中国组织工程研究 ›› 2014, Vol. 18 ›› Issue (15): 2369-2376.doi: 10.3969/j.issn.2095-4344.2014.15.013
• 组织构建与生物活性因子 tissue construction and bioactive factors • 上一篇 下一篇
人脑源性神经营养因子和神经营养素3真核双表达载体的构建与鉴定
栗炳南,李卫东,林俊堂,丰慧根
- 新乡医学院生命科学技术学院,河南省新乡市 453003
-
出版日期:2014-04-09发布日期:2014-04-09 -
通讯作者:栗炳南, 新乡医学院生命科学技术学院,河南省新乡市 453003 -
作者简介:栗炳南,男,1983年生,河南省新乡市人,汉族,2011年中南大学湘雅医学院毕业,博士,讲师,目前主要从事神经系统疾病的发病机制与治疗方面的研究。 -
基金资助:新乡医学院重点领域招标课题(ZD2011-16);河南省教育厅科学技术研究重点项目(13A180850)
Construction and identification of bicistronic eukaryotic expression vector of human brain-derived neurotrophic factor and neurotrophine-3
Li Bing-nan, Li Wei-dong, Lin Jun-tang, Feng Hui-gen
- Department of Life Sciences and Technology, Xinxiang Medical University, Xinxiang 453003, Henan Province, China
-
Online:2014-04-09Published:2014-04-09 -
Contact:Li Bing-nan, Department of Life Sciences and Technology, Xinxiang Medical University, Xinxiang 453003, Henan Province, China -
About author:Li Bing-nan, Ph.D., Lecturer, Department of Life Sciences and Technology, Xinxiang Medical University, Xinxiang 453003, Henan Province, China -
Supported by:the Tender Subject of Key Research Areas of Xinxiang Medical University in 2011, No. ZD2011-16; Key Projects in Scientific Research of Henan Provincial Education Department, No. 13A180850
摘要:
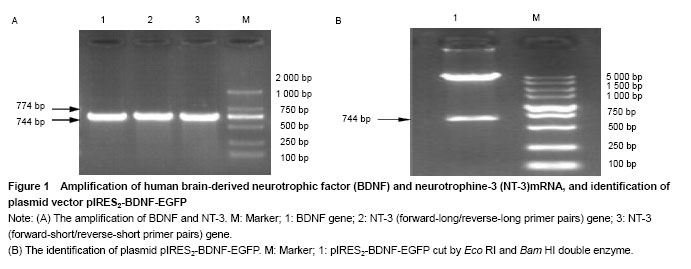
背景:脑源性神经营养因子(brain-derivedneurotrophic factor,BDNF)和神经营养 素3 (Neurotrophines-3,NT-3)在细胞分化过程中有重要作用。病毒载体临床应用存在安全隐患,利用真核表达载体表达蛋白为解决安全性问题提供了一种方法。 目的:构建双基因共表达载体pIRES2-BDNF-NT-3并对其进行鉴定。 方法: BDNF和NT-3基因核心序列是通过直接PCR的方法从外周血单个核细胞的基因组DNA中获取。然后将BDNF的cDNA片段插入到pIRES2-EGFP多克隆位点构建pIRES2-BDNF-EGFP载体,随后将NT-3 cDNA片段以替换EGFP的方式插入pIRES2-BDNF-EGFP中,最后构建成为含有内部核糖体进入位点(IRES)的pIRES2-BDNF-NT-3双基因共表达载体。实验通过双酶切和DNA测序方法对其鉴定,将重组的双基因共表达载体感染HEK293细胞,利用RT-PCR与 Western-blot方法检测双基因的表达。 结果与结论:DNA测序显示,提取的BDNF和NT-3均与基因库报道序列一致。构建的pIRES2-BDNF-NT-3双基因共表达载体经Eco RⅠ/Bam HⅠ切出BDNF条带,经Bam HⅠ/NotⅠ双酶切后切出IRES-NT-3片段,经Eco RⅠ/NotⅠ双酶切后切出BDNF-IRES-NT-3片段。RT-PCR与Western-blot方法检测显示,此载体转染后的HEK293细胞均能表达BDNF和NT-3 mRNA和蛋白。结果证实,实验成功采用IRES序列构建了能分别表达的BDNF和NT-3双基因真核表达载体。
中图分类号:
引用本文
栗炳南,李卫东,林俊堂,丰慧根. 人脑源性神经营养因子和神经营养素3真核双表达载体的构建与鉴定[J]. 中国组织工程研究, 2014, 18(15): 2369-2376.
