2/ASC/Caspase-1 Signaling Pathway Mediated Neuronal Pyroptosis. J Neurotrauma. 2022;39(15-16):1057-1074. [33] 张静.M2小胶质细胞来源外泌体通过调控星形胶质细胞表型促进脊髓损伤恢复的作用及机制研究[D]. 青岛:青岛大学,2024. [34] HUANG JH, CHEN YN, HE H, et al. Schwann cells-derived exosomes promote functional recovery after spinal cord injury by promoting angiogenesis. Front Cell Neurosci. 2022;16: 1077071. [35] REN J, ZHU B, GU G, et al. Schwann cell-derived exosomes containing MFG-E8 modify macrophage/microglial polarization for attenuating inflammation via the SOCS3/STAT3 pathway after spinal cord injury. Cell Death Dis. 2023;14(1):70. [36] PAN D, LI Y, YANG F, et al. Increasing toll-like receptor 2 on astrocytes induced by Schwann cell-derived exosomes promotes recovery by inhibiting CSPGs deposition after spinal cord injury. J Neuroinflammation. 2021;18(1):172. [37] XIONG W, LI C, KONG G, et al. Treg cell-derived exosomes miR-709 attenuates microglia pyroptosis and promotes motor function recovery after spinal cord injury. J Nanobiotechnology. 2022;20(1):529. [38] GAO P, YI J, CHEN W, et al. Pericyte-derived exosomal miR-210 improves mitochondrial function and inhibits lipid peroxidation in vascular endothelial cells after traumatic spinal cord injury by activating JAK1/STAT3 signaling pathway. J Nanobiotechnology. 2023;21(1):452. [39] LI L, MU J, ZHANG Y, et al. Stimulation by Exosomes from Hypoxia Preconditioned Human Umbilical Vein Endothelial Cells Facilitates Mesenchymal Stem Cells Angiogenic Function for Spinal Cord Repair. ACS Nano. 2022;16(7):10811-10823. [40] NIE X, LIU Y, YUAN T, et al. Platelet-rich plasma-derived exosomes promote blood-spinal cord barrier repair and attenuate neuroinflammation after spinal cord injury. J Nanobiotechnology. 2024;22(1):456. [41] 谢艳,吴晓龙,周英杰,等.芍药甘草汤调控血浆外泌体miR-125b-5p对炎性因子及脊髓损伤后肌痉挛的影响[J].中国医院药学杂志,2025,45(4):378-386. [42] YE Z, ZHENG Y, LI N, et al. Repair of spinal cord injury by bone marrow mesenchymal stem cell-derived exosomes: a systematic review and meta-analysis based on rat models. Front Mol Neurosci. 2024;17:1448777. [43] 李森,冯皓宇,杨卓.间充质干细胞外泌体在治疗脊髓损伤中的研究进展[J].颈腰痛杂志,2024,45(6):997-1001. [44] XU Y, ZHU Z, XU X, et al. Neuron-Derived Exosomes Promote the Recovery of Spinal Cord Injury by Modulating Nerve Cells in the Cellular Microenvironment of the Lesion Area. Mol Neurobiol. 2023;60(8):4502-4516. [45] ZHAO X, ZHU J, CHEN S, et al. Neural Stem Cell-Derived Exosomes Improve Neurological Function in Rats with Cerebral Ischemia-Reperfusion Injury by Regulating Microglia-Mediated Inflammatory Response. J Inflamm Res. 2023;16:3079-3092. [46] LI Z, CHEN Z, PENG J. Neural stem cell-derived exosomal FTO protects neuron from microglial inflammatory injury by inhibiting microglia NRF2 mRNA m6A modification. J Neurogenet. 2023;37(3):103-114. [47] FANG Y, QIN Z, ZHANG Y, et al. Implications of microglial heterogeneity in spinal cord injury progression and therapy. Exp Neurol. 