中国组织工程研究 ›› 2026, Vol. 30 ›› Issue (25): 6621-6631.doi: 10.12307/2026.274
• 组织构建综述 tissue construction review • 上一篇 下一篇
中药调控沉默信息调节因子1信号通路干预阿尔茨海默病的新途径
赵芳芳1,郭燕可2,王雪可2,庞伯通1,朱艳强1,秦 阳1,崔应麟2
- 1河南中医药大学,河南省郑州市 450046;2河南省中医院,河南省郑州市 450002
-
收稿日期:2025-08-14修回日期:2025-12-03出版日期:2026-09-08发布日期:2026-04-23 -
通讯作者:崔应麟,主任医师,教授,博士生导师,河南省中医院,河南省郑州市 450002 -
作者简介:赵芳芳,女,1996年生,在读博士,主要从事中医药防治脑病的研究工作。 -
基金资助:国家中医药管理局中医药科学技术研究专项课题(GZY-KJS-2021-017),项目负责人:崔应麟;国家中医药管理局2022年第二届全国名中医传承工作室建设项目(国中医药办人教函[2022]245号),项目负责人:崔应麟;河南省科技攻关项目(252102311014),项目负责人:王雪可
A new approach to intervene in Alzheimer's disease through regulating the silencing information regulator 1 signaling pathway with traditional Chinese medicine
Zhao Fangfang1, Guo Yanke2, Wang Xueke2, Pang Botong1, Zhu Yanqiang1, Qin Yang1, Cui Yinglin2
- 1Henan University of Chinese Medicine, Zhengzhou 450046, Henan Province, China; 2Henan Province Hospital of Traditional Chinese Medicine, Zhengzhou 450002, Henan Province, China
-
Received:2025-08-14Revised:2025-12-03Online:2026-09-08Published:2026-04-23 -
Contact:Cui Yinglin, Chief physician, Professor, Doctoral supervisor, Henan Province Hospital of Traditional Chinese Medicine, Zhengzhou 450002, Henan Province, China -
About author:Zhao Fangfang, MD candidate, Henan University of Chinese Medicine, Zhengzhou 450046, Henan Province, China -
Supported by:Special Scientific Research Project of Traditional Chinese Medicine of National Administration of Traditional Chinese Medicine, No. GZY-KJS-2021-017 (to CYL); Construction Project of the Second National Famous Traditional Chinese Medicine Inheritance Studio in 2022, National Administration of Traditional Chinese Medicine, No. [2022]245 (to CYL); Key Scientific and Technological Project of Henan Province, No. 252102311014 (to WXK)
摘要:
文题释义:
沉默信息调节因子1:属于sirtuin家族成员,功能多样,具有去乙酰化酶活性,可与多种信号通路相关蛋白相互作用。沉默信息调节因子1具有对抗衰老、延长寿命和调节新陈代谢的作用,是线粒体生物发生的重要调节因子,可通过不同的机制调节阿尔茨海默病的病理学进展和表型。
阿尔茨海默病:是最常见的一种神经性病变,是以进行性脑功能失调为特征的疾病。
背景:沉默信息调节因子1作为一种去乙酰化酶,其激活后可调节多种蛋白的转录活性以及许多阿尔茨海默病相关病理过程,包括调节能量代谢、氧化应激、神经炎症、自噬和细胞凋亡等,与阿尔茨海默病的发生、发展及转归密切相关。近年来,大量研究发现,中药单体有效成分及复方可通过激活沉默信息调节因子1信号通路,减少细胞凋亡、保护神经元、抑制淀粉样斑块形成等病理改变,延缓阿尔茨海默病的发展。
目的:探讨沉默信息调节因子1与阿尔茨海默病的关系及中药调控沉默信息调节因子1信号通路治疗阿尔茨海默病的作用机制。
方法:以“阿尔茨海默病,痴呆,炎症,沉默信息调节因子1,氧化应激,炎症因子,信号通路,中药单体,复方”为中文检索词,“Alzheimer’s disease, dementia,SIRT1,inflammation,oxidative stress,inflammatory factors,signaling pathways,TCM monomers,compounds”为英文检索词,检索2015年1月至2025年3月中国知网、万方、维普、中国生物医学文献数据库(SinoMed)和PubMed数据库发表的有关沉默信息调节因子1与阿尔茨海默病及中药调控机制的文献,排除与文章相关性不高及重复、过时的文献,共检索出4 921篇相关文献,最终纳入154篇文献进行综述。
结果与结论:①大量实验研究证实,沉默信息调节因子1在阿尔茨海默病中具有重要作用;②中医方药可通过多种方式调节沉默信息调节因子1信号通路,如阿魏酸钠通过提高阿尔茨海默病大鼠前额叶皮质区沉默信息调节因子1的表达,而改善因缺血缺氧受损的神经细胞,进而改善阿尔茨海默病大鼠的学习记忆能力;牡蛎肽能够增强活性氧,降低海马组织的氧化损伤,缓解神经炎症,增强突触功能,减少神经元损伤和死亡,发挥脑保护作用,改善认知功能障碍。
https://orcid.org/0009-0006-3474-0682 (赵芳芳)
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
中图分类号:
引用本文
赵芳芳, 郭燕可, 王雪可, 庞伯通, 朱艳强, 秦 阳, 崔应麟. 中药调控沉默信息调节因子1信号通路干预阿尔茨海默病的新途径[J]. 中国组织工程研究, 2026, 30(25): 6621-6631.
Zhao Fangfang, Guo Yanke, Wang Xueke, Pang Botong, Zhu Yanqiang, Qin Yang, Cui Yinglin. A new approach to intervene in Alzheimer's disease through regulating the silencing information regulator 1 signaling pathway with traditional Chinese medicine[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(25): 6621-6631.

转移酶催化烟酰胺转化为烟酰胺单核苷酸[9],烟酰胺磷酸核糖转移酶活性水平升高增加细胞烟酰胺腺嘌呤二核苷酸水平以及沉默信息调节因子1 转录活性[10],并通过催化底物的去乙酰化修饰,广泛参与基因表达调控、能量代谢重编程和应激反应等关键生物学过程[11]。首先,沉默信息调节因子1通过激活叉头框蛋白O转录因子增强抗氧化防御系统[12],这一作用与其抑制核因子κB信号通路的抗炎效应共同构成了细胞的保护机制[13]。在此基础上,沉默信息调节因子1进一步通过调节AMP活化蛋白激酶/哺乳动物雷帕霉素靶蛋白(Amp-activated protein kinase,AMPK/mammalian target of rapamycin,mTOR)通路促进自噬,这一过程不仅清除了受损的细胞,另一方面,沉默信息调节因子1介导的肿瘤蛋白p53去乙酰化作用也可显著抑制细胞凋亡[14]。与此同时,沉默信息调节因子1对细胞稳态的调控延伸至能量代谢领域,通过激活过氧化物酶体增殖体激活受体γ共激活因子1α(peroxisome proliferator-activated receptor gamma co-activator-1α,
PGC-1α),它既促进了线粒体的生物发生和功能优化,又协调了糖脂代谢的平衡,从而系统性地维持了细胞的能量稳态[15]。这种多靶点调控特性使沉默信息调节因子1成为连接氧化应激、炎症、自噬、凋亡和能量代谢网络的核心枢纽[16-17],在神经退行性疾病、代谢紊乱和衰老等病理过程中发挥重要的保护作用,具有广阔的干预潜力。
2.2 沉默信息调节因子1在阿尔茨海默病发病过程中的作用
2.2.1 能量稳态的核心作用 大脑作为高耗能器官,其耗氧量约占机体总量的20%[18]。在阿尔茨海默病病理过程中,能量代谢障碍与β-淀粉样蛋白沉积形成恶性循环。早期临床观察显示,轻度认知障碍患者颞顶叶皮质已出现显著的生物能量利用障碍和氧化应激现象[19-20],研究发现,沉默信息调节因子1与过氧化物酶体增殖体激活受体γ共激活因子1α共同调控线粒体能量代谢[21-23]:一方面沉默信息调节因子1通过抑制糖原合成酶激酶3β稳定过氧化物酶体增殖体激活受体γ共激活因子1α,促进线粒体生物发生和ATP生成[24];另一方面,糖原合成酶激酶3β又可磷酸化tau蛋白促进神经原纤维缠结形成。动物实验证实,激活沉默信息调节因子1-过氧化物酶体增殖体激活受体γ共激活因子1α通路不仅能增加线粒体含量和能量代谢效率[25],还能显著减少海马区β-淀粉样蛋白沉积和tau病理,有效改善认知功能并延缓衰老进程[26]。
2.2.2 调控氧化应激 在阿尔茨海默病患者中,能量代谢障碍与氧化应激形成恶性循环,线粒体功能受损导致活性氧过度积累,而活性氧通过抑制沉默信息调节因子1-过氧化物酶体增殖体激活受体γ共激活因子1α轴进一步加重线粒体功能障碍[27]。生理状态下活性氧参与突触信号传导等神经活动[28],但其过量产生会降低沉默信息调节因子1活性,加速神经元衰老[29]。据报道,沉默信息调节因子1的激活可以抵消β-淀粉样蛋白聚集诱导的氧化应激[30]。首先,沉默信息调节因子1通过激活沉默信息调节因子1-叉头框蛋白O1/3(Forkhead Box O proteins1/3,FOXO1/3)-张力素同源物诱导的激酶蛋白1/RBR结构域的E3泛素连接酶信号通路增强线粒体自噬,清除受损线粒体,减少活性氧产生[31]。其次,通过过氧化物酶体增殖体激活受体γ共激活因子1α上调抗氧化基因表达并抑制β-分泌酶,减少β-淀粉样蛋白生成,降低氧化应激水平。此外,沉默信息调节因子1与AMP活化蛋白激酶协同作用,通过沉默信息调节因子1-AMP活化蛋白激酶-过氧化物酶体增殖体激活受体γ共激活因子1α轴调控能量代谢和线粒体生物合成[32]。最后,激活核因子E2相关因子2(nuclear factor erythroid 2-related factor 2,Nrf2)/抗氧化反应元件通路,促进血红素加氧酶1、超氧化物歧化酶等抗氧化基因的表达[33]。这些机制共同构成沉默信息调节因子1调控氧化应激网络,维持氧化还原平衡,减轻β-淀粉样蛋白沉积和tau病理,为阿尔茨海默病治疗提供重要靶点。
2.2.3 神经源性炎症 阿尔茨海默病病理过程中,氧化应激通过激活抑制核因子κB等促炎信号通路,促使小胶质细胞和星形胶质细胞异常活化并释放大量促炎因子,包括如白细胞介素1β、肿瘤坏死因子α和白细胞介素6,形成慢性神经炎症微环境,这种持续的炎症状态通过氧化应激损伤和兴奋性毒性作用,加剧神经元损伤和突触丢失[34],从而加速阿尔茨海默病病程进展[35]。研究表明,沉默信息调节因子1通过双重机制调控Β-分泌酶表达以减少β-淀粉样蛋白生成[36],一方面通过激活过氧化物酶体增殖体激活受体γ共激活因子1α,促进过氧化物酶体增殖物激活受体γ(peroxisome proliferator-activated receptor gamma,PPARγ)去乙酰化,使其与Β-分泌酶基因启动子区的PPAR反应元件结合,进而直接抑制转录[37];另一方面,通过下调核因子κB信号通路间接抑制Β-分泌酶表达[38]。这两种机制协同作用显著降低了β-淀粉样蛋白生成,不仅揭示了沉默信息调节因子1在阿尔茨海默病炎症病理中的关键调控作用,也为靶向沉默信息调节因子1-过氧化物酶体增殖体激活受体γ共激活因子1α/过氧化物酶体增殖物激活受体γ和沉默信息调节因子1-抑制核因子κB通路的治疗策略提供了理论依据。
