中国组织工程研究 ›› 2015, Vol. 19 ›› Issue (42): 6746-6752.doi: 10.3969/j.issn.2095-4344.2015.42.005
• 皮肤粘膜组织构建 skin and mucosal tissue construction • 上一篇 下一篇
利用载有角质细胞生长因子微囊的组织工程皮肤修复裸鼠皮肤缺损
李秉航1,2,邓立欢1,向萌娟1,杨 斌1,2
- 中国医学科学院北京协和医学院整形外科医院,1数字化模拟中心,2颌面整形外科中心,北京市 100144
-
出版日期:2015-10-08发布日期:2015-10-08 -
通讯作者:杨斌,博士,教授,主任医师,博士生导师,中国医学科学院整形外科医院数字化模拟中心,颌面整形外科中心;北京市 100144。 -
作者简介:李秉航,1990年生,山西省临汾市人,汉族,北京协和医学院在读硕士,主要从事组织工程与再生医学方面的研究。 -
基金资助:国家自然科学基金(30772099);北京市自然科学基金(7112111)
Repairing skin defects in nude mice using tissue-engineered skin loading keratinocyte growth factor nanocapsules
Li Bing-hang1, 2, Deng Li-huan1, Xiang Meng-juan1, Yang Bin1, 2
- 1 Digital Simulation Center, Plastic Surgery Hospital, Chinese Academy of Medical Science and Peking Union Medical College, Beijing 100144, China
2 Department of Maxillofacial Surgery, Plastic Surgery Hospital, Chinese Academy of Medical Science and Peking Union Medical College, Beijing 100144, China
-
Online:2015-10-08Published:2015-10-08 -
Contact:Yang Bin, M.D., Professor, Chief physician, Doctoral supervisor, Digital Simulation Center, Plastic Surgery Hospital, Chinese Academy of Medical Science and Peking Union Medical College, Beijing 100144, China; Department of Maxillofacial Surgery, Plastic Surgery Hospital, Chinese Academy of Medical Science and Peking Union Medical College, Beijing 100144, China -
About author:Li Bing-hang, Studying for master’s degree, Digital Simulation Center, Plastic Surgery Hospital, Chinese Academy of Medical Science and Peking Union Medical College, Beijing 100144, China; Department of Maxillofacial Surgery, Plastic Surgery Hospital, Chinese Academy of Medical Science and Peking Union Medical College, Beijing 100144, China -
Supported by:the National Natural Science Foundation of China, No. 30772099; the Natural Science Foundation of Beijing, No. 7112111
摘要:
背景:组织工程皮肤作为一项新兴技术拥有良好的应用前景。有研究表明,角质细胞生长因子可以促进表皮细胞增殖。 目的:观察荷载角质细胞生长因子纳米微囊的新型组织工程皮肤修复裸鼠皮肤缺损的效果和特点。 方法:构建荷载角质细胞生长因子的脱细胞真皮基质复合物;将人表皮干细胞群和成纤维细胞分离、培养,并且进行鉴定;将表皮干细胞群接种于复合物之上,观察其生长状况;将荷载角质细胞生长因子纳米微囊的组织工程皮肤移植于裸鼠皮肤缺损处,将无角质细胞生长因子纳米微囊的组织工程皮肤作为空白组,将其自体皮肤移植修复缺损组作为对照组,于移植后2,4,6周时观察皮片挛缩及组织学愈合情况,并应用抗人角蛋白10及β1-整合素免疫荧光检测修复区表皮和真皮层细胞来源、分化及生长情况。 结果与结论:表皮干细胞在复合物表面生长良好,黏贴紧密,可见有连接成片趋势的小圆形的表皮干细胞及多角形的终末表皮细胞,部分形成克隆团块。移植后第2,4,6周,荷载角质细胞生长因子纳米微囊组织工程皮肤修复裸鼠皮肤缺损的结果均优于空白组及对照组,移植的皮肤边缘与邻近皮肤完全融合,但存在一定程度的挛缩。修复区组织工程皮肤的表皮细胞分层良好并能产生角质层,同时,移植后8,10周,组织工程皮肤切片免疫荧光染色可以鉴别出少量β1-整合素阳性细胞,均为表皮干细胞或短暂扩充细胞。结果证实,荷载角质细胞生长因子纳米胶囊的新型组织工程皮肤修复裸鼠皮肤缺损的效果较好,优于普通组织工程皮肤及自体全厚皮片移植修复。 中国组织工程研究杂志出版内容重点:组织构建;骨细胞;软骨细胞;细胞培养;成纤维细胞;血管内皮细胞;骨质疏松;组织工程
引用本文
李秉航,邓立欢,向萌娟,杨 斌. 利用载有角质细胞生长因子微囊的组织工程皮肤修复裸鼠皮肤缺损[J]. 中国组织工程研究, 2015, 19(42): 6746-6752.
