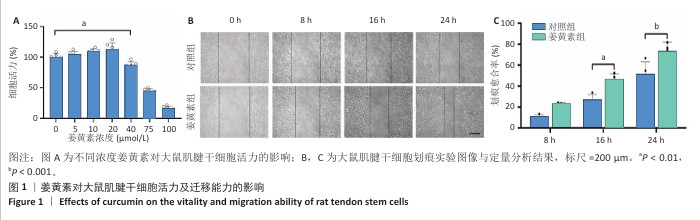
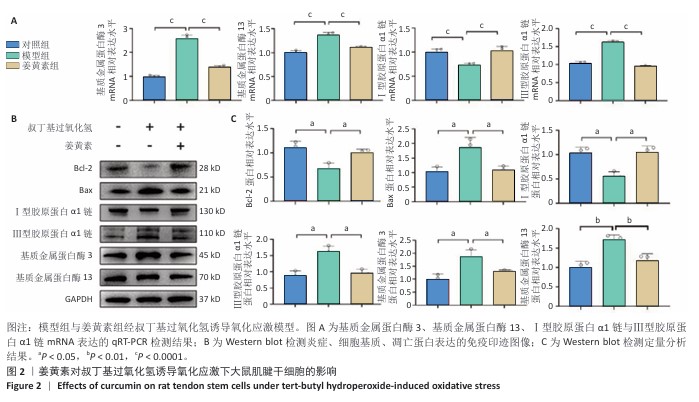
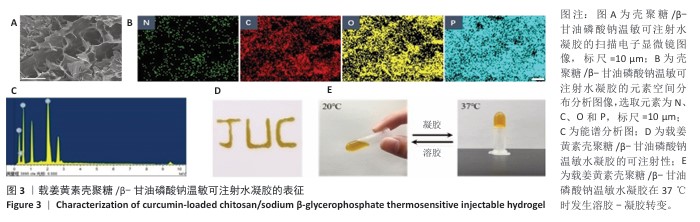
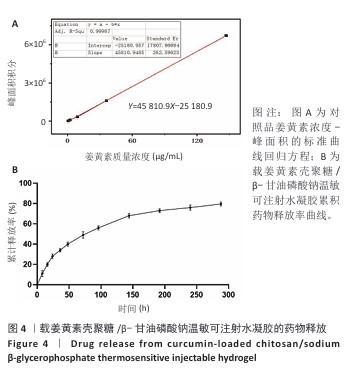
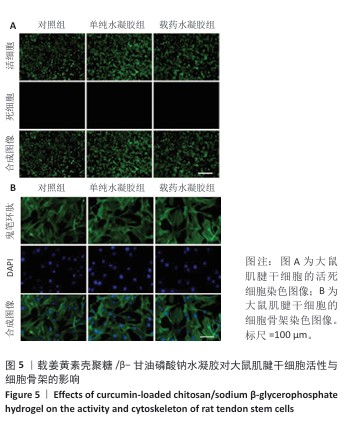
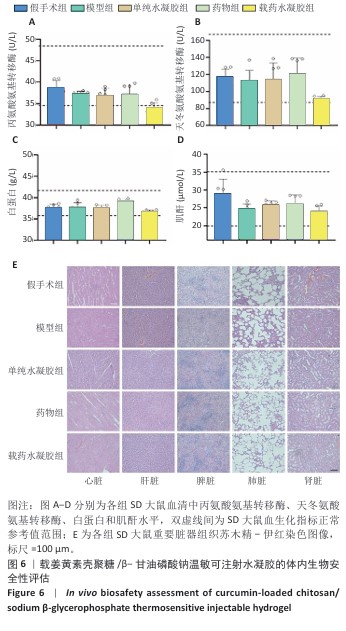
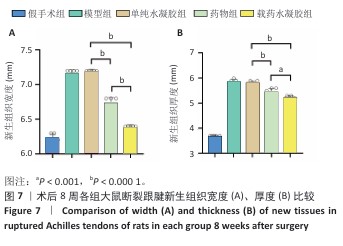
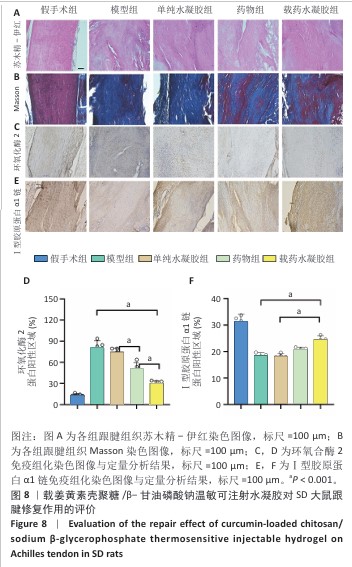
[1] LUO S, HUANG S, LI S. Review of Preclinical and Clinical Studies Supporting the Role of Polydeoxyribonucleotide in the Treatment of Tendon Disorders. Med Sci Monit. 2025;31:e945743.