2.2.4 突触可塑性损伤 在阿尔茨海默病的病理进程中,β-淀粉样蛋白沉积和tau蛋白异常磷酸化通过诱发神经炎症和氧化应激,最终导致突触损伤和认知功能障碍。突触可塑性作为学习记忆的神经基础[39],主要表现为长时程增强和长时程抑制两种形式,分别调控突触连接的增强与减弱[40]。研究发现,阿尔茨海默病患者海马区突触密度显著降低[41],且这种突触病理改变往往先于老年斑和神经原纤维缠结的出现[42-43],沉默信息调节因子1在维持突触可塑性中发挥关键作用[44-46]。动物实验表明,沉默信息调节因子1缺失会导致树突长度、密度及分支显著减少[45],并影响轴突延伸[47];而沉默信息调节因子1可通过上调脑源性神经营养因子表达[48],增加树突棘数量并增强神经元连接,从而改善认知功能。这些发现提示沉默信息调节因子1可能通过调控突触结构和功能来改善阿尔茨海默病相关的记忆和认知障碍。
2.2.5 神经元凋亡 在阿尔茨海默病的病理进程中,慢性神经炎症和氧化应激共同破坏神经元能量代谢稳态,导致线粒体功能障碍和ATP生成不足,进而引发突触可塑性障碍,最终触发神经元凋亡,这一恶性循环不断加剧β-淀粉样蛋白沉积和tau病理,从而加速阿尔茨海默病的进展[34]。研究表明,阿尔茨海默病患者神经元表现出DNA断裂和半胱天冬酶3激活特征,其脑组织中B细胞淋巴瘤2(B-cell Lymphoma 2,Bcl-2)家族成员表达异常,包括抗凋亡蛋白(Bcl-2、Bcl-XL)和促凋亡蛋白(BAX、BAK、BAD、Bcl-2相互作用介导死亡蛋白)的表达失衡,这些改变与β-淀粉样蛋白诱导的神经元死亡密切相关[49]。沉默信息调节因子1通过多重机制发挥神经保护作用:一方面使促凋亡蛋白(Bax、p53)和DNA修复蛋白Ku70去乙酰化,减少线粒体膜上的Bax聚集,抑制凋亡级联反应[50];另一方面通过调控叉头框蛋白O转录因子1/3a和AMP活化蛋白激酶/哺乳动物雷帕霉素靶蛋白、核因子E2相关因子2/血红素加氧酶1等通路,减轻氧化应激损伤,从而维持神经元存活并减少海马区细胞凋亡。
综上,阿尔茨海默病患者脑内沉默信息调节因子1的表达和活性显著降低[51],且药理学激活沉默信息调节因子1在阿尔茨海默病模型中显示出改善认知功能和病理特征的潜力[52]。因此,沉默信息调节因子1通过多靶点调控阿尔茨海默病的核心病理机制,成为阿尔茨海默病治疗的潜在干预靶点[53-54],见图3。
2.3 中药调控沉默信息调节因子1信号通路干预阿尔茨海默病的研究现状 中医对阿尔茨海默病有着深远的认识,早在《黄帝内经》中便有关于认知功能障碍的相关记载。如《灵枢·天年》云:“八十岁,肺气衰,魄离,故言善误”,“九十岁,肾气焦,四肢经脉空虚”等描述,虽未明确提出“痴呆”病名,但其对老年认知衰退的观察与现代阿尔茨海默病的临床表现高度吻合。中医以整体观念、辨证论治为特色,在防治阿尔茨海默病方面积累了丰富经验。药理学研究发现,许多中药有效成分单药及复方
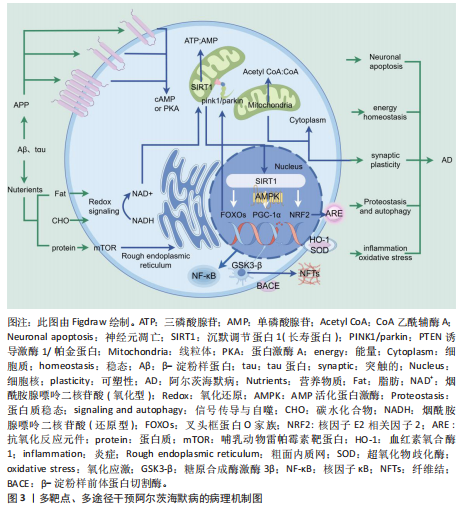
可通过调节沉默信息调节因子1信号通路改善阿尔茨海默病病理过程,为阿尔茨海默病的防治和中医药现代化研究提供了新思路。此文基于现有文献,就中医药通过沉默信息调节因子1通路干预阿尔茨海默病的研究进展作系统梳理。
2.3.1 中药有效成分
(1)酚酸类:阿魏酸钠又名川芎素,主要成分是阿魏酸的钠盐,有抗氧化的功效,对于血管性病变有显著的治疗作用[55]。丁春艳等[56]动物实验研究发现阿魏酸钠通过提高阿尔茨海默病大鼠前额叶皮质区沉默信息调节因子1的表达,间接抑制叉头框蛋白O3A的表达而改善因缺血缺氧受损的神经细胞,进而改善阿尔茨海默病大鼠的学习记忆能力。
丹酚酸 B是从丹参中提取的水溶性生物活性成分,具有抗氧化、抗炎性反应及抗衰老等作用[57]。刘新等[58]通过体外细胞实验发现丹酚酸B可以通过上调沉默信息调节因子1/过氧化物酶体增殖体激活受体γ共激活因子1α信号通路减少活性氧产生及下调丙二醛水平,上调超氧化物岐化酶水平,保护线粒体功能,显著降低β-淀粉样蛋白干预N2A细胞后诱导的氧化应激反应,为抗氧化损伤药物、抗阿尔茨海默病药物筛选提供了实验依据。
二苯乙烯苷是一种具有显著生物活性的多酚类化合物,主要来源于传统中药何首乌的根茎部位。最近的研究表明,二苯乙烯苷对衰老大鼠和淀粉样前体蛋白转基因的阿尔茨海默病模型鼠海马区突触超微结构和学习记忆能力均有所改善,具有清除自由基、抗氧化、抗炎的作用和增强衰老大鼠及淀粉样前体蛋白转基因的阿尔茨海默病模型大鼠的学习记忆能力[59]。王婷[60]进一步动物实验研究发现二苯乙烯苷能够通过清除胞内活性氧、减轻钙超载、稳定线粒体膜电位、抑制氧化应激- c-Jun氨基末端激酶(c-Jun N-terminal Kinase,JNK)-Bax 凋亡信号通路、激活抗衰老蛋白沉默信息调节因子1和抑制核因子κB激活等途径减轻神经元损伤,增强小鼠海马脑片CA1区N-甲基-D-天冬氨酸受体依赖的长时程增强效应和学习记忆能力。
(2)皂苷类:黄芪甲苷是从中草药黄芪中提纯而来的小分子皂苷[61],具有抗衰老、免疫调节作用、抗炎、抗氧化和神经保护作用[62-63]。王绛辉等[64]通过动物实验研究发现黄芪甲苷可通过减少 miR-126表达,激活沉默信息调节因子1/核因子E2相关因子2途径抑制神经细胞凋亡,降低氧化应激反应,提高大鼠行动能力,对神经细胞产生保护作用。
人参皂苷是人参的主要药效成分,有研究证明,人参皂苷 Rg1具有抗衰老、抗氧化和抗炎等多种药理作用[65-66]。胡玲[67]通过体内动物+体外细胞研究发现人参皂苷Rg1能够上调沉默信息调节因子1-核因子E2相关因子2-脑源性神经营养因子通路,明显提高神经干细胞中超氧化物歧化酶、过氧化氢酶的活性,降低丙二醛和一氧化氮含量,上调线粒体活性,增强细胞能量代谢,同时增强抗氧化能力,减少氧化应激损伤,从而延缓小鼠认知障碍。
薯蓣皂苷是从薯蓣中提取出来的生物活性成分[68-69],具有抗肿瘤、调节免疫、抗血小板聚集、调血脂等功效。刘璐等[70]动物实验研究发现薯蓣皂苷可升高大鼠沉默信息调节因子1 mRNA和蛋白表达水平,降低抑制核因子κB mRNA和蛋白表达水平,其原因可能是薯蓣皂苷可刺激机体沉默信息调节因子1的表达,从而靶向抑制抑制核因子κB的表达,延缓机体炎性反应的发生,起到改善大鼠认知功能的作用。
太白楤木总皂苷是从太白楤木根皮中提取出来的总皂苷成分[71]。谭薇等[72]的动物实验研究发现太白楤木总皂苷能够升高沉默信息调节因子1/叉头框蛋白O1/过氧化物酶体增殖体激活受体γ共激活因子1α通路相关蛋白,降低Ace-叉头框蛋白O1、Ace过氧化物酶体增殖体激活受体γ共激活因子1α蛋白表达,缓解脑皮质组织损伤及炎症反应及神经元凋亡,改善小鼠学习与记忆能力,且EX-527可减弱太白楤木总皂苷对小鼠认知功能障碍的改善作用。
(3)黄酮类及多酚类:黄腐酚是由苦参中提取出来的活性成分,具有抗炎、抗氧化应激等作用[73]。孙雪娇[74]通过动物实验研究发现黄腐酚可有效提升沉默信息调节因子1、Nrt2、血红素加氧酶1等抗氧化基因的表达,提高超氧化物歧化酶活性,降低丙二醛含量,减少神经元变性,表明黄腐酚对阿尔茨海默病模型小鼠具有神经保护作用,且其可能是通过调节沉默信息调节因子1/核因子E2相关因子2/血红素加氧酶1信号通路改善氧化应激状态而实现的。
原花青素是从传统中药材莲房中提取而来[75],主要表现为抗氧化、改善血液循环、保护视力、消除水肿、降低胆固醇、增进记忆、减缓衰老以及抗脑卒中等作用。赵思琪[76]通过动物实验研究发现莲房原花青素通过上调阿尔茨海默病模型小鼠海马及皮质沉默信息调节因子1表达水平,增强海马长时程增强效应的产生和维持,减少β-淀粉样蛋白产生和聚集,降低对神经元及突触毒性作用,影响突触可塑性,最终改善认知功能障碍,发挥对阿尔茨海默病的防治作用。
(4)生物碱类:苦参碱是从植物苦参、苦豆子等中药材中分离出的一种生物碱,化学结构属于喹诺嗪类,具有抗心律失常、抗炎、抗病毒、保护神经等药理活性[77]。刘景祎等[78]通过体外细胞实验研究发现苦参碱可抑制AMP活化蛋白激酶/沉默信息调节因子1通路的激活,下调磷酸化AMP活化蛋白激酶、沉默信息调节因子1蛋白表达,减轻β-淀粉样蛋白25-35诱导PC12细胞氧化损伤和炎症反应,并抑制细胞凋亡,减轻阿尔茨海默病小鼠学习记忆功能,保护脑神经。
雷公藤甲素是中药雷公藤的主要活性成分之一,具有抗炎、抗肿瘤及免疫抑制等作用[79],大量研究证实其可在神经系统退行性疾病中发挥积极的神经元保护作用[80]。WAN等[81-83]通过动物实验研究发现雷公藤甲素可上调沉默信息调节因子1蛋白的表达,抑制核因子κB及白细胞介素6、肿瘤坏死因子α水平和半胱天冬酶3蛋白表达,促进海马神经元突触素表达,减少β-淀粉样蛋白生成,减少神经元凋亡,降低组织氧化应激水平及神经元凋亡,提升学习和记忆能力,并且这种作用可通过EX527消除,因此其机制可能是通过调控沉默信息调节因子1蛋白表达实现的。
(5)萜类及内酯类:银杏内酯是得到广泛研究的一种银杏叶提取物,可抑制代谢紊乱、内质网应激、过氧化及炎症等,促进患者神经功能恢复[84]。田新红等[85]动物实验研究发现银杏内酯可降低前列腺素E2、白细胞介素1β以及认知障碍大鼠脑组织小胶质细胞M1型标志物的表达和升高M2型标志物蛋白、沉默信息调节因子1,高迁移率族蛋白B1蛋白表达,抑制炎性因子表达及神经炎症,阻碍神经元凋亡,进而提高大鼠的认知能力。
欧前胡素是伞形科植物蛇床子、白芷、羌活等的有效成分之一[86],是一种呋喃香豆素。研究显示,欧前胡素具有抗炎、抗氧化和神经保护等作用[87-89]。万航娟等[86]动物实验研究发现欧前胡素可上调β-淀粉样蛋白致阿尔茨海默病模型小鼠海马组织中沉默信息调节因子1表达,抑制蛋白激酶R样内质网激酶/真核翻译起始因子2α的磷酸化激活,降低CCAAT/增强子结合蛋白同源蛋白表达,减轻β-淀粉样蛋白诱导内质网应激反应所致的神经损伤防治阿尔茨海默病。
(6)多糖类:刺五加多糖是中药刺五加的主要活性成分[90],具有抗炎、抗氧化等作用,可减轻海马神经递质损伤,提高机体抗氧化能力,进而提高小鼠学习记忆能力。代泓怡等[91]的动物实验研究发现刺五加多糖可以升高沉默信息调节因子1、过氧化物酶体增殖体激活受体γ共激活因子1α表达,改善线粒体功能,进而改善神经损伤;沉默信息调节因子1通路抑制剂EX-527与刺五加多糖同时干预SD大鼠后,沉默信息调节因子1、过氧化物酶体增殖体激活受体γ共激活因子1α表达下调,证实刺五加多糖可通过激活沉默信息调节因子1/过氧化物酶体增殖体激活受体γ共激活因子1α通路,改善大鼠神经损伤,改善大脑学习记忆能力及认知功能障碍。
归芪多糖为当归与黄芪质量比1∶5通过水提醇沉法制备得到,具有降血糖、抗氧化、延缓衰老、保护肝脏功能等多种药理作用[92]。张凯丽[93]的动物实验研究发现归芪多糖可能通过沉默信息调节因子1/过氧化物酶体增殖体激活受体γ共激活因子1α通路减轻衰老大鼠脑组织线粒体的损伤,抑制氧化应激反应,改善线粒体功能,上调线粒体介导的凋亡因子Bcl-2/Bax的比值,并通过沉默信息调节因子1/p53/p21、p16-细胞周期蛋白D1信号通路延缓大鼠衰老。
(7)其他植物提取类:火麻仁提取液是从火麻提炼获得,可通过抗氧化、抗炎、抗血栓和降低血脂等方面的作用而起到改善中枢神经系统病变的作用[94]。黄玲[95]的动物实验研究发现火麻仁提取液多不饱和脂肪酸通过上调沉默信息调节因子1的表达,进而激活沉默信息调节因子1-解整合素-金属蛋白酶1-淀粉样前体蛋白信号通路,使β-淀粉样蛋白的生成减少,从而减弱了β-淀粉样蛋白在衰老大鼠海马区的沉积进而减少对神经元的神经毒性;以及激活沉默信息调节因子1-阴阳1转录因子-cAMP反应元件结合蛋白-脑源性神经营养因子信号通路,使脑源性神经营养因子在海马中的表达增加,加强了突触的可塑性;通过上调NIH/3T3细胞的沉默信息调节因子表达水平而影响细胞衰老信号通路[96],使衰老相关蛋白 p53、p21的表达下降,进而发挥延缓细胞衰老以及抗氧化损伤和神经保护作用,最终起到改善衰老大鼠学习记忆障碍的作用。
丹参酮IA作为一种低毒有效的丹参中药成分,具有抗氧化、抗炎、抗肿瘤、保护线粒体及神经元的作用[97]。史彦锌[98]的体外细胞实验研究发现丹参酮IA可以增加烟酰胺腺嘌呤二核苷酸/还原型烟酰胺腺嘌呤二核苷酸水平,由于沉默信息调节因子1是烟酰胺腺嘌呤二核苷酸依赖性酶,因此也表明丹参酮IA可以提高沉默信息调节因子1的水平,从而抑制哺乳动物雷帕霉素靶蛋白信号通路的激活,并降低活性氧活性,降低氧化应激,增强线粒体自噬水平,抑制线粒体介导的细胞凋亡,改善认知障碍和学习记忆能力,并且给与EX527预处理后烟酰胺腺嘌呤二核苷酸/还原型烟酰胺腺嘌呤二核苷酸的显著降低。因此丹参酮IA是通过烟酰胺腺嘌呤二核苷酸/沉默信息调节因子1/哺乳动物雷帕霉素靶蛋白轴的调节促进线粒体自噬。