2023;359:114239. [48] SONG Y, LI Z, HE T, et al. M2 microglia-derived exosomes protect the mouse brain from ischemia-reperfusion injury via exosomal miR-124. Theranostics. 2019;9(10):2910-2923. [49] MONJE PV, DENG L, XU X. Human Schwann Cell Transplantation for Spinal Cord Injury: Prospects and Challenges in Translational Medicine. Front Cell Neurosci. 2021;15:690894. [50] ZHU S, MA H, HOU M, et al. Schwann Cell-Derived Exosomes Induced Axon Growth after Spinal Cord Injury by Decreasing PTP-sigma Activation on CSPGs via the Rho/ROCK Pathway. Neurochem Res. 2024;49(8): 2120-2130. [51] JU Y, HU Y, YANG P, et al. Extracellular vesicle-loaded hydrogels for tissue repair and regeneration. Mater Today Bio. 2023; 18:100522. [52] YIN P, LIANG W, HAN B, et al. Hydrogel and Nanomedicine-Based Multimodal Therapeutic Strategies for Spinal Cord Injury. Small Methods. 2024;8(1):e2301173. [53] TOPUZ F, UYAR T. Advances in the development of cyclodextrin-based nanogels/microgels for biomedical applications: Drug delivery and beyond. Carbohydr Polym. 2022;297:120033. [54] VALENTINO C, VIGANI B, FEDELI I, et al. Development of alginate-spermidine micro/nanogels as potential antioxidant and anti-inflammatory tool in peripheral nerve injuries. Formulation studies and physico-chemical characterization. Int J Pharm. 2022;626:122168. [55] KAO Y, SONG W, ZHANG R, et al. Synergistic restoration of spinal cord injury through hyaluronic acid conjugated hydrogel-polydopamine nanoparticles combined with human mesenchymal stem cell transplantation. Bioact Mater. 2025;46:569-581. [56] YU H, GAO R, LIU Y, et al. Stimulus-Responsive Hydrogels as Drug Delivery Systems for Inflammation Targeted Therapy. Adv Sci (Weinh). 2024;11(1):e2306152. [57] CHEN H, WANG W, YANG Y, et al. A sequential stimuli-responsive hydrogel promotes structural and functional recovery of severe spinal cord injury. Biomaterials. 2025;316:122995. [58] CHEN Z, ZHANG H, FAN C, et al. Adhesive, Stretchable, and Spatiotemporal Delivery Fibrous Hydrogels Harness Endogenous Neural Stem/Progenitor Cells for Spinal Cord Injury Repair. ACS Nano. 2022;16(2):1986-1998. [59] 蒋昇源,邓博文,刘港,等.导电水凝胶治疗脊髓完全横断大鼠的安全性评估[J].中国组织工程研究,2023,27(21):3314-3319. [60] FAN L, LIU C, CHEN X, et al. Exosomes-Loaded Electroconductive Hydrogel Synergistically Promotes Tissue Repair after Spinal Cord Injury via Immunoregulation and Enhancement of Myelinated Axon Growth. Adv Sci (Weinh). 2022;9(13): e2105586. [61] 彭玲,潘育松,罗文杰,等.3D生物打印支架材料在脊髓损伤修复中的研究进展[J]. 化工新型材料,2025,53(10):273-277. [62] ZHANG Y, WU Z, WU J, et al. Current multi-scale biomaterials for tissue regeneration following spinal cord injury. Neurochem Int. 2024;178:105801. [63] SINGH N, SINGH A, BHATIA D. Self-Assembled DNA–Collagen Bioactive Scaffolds Promote Cellular Uptake and Neuronal Differentiation. ACS Biomater Sci Eng. 2025;11(1):308-321. [64] SUN Y, YANG C, ZHU X, et al. 3D printing collagen/chitosan scaffold ameliorated axon regeneration and neurological recovery after spinal cord injury. J Biomed Mater Res A. 2019;107(9):1898-1908. [65] 漆国栋,江琼,伍亚民,等.