[2] THOMOPOULOS S, PARKS WC, RIFKIN DB, et al. Mechanisms of tendon injury and repair. J Orthop Res. 2015;33(6):832-839.
[3] ZHANG Q, YANG Y, YILDIRIMER L, et al. Advanced technology-driven therapeutic interventions for prevention of tendon adhesion: Design, intrinsic and extrinsic factor considerations. Acta Biomater. 2021;124:15-32.
[4] LIU J, DAI G, XIAO L, et al. Tendon-Derived Stem Cell Sheet Promotes Early-Stage Tendon-Bone Healing. J Musculoskelet Neuronal Interact. 2025;25(1):133-141.
[5] LIANG Y, XU K, ZHANG P, et al. Quercetin reduces tendon adhesion in rat through suppression of oxidative stress. BMC Musculoskelet Disord. 2020;21(1):608.
[6] LEE YW, FU SC, MOK TY, et al. Local administration of Trolox, a vitamin E analog, reduced tendon adhesion in a chicken model of flexor digitorum profundus tendon injury. J Orthop Translat. 2017;10:102-107.
[7] ZHANG W, LI X, COMES FRANCHINI M, et al. Controlled release of curcumin from curcumin-loaded nanomicelles to prevent peritendinous adhesion during Achilles tendon healing in rats. Int J Nanomedicine. 2016;11:2873-2881.
[8] HE Y, YUE Y, ZHENG X, et al. Curcumin, inflammation, and chronic diseases: how are they linked? Molecules. 2015;20(5):9183-9213.
[9] GÜLEÇ A, TÜRK Y, AYDIN BK, et al. Effect of curcumin on tendon healing: an experimental study in a rat model of Achilles tendon injury. Int Orthop. 2018; 42(8):1905-1910.
[10] GOEL A, KUNNUMAKKARA AB, AGGARWAL BB. Curcumin as “Curecumin”: from kitchen to clinic. Biochem Pharmacol. 2008;75(4):787-809.
[11] ARAÚJO CC, LEON LL. Biological activities of Curcuma longa L. Mem Inst Oswaldo Cruz. 2001;96(5):723-728.
[12] AMALRAJ A, PIUS A, GOPI S, et al. Biological activities of curcuminoids, other biomolecules from turmeric and their derivatives - A review. J Tradit Complement Med. 2016;7(2):205-233.
[13] GOPI S, JACOB J, VARMA K, et al. Comparative Oral Absorption of Curcumin in a Natural Turmeric Matrix with Two Other Curcumin Formulations: An Open-label Parallel-arm Study. Phytother Res. 2017;31(12):1883-1891.
[14] KUNNUMAKKARA AB, BORDOLOI D, PADMAVATHI G, et al. Curcumin, the golden nutraceutical: multitargeting for multiple chronic diseases. Br J Pharmacol. 2016; 174(11):1325-1348.
[15] HUSSAIN Z, THU HE, NG SF, et al. Nanoencapsulation, an efficient and promising approach to maximize wound healing efficacy of curcumin: A review of new trends and state-of-the-art. Colloids Surf B Biointerfaces. 2017;150:223-241.
[16] KULAC M, AKTAS C, TULUBAS F, et al. The effects of topical treatment with curcumin on burn wound healing in rats. J Mol Histol. 2013;44(1):83-90.
[17] MEHRABANI D, FARJAM M, GERAMIZADEH B, et al. The healing effect of curcumin on burn wounds in rat. World J Plast Surg. 2015;4(1):29-35.
[18] DUAN Y, CHEN H L, LING M, et al. The Curcumin Analog EF24 Inhibits Proliferation and Invasion of Triple-Negative Breast Cancer Cells by Targeting the Long Noncoding RNA HCG11/Sp1 Axis. Mol Cell Biol. 2022;42(1):e0016321.
[19] RAHMANIAN-DEVIN P, BARADARAN RAHIMI V, ASKARI VR. Thermosensitive chitosan-β-glycerophosphate hydrogels as targeted drug delivery systems: An overview on preparation and their applications. Adv Pharmacol Pharm Sci. 2021; 2021:6640893.
[20] BAJPAI AK, SHUKLA SK, BHANU S, et al. Responsive polymers in controlled drug delivery. Prog Polym Sci. 2008;33(11):1088-1118.
[21] RUEL-GARIEPY E, LEROUX JC. In situ-forming hydrogels—review of temperature-sensitive systems. Eur J Pharm Biopharm. 2004;58(2):409-426.