药物分析发现樱草杜鹃乙酸乙酯、正丁醇提取部位中包含较多黄酮类成分如槲皮素、花旗松素、金丝桃苷、杨梅素等[99],且它们对痴呆症模型均有不同程度的改善作用。屈培珍等[100]的动物实验研究发现樱草杜鹃石油醚提取物能够改善超氧化物歧化酶、谷胱甘肽、过氧化氢酶活力及血红素加氧酶和丙二醛的含量,同时回调凋亡蛋白Bax、半胱天冬酶3和Bcl-2的相对蛋白表达量,明显增加脑内沉默信息调节因子1的平均吸光度值,升高核因子E2相关因子2和血红素加氧酶1的相对蛋白表达量,这提示樱草杜鹃乙酸乙酯可能是通过调控沉默信息调节因子1/核因子E2相关因子2/血红素加氧酶1信号通路抵御脑内氧化应激、降低细胞凋亡而改善模型小鼠认知能力。
双去甲氧基姜黄素能够对抗β-淀粉样蛋白毒性的神经保护作用[101],且抗氧化的能力较母体姜黄素更高,且能增加端粒酶的长度,延长细胞的周期。胡蓉[102]的动物实验研究发现双去甲氧基姜黄素可明显升高淀粉样前体蛋白/早老素1小鼠的沉默信息调节因子1表达以及谷胱甘肽酶活性,降低超氧歧化酶活性,维持神经元形态、增多海马细胞数,改善海马组织形态学结构和氧化应激水平,改善小鼠的学习记忆功能。
牡蛎肽是将牡蛎肉进行酶解、过滤和干燥而得的一种拥有诸多生物活性的小分子肽,具有改善阿尔茨海默病的潜在功效[103]。张文杰[104]的动物实验研究发现牡蛎肽的干预能够增强超氧化物歧化酶、氧化氢酶、脑源性神经营养因子/原肌球蛋白受体激酶B、沉默信息调节因子1-过氧化物酶体增殖体激活受体γ共激活因子1α-核因子E2相关因子1-线粒体转录因子A 信号通路表达,降低海马组织的氧化损伤和白细胞介素1β、白细胞介素6和肿瘤坏死因子α水平,缓解神经炎症,增强突触功能,抑制海马中β-淀粉样蛋白的生成,增强海马的突触可塑性,改善认知功能障碍[103-105]。
掌叶半夏又称虎掌南星[106],为天南星科植物掌叶半夏的块茎,可以镇静镇痛,且具有降压、抑制血小板聚集、抗肿瘤、催吐、泻下与抗氧化的作用,这些药理作用暗示掌叶半夏能降低衰老相关疾病,提高老年人的生活质量。冯协和[107]的体外细胞实验研究发现掌叶半夏可增加沉默信息调节因子1、叉头框蛋白O3基因表达量和叉头框蛋白O3a、人类抗原R和核糖体蛋白S19结合蛋白1的蛋白表达量,降低p53基因和乳腺癌缺失蛋白1蛋白表达量,提高机体的抗氧化能力,减少氧化应激损伤,还可以提高脑组织端粒酶活性,起到稳定端粒长度和结构的作用,延长细胞的生命周期,改善衰老大鼠的学习记忆能力。
迷迭香是一种常见的香草植物,其独特的香味具有激励情绪,强化记忆等作用[108]。施凤飞等[109]的动物实验研究发现迷迭香吸嗅可通过激活沉默信息调节因子1和抑制NOD样受体热蛋白结构域相关蛋白3、半胱天冬酶1信号通路介导的细胞焦亡改善大鼠的认知功能,发挥神经保护作用。
综上所述,通过调节沉默信息调节因子1信号通路干预阿尔茨海默病的中药有效成分涉及黄酮类、酚类、皂苷类、多糖类、生物碱类等。这些中药有效成分可调节沉默信息调节因子1信号通路抑制炎症、氧化应激、内质网应激稳定线粒体的结构和功能,改善能量代谢,减少细胞凋亡,延缓阿尔茨海默病的进展。中药有效成分调控沉默信息调节因子1信号通路改善阿尔茨海默病的过程中涉及多通路间的交互作用,目前的机制研究尚难以体现其全方位的作用。此外,中药有效成分的临床有效性、量效关系等也应深入研究,以便为进一步的新药开发和临床应用提供更可靠的证据。中药有效成分调节沉默信息调节因子1信号通路干预 阿尔茨海默病的作用机制总结见表1。
2.3.2 中药复方
(1)扶正类
补肾填精、益智健脑:补肾健脑汤是一个“温补脾肾”方,临床治疗阿尔茨海默病疗效显著,由开心散和龟鹿二仙胶加减而成,具有补肾健脾,益智开窍功效。现代药理表明,补肾健脑汤中多个中药单体对线粒体自有一定的调控作用,并且可以改善阿尔茨海默病的记忆功能障碍[110-111]。刘永惠等[112]的动物实验研究发现补肾健脑汤可升高ATP、微管相关蛋白1轻链3β、张力素同源物诱导的激酶蛋白1、叉

头框蛋白O3、沉默信息调节因子1、RBR结构域的E3泛素连接酶表达含量,降低活性氧、P62水平,因此其调控机制与激活沉默信息调节因子1-叉头框蛋白O3-张力素同源物诱导的激酶蛋白1-RBR结构域的E3泛素连接酶信号通路有关,进而促进线粒体自噬,最终减轻氧化应激损伤而满足神经元高能量需求,改善小鼠学习认知能力。
补肾益智方是由人参、枸杞、蛇床子、女贞子、丹皮和何首乌6味中药组成的经典复方,该方剂基于中医“肾主骨生髓,髓通于脑”理论,通过填精益髓、补气养血、开窍醒神、活血化瘀等多靶点协同作用,发挥改善认知功能的治疗效果。既往研究发现该方可改善胆碱能系统的功能及Tau蛋白过度磷酸化[113-114],上调谷氨酸和乙酰胆碱受体,加强对谷氨酸的神经毒性的对抗作用[115],可增加神经细胞轴突的长度,增强突触的可塑性[116]。许婷婷[117]的动物实验研究发现补肾益智方可升高沉默信息调节因子1、Bc1-2,降低Bax、半胱天冬酶3水平,改善乙酰胆碱能神经系统,保护神经元,缓解内质网应激,抑制由内质网应激所引起的细胞凋亡,进而改善SAMP8小鼠的认知功能障碍。
生慧汤出自清代名医陈士铎《辨证录·健忘门》,由熟地黄、石菖蒲、远志、山茱萸、柏子仁(去油)、酸枣仁等9味中药组成,具有交通心肾、化痰开窍、调养心神等功效[118]。叶南等[119]的动物实验研究发现生慧汤可降低肿瘤坏死因子α、白细胞介素6、白细胞介素1β含量,升高沉默信息调节因子1含量,因此生慧汤可以抑制海马神经元损伤,其机制可能与调控沉默信息调节因子1/抑制核因子κB信号通路的关键因子和肿瘤坏死因子α、白细胞介素6、白细胞介素1β 等促炎因子的表达有关。
大补元煎源自《景岳全书》,被张景岳誉为“救本培元第一要方”,在培补元气、滋养肾元的同时,滋养大脑,使脑有所养,不至于髓海空虚,从而改善记忆、学习能力。HSIEH等[120]的动物实验研究发现大补元煎会磷酸化AMP活化蛋白激酶a-Thr172,进而激活沉默信息调节因子1,控制胶质细胞,抑制阿尔茨海默病神经炎症,减少神经元凋亡率缓解神经元丢失。
加减薯蓣丸由经典名方薯蓣丸化裁而来,诸药合用,共奏补肾益脑、填精益髓、化痰祛瘀之功。药理学研究发现该方能促进阿尔茨海默病小鼠海马转录因子EB核转移,激活并上调自噬溶酶体途径,清除毒性蛋白β-淀粉样蛋白42,缓解其认知损伤[121]。周剑杰[122]的动物实验研究发现加减薯蓣丸能够上调线粒体生物合成关键因子沉默信息调节因子1/过氧化物酶体增殖体激活受体γ共激活因子1α,上调自噬转录因子叉头框蛋白O1,促进张力素同源物诱导的激酶蛋白1/RBR结构域的E3泛素连接酶通路介导的线粒体自噬途径,有效改善淀粉样前体蛋白/早老素1小鼠的学习记忆能力及海马神经元氧化应激损伤。
补气养血、调和气血:补阳还五汤由黄芪、地龙、桃仁、红花、当归、川芎、芍药组成,是益气活血的代表方[123]。王逸雪[124]的动物实验研究发现补阳还五汤降低大鼠脑内丙二醛、Bax及内质网应激相关通路蛋白,增高内源性抗氧化酶超氧化物歧化酶、氧化氢酶、谷胱甘肽过氧化物酶、Bc1-2、沉默信息调节因子1水平的活性,降低痴呆大鼠氧化应激水平并减少神经细胞凋亡,这些保护效应可能通过调控内质网应激来缓解大鼠学习认知障碍。
当归芍药散出自张仲景《金匮要略》,全方共奏肝脾同调,气血并养,痰瘀共除的功效。现代研究中发现该方对阿尔茨海默病的预防及治疗主要表现在抗氧化损伤,抗细胞凋亡,抑制炎症等方面[125-127]。杨苗等[128]动物实验研究发现当归芍药散可升高磷酸化AMP活化蛋白激酶/AMP活化蛋白激酶、过氧化物酶体增殖体激活受体γ共激活因子1α、沉默信息调节因子1,促进线粒体生物发生,维持线粒体稳态,改善阿尔茨海默病大鼠脑内能量供应,发挥对阿尔茨海默病的防治作用。
黑逍遥散出自《医略六书·女科指要》,诸药合用,全方共奏补肾益髓、疏肝畅情、健脾袪浊、精血并补、气血同治之功[129]。前期研究表明黑逍遥散可从抑制β-淀粉样蛋白沉积、Tau蛋白磷酸化、炎症反应、氧化应激等发病机制防治阿尔茨海默病[130-131]。米彩云等[131]动物实验研究发现黑逍遥散可升高超氧化物歧化酶、谷胱甘肽过氧化物酶4 mRNA 表达,降低丙二醛、Fe2+、长链脂酰辅酶A合成酶4含量减少,抑制神经元铁死亡,改善阿尔茨海默病模型大鼠的认知功能障碍,推测其机制可能与激活沉默信息调节因子1/p53/长链脂酰辅酶A合成酶4信号通路、有效调控关键蛋自有关。
健脾益气、调和脾胃:补肾健脾方由熟地、山药、枸杞子等10余味中药组成,且现代多组研究团队证明补肾健脾类中药能提高超氧化物歧化酶[132]、谷胱甘肽-Px活性、降低丙二醛含量,增强清除自由基能力以发挥抗衰老作用。张楚洁[133]的动物实验研究发现补肾健脾方可能通过降低抑制核因子κB表达,提升沉默信息调节因子1、沉默信息调节因子6表达,抑制白细胞介素1β、肿瘤坏死因子α上调抑制免疫炎症反应,通过上调超氧化物歧化酶发挥抗氧化应激作用,以达到延缓免疫衰老的目的。
开心散出自唐代孙思邈的《备急千金要方》,全方以补为主,标本兼顾,药性平和,且均有益智、安神、宁心之功。现代研究表明开心散对治疗抑郁老年痴呆、记忆减弱等疾病表现均有显著疗效[134]。李少瑜[135]的动物实验研究发现开心散含药血清给药后线粒体融合蛋白1、线粒体融合蛋白2、脑源性神经营养因子、沉默信息调节因子3 蛋白表达量显著升高磷酸化动力相关蛋白1、NOD样受体热蛋白结构域相关蛋白3(Nod-like receptor pyrindomain-containing protein 3,NLRP3)、凋亡相关斑点样蛋白、切割型半胱天冬酶1(Cleaved-Caspase1)、白细胞介素18、白细胞介素1β蛋白表达量显著降低,且细胞活力明显提高,因此防治阿尔茨海默病体现在细胞密度增加、排列紧密、形态恢复正常,通过改善细胞线粒体功能、增加神经营养因子、减轻神经炎症发挥作用。
养心安神:强记汤源自陈士铎《辨证录·健忘门》,由熟地黄、酸枣仁、麦冬和远志四味组成,起补肾化痰、养心安神之功。前期研究发现,强记汤可通过调节AMP活化蛋白激酶/沉默信息调节因子1信号通路D氨基半乳糖胺诱导的认知损伤、神经元凋亡及神经炎[136]。何丽玲等[137]的动物实验研究发现强记汤可增加模型小鼠海马组织中磷酸化AMP活化蛋白激酶α-Thr172、沉默信息调节因子1、过氧化物酶体增殖体激活受体γ共激活因子1、核因子E2相关因子1、核因子E2相关因子2和线粒体转录因子A 蛋自水平,缓解线粒体功能障碍,改善认知功能障碍。
酸枣仁汤出自张仲景《金匮要略》,具有滋阴养血安神益智之功。前期研究发现,酸枣仁汤均具有良好的抗阿尔茨海默病作用。龙清华等[138]的动物实验研究发现酸枣仁汤明显增加ATP、磷酸化AMP活化蛋白激酶α-Thr172、沉默信息调节因子1、核因子E2相关因子1、核因子E2相关因子2、线粒体转录因子A表达并减少活性氧含量,改善阿尔茨海默病小鼠海马区线粒体肿胀、线粒体嵴断裂及线粒体功能障碍,缓解认知损伤。
(2)祛邪类
化痰开窍:洗心汤源自明代陈士铎《辨证录》,由人参、茯神、半夏等中药组成,具有健脾化痰,醒神开窍之功。药理实验研究表明洗心汤及其有效成分可通过改善线粒体功能,调控免疫炎症防治阿尔茨海默病[139-140]。汤子钦等[141]体外细胞实验研究发现洗心汤治疗后小鼠海马中沉默信息调节因子1蛋白表达显著上升,SAMP8海马和血清中炎症相关因子抑制核因子κB、p65、NOD样受体热蛋白结构域相关蛋白3、肿瘤坏死因子α和白细胞介素1β水平均明显下降,以及海马中晚期糖基化终末产物受体、Toll样受体1、Toll样受体2蛋白表达也显著降低。因此洗心汤可通过沉默信息调节因子1相关信号通路改善ATP含量,从而达到抗炎及抗凋亡作用。
活血化瘀、通络解毒:解毒益智方由黄连、益智仁、川芎、地龙、龟板胶、山萸肉、酒大黄7味药组成,具有补益虚损,又祛瘀生新之功效[142]。王田野等[143]的动物实验研究发现解毒益智方能够降低淀粉样前体蛋白/早老素1阿尔茨海默症小鼠的海马组织的丙二醛含量,增加超氧化物歧化酶、沉默信息调节因子1的含量,增强细胞抗氧化能力,上调核因子E2相关因子2的mRNA的表达,通过沉默信息调节因子1/核因子E2相关因子2信号通路对抗阿尔茨海默病小鼠中氧化应激水平失衡起效。
加味脑泰方由黄芪、当归、川芎、地龙等组成,起益气活血化痰之功。前期研究发现,脑泰方通过抗血小板活化、抗血栓、抗炎等作用对缺血缺氧脑组织起到明显的保护作
用[144-145]。易亚乔等[146]的动物实验研究发现加味脑泰方可升高沉默信息调节因子1、核因子κB和 IkBa 蛋白表达,保护缺氧损伤海马神经元继而防治阿尔茨海默病。