Li Bing-hang, Deng Li-huan, Xiang Meng-juan, Yang Bin. Repairing skin defects in nude mice using tissue-engineered skin loading keratinocyte growth factor nanocapsules[J]. Chinese Journal of Tissue Engineering Research, 2015, 19(42): 6746-6752.
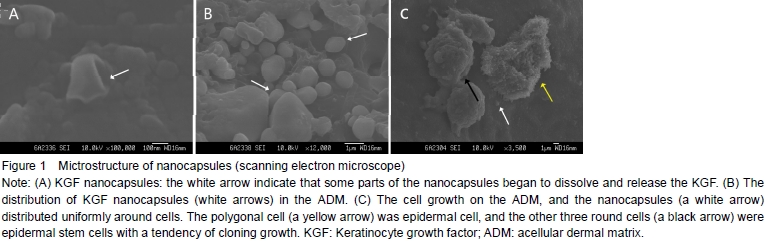
Morphology of KGF nanocapsules and their distribution on the KGF-ADMUnder the scanning electron microscope, the nanocapsules had good appearance with smooth surfaces. They distributed uniformly and adhered to the ADM tightly. The majority of them located on the surface of ADM and a few located deeply (Figures 1A, B).
Growth of epidermal stem cells on the KGF-ADMEpidermal stem cells grew well on the KGF-ADM and exhibited various shapes. There were small round and clumped distributed stem cells and polygonal terminated differentiated cells. They grew well and appeared to have a partly cloning growth or a tendency of merging into pieces. The cells adhered to the KGF-ADM tightly and the nanocapsules also scattered on the KGF-ADM (Figure 1C).

Immunofluorescence of epidermal stem cell populationThe seed cells contained epidermal stem cells which were 17% positive. All nucleuses that showed uniformly blue fluorescence were stained with Hoechst 33342, but most of the adhered cells also had red fluorescence from cell membrane. Some of them had small volume, high nucleoplasm proportion, clumped distribution and higher fluorescent brightness, indicating that they were epidermal stem cells (Figure 2).
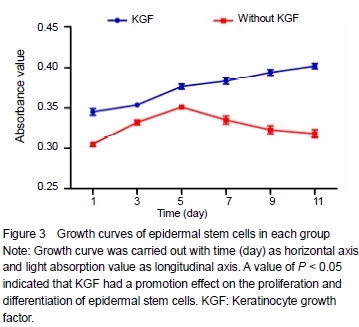
Growth curve of epidermal stem cells cultivated in vitro Drawing growth curve was carried out with time (day) as horizontal axis and absorbance value as longitudinal axis. SPSS 11.0 software was used to complete one-way analysis of variance for the data of the growth curve. A value of P < 0.05 indicated that KGF had a promotion effect on the proliferation and differentiation of epidermal stem cells (Figure 3).