[22] CHO E, QIAO Y, CHEN C, et al. Injectable FHE+BP composites hydrogel with enhanced regenerative capacity of tendon-bone interface for anterior cruciate ligament reconstruction. Front Bioeng Biotechnol. 2023;11:1117090.
[23] SATHESH D, SATHESH KUMAR K, DEVADASAN V, et al. Formulation of a thermo-sensitive hydro-gel for ulcerative colitis treatment. Bioinformation. 2022;18(10):925-937.
[24] FELT O, BURI P, GURNY R. Chitosan: a unique polysaccharide for drug delivery. Drug Dev Ind Pharm. 1998;24(11):979-993.
[25] 王雲蛟.阿司匹林对肌腱干细胞和肌腱病损伤修复的作用和机制研究[D].重庆:中国人民解放军陆军军医大学,2019.
[26] 蒋涛.肌腱干细胞来源外泌体在肌腱损伤修复中的作用及其机制研究[D].南京:南京大学,2019.
[27] GUO D, LI H, LIU Y, et al. Fibroblast growth factor-2 promotes the function of tendon-derived stem cells in Achilles tendon restoration in an Achilles tendon injury rat model. Biochem Biophys Res Commun. 2020;521(1):91-97.
[28] XU K, AL-ANI MK, SUN Y, et al. Platelet-rich plasma activates tendon-derived stem cells to promote regeneration of Achilles tendon rupture in rats. J Tissue Eng Regen Med. 2017;11(4):1173-1184.
[29] AL-ANI M, XU K, SUN Y, et al. Study of Bone Marrow Mesenchymal and Tendon-Derived Stem Cells Transplantation on the Regenerating Effect of Achilles Tendon Ruptures in Rats. Stem Cells Int. 2015;2015:984146.
[30] 汤锦波.各种伤情下屈肌腱的愈合及粘连形成:肌腱愈合[J].手外科杂志, 1992,8(1):31-35.
[31] MULHALL KJ, MCLAUGHLIN R, KAY E, et al. Thermal preconditioning prevents peritendinous adhesions and inflammation. Clin Orthop Relat Res. 2002; 405(405):258-266.
[32] INGOLD I, BERNDT C, SCHMITT S, et al. Selenium utilization by GPX4 is required to prevent hydroperoxide-induced ferroptosis. Cell. 2018;172(3):409-422.e21.
[33] LU S, SONG Y, LUO R, et al. Ferroportin-Dependent Iron Homeostasis Protects against Oxidative Stress-Induced Nucleus Pulposus Cell Ferroptosis and Ameliorates Intervertebral Disc Degeneration In Vivo. Oxid Med Cell Longev. 2021;2021:6670497.
[34] 靳苏香,程洁,钱雯,等.试验相关因素对SD大鼠临床生化学指标的影响[J].毒理学杂志,2019,33(5):366-369,80.
[35] WIEDMEYER CE, RUBEN D, FRANKLIN C. Complete blood count, clinical chemistry, and serology profile by using a single tube of whole blood from mice. J Am Assoc Lab Anim Sci. 2007;46(2):59-64.
[36] BOEHM O, ZUR B, KOCH A, et al. Clinical chemistry reference database for Wistar rats and C57/BL6 mice. Biol Chem. 2007;388(5):547-554.
[37] NOURISSAT G, BERENBAUM F, DUPREZ D. Tendon injury: from biology to tendon repair. Nat Rev Rheumatol. 2015;11(4):223-233.
[38] MAGNUSSON SP, LANGBERG H, KJAER M. The pathogenesis of tendinopathy: balancing the response to loading. Nat Rev Rheumatol. 2010;6(5):262-268.
[39] CHEN L, LIU JP, TANG KL, et al. Tendon derived stem cells promote platelet-rich plasma healing in collagenase-induced rat achilles tendinopathy. Cell Physiol Biochem. 2014;34(6):2153-2168.
[40] DE JONGE S, VAN DEN BERG C, DE VOS RJ, et al. Incidence of midportion Achilles tendinopathy in the general population. Br J Sports Med. 2011;45(13):1026-108.
[41] RILEY G. Tendinopathy--from basic science to treatment. Nat Clin Pract Rheumatol. 2008;4(2):82-89.
[42] MILLAR NL, SILBERNAGEL KG, THORBORG K, et al. Tendinopathy. Nat Rev Dis Primers. 2021;7(1):1.
[43] LIU A, WANG Q, ZHAO Z, et al. Nitric Oxide Nanomotor Driving Exosomes-Loaded Microneedles for Achilles Tendinopathy Healing. ACS Nano. 2021;15(8): 13339-13350.