利水渗湿:泽泻汤由泽泻、白术组成,具有健脾益气、利湿化痰之效[147]。苏世杰等[148]动物实验研究发现泽泻汤能下调海马组织中促炎因子环氧化酶2、诱导型一氧化氮合酶、肿瘤坏死因子α、白介素细胞1β mRNA表达水平,上调磷酸化AMP活化蛋白激酶/AMP活化蛋白激酶、沉默信息调节因子1及过氧化物酶体增殖体激活受体γ共激活因子1α蛋白表达水平,有效改善认知功能障碍,其作用机制可能是激活AMP活化蛋白激酶/沉默信息调节因子1/过氧化物酶体增殖体激活受体γ共激活因子1α通路,改善糖脂代谢紊乱,维持能量代谢稳态,启动线粒体合成,减轻海马神经元的凋亡,起到神经保护作用,改善认知障碍。
交通心肾:交泰丸由黄连、肉桂组成,具有交通心肾、清心安神之功效。蔡伟武[149]的动物实验研究发现交泰丸通过沉默信息调节因子1/肌醇需求酶1/NOD样受体热蛋白结构域相关蛋白3炎性小体通路,增加沉默信息调节因子1蛋白水平,缓解肌醇需求酶1通路的内质网应激水平,减少NOD样受体热蛋白结构域相关蛋白3炎性小体表达,减少炎症的发生,同时有助于β-淀粉样蛋白的清除,减少氧化应激水平的多靶点作用,从而改善阿尔茨海默病模型小鼠的学习和空间记忆能力。
其他类:天地转气汤以肾精亏虚为本,痰蒙清窍为标,从多角度、多因素治疗阿尔茨海默病,适用于多种证型兼有其他证型的阿尔茨海默病患者。殷聚波[150]的动物实验研究发现天地转气汤可降低核因子κB、IkBα含量,通过增加沉默信息调节因子1及相关因子的表达,激活沉默信息调节因子1/核因子κB信号通路,抑制核因子κB蛋白表达,而核因子κB蛋白自身抑制剂IrBα表达增加,进一步减少核因子κB 蛋白表达,减轻海马组织内神经元炎症反应,从而保护神经元、改善大鼠的学习记忆能力。
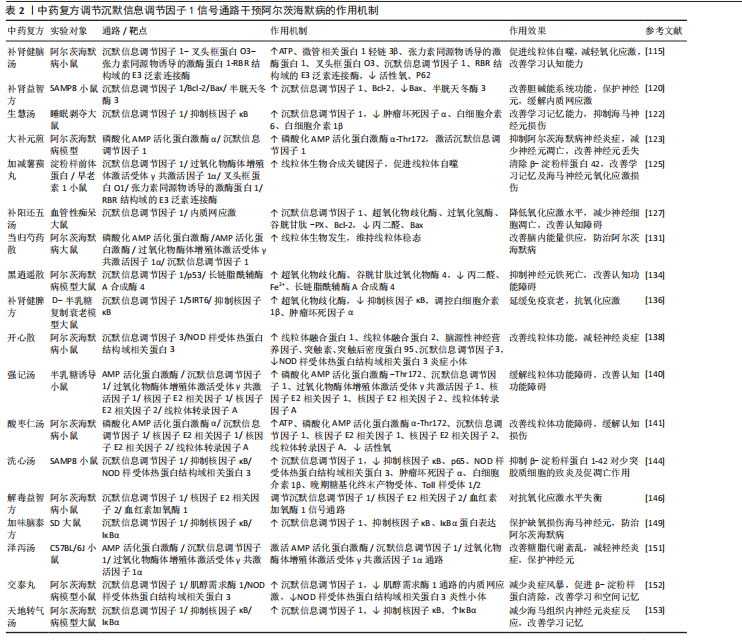
综上,调节沉默信息调节因子1信号通路干预阿尔茨海默病的中药复方的功效集中在益气健脾、温阳利水、活血化瘀等方面,黄芪、茯苓、白术、桂枝、甘草、丹参均为常用药物,其机制主要为调节过氧化物酶体增殖体激活受体γ共激活因子1α、核因子κB、叉头框蛋白O1等下游通路,调控线粒体稳态,抑制炎症反应和氧化应激。中药复方是中医整体观念、辨证论治特色的重要体现。配伍原则是解析中药复方内在联系的核心,也是复方发挥作用的关键,但目前中药复方基于沉默信息调节因子1信号通路治疗阿尔茨海默病的相关研究尚缺乏关于配伍关系的深入探讨,未来可进一步结合药物入血成分在体内的动态变化解析其作用机制,为临床应用提供更可靠的证据支撑。中药复方调节沉默信息调节因子1信号通路干预阿尔茨海默病的作用机制总结见表2。
| [1] 麻煜程, 李汶阳, 夏雨田,等. 阿尔兹海默病多靶点药物研究进展[J]. 西部医学,2025,37(8): 1242-1248. [2] VAZ M, SILVESTRE S. Alzheimer’s disease: Recent treatment strategies. Eur J Pharmacol. 2020; 887:173554. [3] LI X, FENG X, SUN X, et al. Global, regional, and national burden of Alzheimer’s disease and other dementias, 1990-2019. Front Aging Neurosci. 2022;14:937486. [4] ZHAO Y, ZHANG J, ZHENG Y, et al. NAD(+) improves cognitive function and reduces neuroinflammation by ameliorating mitochondrial damage and decreasing ROS production in chronic cerebral hypoperfusion models through Sirt1/PGC-1α pathway. J Neuroinflammation. 2021;18(1):207. [5] WEN Q, WANG Y, PAN Q, et al. MicroRNA-155-5p promotes neuroinflammation and central sensitization via inhibiting SIRT1 in a nitroglycerin-induced chronic migraine mouse model. J Neuroinflammation. 2021;18(1):287. [6] TANG BL. Sirt1 and the Mitochondria. Mol Cells. 2016;39(2):87-95. [7] ZHANG F, WANG S, GAN L, et al. Protective effects and mechanisms of sirtuins in the nervous system. Prog Neurobiol. 2011;95(3):373-395. [8] YANG Y, LIU Y, WANG Y, et al. Regulation of SIRT1 and Its Roles in Inflammation. Front Immunol. 2022;138:31168. [9] PANG N, HU Q, ZHOU Y, et al. Nicotinamide Adenine Dinucleotide Precursor Suppresses Hepatocellular Cancer Progression in Mice. Nutrients. 2023;15(6):1447 [10] GARTEN A, SCHUSTER S, PENKE M, et al. Physiological and pathophysiological roles of NAMPT and NAD metabolism. Nat Rev Endocrinol. 2015;11(9):535-546. [11] WU YJ, FANG WJ, PAN S, et al. Regulation of Sirt1 on energy metabolism and immune response in rheumatoid arthritis. Int Immunopharmacol. 2021;101(Pt A):108175. [12] DILMAC S, KUSCU N, CANER A, et al. SIRT1/FOXO Signaling Pathway in Breast Cancer Progression and Metastasis. Int J Mol Sci. 2022; 23(18)10227. [13] CHEN C, ZHOU M, GE Y, et al. SIRT1 and aging related signaling pathways. Mech Ageing Dev. 2020;187:111215. [14] XU C, WANG L, FOZOUNI P, et al. SIRT1 is downregulated by autophagy in senescence and ageing. Nat Cell Biol. 2020;22(10):1170-1179. [15] VARGHESE B, CHIANESE U, CAPASSO L, et al. SIRT1 activation promotes energy homeostasis and reprograms liver cancer metabolism. J Transl Med. 2023;21(1):627. [16] FIORENTINO F, FABBRIZI E, MAI A, et al. Activation and inhibition of sirtuins: From bench to bedside. Med Res Rev. 2025;45(2):484-560. [17] PARK JH, BURGESS JD, FAROQI AH, et al. Alpha-synuclein-induced mitochondrial dysfunction is mediated via a sirtuin 3-dependent pathway. Mol Neurodegener. 2020;15(1):5. [18] GOLDBERG DJ, ZAK V, GOLDSTEIN BH, et al. Results of the FUEL Trial. Circulation. 2020;141(8): 641-651. [19] DEWANJEE S, CHAKRABORTY P, BHATTACHARYA H, et al. Altered glucose metabolism in Alzheimer’s disease: Role of mitochondrial dysfunction and oxidative stress. Free Radic Biol Med. 2022; 193(Pt 1):134-157. [20] MARTINO ADAMI PV, GALEANO P, WALLINGER ML, et al. Worsening of memory deficit induced by energy-dense diet in a rat model of early-Alzheimer’s disease is associated to neurotoxic Aβ species and independent of neuroinflammation. Biochim Biophys Acta Mol Basis Dis. 2017; 1863(3):731-743. [21] LI Y, WANG P, YANG X, et al. SIRT1 inhibits inflammatory response partly through regulation of NLRP3 inflammasome in vascular endothelial cells. Mol Immunol. 2016;77:148-156. [22] KAARNIRANTA K, KAJDANEK J, MORAWIEC J, et al. PGC-1α Protects RPE Cells of the Aging Retina against Oxidative Stress-Induced Degeneration through the Regulation of Senescence and Mitochondrial Quality Control. The Significance for AMD Pathogenesis. Int J Mol Sci. 2018;19(8): 2317. [23] JIAO W, HU F, LI J, et al. Qiangji Jianli Decoction promotes mitochondrial biogenesis in skeletal muscle of myasthenia gravis rats via AMPK/PGC-1α signaling pathway. Biomed Pharmacother. 2020;129:110482. [24] XU W, LUO Y, YIN J, et al. Targeting AMPK signaling by polyphenols: a novel strategy for tackling aging. Food Funct. 2023;14(1):56-73. [25] REDHWAN MAM, M GH, SAMADDAR S, et al. Chitosan/siRNA nanoparticles targeting PARP-1 attenuate Neuroinflammation and apoptosis in hyperglycemia-induced oxidative stress in Neuro2a cells. Int J Biol Macromol. 2024;282(Pt 2): 136964. [26] 胡媛媛, 姜广建, 祝嘉健,等. 藜麦复合物调控SIRT1/PGC-1α通路改善糖尿病小鼠肝细胞损伤[J]. 湖南中医药大学学报,2021,41(12): 1863-1868. [27] WANG F, WANG J, SHEN Y, et al. Iron Dyshomeostasis and Ferroptosis: A New Alzheimer’s Disease Hypothesis? Front Aging Neurosci. 