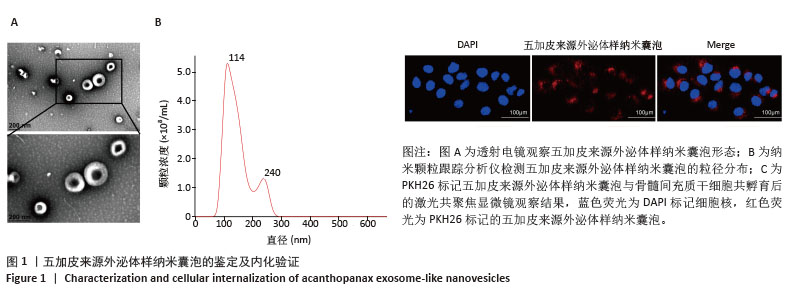
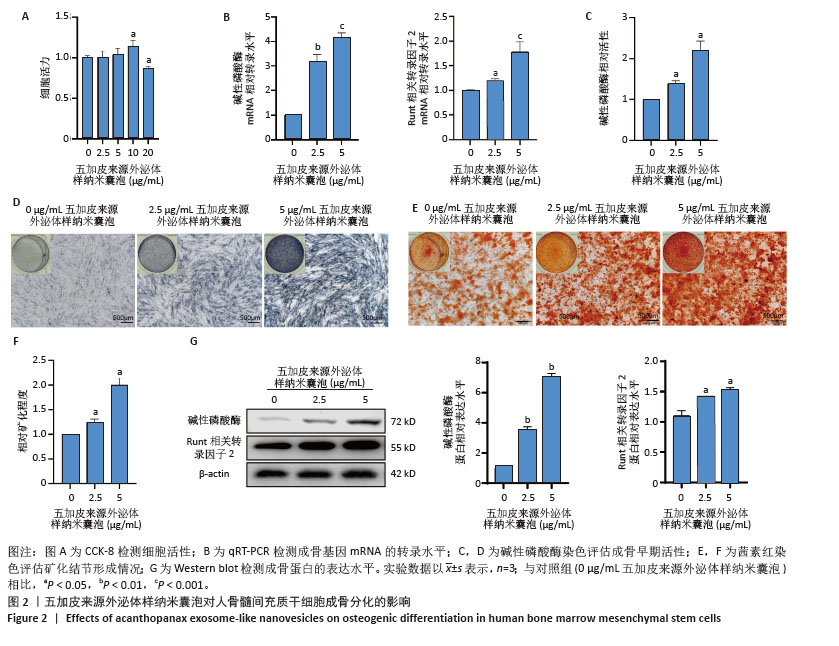
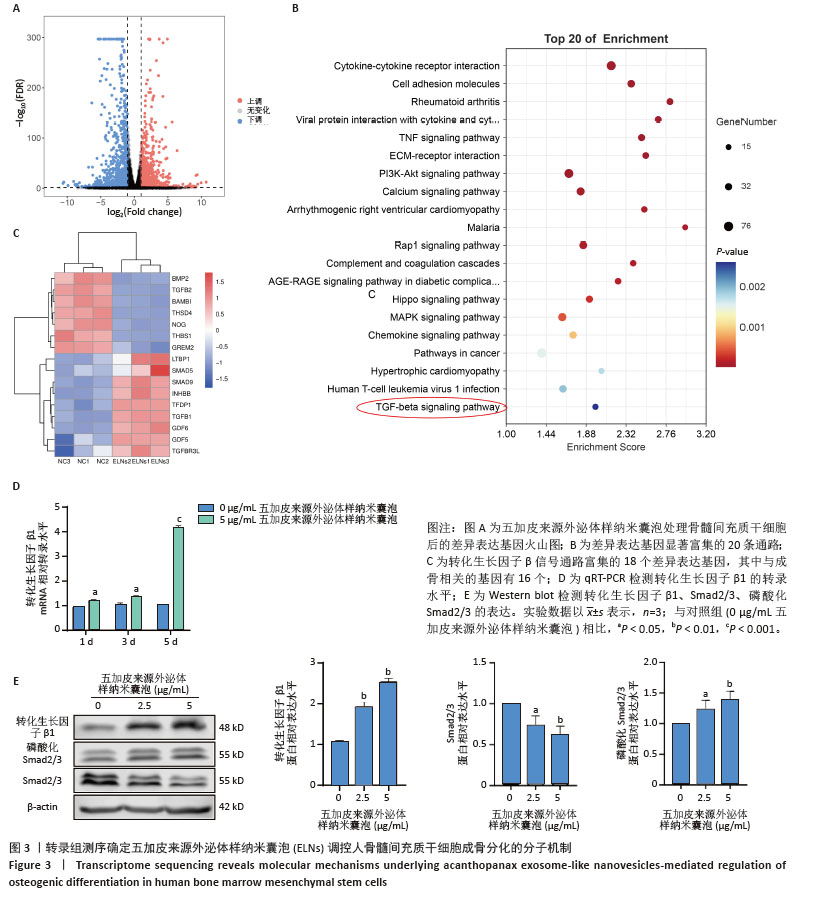
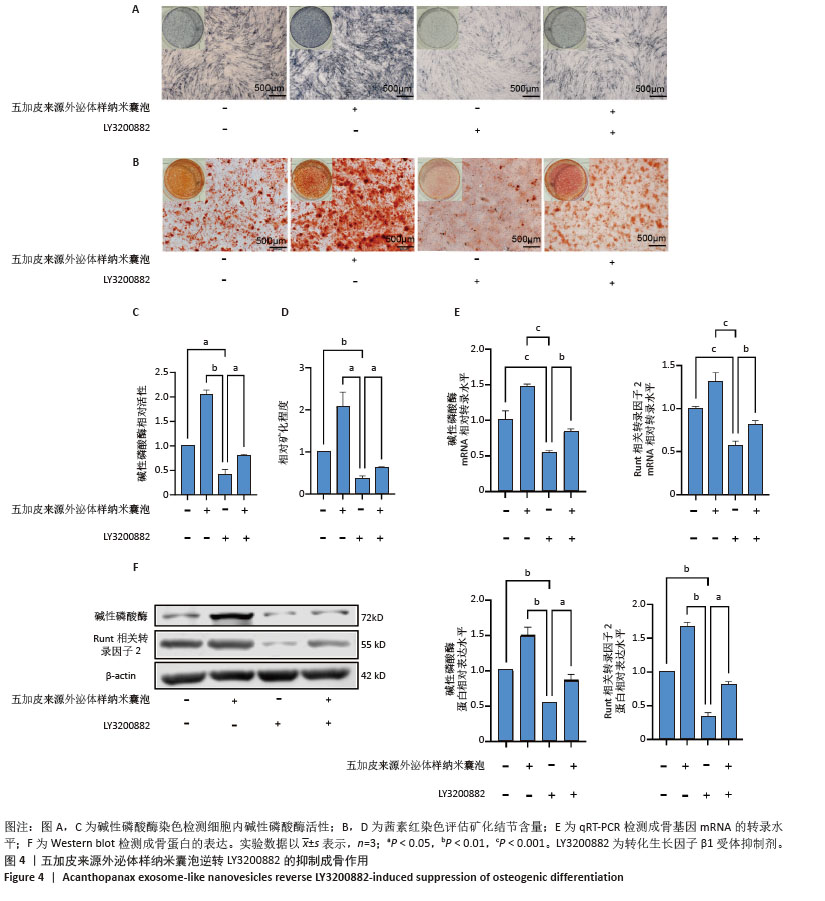
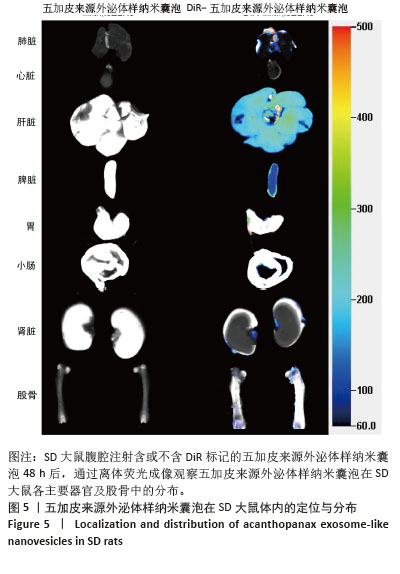
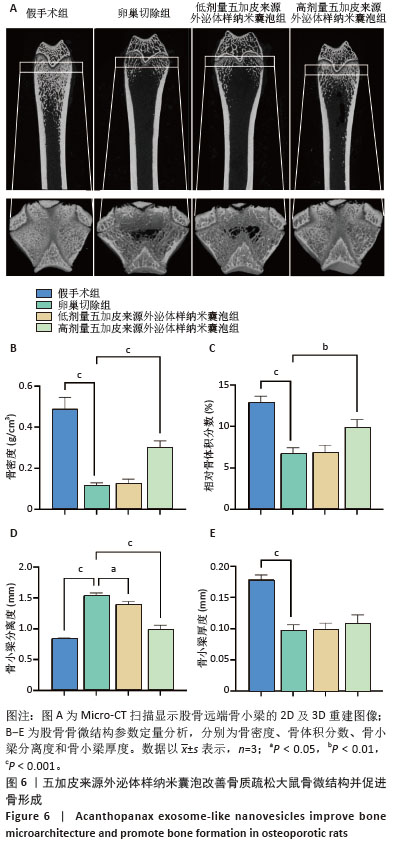
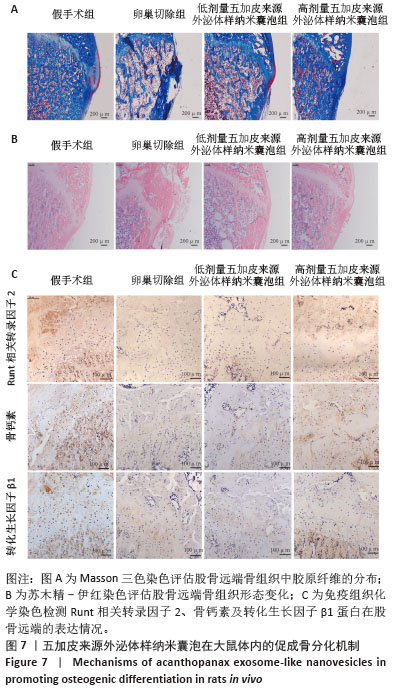