Gross and hematoxylin-eosin observation of the skin in repairing area Two weeks after transplantation, the repairing area of experimental group and blank group both had healed basically without obvious difference, but the skin tissue in the repairing area did not healed yet in the control group and there was some obvious contraction. Skin sections observed under the microscope all had inflammatory cells gathered and significant inflammation reaction in the three groups. Four weeks later, the skin grafts showed contraction in each group, especially in the control group. The grafts were softer than those at 2 weeks and the color was close to the surrounding skin tissues. Under the microscope, the number of cell layers in the experimental group and blank group was similar, corneous surface had formed and dermis became thicker. In the dermis, fibroblasts could been seen, collagen fibers were arranged orderly and inflammatory cells and small blood vessels had decreased, but the cells of epidermal layer in the blank group were connected loosely with the basis and some of them fell off. In the control group, the epidermal layer cells were connected tightly with the neighborhood and the thickness of dermis was close to the normal tissue. After 6 weeks, the color of the grafts in all groups had no difference from the surrounding tissues, and in the experimental group, the skin had less contraction compared with the control group. Laboratory observation showed that the number of epidermal cells in the experimental and blank group were more close with normal skin tissue which contained the stratum basale, prickle cell layer, granular layer and stratum corneum. There were more fibroblasts than before and collagen fibers arranged much orderly. Epidermal cells were connected with the basis more tightly and the normal cutin was seen. The number of epidermal cell layers in the control group was less than that in the experimental group, and the skin graft in the control group had an obvious contraction (Figure 4).
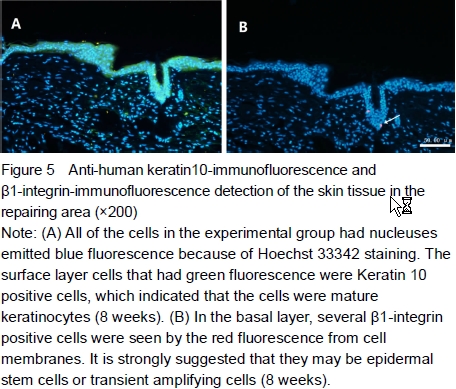
K10-immunofluorescence and β1-integrin-immunofluorescence detection of the skin tissue in the repairing areaAt 8 weeks after transplantation, the skin tissue in the repairing area of experimental group was incised for anti-human K10-FITC-immunofluorescence and β1-integrin Cy3-immunofluorescence detection. The skin tissues had good layers and expressed normal keratin which emitted uniform green fluorescence. In some of the sections, a few of β1-integrin positive cells were seen which probably epidermal stem cells or transient amplifying cells (Figure 5).
| [1] Yang B, Hong QQ, Qiu RS, et al. Fast reconstruction of compostite tissue-engineered skin in vitro. Zhongguo Xiufu Chongjian Waike Zazhi. 2007;21:406-410.
[2] Cao YL, Cai X, Cui L, et al. Repair of porcine full-thickness skin defects with autologous tissue engineered skin. Zhonghua Waike Zazhi. 2002;40:24-26.
[3] Yang GH, Cui L, Liu W, et al. Repairment of full-thickness wounds utilizing tissue engineering techniques. Zhonghua Shiyan Waike Zazhi. 2003;20:408-410.
[4] Eaglstein WH, Alvares OM, Auletta M, et al. Acute ecxisional wounds treated with a tissue-engineered skin (Apligraf). Dermatol Surg. 1999;25:195.
[5] Cotsarelis G, Kaur P, Dhouailly D, et al. Epithelial stem cells in the skin: definition , markers, localization and functions. Exp Dermatol. 1999;8:80-88.
[6] Morasso I, Marjana TC. Epidermal stem cells: the cradle of epidermal determination, differentiation and wound healing, Biol Cell. 2005;97(3):173-183.
[7] Michel M, Torok N, Godbout MJ, et al. Keratin 19 as a biochemical marker of skin stem cells in vivo and in vitro: keratin 19 expressing cells are differentially localized in function of anatomic sites, and their number varies with donor age and culture stage. J Cell Sci. 1996;109: 1017-1028.
[8] Raghavan S, Bauer C, Mundschau G, et al. Conditional ablation of beta1 integrin in skin. Severe defects in epidermal proliferation, basement membrane formation, and hair follicle invagination. J Cell Biol. 2000;150(5): 1149-1160.
[9] Kaur P, Li A. Adhesive properties of human basal epidermal cells: an analysis of keratinocyte stem cells, transit amplifying cells, and postmitotic differentiating cells. J Invest Dermatol. 2000;114:413-420.
[10] Yang B, Hong QQ, Xu L, et al. A Study on rapid and efficient separation of epidermal stem cell. Zuzhi Gongcheng yu Chongjian Waike Zazhi. 2005;1(5):268-271.