种子细胞与生物支架在脊髓组织工程中的实验研究进展[J].中国康复理论与实践,2021,27(6): 677-686. [66] YOU R, ZHANG Q, LI X, et al. Multichannel Bioactive Silk Nanofiber Conduits Direct and Enhance Axonal Regeneration after Spinal Cord Injury. ACS Biomater Sci Eng. 2020;6(8):4677-4686. [67] WANG Z, DUAN H, HAO F, et al. Circuit reconstruction of newborn neurons after spinal cord injury in adult rats via an NT3-chitosan scaffold. Prog Neurobiol. 2023; 220:102375. [68] XIAO Z, YAO Y, WANG Z, et al. Local Delivery of Taxol From FGL-Functionalized Self-Assembling Peptide Nanofiber Scaffold Promotes Recovery After Spinal Cord Injury. Front Cell Dev Biol. 2020;8:820. [69] XIE Q, YAN C, LIU G, et al. In Situ Triggered Self-Contraction Bioactive Microgel Assembly Accelerates Diabetic Skin Wound Healing by Activating Mechanotransduction and Biochemical Pathway. Adv Mater. 2024; 36(38): e2406434. [70] MENG H, SU J, SHEN Q, et al. A Smart MMP-9-responsive Hydrogel Releasing M2 Macrophage-derived Exosomes for Diabetic Wound Healing. Adv Healthc Mater. 2025; 14(10):e2404966. [71] YANG Q, LIU G, CHEN G, et al. Novel injectable adhesive hydrogel loaded with exosomes for holistic repair of hemophilic articular cartilage defect. Bioact Mater. 2024;42:85-111. [72] ZHENG J, HE J, WU J, et al. Polyphenol-Mediated Electroactive Hydrogel with Armored Exosomes Delivery for Bone Regeneration. ACS Nano. 2025;19(18):17796-17812. [73] GUERRERO J, MAEVSKAIA E, PFISTER P, et al. Mineralized osteoblast-derived exosomes and 3D-printed ceramic-based scaffolds for enhanced bone healing: A preclinical exploration. Acta Biomater. 2025;200: 686-702. [74] ZHENG Y, FU L, ZHANG Z, et al. Three-Dimensional Bioprinting of Growth Differentiation Factor 5-Preconditioned Mesenchymal Stem Cell-Derived Exosomes Facilitates Articular Cartilage Endogenous Regeneration. ACS Nano. 2025;19(16): 15281-15301. [75] SUN M, LI Q, YU H, et al. Cryo-self-assembled silk fibroin sponge as a biodegradable platform for enzyme-responsive delivery of exosomes. Bioact Mater. 2022;8:505-514. [76] RAMER LM, RAMER MS, BRADBURY EJ. Restoring function after spinal cord injury: towards clinical translation of experimental strategies. Lancet Neurol. 2014;13(12): 1241-1256. [77] MA Y, YU X, PAN J, et al. Exosomes: a promising microenvironment modulator for spinal cord injury treatment. Int J Biol Sci. 2025;21(8): 3791-3824. [78] YU Q, JIANG X, LIU X, et al. Glutathione-modified macrophage-derived cell membranes encapsulated metformin nanogels for the treatment of spinal cord injury. Biomater Adv. 2022;133:112668. [79] WU W, JIA S, XU H, et al. Supramolecular Hydrogel Microspheres of Platelet-Derived Growth Factor Mimetic Peptide Promote Recovery from Spinal Cord Injury. ACS Nano. 2023;17(4):3818-3837. [80] 韩悦.