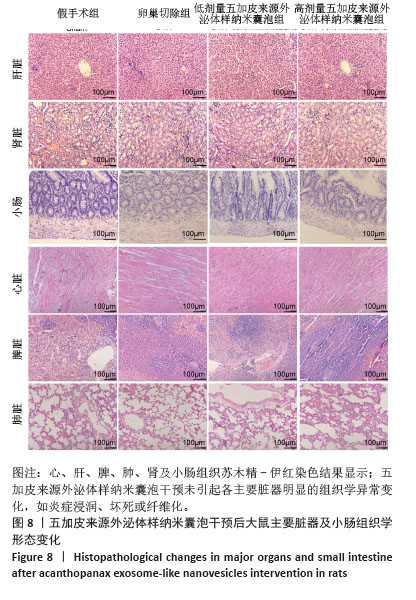
[1] WANG J, SHU B, TANG DZ, et al. The prevalence of osteoporosis in China: a community-based cohort study. Front Public Health. 2023;11:1084005.
[2] 邹顺一,易进,曾浩,等.绝经后骨质疏松症:相关信号通路的可视化分析[J].中国组织工程研究,2026,30(16):4229-4239.
[3] LEBOFF MS, GREENSPAN SL, INSOGNA KL, et al. The clinician’s guide to prevention and treatment of osteoporosis. Osteoporos Int. 2022;33(10):2049-2102.
[4] AIBAR-ALMAZÁN A, VOLTES-MARTÍNEZ A, CASTELLOTE-CABALLERO Y, et al. Current status of the diagnosis and management of osteoporosis. Int J Mol Sci. 2022;23(16):9465.