[11] Yang B, Hong QQ, Xu L, et al. Fast harvest of seed cells for composite tissue engineered skin at one time. Zhongguo Xiufu Chongjian Waike Zazhi. 2006;20(7):754-757.
[12] Andreadis ST, Hamoen KE, Yarmush ML, et al. Keratinocyte growth factor induces hyperproliferation and delays differentiation in a skin equivalent model system. FASEB J. 2001;15:898-906.
[13] Pan ZZ, Devaux Y, Ray P. Ribosomal S6 kinase as a mediator of KGF induced activation of Akt in epithelial cells. Molecul Biol Cell. 2004;15:3106-3113.
[14] Kopp J, Guang YW, Peter K, et al. Accelerated wound healing by in vivo application of keratinocytes overexpressing KGF. Mol Ther. 2004;10(1):86-96.
[15] Farrell CL, Rex KL, Chen JN, et al. The effects of keratinocyte growth factor in preclinical models of mucositis. Cell Prolif. 2002;35(Suppl 1):78-85.
[16] Melissa J, Mc G, Sun D, et al. Epidermal growth factor-induced cell proliferation in the dult rat striatum. Brain Res. 2004;1007:29-38.
[17] Yang B, Qiu RS, Quan DP, et al. Construction and characteristics of acellular dermal matrix loaded with keratinocyte growth factor nanocapsules and its effects on the growth of epidermal stem cells. Zhongguo Zuzhi Gongcheng Yanjiun yu Linchuang Kangfu. 2007;11(18): 3551-3554.
[18] Kopp J, Wang GY, Kulmburg P, et al. Accelerated wound healing by in vivo application of keratinocytes overexpressing KGF. Mol Ther. 2004;10(1):86-96.
[19] Witte RP, Kao WJ. Keratinocyte-fibroblast paracrine interaction: the effects of substrate and culture condition. Biomaterials. 2005;26(17):3673-3682.
[20] Schwarz C, Mehnert W. Solid lipid nanoparticles (SLN) for controlled drug delivery. II. Drug incorporation and physicochemical characterization. J Microencapsul 1999; 16(2):205-213.
[21] Song CX. Formulation and characterization of biodegradable nanoparticles for intravascular local drug delivery. J Control Release. 1997;43:197-212.
[22] Wainwright DJ. Use of an acellulor allogroft dermal matrix (Alloderm) in the management of full-thickness burns. Burns. 1995;21(4):243.
[23] Rnnekampff HO. Acellular human dermis promotes cultured keratinote engraftment. J Burn Care Rehabil. 1997;18(6):535.
[24] Watt FM. Epidermal stem cells: markers, patterning and the control of stem cell fate. Philos Trans R Soc Lond B Biol Sci. 1998;353(1370):831-837.
[25] Bader RA, Kao WJ. Modulation of the keratinocyte-fibroblast paracrine relationship with gelatin-based semi-interpenetrating networks containing bioactive factors for wound repair.J Biomater Sci Polym Ed. 2009;20(7-8):1005-1030.
[26] Ceccarelli S, Cardinali G, Aspite N, et al. Cortactin involvement in the keratinocyte growth factor and fibroblast growth factor 10 promotion of migration and cortical actin assembly in human keratinocytes. Exp Cell Res. 2007;313(9):1758-77.