2022;14:830569. [28] GREWAL AK, SINGH TG, SHARMA D, et al. Mechanistic insights and perspectives involved in neuroprotective action of quercetin. Biomed Pharmacother. 2021;140:111729. [29] CONTI V, CORBI G, SIMEON V, et al. Aging-related changes in oxidative stress response of human endothelial cells. Aging Clin Exp Res. 2015;27(4): 547-553. [30] YE F, WU A. The Protective Mechanism of SIRT1 in the Regulation of Mitochondrial Biogenesis and Mitochondrial Autophagy in Alzheimer’s Disease. J Alzheimers Dis. 2021;82(1):149-157. [31] ZHAO N, XIA J, XU B. Physical exercise may exert its therapeutic influence on Alzheimer’s disease through the reversal of mitochondrial dysfunction via SIRT1-FOXO1/3-PINK1-Parkin-mediated mitophagy. J Sport Health Sci. 2021; 10(1):1-3. [32] SHI D, HAO Z, QI W, et al. Aerobic exercise combined with chlorogenic acid exerts neuroprotective effects and reverses cognitive decline in Alzheimer’s disease model mice (APP/PS1) via the SIRT1/ /PGC-1α/PPARγ signaling pathway. Front Aging Neurosci. 2023;151:269952. [33] RYU JS, KANG HY, LEE JK. Effect of Treadmill Exercise and Trans-Cinnamaldehyde against d-Galactose- and Aluminum Chloride-Induced Cognitive Dysfunction in Mice. Brain Sci. 2020; 10(11):793. [34] ARIOZ BI, TASTAN B, TARAKCIOGLU E, et al. Melatonin Attenuates LPS-Induced Acute Depressive-Like Behaviors and Microglial NLRP3 Inflammasome Activation Through the SIRT1/Nrf2 Pathway. Front Immunol. 2019;10:1511. [35] LIN J Y, KUO WW, BASKARAN R, et al. Swimming exercise stimulates IGF1/ PI3K/Akt and AMPK/SIRT1/PGC1α survival signaling to suppress apoptosis and inflammation in aging hippocampus. Aging (Albany NY). 2020;12(8):6852-6864. [36] DHAPOLA R, SHARMA P, KUMARI S, et al. Environmental Toxins and Alzheimer’s Disease: a Comprehensive Analysis of Pathogenic Mechanisms and Therapeutic Modulation. Mol Neurobiol. 2024;61(6):3657-3677. [37] KOO JH, KANG EB, OH YS, et al. Treadmill exercise decreases amyloid-β burden possibly via activation of SIRT-1 signaling in a mouse model of Alzheimer’s disease. Exp Neurol. 2017;288:142-152. [38] ABU SHELBAYEH O, ARROUM T, MORRIS S, et al. PGC-1α Is a Master Regulator of Mitochondrial Lifecycle and ROS Stress Response. Antioxidants (Basel). 2023;12(5):1075. [39] RAMIREZ S. Crystallizing a memory. Science. 2018; 360(6394):1182-1183. [40] CHEYNE JE, MONTGOMERY JM. The cellular and molecular basis of in vivo synaptic plasticity in rodents. Am J Physiol Cell Physiol. 2020;318(6): c1264-c1283. [41] BASTIN C, BAHRI MA, MEYER F, et al. In vivo imaging of synaptic loss in Alzheimer’s disease with [18F]UCB-H positron emission tomography. Eur J Nucl Med Mol Imaging. 2020;47(2):390-402. [42] LI S, SELKOE DJ. A mechanistic hypothesis for the impairment of synaptic plasticity by soluble Aβ oligomers from Alzheimer’s brain. J Neurochem. 2020;154(6):583-597. [43] 曹颖, 官志忠. 阿尔茨海默病发病机制中突触病理及突触囊泡内吞蛋白的改变[J]. 中华老年心脑血管病杂志,2010,12(1):92-94. [44] GAO J, WANG WY, MAO YW, et al. A novel pathway regulates memory and plasticity via SIRT1 and miR-134. Nature. 2010;466(7310):1105-1109. [45] MICHáN S, LI Y, CHOU MM, et al. SIRT1 is essential for normal cognitive function and synaptic plasticity. J Neurosci. 2010;30(29):9695-9707. [46] HERSKOVITS AZ, GUARENTE L. SIRT1 in neurodevelopment and brain senescence. Neuron. 2014;81(3):471-483. [47] CHIBAATAR E, LE K, ABDOULAYE IA, et al. Melatonin Ameliorates Lipopolysaccharide-Induced Microglial Inflammation via Triggering SIRT1/HMGB1 Signaling Axis. J Mol Neurosci. 2021;71(4):691-701. [48] VOGTMANN R, BAO M, DEWAN MV, et al. Growth-Restricted Fetuses and Offspring Reveal Adverse Sex-Specific Metabolic Responses in Preeclamptic Mice Expressing Human sFLT1. Int J Mol Sci. 2023; 24(8):6885. [49] ZHANG L, QIAN Y, LI J, et al. BAD-mediated neuronal apoptosis and neuroinflammation contribute to Alzheimer’s disease pathology. iScience. 2021;24(9):102942. [50] KITADA M, OGURA Y, KOYA D. The protective role of Sirt1 in vascular tissue: its relationship to vascular aging and atherosclerosis. Aging (Albany NY). 2016;8(10):2290-2307. [51] DO J, KIM N, JEON SH, et al. Trans-Cinnamaldehyde Alleviates Amyloid-Beta Pathogenesis via the SIRT1-PGC1α-PPARγ Pathway in 5XFAD Transgenic Mice. Int J Mol Sci. 2020;21(12):4492. [52] LI HR, LIU Q, ZHU CL, et al. β-Nicotinamide mononucleotide activates NAD+/SIRT1 pathway and attenuates inflammatory and oxidative responses in the hippocampus regions of septic mice. Redox Biol. 2023;63:102745. [53] LU C, ZHAO H, LIU Y, et al. Novel Role of the SIRT1 in Endocrine and Metabolic Diseases. Int J Biol Sci. 2023;19(2):484-501. [54] SHIN M K, VÁZQUEZ-ROSA E, KOH Y, et al. Reducing acetylated tau is neuroprotective in brain injury. Cell. 2021;184(10):2715-2732.e2723. [55] XU SH, YIN MS, LIU B, et al. Tetramethylpyrazine-2’-O-sodium ferulate attenuates blood-brain barrier disruption and brain oedema after cerebral ischemia/reperfusion. Hum Exp Toxicol. 2017; 36(7):670-680. [56] 丁春艳, 丁坤, 程博为, 等. 阿魏酸钠通过SIRT1/FOXO3A改善血管性痴呆大鼠学习记忆障碍的机制研究[J]. 右江民族医学院学报,2024,46(3):283-288. [57] HE Y, JIA K, LI L, et al. Salvianolic acid B attenuates mitochondrial stress against Aβ toxicity in primary cultured mouse neurons. Biochem Biophys Res Commun. 2018;498(4):1066-1072. [58] 刘新, 赵媛. 丹酚酸B通过SIRT1/PGC-1α通路对Aβ1-42干预N2A细胞保护作用研究[J]. 脑与神经疾病杂志,2024,32(8):491-497. [59] DASKALAKI AD, KALLERGI E, KOLAXI A, et al. Local biogenesis of autophagic vesicles in neuronal dendrites facilitates long-term synaptic depression. Autophagy. 