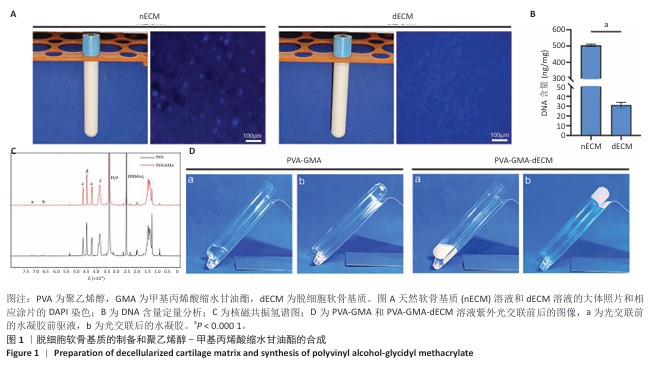
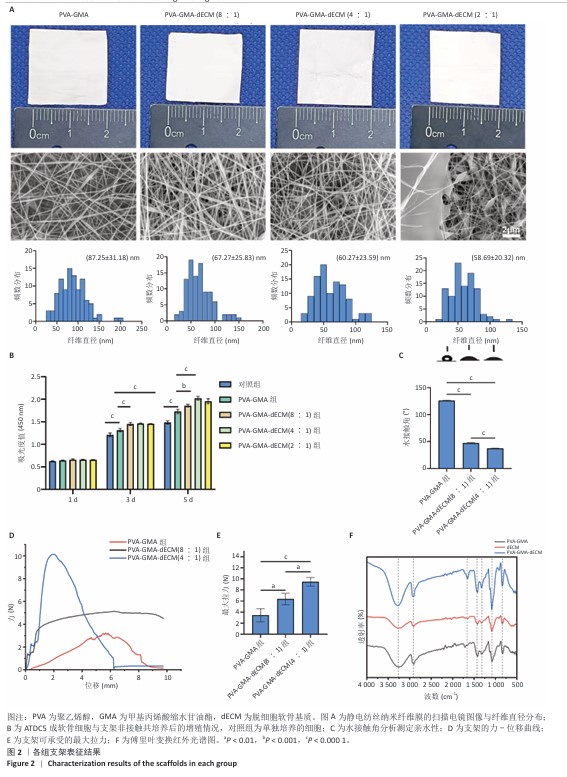
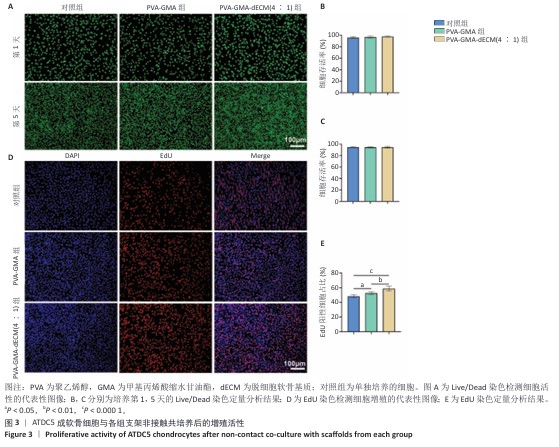
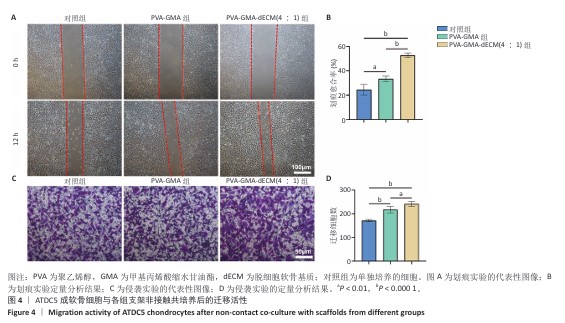
[1] HE S, DENG H, LI P, et al. Arthritic microenvironment-dictated fate decisions for stem cells in cartilage repair. Adv Sci. 2023;10(27): e2207715.
[2] WANG Z, LI X, JIANG Y, et al. Preparation of hydrogel microsphere and its application in articular cartilage injury. Mater Today Bio. 2025; 31:101641.
[3] SHI J, LIU Y, LING Y, et al. Polysaccharide-protein based scaffolds for cartilage repair and regeneration. Int J Biol Macromol. 2024;274(Pt 2): 133495.