Li Bing-nan, Li Wei-dong, Lin Jun-tang, Feng Hui-gen. Construction and identification of bicistronic eukaryotic expression vector of human brain-derived neurotrophic factor and neurotrophine-3[J]. Chinese Journal of Tissue Engineering Research, 2014, 18(15): 2369-2376.
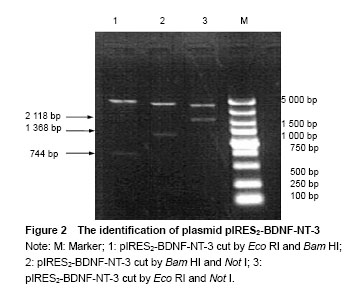
Identification of plasmid pIRES2-BDNF-NT-3 The plasmid pIRES2-BDNF-NT-3 was cut by Eco RI and Bam HI double enzyme. And a fragment about 744 bp would be obtained. This indicated BDNF gene was inserted into the plasmid pIRES2-BDNF-NT-3. The plasmid pIRES2-BDNF-NT-3 was cut by Bam HI and Not I double enzyme. And a fragment about 1 368 bp would be obtained. This indicated IRES-NT-3 gene was inserted into the plasmid pIRES2-BDNF-NT-3. The plasmid pIRES2-BDNF-NT-3 was cut by Eco RI and Not I double enzyme. And a fragment about 2 118 bp would be obtained. This indicated that BDNF-IRES-NT-3 gene was inserted into the plasmid pIRES2-BDNF-NT-3. The sequence of the plasmid pIRES2-BDNF-NT-3 was in accordance with gene sequence in Gene Bank (Figure 2).
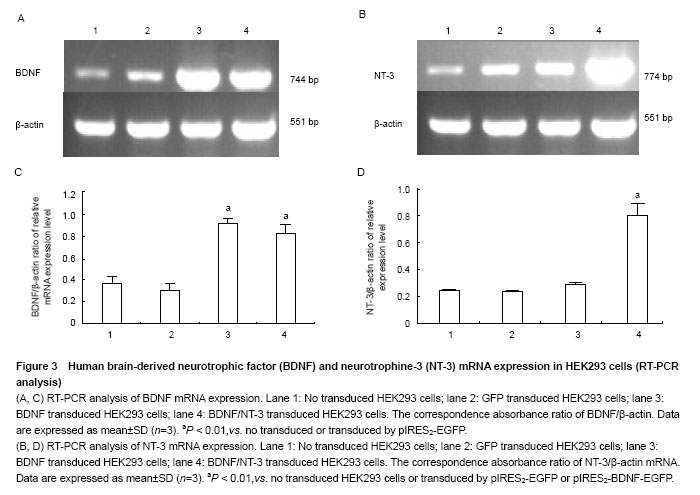
RT-PCR analysis of BDNF and NT-3 mRNA expression in HEK293 cells
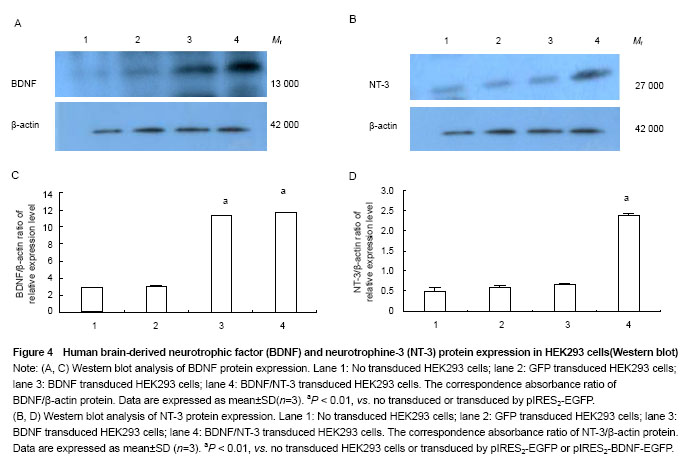
Western blot analysis of BDNF and NT-3 expression in HEK293 cells
| [1]Tancredo JR, Maria RM, Azevedo ER, et al. Clinical assessment of spasticity in individuals with spinal cord injury. Acta Ortop Bras. 2013;21(6):310-314. [2]Fouad K, Bennett DJ, Vavrek R, et al. Long-term viral brain-derived neurotrophic factor delivery promotes spasticity in rats with a cervical spinal cordhemisection. Front Neurol. 2013;19(4):187-197. [3]Hou S, Nicholson L, van Niekerk E, et al. Dependence of regenerated sensory axons on continuous neurotrophin-3 delivery. J Neurosci. 2012;32(38): 13206-13220. [4]Blesch A, Fischer I, Tuszynski MH. Gene therapy, neurotrophic factors and spinal cord regeneration. Handb Clin Neurol. 2012;109(1):563-574. [5]Franz S, Weidner N, Blesch A. Gene therapy approaches to enhancing plasticity and regeneration after spinal cord injury. Exp Neurol. 2012;235(1):62-69. [6]Rauskolb S, Zagrebelsky M, Dreznjak A, et al. Global deprivation of brain-derived neurotrophic factor in the CNS reveals an area-specific requirement for dendritic growth. J Neurosci. 