脂肪间充质干细胞源性外泌体复合RGD肽修饰水凝胶修复脊髓损伤的作用机制研究[D].昆明:云南中医药大学,2024. [81] GUAN P, FAN L, ZHU Z, et al. M2 microglia-derived exosome-loaded electroconductive hydrogel for enhancing neurological recovery after spinal cord injury. J Nanobiotechnology. 2024;22(1):8. [82] LIU W, LIU Q, LI Z, et al. Multifunctional magneto-electric and exosome-loaded hydrogel enhances neuronal differentiation and immunoregulation through remote non-invasive electrical stimulation for neurological recovery after spinal cord injury. Bioact Mater. 2025;48:510-528. [83] ZHOU X, LV Y, XIE H, et al. RNA sequencing of exosomes secreted by fibroblast and Schwann cells elucidates mechanisms underlying peripheral nerve regeneration. Neural Regen Res. 2024;19(8):1812-1821. [84] SUN Y, LIU Q, QIN Y, et al. Exosomes derived from CD271(+)CD56(+) bone marrow mesenchymal stem cell subpopoulation identified by single-cell RNA sequencing promote axon regeneration after spinal cord injury. Theranostics. 2024;14(2):510-527. [85] HUBBERT C, GUARDIOLA A, SHAO R, et al. HDAC6 is a microtubule-associated deacetylase. Nature. 2002;417(6887): 455-458. [86] QIN T, LI C, XU Y, et al. LOCAL DELIVERY OF EGFR(+)NSCS-DERIVED EXOSOMES PROMOTES NEURAL REGeneration post spinal cord injury via miR-34a-5p/HDAC6 pathway. Bioact Mater. 2024;33:424-443. [87] FANG A, WANG Y, GUAN N, et al. Porous microneedle patch with sustained delivery of extracellular vesicles mitigates severe spinal cord injury. Nat Commun. 2023;14(1):4011. [88] CHENG J, CHEN Z, LIU C, et al. Bone mesenchymal stem cell-derived exosome-loaded injectable hydrogel for minimally invasive treatment of spinal cord injury. Nanomedicine (Lond). 2021;16(18): 1567-1579. [89] WANG G, LI Q, LIU S, et al. An injectable decellularized extracellular matrix hydrogel with cortical neuron-derived exosomes enhances tissue repair following traumatic spinal cord injury. Mater Today Bio. 2024;28: 101250. [90] TRACY EP, STIELBERG V, ROWE G, et al. State of the field: cellular and exosomal therapeutic approaches in vascular regeneration. Am J Physiol Heart Circ Physiol. 2022;322(4):H647-H680. [91] GU C, LI Y, LIU J, et al. Neural stem cell-derived exosomes-loaded adhesive hydrogel controlled-release promotes cerebral angiogenesis and neurological function in ischemic stroke. Exp Neurol. 2023;370:114547. [92] MU J, LI L, WU J, et al. Hypoxia-stimulated mesenchymal stem cell-derived exosomes loaded by adhesive hydrogel for effective angiogenic treatment of spinal cord injury. Biomater Sci. 2022;10(7):1803-1811. [93] YUN Z, WU J, SUN X, et al. Neural-enhancing PRP/Alg/GelMA triple-network hydrogel for neurogenesis and angiogenesis after spinal cord injury via PI3K/AKT/mTOR signaling pathway. Theranostics. 