[27] Marti GP, Mohebi P, Liu L, et al. KGF-1 for wound healing in animal models. Methods Mol Biol. 2008;423:383-391. |
| [1] | 张同同, 王中华, 文 杰, 宋玉鑫, 刘 林. 3D打印模型在颈椎肿瘤手术切除与重建中的应用[J]. 中国组织工程研究, 2021, 25(9): 1335-1339. |
| [2] | 曾燕华, 郝延磊. 许旺细胞体外培养及纯化的系统性综述[J]. 中国组织工程研究, 2021, 25(7): 1135-1141. |
| [3] | 徐东紫, 张 婷, 欧阳昭连. 心脏组织工程领域全球专利竞争态势分析[J]. 中国组织工程研究, 2021, 25(5): 807-812. |
| [4] | 吴子健, 胡昭端, 谢有琼, 王 峰, 李 佳, 李柏村, 蔡国伟, 彭 锐. 3D打印技术与骨组织工程研究文献计量及研究热点可视化分析[J]. 中国组织工程研究, 2021, 25(4): 564-569. |
| [5] | 常文辽, 赵 杰, 孙晓亮, 王 锟, 吴国锋, 周 剑, 李树祥, 孙 晗. 人工骨膜的材料选择、理论设计及生物仿生功能[J]. 中国组织工程研究, 2021, 25(4): 600-606. |
| [6] | 刘 旒, 周箐竹, 龚 桌, 刘博言, 杨 斌, 赵 娴. 胶原/无机材料构建组织工程骨的特点及制造技术[J]. 中国组织工程研究, 2021, 25(4): 607-613. |
| [7] | 刘 飞, 崔宇韬, 刘 贺. 局部抗生素递送系统治疗骨髓炎的优势与问题[J]. 中国组织工程研究, 2021, 25(4): 614-620. |
| [8] | 李晓壮, 段 浩, 王伟舟, 唐志宏, 王旸昊, 何 飞. 骨组织工程材料治疗骨缺损疾病在体内实验中的应用[J]. 中国组织工程研究, 2021, 25(4): 626-631. |
| [9] | 张振坤, 李 喆, 李 亚, 王莹莹, 王亚苹, 周馨魁, 马珊珊, 关方霞. 海藻酸盐基水凝胶/敷料在创面愈合中的应用:持续、动态与顺序释放[J]. 中国组织工程研究, 2021, 25(4): 638-643. |
| [10] | 陈佳娜, 邱燕玲, 聂敏海, 刘旭倩. 组织工程支架材料修复口腔颌面部软组织缺损[J]. 中国组织工程研究, 2021, 25(4): 644-650. |
| [11] | 邢 浩, 张永红, 王 栋. 长骨大段骨缺损修复方法的优势与不足[J]. 中国组织工程研究, 2021, 25(3): 426-430. |
| [12] | 王 皓, 陈明学, 李俊康, 罗旭江, 彭礼庆, 李 获, 黄 波, 田广招, 刘舒云, 眭 翔, 黄靖香, 郭全义, 鲁晓波. 脱细胞猪皮基质构建组织工程半月板支架[J]. 中国组织工程研究, 2021, 25(22): 3473-3478. |
| [13] | 莫剑玲, 何少茹, 冯博文, 简敏桥, 张晓晖, 刘财盛, 梁一晶, 刘玉梅, 陈 亮, 周海榆, 刘艳辉. 构建预血管化细胞膜片及血管形成相关因子的表达[J]. 中国组织工程研究, 2021, 25(22): 3479-3486. |
| [14] | 刘 畅, 李大同, 刘 元, 孔令擘, 郭 瑞, 杨利学, 郝定均, 贺宝荣. 急性症状性骨质疏松性胸腰椎压缩骨折椎体强化手术后疗效欠佳:与骨水泥、骨密度、邻近骨折的关系[J]. 中国组织工程研究, 2021, 25(22): 3510-3516. |
| [15] | 刘利永, 周 雷. 组织工程用水凝胶研发现状和发展趋势:基于专利信息的分析[J]. 中国组织工程研究, 2021, 25(22): 3527-3533. |
on tissue-engineered skin focus on tissue-engineered skin composite which choose autologous cells as seed cells and can be constructed in a short time. Studies on epidermal stem cells which serve as the seed cells are increasing, and the biological characteristics, markers[5-9], isolation and culture methods[1, 10-11] are been understanding deeply. On the other hand, a lot of growth factors in the skin tissue play a complicated regulatory role in skin repair and cell proliferation or differentiation, among which, keratinocyte growth factor (KGF) has the closest relation with epidermal stem cells[12-13]. It can promote proliferation of epidermal stem cells and delay them to differentiate into final terminal cells [14-16]. Therefore, in our research, we constructed a new kind of tissue-engineered skin which choose acellular dermal matrix containing KGF nanocapsules as vector and epidermal stem cells as seed cells. The stem cells can proliferate which are affected persistently by KGF nanocapsules in a biological release system. Here, we used this new kind of tissue-engineered skin to repair full-thickness skin defects on the nude mice and observed the healing effect.
Design
A randomized controlled animal experiment.