[4] HUNTER CDR, FEATHERALL J, MCNAMARA N, et al. High-energy and polytraumatic multiligament knee injuries requiring surgical treatment are associated with more chondral damage yet similar rates of meniscal injury. Arthroscopy. 2025;41(10):4128-4135.
[5] SARIS D, PRICE A, WIDUCHOWSKI W, et al. Matrix-Applied Characterized Autologous Cultured Chondrocytes Versus Microfracture: Two-Year Follow-up of a Prospective Randomized Trial. Am J Sports Med. 2014;42(6):1384-1394.
[6] FU X, ZHANG Z, WANG Y, et al. Visualized trends and bibliometric analysis in ankle cartilage repair from 2004 to 2024. Front Med. 2024; 11:1503707.
[7] VOLOVA LT, KOTELNIKOV GP, SHISHKOVSKY I, et al. 3D bioprinting of hyaline articular cartilage: biopolymers hydrogels, and bioinks. Polymers (Basel). 2023;15(12):2695.
[8] HINCKEL BB, THOMAS D, VELLIOS EE, et al. Algorithm for treatment of focal cartilage defects of the knee: classic and new procedures. Cartilage. 2021;13(1_suppl):473S-495S.
[9] CHEN Y, CHEN LF, WANG Y, et al. Engineered dECM-based microsystem promotes cartilage regeneration in osteoarthritis by synergistically enhancing chondrogenesis of BMSCs and anti-inflammatory effect. Compos Part B Eng. 2025;290:111974.
[10] CHEN M, LI Y, LIU S, et al. Hierarchical macro-microporous WPU-ECM scaffolds combined with microfracture promote in situ articular cartilage regeneration in rabbits. Bioact Mater. 2020;6(7):1932-1944.
[11] GAHARWAR AK, SINGH I, KHADEMHOSSEINI A. Engineered biomaterials for in situ tissue regeneration. Nat Rev Mater. 2020;5: 686-705.
[12] FU L, LI L, BIAN Q, et al. Cartilage-like protein hydrogels engineered via entanglement. Nature. 2023;618(7966):740-747.
[13] COBURN JM, GIBSON M, MONAGLE S, et al. Bioinspired nanofibers support chondrogenesis for articular cartilage repair. Proc Natl Acad Sci U S A. 2012;109(25):10012-10017.
[14] RUMON MMH, AKIB AA, SARKAR SD, et al. Polysaccharide-based hydrogels for advanced biomedical engineering applications. ACS Polym Au. 2024;4(6):463-486.
[15] DING Y, HUANG Y, ZHANG F, et al. Biological augmentation using electrospun constructs with dual growth factor release for rotator cuff repair. ACS Appl Bio Mater. 2025;8(3):2548-2557.
[16] FOROUGH MR, EMADI R, AHMADIAN M, et al. Fabrication and characterization of polycaprolactone-baghdadite nanofibers by electrospinning method for tissue engineering applications. Materials (Basel). 2024;17(17):4187.
[17] ARASH A, DEHGAN F, ZAMANLUI BENISI S, et al. Polysaccharide base electrospun nanofibrous scaffolds for cartilage tissue engineering: challenges and opportunities. Int J Biol Macromol. 2024;277(Pt 1): 134054.
[18] SABERIAN E, JENCA A, ZAFARI Y, et al. Scaffold application for bone regeneration with stem cells in dentistry: literature review. Cells. 2024;13(12):1065.
[19] LIANG Y, WEI S, ZHANG A. Bioengineered tracheal graft with enhanced vascularization and mechanical stability for functional airway reconstruction. Regen Ther. 2025;29:364-380.
[20] YUAN S, YANG X, WANG X, et al. Injectable xenogeneic dental pulp decellularized extracellular matrix hydrogel promotes functional dental pulp regeneration. Int J Mol Sci. 2023;24(24):17483.
[21] YUAN Z, LYU Z, LIU X, et al. Mg-BGNs/DCECM composite scaffold for cartilage regeneration: a preliminary in vitro study. Pharmaceutics. 2021;13(10):1550.
[22] MARTIN AR, PATEL JM, ZLOTNICK HM, et al. Emerging therapies for cartilage regeneration in currently excluded ‘red knee’ populations. NPJ Regen Med. 2019;4:12.
[23] NGUYEN TU, WATKINS KE, KISHORE V. Photochemically crosslinked cell-laden methacrylated collagen hydrogels with high cell viability and functionality. J Biomed Mater Res A. 2019;107(7):1541-1550.