2025;15(9):3837-3861. [94] CHANG Q, HAO Y, WANG Y, et al. Bone marrow mesenchymal stem cell-derived exosomal microRNA-125a promotes M2 macrophage polarization in spinal cord injury by downregulating IRF5. Brain Res Bull. 2021; 170:199-210. [95] LI X, ZHANG J, ZHANG Y, et al. Conjugated therapy with coaxially printed neural stem cell-laden microfibers and umbilical cord mesenchymal stem cell derived exosomes on complete transactional spinal cord defects. Mater Today Bio. 2025;32:101639. [96] XIAO Y, HU X, JIANG P, et al. Thermos-responsive hydrogel system encapsulated engineered exosomes attenuate inflammation and oxidative damage in acute spinal cord injury. Front Bioeng Biotechnol. 2023;11: 1216878. [97] LIU Z, YANG Y, HE L, et al. High-dose methylprednisolone for acute traumatic spinal cord injury: A meta-analysis. Neurology. 2019; 93(9):e841-e850. [98] ZHU B, GU G, REN J, et al. Schwann Cell-Derived Exosomes and Methylprednisolone Composite Patch for Spinal Cord Injury Repair. ACS Nano. 2023;17(22):22928-22943. [99] LI L, ZHANG Y, MU J, et al. Transplantation of Human Mesenchymal Stem-Cell-Derived Exosomes Immobilized in an Adhesive Hydrogel for Effective Treatment of Spinal Cord Injury. Nano Lett. 2020;20(6):4298-4305. [100] RAN N, LI W, ZHANG R, et al. Autologous exosome facilitates load and target delivery of bioactive peptides to repair spinal cord injury. Bioact Mater. 2023;25:766-782. [101] LIU X, ZHANG L, XU Z, et al. A functionalized collagen-I scaffold delivers microRNA 21-loaded exosomes for spinal cord injury repair. Acta Biomater. 2022;154:385-400. [102] HE X, YANG L, DONG K, et al. Biocompatible exosome-modified fibrin gel accelerates the recovery of spinal cord injury by VGF-mediated oligodendrogenesis. J Nanobiotechnology. 2022;20(1):360. [103] ZHANG L, FAN C, HAO W, et al. NSCs Migration Promoted and Drug Delivered Exosomes-Collagen Scaffold via a Bio-Specific Peptide for One-Step Spinal Cord Injury Repair. Adv Healthc Mater. 2021;10(8):e2001896. [104] ZAREPOUR A, HOOSHMAND S, GOKMEN A, et al. Spinal Cord Injury Management through the Combination of Stem Cells and Implantable 3D Bioprinted Platforms. Cells. 2021;10(11):3189. [105] 张羽.间充质干细胞来源外泌体联合水凝胶的局部递送体系用于脊髓损伤治疗的研究[D].杭州:浙江大学,2020. [106] LI Y, LUO W, MENG C, et al. Exosomes as promising bioactive materials in the treatment of spinal cord injury. Stem Cell Res Ther. 2024; 15(1):335. |
| [1] | 苏湄佳, 李红玉, 刘 琪, 夏 冰, 蔡 婧, 黄景辉. 口服生姜外泌体样纳米颗粒促进坐骨神经损伤修复的作用与机制[J]. 中国组织工程研究, 2026, 30(32): 8391-8401. |
| [2] | 范 筱, 李慧云, 窦馥国, 张 楠, 张新颜. 纳米磷酸铜水凝胶敷料促进小鼠皮肤创面愈合[J]. 中国组织工程研究, 2026, 30(32): 8413-8419. |
| [3] | 刘邦定, 唐永亮, 李 妮, 任 波. 载槲皮素水凝胶材料治疗感染性骨缺损[J]. 中国组织工程研究, 2026, 30(32): 8427-8435. |
| [4] | 陈世超, 邓云艺, 赵任圣洁, 余 科, 李广文. 负载槲皮素-纳米银粒子光敏水凝胶应用于感染创口的抗菌性能[J]. 中国组织工程研究, 2026, 30(32): 8436-8442. |
| [5] | 李明慧, 郄浩宇, 潘 敏, 毕蕊洁, 吕晓萌, 张浩雅, 韩逸飞. 基于水凝胶载体的类风湿性关节炎治疗递送系统[J]. 中国组织工程研究, 2026, 30(32): 8496-8501. |