Time and setting
The experiments were performed at the Research Center of Plastic Surgery Institute of
Materials
Fifteen nude mice (6-8 weeks old, female, 15
Foreskin samples were collected from outpatient operating room of the Plastic Surgery Hospital of Chinese
Poly (lactic-co-glycolic acid) (PLGA) was synthetized in the Macromolecular Institute of
Methods
Construction of KGF-ADM
The KGF-ADM was prepared with the method of phacoemulsification-solvent evaporation-low temperature drying and collagenase combined with trypsin digestion according to the report from Yang et al [17]. The nanocapsule morphology, distribution and connection with ADM were observed under a scanning electron microscope.
Isolation and identification of seed cells
The epidermal stem cells and fibroblasts were isolated in the same time from foreskin tissues with the method of collagenase combined with type I trypsin digestion, and their morphology was observed according to the report from Yang et al [11]. K19 of the epidermal stem cell population was identified with FITC-immunofluorescence and flow cytometry. The vimentin of fibroblasts was also detected by FITC-immunofluorescence, then these two kinds of cells were cultivated for 7 days and the growth state was observed respectively. Drawing growth curves of epidermal stem cell population added with KGF: Single cell suspension of epidermal stem cells was prepared and seeded in 96-well plates in three groups in which one was added with KGF, one was not and another one was simple fibroblast feeder layer cells. The absorption values of three wells were determined and averaged. Drawing growth curve was carried out with time (day) as horizontal axis and absorbance value as longitudinal axis. Blank well seeded with simple fibroblast feeder layer cells was used for zero adjustment.
Construction of tissue-engineered skin loading KGF nanocapsules
The single cell suspension of epidermal stem cells was cultivated on the aseptic KGF-ADM in 1×105/cm2. Then the KGF-ADM was kept in a culture incubator of
Constructing an animal model of skin defects
Full-thickness skin samples,
Detection of the skin in repairing area
Gross appearance was observed when dressing was tied off to evaluate the healing. The skin samples of 2, 4 and 6 weeks after transplantation were collected respectively and fixed in 10% formalin for further use.
Hematoxylin-eosin staining
The skin samples were embedded in paraffin for 4 μm section and then the hematoxylin-eosin staining was carried out. The staining results were observed with a light microscope.
Keratin 10 (K10)-FITC-immunofluorescence
Mouse anti-human cytokeratin10 monoclonal antibody
was chosen as primary antibody, FITC fluorescein-labeled goat anti-mouse IgG monoclonal antibody was chosen as secondary antibody and Hoechst 33342 was nuclear stain in immunofluorescence. PBS instead of primary antibody was blank control, human foreskin tissue was positive control and skin autograft was negative control. The steps were followed by the instruction.
β1-integrin-Cy3-immunofluorescenc
Rabbit anti-human β1-integrin polyclonal antibody was chosen as primary antibody, Cy3 fluorescein-labeled goat-anti-rabbit IgG polyclonal antibody as secondary antibody and hoechst33342 was nuclear stain.
Statistical processing
SPSS 11.0 software was used in the experimental data statistical processing. Differences between groups were assessed using one-way analysis of variance for the data of the growth curve. The result which was P < 0.05 indicated that KGF had a promotion effect on the proliferation and differentiation of epidermal stem cells.
