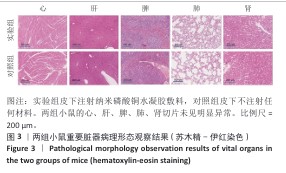
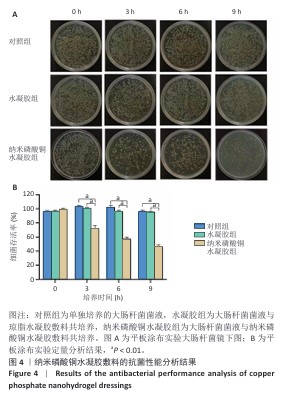
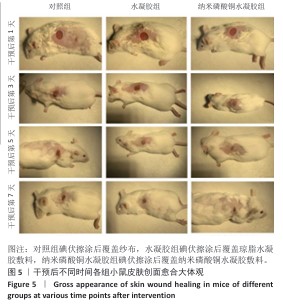
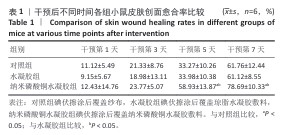
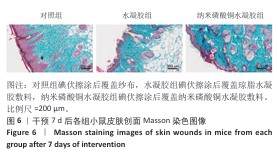
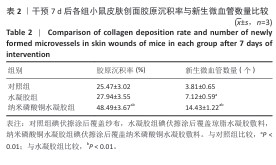
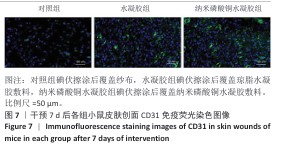
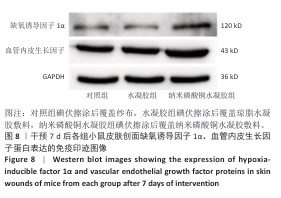
[1] BONIFANT H, HOLLOWAY S. A review of the effects of ageing on skin integrity and wound healing. Br J Community Nurs. 2019;24(Sup3):S28-S33.
[2] BASHARAT S, MALIK S, SAMAD HUA, et al. Diving deep into healing: the promising role of fish skin in wound recovery. Wounds. 2024;36(8):274-280.
[3] NUSSBAUM SR, CARTER MJ, FIFE CE, et al. An Economic Evaluation of the Impact, Cost, and Medicare Policy Implications of Chronic Nonhealing Wounds. Value Health. 2018;21(1):27-32.
[4] YU H, GONG W, MEI J, et al. The efficacy of a paeoniflorin-sodium alginate-gelatin skin scaffold for the treatment of diabetic wound: An in vivo study in a rat model. Biomed Pharmacother. 2022;151:113165.
[5] LIHAO Q, TINGTING L, JIAWEI Z, et al. 3D bioprinting of Salvianolic acid B-sodium alginate-gelatin skin scaffolds promotes diabetic wound repair via antioxidant, anti-inflammatory, and proangiogenic effects. Biomed Pharmacother. 2024;171:116168.
[6] ZHOU G, ZHU J, JIN L, et al. Salvianolic-Acid-B-Loaded HA Self-Healing Hydrogel Promotes Diabetic Wound Healing through Promotion of Anti-Inflammation and Angiogenesis. Int J Mol Sci. 2023;24(7):6844.
[7] JOHARI N, RAHIMI F, AZAMI H, et al. The impact of copper nanoparticles surfactant on the structural and biological properties of chitosan/sodium alginate wound dressings. Biomater Adv. 2024;162:213918.
[8] VALENCIA-GÓMEZ LE, MUZQUIZ-RAMOS EM, FAUSTO-REYES AD, et al. O-carboxymethyl chitosan/gelatin/silver-copper hydroxyapatite composite films with enhanced antibacterial and wound healing properties. J Biomater Appl. 2022;37(5):773-785.
[9] SUN BK, SIPRASHVILI Z, KHAVARI PA. Advances in skin grafting and treatment of cutaneous wounds. Science. 2014;346(6212):941-945.
[10] LEE PC, LI CZ, LU CT, et al. Microcurrent Cloth-Assisted Transdermal Penetration and Follicular Ducts Escape of Curcumin-Loaded Micelles for Enhanced Wound Healing. Int J Nanomedicine. 2023;18:8077-8097.
[11] CHEN Y, ZHAO Q, HAN J, et al. Dual Drug Loaded pH-sensitive Micelles for Efficient Bacterial Infection Treatment. Pharm Res. 2022;39(6):1165-1180.
[12] PEÑA OA, MARTIN P. Cellular and molecular mechanisms of skin wound healing. Nat Rev Mol Cell Biol. 2024;25(8):599-616.
[13] SORG H, SORG CGG. Skin Wound Healing: Of Players, Patterns, and Processes. Eur Surg Res. 2023;64(2):141-157.
[14] HASSANSHAHI A, MORADZAD M, GHALAMKARI S, et al. Macrophage-Mediated Inflammation in Skin Wound Healing. Cells. 2022;11(19):2953.