[5] 王瑞.选择性雌激素受体调节剂治疗绝经后女性骨质疏松症的效果及骨代谢指标研究[J].医学理论与实践,2025,38(15):2603-2605.
[6] IMAMUDEEN N, BASHEER A, IQBAL AM, et al. Management of osteoporosis and spinal fractures: contemporary guidelines and evolving paradigms. Clin Med Res. 2022;20(2):95-106.
[7] 王露,刘伟兵,钟嘉伟,等.骨质疏松症药物治疗的现状和研究进展[J].中国现代医生,2024,62(27):124-128.
[8] ENSRUD KE, CRANDALL CJ. Osteoporosis. Ann Intern Med. 2024;177(1):ITC1-ITC6.
[9] REID IR, BILLINGTON EO. Drug therapy for osteoporosis in older adults. Lancet. 2022;399(10329):1080-1092.
[10] XU H, XU J, CHEN F, et al. Acanthopanax senticosus aqueous extract ameliorates ovariectomy-induced bone loss by inhibiting NF-κB ligand-induced osteoclastogenesis. Food Funct. 2020;11(11):9696-9709.
[11] HWANG YC, JEONG IK, AHN KJ, et al. Effects of Acanthopanax senticosus extract on bone turnover and bone mineral density in postmenopausal women. J Bone Miner Metab. 2009;27(5):584-590.
[12] BOHN B, NEBE CT, BIRR C. Durchflußzytometrische Untersuchungen auf immunmodulatorische Wirkungen von Eleutherococcus senticosus-Extrakt. Arzneim Forsch. 1987;37:1193-1196.
[13] LIM DW, KIM JG, LEE Y, et al. Preventive effects of Eleutherococcus senticosus bark extract in OVX-induced osteoporosis. Molecules. 2013;18(7):7998-8008.
[14] LIU J, ZHANG Z, GUO Q, et al. Syringin prevents bone loss via TRAF6-mediated inhibition of NF-κB and activation of PI3K/AKT. Phytomedicine. 2018;42:43-50.
[15] JIN Z, NA J, LIN X, et al. Plant-derived exosome-like nanovesicles: a novel nanotool for disease therapy. Heliyon. 2024;10(9):e30630.
[16] 韩菲,马小梅,石旭柳,等.柑橘属植物来源的外泌体样纳米颗粒及其疾病治疗研究进展[J].中草药,2024,55(19):6768-6778.
[17] FENG H, YUE Y, ZHANG Y, et al. Plant-derived exosome-like nanoparticles: emerging nanosystems for enhanced tissue engineering. Int J Nanomedicine. 2024;19:1189-1204.
[18] 秦启顺,王兴盛,徐世红,等.中药及其活性成分诱导骨髓间充质干细胞成骨分化相关信号通路的研究进展[J].生物医学工程与临床,2024,28(6): 884-892.
[19] 药文鑫,王毅,蔡林,等.植物源外泌体样纳米囊泡刺激成骨细胞、抑制破骨细胞并促进成骨[J].中国组织工程研究,2025,29(31):6765-6771.
[20] HWANG JH, PARK YS, KIM HS, et al. Yam-derived exosome-like nanovesicles stimulate osteoblast formation and prevent osteoporosis in mice. J Control Release. 2023;355:184-198.
[21] ZHAN W, DENG M, HUANG X, et al. Pueraria lobata-derived exosome-like nanovesicles alleviate osteoporosis by enhancing autophagy. J Control Release. 2023;364:644-653.
[22] ZHANG X, WANG G, WANG W, et al. Bone marrow mesenchymal stem cells paracrine TGF-β1 to mediate osteoblast activity in bone repair. Cytokine. 2023; 164:156139.
[23] YIN B, YANG M, WANG B, et al. Total flavonoids isolated from Eucommia ulmoides can alleviate bone loss and regulate intestinal microbiota in ovariectomized rats. Front Pharmacol. 2025;16:1513863.
[24] WAN Y, HU C, HOU Y, et al. OPG gene-modified adipose-derived stem cells improve bone formation around implants in osteoporotic rat maxillae. Heliyon. 2023;9(10):e19474.