The research of tissue-engineered skin has made a great development in plastic surgery field, but as above-mentioned, the tissue-engineered skin and skin substitutes reported in literature or used in a commercialized market have a lot of disadvantages. So, in this experiment, we evaluated the effect of a new kind of tissue-engineered skin, ADM-KGF, and epidermal stem cells in repairing skin defects of nude mice. We also gave detailed discussions on the results, advantages and problems. KGF which belongs to fibroblast growth factor family (FGF-7) is a kind single polypeptide chain synthesized by fibroblasts and purified from fibroblast culture medium. Different from other members of FGF, KGF acts on epithelial cells exclusively as a paracrine regulation factors promoting the growth of epidermal cells. Local application of KGF can keep the proliferation and differentiation of epidermal cells and complete the covering and healing of wound surfaces through epithelization[6, 18-24]. KGF is massively produced in the early stages of wound healing[25]. Keratinocyte proliferation, migration, and cytoskeletal rearrangement are largely controlled by KGF that is released by the underlying fibroblasts[26]. The growth curve in this study showed that the growth of epidermal cells added with KGF was better than the group without KGF, which is similar to the results of previous research and confirms the proliferation effect of KGF again[6, 18-19, 25-27]. KGF was often added in culture medium or sprayed on defect surfaces directly in the previous experiments. Because of the short half-time, KGF needs multiple doses or continuous administration to keep its concentration, which increase the burden of laboratory technicians and the tediousness of treatment process, and meanwhile, some KGF cannot act directly on epidermal cells, which also induce waste to some extent. Therefore, KGF release system is needed and the nanocapsule is a nice choice to construct controlled release system of growth factors[20-21]. In this study, we first encapsulated KGF in the PLGA nanocapsules, cross-linked the capsules with ADM, and then epidermal cells were seeded on it. All of these can make KGF release last for 4 weeks or more so that KGF can act on cells slowly and thoroughly. This method has not been reported yet and our study showed that tissue-engineered skin loading KGF nanocapsules was better than blank group and control group in the repair of skin defects, which indicates that KGF encapsulated in the nonocapsules can have a continuous and slow action. Based on our previous research[17], we constructed KGF nanocapsules successfully. The capsules have an integrated shape and a smooth surface, what is more, they also have an ability of connecting tightly with ADM without exfoliation. Most of disadvantages of polypeptide growth factors, such as short half-time, discharged and diluted by wound exudates, degraded by protease easily and decreased efficacy, can be solved through this method. Because the biocompatibility of macromolecular chemical PLGA which degrade into CO2 and H2O, this ideal release system can release growth factors continuously and without poison and harm to the human body. In addition, the use of ADM is based on the following experimental and clinic reason[3, 22-23]: without the keratinocytes, langerhans cells and other cells that have immunogenicity, the ADM is suitable to be dermal scaffold of tissue-engineered skin which has no immunogenicity and cannot induce immunological rejection. This is why we choose ADM as the dermal scaffold to utilize its native structure and molecular information. As seed cells of tissue-engineered skin, the epidermal stem cells have two significant biological characteristics[5]: slow cell cycle, shorter time for cell division; and powerful ability of proliferation, clonal growth in vitro culture. The DNA in cells has lest mutation and has enough time for self-repair and self-renew. Some research has indicated that β1-integrin [24] and K19[7] are two most reliable surface markers for cell selection. So in this study, we used β1-integrin to confirm the existence of epidermal stem cell and used K10 as a marker of mature keratinocyte. Testified by immunofluorescence assay and flow cytometry detection, there were a lot of epidermal stem cells[1, 10-11] in epidermal cells obtained through collagenase type I combined with trypsin digestion method. The epidermal stem cells seeded on the KGF-ADM grew well and could be affected by KGF continuously. They were not only connected with ADM tightly and had an active proliferation, but also combined with PLGA tightly and grew well on it. This phenomenon is not seen in previous report and manifests that our method is a reliable way and the extracellular matrix constructed is suitable for cell adhesion or growth. The KGF in nanocapsules can act on epidermal cells and epidermal stem cells adhered on the cyst wall directly through the ways like leaching or infiltration of cyst wall, dissolution of PLGA and