2010;30(5):1739-1749. [7]Huang T, Krimm RF. BDNF and NT4 play interchangeable roles in gustatory development. Dev Biol. 2014;386(2): 308-320. [8]Angelucci F, Ricci V, Gelfo F, et al. BDNF serum levels in subjects developing or not post-traumatic stress disorder after trauma exposure. Brain Cogn. 2014;84(1):118-122. [9]Fon D, Zhou K, Ercole F, et al. Nanofibrous scaffolds releasing a small molecule BDNF-mimetic for the re-direction of endogenous neuroblast migration in the brain. Biomaterials. 2014;35(9):2692-2712. [10]Li ST, Pan J, Hua XM, et al. Endothelial nitric oxide synthase protects neurons against ischemic injury through regulation of brain-derived neurotrophic factor expression. CNS Neurosci Ther. 2014;20(2):154-164. [11]Franz S, Weidner N, Blesch A. Gene therapy approaches to enhancing plasticity and regeneration after spinal cord injury. Exp Neurol. 2012;235(1):62-69. [12]Andero R, Choi DC, Ressler KJ. BDNF-TrkB receptor regulation of distributed adult neural plasticity, memory formation, and psychiatric disorders. Prog Mol Biol Transl Sci. 2014;122(1):169-192. [13]Morcuende S, Muñoz-Hernández R, Benítez-Temiño B, et al. Neuroprotective effects of NGF, BDNF, NT-3 and GDNF on axotomized extraocular motoneurons in neonatal rats. Neuroscience. 2013;250(1):31-48. [14]Zanin JP, Battiato NL, Rovasio RA. Neurotrophic factor NT-3 displays a non-canonical cell guidance signaling function for cephalic neural crest cells. Eur J Cell Biol. 2013;92(8-9): 264-279. [15]Sun Z, Guo Q, Xu X, et al. Protective effects of pretreatment with neurotrophin-3 on intrathecal ropivacaine in rats. Zhong Nan Da Xue Xue Bao Yi Xue Ban. 2014;39(1):17-22. [16]Tang Y, He H, Cheng N, et al. PDGF, NT-3 and IGF-2 in combination induced transdifferentiation of muscle-derived stem cells into schwann cell-like cells. PLoS One. 2014;9(1): 73402-73410. [17]Yang J, Yan Y, Xia Y, et al. Neurotrophin 3 transduction augments remyelinating and immunomodulatory capacity of neural stem cells. Mol Ther. 2013;22(2):440-450. [18]Ishimaru N, Fukuchi M, Hirai A, et al. Differential epigenetic regulation of BDNF and NT-3 genes by trichostatin A and 5-aza-2'-deoxycytidine in Neuro-2a cells. Biochem Biophys Res Commun. 2010;394(1):173-177. [19]Xie BH, Xie YF. Twin PCRs: a simple and efficient method for directional cloning of PCR products. World J Microbiol Biotechnol. 2011;27(9):2223-2225. [20]Sakai K, Yamamoto A, Matsubara K, et al. Human dental pulp-derived stem cells promote locomotor recovery after complete transection of the rat spinalcord by multiple neuro-regenerative mechanisms. J Clin Invest. 2012;122(1): 80-90. [21]Massa SM, Yang T, Xie Y, et al. Small molecule BDNF mimetics activate TrkB signaling and prevent neuronal degeneration in rodents. J Clin Invest. 