[25] 王丹,邵子芹,王雅雯,等.巴戟天-丹参对去卵巢大鼠骨质疏松影响的研究[J].中国骨质疏松杂志,2024,30(12):1751-1756.
[26] 李敏,扎克艳•地力夏提,胡君萍,等.酒花提取物调控Wnt/β-catenin信号通路对去卵巢骨质疏松大鼠的作用及机制研究[J].中药新药与临床药理,2024,35(9):1337-1343.
[27] YAKYMOVYCH I, YAKYMOVYCH M, HAMIDI A, et al. The type II TGF-β receptor phosphorylates Tyr182 in the type I receptor to activate downstream Src signaling. Sci Signal. 2022;15(760):eabp9521.
[28] LIU H, CHEN YG. The interplay between TGF-β signaling and cell metabolism. Front Cell Dev Biol. 2022;10:846723.
[29] FREGNANI A, SAGGIN L, GIANESIN K, et al. CK1α/RUNX2 axis in the bone marrow microenvironment: a novel therapeutic target in multiple myeloma. Cancers (Basel). 2022;14(17):4173.
[30] LIU Y, XIONG W, LI J, et al. Application of dental pulp stem cells for bone regeneration. Front Med (Lausanne). 2024;11:1339573.
[31] GAO M, ZHANG Z, SUN J, et al. The roles of circRNA-miRNA-mRNA networks in the development and treatment of osteoporosis. Front Endocrinol (Lausanne). 2022;13:945310.
[32] NAM B, PARK H, LEE YL, et al. TGFβ1 suppressed matrix mineralization of osteoblasts differentiation by regulating SMURF1-C/EBPβ-DKK1 axis. Int J Mol Sci. 2020;21:9771.
[33] ZHANG XF, WANG ZX, ZHANG BW, et al. TGF-β1-triggered BMI1 and SMAD2 cooperatively regulate miR-191 to modulate bone formation. Mol Ther Nucleic Acids. 2024;35(2):102164.
[34] LU X, LI W, WANG H, et al. The role of the Smad2/3/4 signaling pathway in osteogenic differentiation regulation by ClC-3 chloride channels in MC3T3-E1 cells. J Orthop Surg Res. 2022;17(1):338.
[35] LAMORA A, TALBOT J, MULLARD M, et al. TGF-β signaling in bone remodeling and osteosarcoma progression. J Clin Med. 2016;5(11):96.
[36] CHEN Y, MEI L, QIAN Y, et al. Integrated bioinformatic analysis of protein landscape in gingival crevicular fluid unveils sequential bioprocess in orthodontic tooth movement. Prog Orthod. 2024;25(1):37.
[37] INOUE M, NAGAI-YOSHIOKA Y, YAMASAKI R, et al. Mechanisms involved in suppression of osteoclast supportive activity by TGF-β1 via the ubiquitin-proteasome system. PLoS One. 2022;17(2):e0262612.
[38] WU Z, LI W, JIANG K, et al. Regulation of bone homeostasis: signaling pathways and therapeutic targets. MedComm (2020). 2024;5(8):e657.
[39] XIA Y, INOUE K, DU Y, et al. TGFβ reprograms TNF stimulation of macrophages towards a non-canonical pathway driving inflammatory osteoclastogenesis. Nat Commun. 2022;13(1):3920.
[40] 朱文潇, 王向阳, 刘永辉, 等.平乐三七接骨丸对桡骨骨折兔模型转化生长因子-β/骨形态发生蛋白信号通路表达的影响[J]. 陕西中医,2022,43(9): 1175-1179.
[41] XIA C, GE Q, FANG L, et al. TGF-β/Smad2 signalling regulates enchondral bone formation of Gli1+ periosteal cells during fracture healing. Cell Prolif. 2020;53(11):e12904.
[42] GELIBTER S, MAROSTICA G, MANDELLI A, et al. The impact of storage on extracellular vesicles: a systematic study. J Extracell Vesicles. 2022;11(2):e12162. |