2022;18(8):2011-2012. [60] 王婷. 何首乌二苯乙烯苷对缺血性脑损伤和学习记忆的影响及其机制[D]. 武汉:华中科技大学,2009. [61] 方建, 李晓晖, 陈文武. 黄芪甲苷基于MEK5/ERK5信号通路对阿尔茨海默病大鼠小胶质细胞活性的影响[J]. 上海中医药杂志,2021,55(10): 73-78. [62] JIANG M, NI J, CAO Y, et al. Astragaloside IV Attenuates Myocardial Ischemia-Reperfusion Injury from Oxidative Stress by Regulating Succinate, Lysophospholipid Metabolism, and ROS Scavenging System. Oxid Med Cell Longev. 2019; 2019:9137654. [63] ZHANG L, DENG S. Effects of astragaloside IV on inflammation and immunity in rats with experimental periodontitis. Braz Oral Res. 2019; 33:e032. [64] 王绛辉, 郑凯, 张倩,等. 黄芪甲苷通过减少miR-126表达调节SIRT1/Nrf2通路改善老年痴呆机制[J]. 中国老年学杂志,2025,45(6):1353-1360. [65] ZHANG H, SU Y, SUN Z, et al. Ginsenoside Rg1 alleviates Aβ deposition by inhibiting NADPH oxidase 2 activation in APP/PS1 mice. J Ginseng Res. 2021;45(6):665-675. [66] XU TZ, SHEN XY, SUN LL, et al. Ginsenoside Rg1 protects against H2O2‑induced neuronal damage due to inhibition of the NLRP1 inflammasome signalling pathway in hippocampal neurons in vitro. Int J Mol Med. 2019;43(2):717-726. [67] 胡玲. 人参皂苷Rg1调控Sirt1-Nrf2-BDNF通路延缓脑衰老的机制研究[D]. 重庆:重庆医科大学,2023. [68] 李文砚, 黄静丽, 李长洲,等. 薯蓣皂苷元生物合成途径及其关键基因研究进展[J]. 植物生理学报,2024,60(8):1221-1228. [69] ZHOU C, LI X, ZHOU Z, et al. Comparative Transcriptome Analysis Identifies Genes Involved in Diosgenin Biosynthesis in Trigonella foenum-graecum L. Molecules. 2019;24(1):140 [70] 刘璐, 丁力, 方晓霞,等. 薯蓣皂苷对阿尔茨海默病模型大鼠认知功能的影响[J]. 中国药业, 2022,31(11):55-59. [71] WIDJAJA AA, LIM WW, VISWANATHAN S, et al. Inhibition of IL-11 signalling extends mammalian healthspan and lifespan. Nature. 2024;632(8023): 157-165. [72] 谭薇, 吴莲珍, 杨晨曦. 太白楤木总皂苷介导SIRT1/FOXO1/PGC-1α通路对创伤性脑损伤小鼠认知功能障碍的影响[J]. 中国药学杂志,2024, 59(1):39-44. [73] 王海波, 刘静蕊, 高莹莹,等. 异黄腐酚药理作用及机制的研究进展[J]. 中南药学,2025,23(1): 196-202. [74] 孙雪娇. 黄腐酚对阿尔茨海默病模型大鼠认知损害的神经保护作用及机制研究[D]. 石家庄:河北医科大学,2016. [75] XU Z, DU P, MEISER P, et al. Proanthocyanidins: oligomeric structures with unique biochemical properties and great therapeutic promise. Nat Prod Commun. 2012;7(3):381-388. [76] 赵思琪. 莲房原花青素对阿尔茨海默病小鼠认知功能的改善作用及其机制研究[D]. 武汉: 武汉科技大学,2019. [77] CHHABRA S, MEHAN S. Matrine exerts its neuroprotective effects by modulating multiple neuronal pathways. Metab Brain Dis. 2023;38(5): 1471-1499. [78] 刘景祎, 李富慧, 宋彦. 基于AMPK/SIRT1通路探究苦参碱对阿尔茨海默病Aβ25-35诱导PC12细胞氧化损伤和凋亡的影响[J]. 中国医科大学学报,2024,53(8):741-746+751. [79] 崔进, 陈晓, 苏佳灿. 雷公藤甲素药理作用研究新进展[J]. 中国中药杂志,2017,42(14):2655-2658. [80] CHENG S, LEBLANC KJ, LI L. Triptolide preserves cognitive function and reduces neuropathology in a mouse model of Alzheimer’s disease. PLoS One. 2014;9(9):e108845. [81] WAN B, HU X, NIE J, et al. Effects of triptolide on degeneration of dendritic spines induced by Aβ1-40 injection in rat hippocampus. Neurol Sci. 2014; 35(1):35-40. [82] OBERSTEIN TJ, TAHA L, SPITZER P, et al. Imbalance of Circulating T(h)17 and Regulatory T Cells in Alzheimer’s Disease: A Case Control Study. Front Immunol. 2018;9:1213. [83] 姚鹏, 陈勇, 徐国海. 雷公藤甲素对血管性痴呆大鼠认知功能的影响及对SIRT1/NF-κB信号通路的作用[J]. 中国中药杂志,2019,44(16):3423-3428. [84] 李静, 曹福彬, 高嘉潞,等. 银杏内酯B对急性脑梗死患者血清IL-6水平、脑血流量的影响及其临床疗效[J]. 中国老年学杂志,2021,41(20): 4360-4362. [85] 田新红, 郝莉, 游言文,等. 银杏内酯B对阿尔茨海默病大鼠海马Synapsin-1、Beclin1和LC3表达的影响[J]. 中国中医基础医学杂志, 2017,23(12):1701-1704+1759. [86] 万航娟, 罗丽, 何蔚. 欧前胡素对阿尔茨海默病模型小鼠SIRT1和磷酸化PERK/eIF2α及CHOP蛋白表达的影响[J]. 赣南医学院学报, 2022,42(5):443-449. [87] CHOWDHURY AA, GAWALI NB, SHINDE P, et al. Imperatorin ameliorates lipopolysaccharide induced memory deficit by mitigating proinflammatory cytokines, oxidative stress and modulating brain-derived neurotropic factor. Cytokine. 2018;110:78-86. [88] ZHENG XX, ZHANG KY, LI YC, et al. Imperatorin ameliorates learning and memory deficits through BDNF/TrkB and ERK/CaMKIIα/CREB signaling in prenatally-stressed female offspring. Phytother Res, 2020;34(9):2408-2418. [89] GE JW, DENG SJ, XUE ZW, et al. Imperatorin inhibits mitogen-activated protein kinase and nuclear factor kappa-B signaling pathways and alleviates neuroinflammation in ischemic stroke. CNS Neurosci Ther. 2022;28(1):116-125. [90] 邹婷, 刘友昊, 高晓岩,等. 刺五加精制多糖对尼古丁诱导的学习记忆障碍小鼠的影响[J]. 实用临床医药杂志,2023,27(22):67-70+76. [91] 代泓怡, 狄岩, 李翔, 等. 刺五加多糖调控SIRT1/PGC-1α通路对睡眠剥夺大鼠海马神经元损伤的影响[J]. 中医药导报,2025,31(1):48-52. [92] 蒲秀瑛,赵小亮,王鸣刚,等. SIRT1在衰老细胞(脑)线粒体损伤中的作用及归芪多糖延缓衰老的分子机制研究[Z].兰州理工大学, 2023. [93] 张凯丽. 基于SIRT1和线粒体的归芪多糖延缓大鼠脑组织衰老的分子作用机制[D]; 兰州:兰州理工大学,2021. [94] PARK Y, MOON HJ, KIM SH. N-3 polyunsaturated fatty acid consumption produces neurobiological effects associated with prevention of depression in rats after the forced swimming test. J Nutr Biochem. 2012;23(8):924-928. [95] 黄玲. SIRT1介导的火麻仁提取液改善衰老大鼠学习记忆障碍的机制[D]. 南宁:广西医科大学, 2018. [96] 陈宁园. 火麻仁提取液对D-半乳糖致衰老大鼠空间学习和记忆的干预作用及其机制研究[D]. 南宁:广西医科大学,2017. [97] JIN X, LIU MY, ZHANG DF, et al. Natural products as a potential modulator of microglial polarization in neurodegenerative diseases. Pharmacol Res. 2019;145:104253. [98] 史彦锌. 丹参酮ⅡA通过NAD+/Sirt1/mTOR促进AD细胞模型线粒体自噬的机制研究 [D]. 沈阳:沈阳医学院,2024. [99] 吴奶珠, 黄帅, 王友松,等. 藏药樱草杜鹃的黄酮类成分研究[J]. 中草药,2011,42(7):1279-1281. [100] 屈培珍, 魏江平, 刘毅,等. 基于SIRT1/NRF2/HO-1信号探讨樱草杜鹃石油醚提取物抗痴呆的作用及机制[J]. 中药新药与临床药理,2023, 34(3):341-348. [101] XIAO Z, LIN L, LIU Z, et al. Potential therapeutic effects of curcumin: relationship to microtubule-associated proteins 2 in Aβ1-42 insult. Brain Res. 2010;1361:115-123. [102] 胡蓉. 双去甲氧姜黄素通过上调Sirt1抗氧化应激拮抗阿尔茨海默病[D]. 衡阳:南华大学, 2018. [103] CAI Y, LIU J, WANG B, et al. Microglia in the Neuroinflammatory Pathogenesis of Alzheimer’s Disease and Related Therapeutic Targets. Front Immunol. 