2010;120(5): 1774-1785. [22]Deogracias R, Yazdani M, Dekkers MP, et al. Fingolimod, a sphingosine-1 phosphate receptor modulator, increases BDNF levels and improves symptoms of amouse model of Rett syndrome. Proc Natl Acad Sci U S A. 2012;109(35): 14230-14235. [23]Bonner JF, Blesch A, Neuhuber B, et al. Promoting directional axon growth from neural progenitors grafted into the injured spinal cord. J Neurosci Res. 2010;88(6):1182-1192. [24]Franz S, Weidner N, Blesch A. Gene therapy approaches to enhancing plasticity and regeneration after spinal cord injury. Exp Neurol. 2012;235(1):62-69. [25]Ding Y, Yan Q, Ruan JW, et al. Electroacupuncture promotes the differentiation of transplanted bone marrow mesenchymal stem cells overexpressing TrkC into neuron-like cells in transected spinal cord of rats. Cell Transplant. 2013;22(1): 65-86. [26]Gelain F, Panseri S, Antonini S, et al. Transplantation of nanostructured composite scaffolds results in the regeneration of chronically injured spinalcords. ACS Nano. 2011;5(1):227-236. [27]Overman JJ, Carmichael ST. Plasticity in the injured brain: more than molecules matter. Neuroscientist. 2014;20(1): 15-28. [28]García-Alías G, Petrosyan HA, Schnell L, et al. Chondroitinase ABC combined with neurotrophin NT-3 secretion and NR2D expression promotes axonalplasticity and functional recovery in rats with lateral hemisection of the spinal cord. J Neurosci. 2011;31(49):17788-17799. [29]Jenkins SI, Pickard MR, Granger N, et al. Magnetic nanoparticle-mediated gene transfer to oligodendrocyte precursor cell transplant populations isenhanced by magnetofection strategies. ACS Nano. 2011;5(8):6527-6538. [30]Koo HM, Lee SM, Kim MH. Spontaneous wheel running exercise induces brain recovery via neurotrophin-3 expression following experimental traumatic brain injury in rats. J Phys Ther Sci. 2013;25(9):1103-1107. [31]Beggs S, Alvares D, Moss A, et al. A role for NT-3 in the hyperinnervation of neonatally wounded skin. Pain. 2012; 153(10):2133-2139. [32]Smith GM, Falone AE, Frank E. Sensory axon regeneration: rebuilding functional connections in the spinal cord. Trends Neurosci. 2012;35(3):156-163. |
| [1] | 张同同, 王中华, 文 杰, 宋玉鑫, 刘 林. 3D打印模型在颈椎肿瘤手术切除与重建中的应用[J]. 中国组织工程研究, 2021, 25(9): 1335-1339. |
| [2] | 王晗月, 李富荣, 杨晓菲, 胡巢凤. 高效靶向激活肝细胞内源基因直接重编程为胰岛样细胞[J]. 中国组织工程研究, 2021, 25(7): 1056-1063. |
| [3] | 曾燕华, 郝延磊. 许旺细胞体外培养及纯化的系统性综述[J]. 中国组织工程研究, 2021, 25(7): 1135-1141. |
| [4] | 徐东紫, 张 婷, 欧阳昭连. 心脏组织工程领域全球专利竞争态势分析[J]. 中国组织工程研究, 2021, 25(5): 807-812. |