| [6] | 黎梓楷, 张程程, 熊嘉颖, 杨曦瑞, 阳 晶, 石海山. 奥硝唑药物对牙髓再生根管内血管化的潜在影响[J]. 中国组织工程研究, 2025, 29(在线): 1-7. |
| [7] | 赵文琪, 于海驰, 宋艺儒, 袁天阳, 刘钦毅. 富血小板血浆及水凝胶治疗脊髓损伤[J]. 中国组织工程研究, 2025, 29(10): 2189-2200. |
| [8] | 余幸鸽, 林开利. 纳米复合水凝胶在骨组织工程中的应用[J]. 中国组织工程研究, 2020, 24(34): 5441-5446. |
| [9] | 方妤露, 易兵成, 沈炎冰, 唐 寒, 张彦中. 玉米壳纤维增强壳聚糖基水凝胶应用于软骨组织工程支架的潜力[J]. 中国组织工程研究, 2020, 24(34): 5493-5501. |
| [10] | 谭 静, 易国栋. 萝卜硫素对H2O2诱导人肝细胞氧化应激损伤模型的保护作用[J]. 中国组织工程研究, 2020, 24(31): 5052-5056. |
| [11] | 袁一鸣, 王 艳, 陈程程, 赵明月, 裴 飞. 外泌体在周围神经损伤进程中的效应[J]. 中国组织工程研究, 2020, 24(31): 5079-5084. |
| [12] | 荀 翀, 王 强, 李长洲, 刘晓峰. 自体骨髓干细胞移植对脊髓损伤潜在分子靶点治疗机制的生物信息学分析[J]. 中国组织工程研究, 2020, 24(31): 4927-4933. |
| [13] | 林嘉颖, 陈淑仪, 陈胜锋, 王丙云, 陈志胜, 刘璨颖, 白银山, 计慧琴, 谢仕廷. 犬脂肪间充质干细胞及其外泌体修复庆大霉素致犬肾小管上皮细胞损伤[J]. 中国组织工程研究, 2020, 24(25): 3959-3965. |
外泌体内携带包含蛋白质、mRNA、微小RNA(microRNA,miRNA)、DNA、脂质在内的多种生物分子[5],可透过血-脊髓屏障并能调节邻近神经元的活动,影响细胞的增殖、分化和凋亡以及神经网络的形成,在神经系统疾病恢复中扮演着重要角色[6]。外泌体由机体自身产生,作用于体内并不会发生强烈的免疫排斥反应,它携带的多种细胞因子能够较好地抑制脊髓损伤后的炎症反应、积极促进血管再生、修复破损的血-脊髓屏障,提高运动功能[7]。但单一外泌体注射治疗的效率较低,不能最大限度地发挥治疗效果。目前已有大量研究证明,将外泌体与生物材料相结合能极大促进脊髓损伤后的功能恢复[8]。
生物材料主要包含水凝胶以及生物支架,分为天然和合成两大类[9]。研究发现,生物材料在脊髓损伤修复领域展现出双重治疗优势:不仅能够为轴突再生过程提供三维结构支撑,还表现出显著的抗炎特性[10]。生物材料可精准负载神经营养因子、小分子药物及干细胞等治疗组分,进而调控损伤局部微环境中抑制性因子的表达,促进神经再生微环境的动态平衡重建[11]。因此,将生物材料与外泌体相结合能更大程度地发挥治疗作用,促进损伤后的组织修复。该文综述外泌体与生物材料结合在脊髓损伤治疗中的作用机制及其研究进展,为后期脊髓损伤治疗提供新的思路。
1.2 资料来源
1.2.1 检索人及检索时间 由第一作者在2025年3月进行检索。
1.2.2 检索文献时限 1976年6月至2025年7月。
1.2.3 检索数据库 中国知网、PubMed、Web of Science数据库。
1.2.4 检索词 英文检索词为“spinal cord injury,exosomes,hydrogel,biological scaffold,neuroinflammation,oxidative stress,axonal regeneration,angiogenesis ”,中文检索词为“脊髓损伤,外泌体,水凝胶,生物支架,神经炎症,氧化应激,轴突再生,血管再生”。
1.2.5 检索文献类型 研究原著、综述、述评和病例报告。
1.2.6 手工检索情况 无。
1.2.7 检索策略 以PubMed数据库检索策略为例,见图2。
1.2.8 检索文献量 共检索到文献710篇,其中中文文献60篇、英文文献650篇。
1.3 入选标准
纳入标准:①有关促进脊髓损伤症状恢复的实验性或基础研究;②与外泌体治疗脊髓损伤同一领域中论点、论据可靠的文献;③与外泌体和生物材料相关且在脊髓损伤或中枢神经系统中能表现较大影响力的文献。
排除标准:①与神经损伤修复关联性小且侧重于材料构建方面的文献;②重复性研究且研究目的无相关性的文献;③资料无法提取的部分文献。
1.4 质量评估与数据提取 选择与文章内容相关性大、新颖且具有价值的文章进行分析讨论,排除与研究目的相关性差及内容陈旧、重复的文献,最终纳入106篇符合标准的文献进行综述,包括中文文献10篇、英文文献96篇。文献筛选流程见图3。
3.2 该综述区别于他人他篇的特点 文章主要探讨了与生物材料结合的外泌体在脊髓损伤中的作用与最新研究进展,外泌体具有促进神经细胞间的信息传递、血管生成、轴突再生以及抑制神经炎凋亡等生物特性,为治疗脊髓损伤奠定了基础[106]。相较于以往综述,该文更加关注外泌体与新兴生物材料结合的研究方向,包括与水凝胶及生物支架等生物材料结合,提出了可以将它们作为新的治疗手段,改善外泌体在体内存活率低、易被耗竭的问题,极大提高脊髓损伤后的治愈率。
3.3 该综述的局限性 ①通过检索国内外相关文献发现,目前搭载生物材料外泌体对脊髓损伤中具体细胞因子和信号通路的影响机制未完全阐明;②大多数实验都基于动物模型或者细胞培养,缺少临床试验数据,特别是国内的临床试验和基础实验研究更少;③外泌体对脊髓损伤的治疗仍然存在分歧。
3.4 该综述的意义 此综述较全面地讨论了目前外泌体治疗脊髓损伤的作用机制和最新研究进展,外泌体主要通过自身携带的miRNA及长链非编码RNA以介导不同的信号通路,在脊髓损伤治疗中发挥不同作用;生物材料具有高稳定性、高生物相容性的特点,这使它们成为外泌体发挥作用的高效载体;外泌体与生物材料的结合能更好地提高脊髓损伤后的治愈率。
该文主要聚焦于与生物材料结合的细胞外泌体,目前对于脊髓损伤的研究,主要聚焦于脊髓损伤后继发性损伤的修复,如何更好地缓解继发性损伤带来的伤害是目前研究的重点问题。该文针对治疗脊髓损伤的新兴方法进行综述,即细胞外泌体与生物材料相结合,这种方法既能避免发生传统干细胞移植带来的免疫排斥反应等问题,且易透过血脊髓屏障,又能为损伤部位提供有效组织支撑,较好地改善了脊髓损伤后的一系列继发症状。单一的外泌体治疗在体内生物利用率较低,无法精准到达损伤部位,治疗效果较差。在回顾文献过程中发现,外泌体结合生物材料的治疗方法恢复情况远优于传统治疗手段,患者生存率与治愈率更高,提示在未来,外泌体结合生物材料将成为治疗脊髓损伤重要技术之一。
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||