[44] DE MOS M, VAN EL B, DEGROOT J, et al. Achilles tendinosis: changes in biochemical composition and collagen turnover rate. Am J Sports Med. 2007; 35(9):1549-1556.
[45] VINCENTI MP. The matrix metalloproteinase (MMP) and tissue inhibitor of metalloproteinase (TIMP) genes. Transcriptional and posttranscriptional regulation, signal transduction and cell-type-specific expression. Methods Mol Biol. 2001;151:121-148.
[46] MARCOS RL, ARNOLD G, MAGNENET V, et al. Biomechanical and biochemical protective effect of low-level laser therapy for Achilles tendinitis. J Mech Behav Biomed Mater. 2014;29:272-285.
[47] DUAN Y, LI K, WANG H, et al. Preparation and evaluation of curcumin grafted hyaluronic acid modified pullulan polymers as a functional wound dressing material. Carbohydr Polym. 2020;238:116195.
[48] ZHOU Y, MING J, DENG M, et al. Chemically modified curcumin (CMC2.24) alleviates osteoarthritis progression by restoring cartilage homeostasis and inhibiting chondrocyte apoptosis via the NF-kappaB/HIF-2alpha axis. J Mol Med (Berl). 2020;98(10):1479-1491.
[49] BLAINE TA, KIM YS, VOLOSHIN I, et al. The molecular pathophysiology of subacromial bursitis in rotator cuff disease. J Shoulder Elbow Surg. 2005; 14(1 Suppl S):84S-89S.
[50] SHOHRATI M, ABEDI F, BAGHERI M, et al. Effects of curcumin on vascular smooth muscle cells: implications for health and disease. Pharmacol Rep. 2025. doi: 10.1007/s43440-025-00744-3.
[51] HAYDEN MS, GHOSH S. NF-κB, the first quarter-century: remarkable progress and outstanding questions. Genes Dev. 2012;26(3):203-234.
[52] PASPARAKIS M. Regulation of tissue homeostasis by NF-kappaB signalling: implications for inflammatory diseases. Nat Rev Immunol. 2009;9(11):778-788.
[53] LAWRENCE T. The nuclear factor NF-kappaB pathway in inflammation. Cold Spring Harb Perspect Biol. 2009;1(6):a001651.
[54] YANYAN Y, SEUNG CHEOL K, TAO Y, et al. Functional roles of p38 mitogen-activated protein kinase in macrophage-mediated inflammatory responses. Mediators Inflamm. 2014;2014:352371.
[55] RAMAN M, CHEN W, COBB MH. Differential regulation and properties of MAPKs. Oncogene. 2007;26(22):3100-3112.
[56] LI J, TIANZHU L, KEXIN L, et al. Inflammation-related signaling pathways in tendinopathy. Open Life Sci. 2023;18(1):20220729.
[57] MULLER SA, DURSELEN L, HEISTERBACH P, et al. Effect of a Simple Collagen Type I Sponge for Achilles Tendon Repair in a Rat Model. Am J Sports Med. 2016;44(8):1998-2004.
[58] ZHANG K, ZHANG S, LI Q, et al. Effects of celecoxib on proliferation and tenocytic differentiation of tendon-derived stem cells. Biochem Biophys Res Commun. 2014;450(1):762-766.
[59] ABDOLLAHIYAN P, OROOJALIAN F, MOKHTARZADEH A. The triad of nanotechnology, cell signalling, and scaffold implantation for the successful repair of damaged organs: An overview on soft-tissue engineering. J Control Release. 2021;332:460-492.
[60] VERONESI F, BORSARI V, CONTARTESE D, et al. The clinical strategies for tendon repair with biomaterials: A review on rotator cuff and Achilles tendons. J Biomed Mater Res B Appl Biomater. 2020;108(5):1826-1843.
[61] ABDUL ALIM M, DOMEIJ-ARVERUD E, NILSSON G, et al. Achilles tendon rupture healing is enhanced by intermittent pneumatic compression upregulating collagen type I synthesis. Knee Surg Sports Traumatol Arthrosc. 2018;26: 2021-2029.
[62] YAO L, BESTWICK CS, BESTWICK L, et al. Non-immortalized human tenocyte cultures as a vehicle for understanding cellular aspects to tendinopathy. Transl Med UniSa. 2011;1:173.
[63] MAO Z, FAN B, WANG X, et al. A Systematic Review of Tissue Engineering Scaffold in Tendon Bone Healing in vivo. Front Bioeng Biotechnol. 2021;9:621483.
[64] YANG L, TANG C, CHEN Y, et al. Pharmacological Inhibition of Rac1 Activity Prevents Pathological Calcification and Enhances Tendon Regeneration. ACS Biomater Sci Eng. 2019;5(7):3511-3522.
|