| [5] | 吴子健, 胡昭端, 谢有琼, 王 峰, 李 佳, 李柏村, 蔡国伟, 彭 锐. 3D打印技术与骨组织工程研究文献计量及研究热点可视化分析[J]. 中国组织工程研究, 2021, 25(4): 564-569. |
| [6] | 常文辽, 赵 杰, 孙晓亮, 王 锟, 吴国锋, 周 剑, 李树祥, 孙 晗. 人工骨膜的材料选择、理论设计及生物仿生功能[J]. 中国组织工程研究, 2021, 25(4): 600-606. |
| [7] | 刘 旒, 周箐竹, 龚 桌, 刘博言, 杨 斌, 赵 娴. 胶原/无机材料构建组织工程骨的特点及制造技术[J]. 中国组织工程研究, 2021, 25(4): 607-613. |
| [8] | 刘 飞, 崔宇韬, 刘 贺. 局部抗生素递送系统治疗骨髓炎的优势与问题[J]. 中国组织工程研究, 2021, 25(4): 614-620. |
| [9] | 李晓壮, 段 浩, 王伟舟, 唐志宏, 王旸昊, 何 飞. 骨组织工程材料治疗骨缺损疾病在体内实验中的应用[J]. 中国组织工程研究, 2021, 25(4): 626-631. |
| [10] | 张振坤, 李 喆, 李 亚, 王莹莹, 王亚苹, 周馨魁, 马珊珊, 关方霞. 海藻酸盐基水凝胶/敷料在创面愈合中的应用:持续、动态与顺序释放[J]. 中国组织工程研究, 2021, 25(4): 638-643. |
| [11] | 陈佳娜, 邱燕玲, 聂敏海, 刘旭倩. 组织工程支架材料修复口腔颌面部软组织缺损[J]. 中国组织工程研究, 2021, 25(4): 644-650. |
| [12] | 邢 浩, 张永红, 王 栋. 长骨大段骨缺损修复方法的优势与不足[J]. 中国组织工程研究, 2021, 25(3): 426-430. |
| [13] | 陈思奇, 先德彬, 徐荣胜, 覃中杰, 张 磊, 夏德林. 羟基磷灰石-磷酸三钙支架复合骨髓间充质干细胞和人脐静脉内皮细胞对大鼠颅骨缺损修复早期成血管的影响[J]. 中国组织工程研究, 2021, 25(22): 3458-3465. |
| [14] | 刘 鋆, 杨 龙, 王伟宇, 周玉虎, 吴 颖, 卢 涛, 舒莉萍, 马敏先, 叶 川. 聚3-羟基丁酸酯4-羟基丁酸酯/聚乙二醇/氧化石墨烯组织工程支架的制备和性能评价[J]. 中国组织工程研究, 2021, 25(22): 3466-3472. |
| [15] | 王 皓, 陈明学, 李俊康, 罗旭江, 彭礼庆, 李 获, 黄 波, 田广招, 刘舒云, 眭 翔, 黄靖香, 郭全义, 鲁晓波. 脱细胞猪皮基质构建组织工程半月板支架[J]. 中国组织工程研究, 2021, 25(22): 3473-3478. |
Design
HEK293 cells are kept in Stem Cells and Biological Treatment Center at Xinxiang Medical University, China.
.jpg)
Two primer pairs were designed to amplify human NT-3 from the genomic DNA. Forward-long primer was 4 bases longer than forward-short at the 5' end. So did primer reverse-long than reverse-short. The underlined bases in forward primers were introduced parts of sequence of Bst XI site, and in reverse primers of Not I site, which will be used to assemble the Bst XI and Not I sticky ends. The length of amplified fragment was 774 bp.
.jpg)
.jpg)
The mRNA and protein expressions of NT-3 and BDNF were detected by RT-PCR and western blot assay, respectively.
Experimental data were analyzed using SPSS 13.0 statistical software (SPSS, Chicago, IL, USA) and the results are expressed as mean±SD. Data comparisons between groups were tested by one-way analysis of variance, P < 0.05 was considered statistically significant
在基因治疗中,联合应用多个具有协同作用的治疗基因通常可产生较单基因更为理想的效果。但是与国内外同类研究水平的比较以往进行多基因联合治疗的方法存在不同缺陷,利用携带不同基因的独立载体系统同时转染靶细胞的方法,虽然可自由调节各表达载体的比例,但各基因的总表达效率低下;而用传统方法构建的多启动子表达载体,因为每个治疗基因都需要一个包括启动子、终止子等表达元件在内的完整基因表达盒,使得载体过于庞大,操作困难;而2个基因融合表达的策略可能因蛋白结构相互影响而导致功能丧失。利用内部核糖体进入位点构建多基因共表达载体,则可大大提高转移及表达效率。
实验成功采用内部核糖体进入位点序列构建了能够分别表达的脑源性神经营养因子与神经营养素-3双基因真核表达载体,既增加了外源基因的表达,同时又可以减少基因治疗载体的用量,减少了不安全因素,为临床的进一步应用带来了希望。
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||