2022;138:56376. [104] 张文杰. 有氧运动联合牡蛎肽对AD样大鼠神经精神症状的干预作用及机制研究[D]. 扬州:扬州大学,2024. [105] LLANOS-GONZÁLEZ E, HENARES-CHAVARINO ÁA, PEDRERO-PRIETO CM, et al. Interplay Between Mitochondrial Oxidative Disorders and Proteostasis in Alzheimer’s Disease. Front Neurosci. 2019;13:1444. [106] 张庚, 张笑玲, 孟义江,等. 掌叶半夏开花特性及繁育生物学研究[J]. 河北农业大学学报, 2020,43(2):47-52. [107] 冯协和. 掌叶半夏提取物抗衰老活性及作用机制研究[D]. 武汉:湖北中医药大学,2016. [108] 丁桃, 胥玉林, 涂梦婷,等. 芳香疗法临床应用研究进展[J]. 亚太传统医药,2023,19(7):226-230. [109] 施凤飞, 鲍杰伟, 张衍辉,等. 迷迭香吸嗅通过SIRT1、NLRP3、Caspase-1信号通路介导细胞焦亡改善血管性认知障碍作用机制[J]. 医学理论与实践,2024,37(17):2881-2884. [110] 蒋怡萱, 谢晓琴, 李和梅,等. 人参皂苷Rg1对Aβ25-35损伤的PC12细胞中线粒体自噬的调节作用[J]. 中华中医药杂志,2022,37(3):1369-1373. [111] 康健. 基于BACE1靶点和线粒体自噬研究β-细辛醚对APP/PS1小鼠的作用机制[D]. 广州:广州中医药大学,2021. [112] 刘永惠,李少为,郑清莲.补肾健脑方对血管性痴呆大鼠乙酰胆碱和海马区ERK1和ERK2表达的影响[J]. 中国中西医结合杂志,2012,32(4): 504-509. [113] HOU XQ, ZHANG L, YANG C, et al. Alleviating effects of Bushen-Yizhi formula on ibotenic acid-induced cholinergic impairments in rat. Rejuvenation Res. 2015;18(2):111-127. [114] 张魁华,赖世隆,胡镜清,等.补肾益智方对AD动物模型大脑神经元Tau蛋白及其相关酶类的影响[J]. 中国中医基础医学杂志,2002, 8(6):30-32. [115] 张晓东. “补肾益智方”改善老年痴呆长时程增强现象的实验研究[D]. 广州: 广州中医药大学,2000. [116] HOU XQ, WU DW, ZHANG CX, et al. Bushen‑Yizhi formula ameliorates cognition deficits and attenuates oxidative stress‑related neuronal apoptosis in scopolamine‑induced senescence in mice. Int J Mol Med. 2014;34(2):429-439. [117] 许婷婷. 补肾益智方通过SIRT1/ER stress通路改善SAMP8鼠的认知功能障碍[D]. 广州:广州中医药大学,2018. [118] 梁迷, 林森相, 马捷,等. 四逆散及生慧汤拆方对神经突触可塑性的作用研究[J]. 北京中医药大学学报,2014,37(5):314-317+329+363. [119] 叶南, 邵玉萍, 王平. 基于SIRT1/NF-κB信号通路探讨有氧运动结合生慧汤对睡眠剥夺大鼠学习记忆的影响[J]. 世界科学技术-中医药现代化,2021,23(9):2994-3002. [120] HSIEH MH, CUI ZY, YANG AL, et al. Cerebral Cortex Apoptosis in Early Aged Hypertension: Effects of Epigallocatechin-3-Gallate. Front Aging Neurosci. 2021;137:05304. [121] 邱静, 鄢文静, 张雨婷,等. 加减薯蓣丸调节转录因子EB介导的自噬溶酶体通路改善APP/PS1小鼠学习记忆能力研究[J]. 安徽中医药大学学报,2020,39(1):60-64. [122] 周剑杰. 基于线粒体质量调控探讨加减薯蓣丸减轻APP/PS1小鼠神经元氧化应激损伤的作用及机制[D]. 武汉:湖北中医药大学,2022. [123] 罗艳薇, 梁正, 王晶,等. 补阳还五汤治疗泛血管疾病的药理作用及临床研究进展[J]. 实用中医内科杂志,2025,39(11):113-116. [124] 王逸雪. 补阳还五汤改善血管性痴呆大鼠认知障碍的实验研究[D]. 广州:广州中医药大学,2023. [125] 余婧萍, 贺春香, 成绍武,等. 当归芍药散通过调控NF-κB炎性通路改善H2O2诱导的SH-SY5Y细胞氧化损伤的作用[J]. 中国实验方剂学杂志, 2020,26(10):1-7. [126] 贺春香, 余婧萍, 李富周,等. 当归芍药散含药血清对Aβ1-42诱导的SH-SY5Y细胞周期和凋亡的影响[J]. 中成药,2020,42(11):2875-2882. [127] FU X, LIU Q, SUN X, et al. Research Advances in the Treatment of Alzheimer’s Disease with Polysaccharides of Danggui-Shaoyao-San. J Alzheimers Dis. 2022;85(1):7-19. [128] 杨苗, 于文静, 贺春香,等. 当归芍药散对AD大鼠线粒体稳态及AMPK/SIRT1/PGC-1α信号通路的影响[J]. 中国实验方剂学杂志,2023,29(3): 9-16. [129] 王虎平, 米彩云, 李明成,等. 基于“三阴并调”探讨黑逍遥散对AD模型大鼠海马神经元及PP2A的影响及作用机制[J]. 中药药理与临床, 2022,38(4):40-45. [130] 王虎平, 吕育洁, 胡韵韵,等. 基于RAS/RAF/MEK/ERK信号通路探讨黑逍遥散干预AD模型大鼠氧化应激的作用机制[J]. 中国实验方剂学杂志,2024,30(17):35-42. [131] 米彩云, 彭超, 周君,等. 黑逍遥散调控p38MAPK/ATF2/COX-2信号通路对阿尔茨海默病大鼠神经炎症的影响[J]. 中成药,2024,46(2): 590-595. [132] 李少康, 赵赟, 高娜,等. 补肾类方药及中药有效成分抗衰老的作用机制研究进展[J]. 天津中医药,2023,40(4):537-544. [133] 张楚洁. 基于NF-κB/SIRT1/SIRT6信号通路探究补肾健脾方对肾虚证、脾虚证衰老大鼠延缓免疫衰老机制[D]. 长沙:湖南中医药大学,2023. [134] 王常瞵, 高鹏, 姜晶晶,等. 开心散及其组成药材指纹/特征图谱的研究概况[J]. 山东中医药大学学报,2020,44(1):98-104. [135] 李少瑜. 基于SIRT3/NLRP3通路探究开心散改善APP/PS1小鼠认知障碍的机制研究 [D]. 广州:广州中医药大学,2023. [136] HE LL, WANG YC, AI YT, et al. Qiangji Decoction Alleviates Neurodegenerative Changes and Hippocampal Neuron Apoptosis Induced by D-Galactose via Regulating AMPK/SIRT1/NF-κB Signaling Pathway. Front Pharmacol. 2021;12: 735812. [137] 何丽玲, 胡慧. 强记汤通过激活AMPKα/SIRT1/PGC-1α信号通路减轻D-半乳糖诱导的认知损伤和线粒体功能障碍[J]. 中国病理生理杂志, 2024,40(10):1906-1915. [138] 龙清华, 朱麒行, 麦合丽娅·艾斯卡尔,等. 酸枣仁汤通过激活AMPK/SIRT1/PGC-1α信号通路改善阿尔茨海默病模型小鼠线粒体功能障碍[J]. 中国药理学通报,2023,39(7):1256-1262. [139] YANG WT, ZHENG XW, CHEN S, et al. Chinese herbal medicine for Alzheimer’s disease: Clinical evidence and possible mechanism of neurogenesis. Biochem Pharmacol. 2017; 141:143-155. [140] WU JJ, YANG Y, WAN Y, et al. New insights into the role and mechanisms of ginsenoside Rg1 in the management of Alzheimer’s disease. Biomed Pharmacother. 2022;152:113207. [141] 汤子钦, 卢建政, 文晓东,等. 导痰洗心汤调控AMPK/SIRT1/NF-κB信号通路蛋白表达抑制阿尔茨海默病少突胶质细胞凋亡[J]. 中国中西医结合杂志,2024,44(8):966-973. [142] 朱晓婷. 解毒益智方对阿尔茨海默病双转基因小鼠行为学及大脑皮层内β-淀粉样蛋白沉积及BACE1表达影响的研究[D]. 长春:长春中医药大学,2021. [143] 王田野, 张鹏起, 朱晓婷,等. 基于降低氧化应激损伤探究解毒益智方改善APP/PS1阿尔茨海默病小鼠神经毒性作用机制[J]. 中国老年学杂志,2022,42(6):1440-1445. [144] 周瑜, 方锐, 王国佐,等. 脑泰方对SD大鼠凝血功能的影响[J]. 中华中医药学刊,2015,33(7): 1603-1605. [145] 石咏梅, 马英民, 廖君, 等. 内质网应激PERK通路在脑泰方提取物保护局灶性脑缺血大鼠海马神经元中的作用[J]. 中国老年学杂志, 2017,37(22): 5512-5515. [146] 易亚乔, 刘检, 刘林, 等. 加味脑泰方对大鼠缺氧/复氧损伤海马神经元炎性通路SIRT1/NF-κB的影响[J]. 中国中医药信息杂志,2019,26(3): 45-50. [147] 康舒宇, 解瑶, 许田甜, 等. 基于方证代谢组学的泽泻汤治疗眩晕症的药效物质基础研究[J]. 中草药,2025,56(8):2687-2699. [148] 苏世杰, 林东新, 刘壮壮,等. 基于AMPK/SIRT1/PGC-1α通路探讨泽泻汤改善高脂饮食诱导小鼠认知障碍的作用机制[J]. 中华中医药杂志,2023,38(9):4154-4160. [149] 蔡伟武. 交泰丸改善APP/PS1小鼠的认知功能障碍及其机制研究[D]. 广州:广州中医药大学, 2021. [150] 殷聚波. 天地转气汤对阿尔茨海默病模型大鼠学习记忆能力及海马SIRT1、NF-κB、IκBα蛋白水平的影响[D]. 长沙:湖南中医药大学,2023. [151] YANG XR, WANG R, QIN HP, et al. [Neuroprotective role of silent information regulator 1 in Alzheimer’s disease]. Sheng Li Xue Bao. 2011;63(4):396-400. [152] BONDA DJ, LEE HG, CAMINS A, et al. The sirtuin pathway in ageing and Alzheimer disease: mechanistic and therapeutic considerations. Lancet Neurol. 2011;10(3):275-279. [153] ZHU C, ZHANG Z, ZHU Y, et al. Study on the role of Dihuang Yinzi in regulating the AMPK/SIRT1/PGC-1α pathway to promote mitochondrial biogenesis and improve Alzheimer’s disease. J Ethnopharmacol. 2025;337(Pt 2):118859. [154] MOUSSA C, HEBRON M, HUANG X, et al. Resveratrol regulates neuro-inflammation and induces adaptive immunity in Alzheimer’s disease. J Neuroinflammation. 2017;14(1):1. |