[24] HUA Y, HUO Y, BAI B, et al. Fabrication of biphasic cartilage-bone integrated scaffolds based on tissue-specific photo-crosslinkable acellular matrix hydrogels. Mater Today Bio. 2022;17:100489.
[25] ELOMAA L, ALMALLA A, KESHI E, et al. Rise of tissue- and species-specific 3D bioprinting based on decellularized extracellular matrix-derived bioinks and bioresins. Biomater Biosyst. 2023;12:100084.
[26] MEINERT C, WEEKES A, CHANG CW, et al. Crosslinking substrate regulates frictional properties of tissue-engineered cartilage and chondrocyte response to loading. Commun Mater. 2025;6:55.
[27] 滕建祥,朱骥生,袁代柱,等.双喷头静电纺丝法制备载软骨脱细胞基质的复合纳米纤维支架[J].中国组织工程研究,2023, 27(34):5448-5454.
[28] LIN CJ, LIN HL, YOU WC, et al. Composite hydrogels of ultrasound-assisted-digested formic acid-decellularized extracellular matrix and sacchachitin nanofibers incorporated with platelet-rich plasma for diabetic wound treatment. J Funct Biomater. 2023;14(8):423.
[29] HUANG T, ZENG Y, LI C, et al. Preparation and investigation of cellulose acetate/gelatin Janus nanofiber wound dressings loaded with zinc oxide or curcumin for enhanced antimicrobial activity. Membranes (Basel). 2024;14(5):95.
[30] ZHANG B, PEI Z, HE W, et al. 3D-printed porous zinc scaffold combined with bioactive serum exosomes promotes bone defect repair in rabbit radius. Aging (Albany NY). 2024;16(11):9625-9648.
[31] SUN J, CAO W, PAN S, et al. Porous organic materials in tissue engineering: recent advances and applications for severed facial nerve injury repair. Molecules. 2024;29(3):566.
[32] EVUARHERHE A JR, CONDRON NB, KNAPIK DM, et al. Effect of Mechanical Mincing on Minimally Manipulated Articular Cartilage for Surgical Transplantation. Am J Sports Med. 2022;50(9):2515-2525.
[33] WANG X, ZHANG Z, QIN C, et al. Shape-memory responses compared between random and aligned electrospun fibrous mats. Front Bioeng Biotechnol. 2023;11:1130315.
[34] QIAO S, PEIJIE T, NAN J. Crosslinking strategies of decellularized extracellular matrix in tissue regeneration. J Biomed Mater Res A. 2024;112:640-671.
[35] 李亚超,査刘生.可光交联的可注射水凝胶研究[J].石油化工, 2019,48(11): 1140-1145.
[36] LI T, LIU J, GUO M, et al. Synthesis of biocompatible BSA-GMA and two-photon polymerization of 3D hydrogels with free radical type I photoinitiator. Int J Bioprint. 2023;9(5):752.
[37] HERBANU A, ANA ID, ARDHANI R, et al. Fibrous PVA matrix containing strontium-substituted hydroxyapatite nanoparticles from golden apple snail (Pomacea canaliculata L.) shells for bone tissue engineering. Bioengineering (Basel). 2023;10(7):844.
[38] WANG X, XU T, WANG F. Coaxial bioprinting of Schwann cells and neural stem cells in a three-dimensional microenvironment for the repair of peripheral nerve defects. J Biomed Mater Res A. 2025;113:e37943.
[39] GOLEBIOWSKA AA, INTRAVIAI JT, SATHE VM, et al. Decellularized extracellular matrix biomaterials for regenerative therapies: advances, challenges and clinical prospects. Bioact Mater. 2023;32:98-123.
[40] SONG Z, ZHANG X, XU Y, et al. 3D-printed polycaprolactone combined with cartilage acellular matrix tissue engineered cartilage scaffold manufactured by low-temperature deposition manufacturing. Front Bioeng Biotechnol. 2025;13:1604515.
[41] GUO X, LIU B, ZHANG Y, et al. Decellularized extracellular matrix for organoid and engineered organ culture. J Tissue Eng. 2024;15: 20417314241300386.
[42] VAN DER KRAAN PM. The changing role of TGFβ in healthy, ageing and osteoarthritic joints. Nat Rev Rheumatol. 2017;13(3):155-163.
[43] JIANG S, GUO W, TIAN G, et al. Clinical application status of articular cartilage regeneration techniques: tissue-engineered cartilage brings new hope. Stem Cells Int. 2020;2020:5690252.
|