[15] XIONG M, ZHANG Q, HU W, et al. The novel mechanisms and applications of exosomes in dermatology and cutaneous medical aesthetics. Pharmacol Res. 2021;166:105490.
[16] YANG J, HUANG Z, TAN J, et al. Copper ion/gallic acid MOFs-laden adhesive pomelo peel sponge effectively treats biofilm-infected skin wounds and improves healing quality. Bioact Mater. 2024;32:260-276.
[17] STRICKLAND AD, OZTURK M, CONTI T, et al. Copper-based dressing: Efficacy in a wound infection of ex vivo human skin. Tissue Cell. 2023;84:102196.
[18] SHRESTHA S, WANG B, DUTTA PK. Commercial Silver-Based Dressings: In Vitro and Clinical Studies in Treatment of Chronic and Burn Wounds. Antibiotics (Basel). 2024;13(9):910.
[19] SALVO J, SANDOVAL C. Role of copper nanoparticles in wound healing for chronic wounds: literature review. Burns Trauma. 2022;10:tkab047.
[20] BUTSYK A, VARAVA Y, MOSKALENKO R, et al. Copper Nanoparticle Loaded Electrospun Patches for Infected Wound Treatment: From Development to In-Vivo Application. Polymers (Basel). 2024;16(19):2733.
[21] ASSAF S, KELLY O. Nutritional Dermatology: Optimizing Dietary Choices for Skin Health. Nutrients. 2024;17(1):60.
[22] QI L, ZHANG C, WANG B, et al. Progress in Hydrogels for Skin Wound Repair. Macromol Biosci. 2022;22(7):e2100475.
[23] WU Z, LU D, SUN S, et al. Material Design, Fabrication Strategies, and the Development of Multifunctional Hydrogel Composites Dressings for Skin Wound Management. Biomacromolecules. 2025;26(3):1419-1460.
[24] SANGHA MS, DEROIDE F, MEYS R. Wound healing, scarring and management. Clin Exp Dermatol. 2024;49(4):325-336.
[25] JIANG D, GUO R, MACHENS HG, et al. Diversity of Fibroblasts and Their Roles in Wound Healing. Cold Spring Harb Perspect Biol. 2023;15(3):a041222.
[26] TALBOTT HE, MASCHARAK S, GRIFFIN M, et al. Wound healing, fibroblast heterogeneity, and fibrosis. Cell Stem Cell. 2022;29(8):1161-1180.
[27] NARASIMHAN BN, FRALEY SI. Degradability tunes ECM stress relaxation and cellular mechanics. bioRxiv [Preprint]. 2024:2024.07.28.605514. doi:10.1101/2024.07.28.605514.
[28] MORETTI L, STALFORT J, BARKER TH, et al. The interplay of fibroblasts, the extracellular matrix, and inflammation in scar formation. J Biol Chem. 2022; 298(2):101530.
[29] ARAUJO TAT, ALMEIDA MC, AVANZI I, et al. Collagen membranes for skin wound repair: A systematic review. J Biomater Appl. 2021;36(1):95-112.
[30] VAN ORTEN A, GOETZ W, BILHAN H. A Novel Prehydrated Porcine-Derived Acellular Dermal Matrix: A Histological and Clinical Evaluation. Int J Biomater. 2024;2024:7322223.
[31] MAUROUX A, GOFFLO S, BREUGNOT J, et al. Angiogenesis and full thickness wound repair in a cell sheet-based vascularized skin substitute. Acta Biomater. 2024;187:123-137.
[32] SONG H, HAO D, ZHOU J, et al. Development of pro-angiogenic skin substitutes for wound healing. Wound Repair Regen. 2024;32(3):208-216.
[33] LI Q, SONG H, LI S, et al. Macrophage metabolism reprogramming EGCG-Cu coordination capsules delivered in polyzwitterionic hydrogel for burn wound healing and regeneration. Bioact Mater. 2023;29:251-264.
[34] HU Y, XIONG Y, ZHU Y, et al. Copper-Epigallocatechin Gallate Enhances Therapeutic Effects of 3D-Printed Dermal Scaffolds in Mitigating Diabetic Wound Scarring. ACS Appl Mater Interfaces. 2023;15(32):38230-38246.
[35] KUMAR S, WANG G, LIU W, et al. Hypoxia-Induced Mitogenic Factor Promotes Cardiac Hypertrophy via Calcium-Dependent and Hypoxia-Inducible Factor-1α Mechanisms. Hypertension. 2018;72(2):331-342.
[36] ZHENG L, HAN P, LIU J, et al. Role of copper in regression of cardiac hypertrophy. Pharmacol Ther. 2015;148:66-84.
[37] CHEN N, GUO L, WANG L, et al. Sleep fragmentation exacerbates myocardial ischemia‒reperfusion injury by promoting copper overload in cardiomyocytes. Nat Commun. 2024;15(1):3834.
[38] WANG J, WANG J, YU J, et al. Copper and Melanoma Risk: Results from NHANES 2007-2018 and Mendelian Randomization Analyses. Biol Trace Elem Res. 2024; 202(11):4909-4922.
|