others. The experimental findings also prove that PLGA has a good biocompatibility and the method is completely feasible and effective by which we combined natural biomaterials ADM with chemosynthetic macromolecular degradable biomaterials PLGA to construct the dermal matrix loading nanocapsules.In the experiment, we used the tissue-engineered skin constructed according to the above-mentioned method to repair full-thickness skin defects of nude mice. We had a 6-8 weeks continuous observation on the restoration of skin transplantation. The experiment was carried out in three groups: one was experimental group which was applied with tissue-engineered skin lading KGF nanocapsules, one was blank group which was applied with tissue-engineered skin without KGF nanocapsules, and the last one was control group in which autologous full-thickness skin of nude mice was used. Two weeks after transplantation, the repairing areas of three groups were healed basically. At 4 weeks, the color of skin grafts in each group was as same as the surrounding tissues, but there were some contractions in the three groups, especially in the control group. Six weeks later, the color of skin grafts in each group had no difference from the normal skin, and the skin graft in the experimental group had no contraction anymore, but in the control group the contraction was still obvious. Under the microscope, the experimental group had the best healing: the layer number of epidermal cells was similar to the normal skin, the structure was similar to the stratified squamous epithelium, the connection between cells or epidermal cells and dermis was tight, the stratum corneum which could play a barrier function of skin was produced and the new collagen in the dermis was arranged orderly. These results indicate that epidermal cells could proliferate and differentiate continuously since they were affected by KGF. On the other hand, there was abscission in some epidermal cell layer areas of blank group which was probably due to the lack of KGF. Detected by anti-human K10 FITC-immunofluorescence, the transplanted skin in experimental group had good cell layers in the epidermis and expressed normal keratin with green fluorescence, confirming that the defect areas were repaired by the tissue-engineered skin. We detected skins graft at 8 and 10 weeks after transplantation using anti-human β1-integrin-immunofluorescence, and found β1-integrin positive cells in some sections. Based on this, we inferred that there were still epidermal stem cells or transient amplifying cells which had the ability of proliferation and differentiation. This result reveals again that the epidermal stem cells as seed cells can keep the self-renew and growth of tissue-engineered skin for a long time. In the traditional skin transplantation model, autologous full-thickness skin is always used as “gold standard”, but in this study, we found the repairing effect of experimental group was better than the skin autograft. The tissue-engineered skin combined with epidermal stem cells maybe hold a promise of being a new kind biomaterial to repair skin defects completely. In addition, we also observed that the contraction of auto-skin transplantation group was worse than the other two groups, which is probably because the skin autograft of nude mice was looser than the ADM used in the experimental and blank groups. As we known, ADM is much thicker than the full-thickness skin of nude mice. When taking bandaging fixation, the ADM is unease to contract because of some supporting effect. So, the long term effect of ADM is better than the skin autograft.In short, the findings of this experiment and our previous research[1, 10-11, 17] all show that: we can construct a new kind tissue-engineered skin which is suitable for skin burn treatment and defects which can be repaired within 1-2 weeks by using KGF nanocapsules controlled release system and epidermal stem cells. The effect of this kind tissue-engineered skin in repairing full-thickness skin defect is better than common tissue-engineered skin and similar to auto full-thickness skin. In the meanwhile, there are still epidermal stem cells or transient amplifying cells existing in the tissue-engineered skin that have proliferative and differentiative potentiality to keep long-term autogenous renew and metabolism of the transplanted skin successively.
实验在传统组织工程皮肤中加入角质细胞生长因子,并且创新性地应用纳米微囊来构建缓释系统,以达到持续稳定的作用效果。利用天然生物材料脱细胞真皮基质搭载纳米微囊构建新型组织工程皮肤支架,结果证明此方法可行且有效。并通过组织学检测和动物实验研究该组织工程皮肤修复皮肤缺损的效果,结果也证实其优于传统组织工程皮肤和自体皮肤移植。 中国组织工程研究杂志出版内容重点:组织构建;骨细胞;软骨细胞;细胞培养;成纤维细胞;血管内皮细胞;骨质疏松;组织工程
文章特点: 文章概要:实验尝试以表皮干细胞为种子细胞,利用搭载角质细胞生长因子纳米微囊的脱细胞真皮基质构建新型的组织工程皮肤。利用新型组织工程皮肤修复裸鼠皮肤缺损,通过与传统组织工程皮肤及自体皮肤对比,研究其修复的效果与特点,以此来对新型组织工程皮肤的构建方法、组织学特点及修复皮肤缺损效果进行评价。 文章要点:组织工程皮肤是多个领域的研究热点,对整形美容和修复重建等方面具有重要的临床意义,而现有的组织工程皮肤存在诸多不足,限制其进一步发展和应用。实验通过以纳米微囊的形式在组织工程皮肤中加入角质细胞生长因子,通过其对表皮干细胞的持续稳定的调节作用改善组织工程皮肤的特性,并通过实验证明其在修复裸鼠皮肤全层缺损中取得了良好的效果。 中国组织工程研究杂志出版内容重点:组织构建;骨细胞;软骨细胞;细胞培养;成纤维细胞;血管内皮细胞;骨质疏松;组织工程
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||