| [1] | 董春阳, 周天恩, 莫孟学, 吕文权, 高 明, 朱瑞凯, 高志伟. 二甲双胍联合血水草敷料治疗深Ⅱ度烧伤创面的作用机制[J]. 中国组织工程研究, 2026, 30(8): 2001-2013. |
| [2] | 杨学涛, 朱梦菡, 张宸熙, 孙一民, 叶 玲. 抗氧化纳米材料在口腔中的应用和不足[J]. 中国组织工程研究, 2026, 30(8): 2044-2053. |
| [3] | 刘安婷, 陆江涛, 张文杰, 贺 玲, 唐宗生, 陈晓玲. 血小板裂解物调控腺苷酸活化蛋白激酶抑制镉诱导的神经细胞凋亡[J]. 中国组织工程研究, 2026, 30(7): 1800-1807. |
| [4] | 李 豪, 陶红成, 曾 平, 刘金富, 丁 强, 牛驰程, 黄 凯, 康宏誉. 丝裂原活化蛋白激酶信号通路调控骨关节炎的发生发展:指导中药靶点治疗[J]. 中国组织工程研究, 2026, 30(6): 1476-1485. |
| [5] | 刘 煜, 雷森林, 周锦涛, 刘 辉, 李先辉. 有氧和抗阻运动改善肥胖相关认知障碍的作用机制[J]. 中国组织工程研究, 2026, 30(5): 1171-1183. |
| [6] | 刘新月, 李春年, 李一卓, 徐世芳. 口腔牙槽骨缺损的再生修复[J]. 中国组织工程研究, 2026, 30(5): 1247-1259. |
| [7] | 杨 肖, 白月辉, 赵甜甜, 王东昊, 赵 琛, 袁 硕. 颞下颌关节骨关节炎软骨退变:机制及再生的挑战[J]. 中国组织工程研究, 2026, 30(4): 926-935. |
| [8] | 穆秉桃, 郭敏芳, 胡芬琦, 刘淇源, 贾 辉, 徐明源, 陈佳媛, 张慧宇, 孟 涛, 尉杰忠. 雷公藤甲素缓解过氧化氢诱导SH-SY5Y细胞凋亡的线粒体动力学机制[J]. 中国组织工程研究, 2026, 30(34): 8986-8993. |
| [9] | 邹玉雄, 刘 英, 刘晓蒙, 顾 楠, 朱 玥, 朱晋台, 李书明, 敖梅英, 刘 潜, 何 媛. 儿童脑性瘫痪中药用药规律:基于病历与文献的分析[J]. 中国组织工程研究, 2026, 30(34): 9081-9087. |
| [10] | 袁晶晶, 张晓敏, 杜朋洋, 王维峰. 玛仕度肽改善APP/PS1/Tau三转基因小鼠的认知能力[J]. 中国组织工程研究, 2026, 30(34): 8962-8969. |
| [11] | 汪陈莫及, 吴亚东, 高 迪, 王 浩, 李念虎. 山柰酚调控线粒体自噬抑制大鼠椎间盘退变的作用机制[J]. 中国组织工程研究, 2026, 30(29): 7529-7538. |
| [12] | 王绍娜, 李瑞洋, 安兰花, 张金生, . 非编码RNA在阿尔茨海默病中的作用及中医药干预[J]. 中国组织工程研究, 2026, 30(29): 7663-7672. |
| [13] | 田轩赫, 仝思宇, 滕 飞, 钟 帅, 赵啸虎, 张玉娅, 刘 源, 姜 萍. 痛风的潜在靶点及药物预测:可成药基因识别[J]. 中国组织工程研究, 2026, 30(29): 7706-7714. |
| [14] | 黎征鹏, 邵微刚, 曾 浩, 向科霖, 张博涛, 邹顺一, 陈 胜, 祁 文, . 骨关节炎特征性基因及靶向药食同源中药预测:生信分析及动力学模拟鉴定[J]. 中国组织工程研究, 2026, 30(29): 7739-7748. |
| [15] | 雍 侨, 孙 鑫, 汪国友, 张 磊, 沈骅睿, 刘 欢, 关钛元. 少阳生骨方抑制氧化应激延缓膝骨关节炎大鼠软骨衰老[J]. 中国组织工程研究, 2026, 30(28): 7251-7259. |
沉默信息调节因子1(Silent information regulator 1,SIRT1)作为烟酰胺腺嘌呤二核苷酸依赖性去乙酰化酶,通过去乙酰化活性调控多种蛋白的表达,从而在阿尔茨海默病中发挥多重保护作用。研究表明,沉默信息调节因子1能够改善阿尔茨海默病中的慢性神经炎症微环境[4],促进线粒体生物合成并减少活性氧积累[5]、同时激活自噬相关蛋白[6],进而加速β-淀粉样蛋白聚集体和异常tau蛋白的降解,最终改善突触可塑性和记忆功能。基于这些发现,以沉默信息调节因子1为靶点,通过调控氧化应激、炎症反应、自噬及能量代谢等生物过程[7],可能为阿尔茨海默病的临床治疗提供新的干预策略。
近年来,中医药在防治阿尔茨海默病方面取得优异成果,大量研究表明中医药通过激活沉默信息调节因子1信号通路,显著增强神经元活性,有效抑制β-淀粉样蛋白异常聚集并调控tau蛋白磷酸化水平,实现对阿尔茨海默病的治疗作用,但尚缺乏对此的相关综述。鉴于此,此文在中国知网、万方、维普、中国生物医学文献数据库(SinoMed)、PubMed等数据库中进行文献检索,从沉默信息调节因子1入手简要介绍其基本功能,阐述沉默信息调节因子1信号通路在阿尔茨海默病发病机制中的作用,并系统总结中医药调控沉默信息调节因子1信号通路干预阿尔茨海默病的研究现状,以期为阿尔茨海默病的深入研究及临床新药开发提供思路。
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
1.1 资料来源
1.1.1 检索人及检索时间 第一作者在2025年3月进行文献检索。
1.1.2 检索文献时限 文章发表时间为2015年1月至2025年3月。
1.1.3 检索数据库 中国知网、万方、维普、中国生物医学文献数据库(SinoMed)和PubMed数据库。
1.1.4 检索词 中文检索词为“阿尔茨海默病,痴呆,炎症,沉默信息调节因子1,氧化应激,炎症因子,信号通路,中药单体,复方”;英文检索词为“Alzheimer’s disease,dementia,SIRT1,inflammation,oxidative stress,inflammatory factors,signaling pathways,TCM monomers,compounds”。
1.1.5 检索文献类型 研究原著、综述、学位论文和荟萃分析。
1.1.6 检索策略 以PubMed数据库检索策略为例,见图1。
1.2 入选标准
1.2.1 纳入标准 ①沉默信息调节因子1与阿尔茨海默病相关的文章;②中药调控沉默信息调节因子1治疗阿尔茨海默病相关的文章。
1.2.2 排除标准 ①与研究主题不相符或研究目的无关的文献;②通过Endnote 21软件查重并人工复核重复及过时的文献。
1.3 质量评估 采用PRISMA流程图规范筛选过程,经排除无关、重复、过时的文献后,共检索出4 921篇文献,其中中国知网2 892篇、PubMed 数据库2 029篇,采用改良的
Newcastle-Ottawa量表(NOS)对纳入文献进行评价,排除与文章相关性不高的文献,根据文章摘要及标题再次进行筛选,最终纳入观点新
颖、研究性强的154篇文献进行综述。文献检索流程图见图2。
3.1 该领域研究的贡献和存在的问题 Sirtuin家族蛋白在多种脑功能中发挥关键作用,包括神经发生、调节氧化应激、突触可塑性、小胶质细胞激活以及认知和情绪调节[151]。在7种亚型中,沉默信息调节因子1因在神经系统疾病中的广泛参与性,已成为阿尔茨海默病研究的重要靶点[152]。研究表明,沉默信息调节因子1激活剂通过调控线粒体功能、减少氧化应激、抑制神经炎症和促进自噬等多种机制发挥治疗阿尔茨海默病的作用[152]。目前,多项针对沉默信息调节因子1调节剂治疗阿尔茨海默病的临床试验正在进行中[153-154],这为阿尔茨海默病的临床干预提供了新的潜在策略。然而,尽管沉默信息调节因子1在阿尔茨海默病治疗中展现出广阔前景,仍存在以下问题:①目前关于中医药调控沉默信息调节因子1信号通路干预阿尔茨海默病的研究多基于细胞和动物实验,临床转化证据不足,缺乏统一的证候评价标准;②机制研究多采用Western blot等传统技术,缺乏单细胞测序、空间转录组等时空分辨技术的应用;③中医药多靶点、整体调控的特点导致其机制研究面临技术复杂性,现有实验模型可能难以全面模拟人体内环境。
3.2 该综述区别于他人他篇的特点 该综述以沉默信息调节因子1为切入点,系统总结了沉默信息调节因子1的生物学功能及其与阿尔茨海默病病理生理的关联,全面阐述了近年来沉默信息调节因子1激活剂通过多机制干预阿尔茨海默病的研究进展,并对其临床应用前景进行了展望。
3.3 该综述的局限性 该综述的局限性主要体现在以下几个方面:①尽管文章系统总结了沉默信息调节因子1在阿尔茨海默病中的作用及中医药的调控机制,但受限于文献检索范围,部分相关研究可能未被纳入;②目前关于中医药调控沉默信息调节因子1信号通路干预阿尔茨海默病的研究多基于细胞和动物实验,临床转化证据不足,缺乏大样本随机对照试验的支持;③在分析中药复方及单体成分的作用时,对个体差异、药物配伍相互作用及中医证型异质性的考量仍需加强。
3.4 临床转化潜力 基于此文的研究提出从基础研究到临床应用的系统框架,但仍存在几个关键问题:首先,各阶段间的转化标准不够明确,缺乏量化的评估指标来指导研究进程;其次,临床前阶段采用的诱导多能性干细胞类器官模型难以完全模拟阿尔茨海默病复杂的病理微环境,可能影响药物筛选的准确性;再者,临床阶段的适应性设计虽能提高效率,但未充分考虑阿尔茨海默病患者异质性对试验结果的影响;最后,产业化阶段面临中药复方质量控制困难、知识产权保护以及剂型优化等实际挑战,缺乏具体的解决方案。这些局限性可能影响整个转化过程的顺利实施和最终成果的可靠性。
3.5 综述的重要意义 阿尔茨海默病是威胁全球老年人群健康的重大神经退行性疾病,沉默信息调节因子1作为调控线粒体功能、氧化应激和神经炎症的关键靶点,其激活为阿尔茨海默病治疗提供了新思路。此次综述阐明了中医药通过沉默信息调节因子1及其下游通路(如抑制核因子κB、叉头框蛋白O、过氧化物酶体增殖体激活受体γ共激活因子1α等)多维度干预阿尔茨海默病的潜力,凸显了中医药整体调节和耐药性低的优势。这一研究不仅为阿尔茨海默病的基础机制探索补充了新的理论依据,也为开发基于沉默信息调节因子1的中药创新疗法奠定了科学基础。
综上所述,尽管中医药通过多靶点调控沉默信息调节因子1信号通路在阿尔茨海默病治疗中展现出独特优势,但目前研究仍存在以下关键问题:首先,现有研究多集中于单一靶点,对沉默信息调节因子1与其他信号通路(如AMP活化蛋白激酶/哺乳动物雷帕霉素靶蛋白、脑源性神经营养因子/原肌球蛋白受体激酶B)的交互作用机制研究不足;其次,阿尔茨海默病中神经元死亡形式复杂,沉默信息调节因子1与其他程序性细胞死亡方式(如凋亡、坏死性凋亡)的关联性尚未阐明;第三,约85%的研究仍停留在细胞和动物实验阶段,临床转化证据薄弱,特别是缺乏针对不同中医证型(如肾虚证、痰瘀证)的精准临床研究;最后,现有中药单体研究难以体现中医整体观,而复方研究又面临成分复杂、质量控制困难等挑战。针对这些问题,未来研究应着重从以下方面突破:①综合运用网络药理学、分子对接、高通量质谱分析等现代科学技术,从基因-转录-蛋白-代谢不同角度对具体的药物作用机制及有效靶点进行研究;②建立基于中医证候的类器官模型,阐明沉默信息调节因子1在不同证型阿尔茨海默病中的动态变化规律;③以中医理论为基础,开展高质量的临床研究,将体内外实验与临床相结合,全方位、多层次探讨中药调控沉默信息调节因子1信号通路治疗阿尔茨海默病的机制及疗效,从而为中药靶向应用提供理论基础,为治疗的安全性及有效性提供有利的证据。
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
该综述以沉默信息调节因子1为切入点,系统总结了沉默信息调节因子1的生物学功能及其与阿尔茨海默病病理生理的关联,全面阐述了近年来沉默信息调节因子1激活剂通过多机制干预阿尔茨海默病的研究进展,并对其临床应用前景进行了展望。#br# 该综述的局限性主要体现在以下几个方面:①尽管文章系统总结了沉默信息调节因子1在阿尔茨海默病中的作用及中医药的调控机制,但受限于文献检索范围,部分相关研究可能未被纳入;②目前关于中医药调控沉默信息调节因子1信号通路干预阿尔茨海默病的研究多基于细胞和动物实验,临床转化证据不足,缺乏大样本随机对照试验的支持;③在分析中药复方及单体成分的作用时,对个体差异、药物配伍相互作用及中医证型异质性的考量仍需加强。
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||