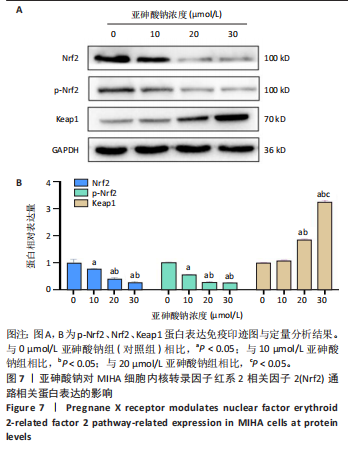
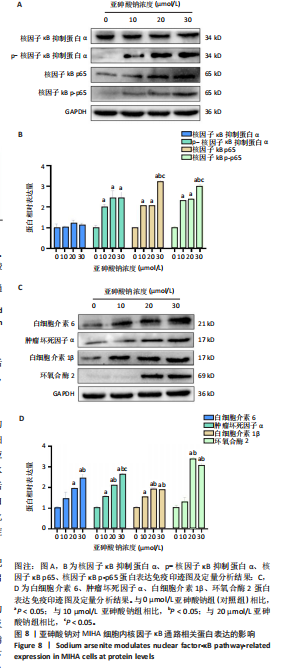
[1] PALMA-LARA I, MARTÍNEZ-CASTILLO M, QUINTANA-PÉREZ JC, et al. Arsenic exposure: A public health problem leading to several cancers. Regul Toxicol Pharmacol. 2020;110:104539.
[2] UPADHYAY MK, SHUKLA A, YADAV P, et al. A review of arsenic in crops, vegetables, animals and food products. Food Chem. 2019;276:608-618.
[3] KHAN MI, AHMAD MF, AHMAD I, et al. Arsenic Exposure through Dietary Intake and Associated Health Hazards in the Middle East. Nutrients. 2022;14(10):2136.
[4] ISLAM MS, MUSTAFA RA, PHOUNGTHONG K, et al. Arsenic in the foodstuffs: potential health appraisals in a developing country, Bangladesh. Environ Sci Pollut Res Int. 2023;30(10):26938-26951.
[5] HIRANO S. Biotransformation of arsenic and toxicological implication of arsenic metabolites. Arch Toxicol. 2020;94(8):2587-2601.
[6] MA G, YAN X, WANG C, et al. Mechanism of arsenic-induced liver injury in rats revealed by metabolomics and ionomics based approach. Ecotoxicol Environ Saf. 2025;293:118038.
[7] XING Y, YAN J, NIU Y. PXR: a center of transcriptional regulation in cancer. Acta Pharm Sin B. 2020;10(2):197-206.
[8] LV Y, LUO YY, REN HW, et al. The role of pregnane X receptor (PXR) in substance metabolism. Front Endocrinol (Lausanne). 2022;13:959902.
[9] ZHANG J, HUANG Y, LI H, et al. B3galt5 functions as a PXR target gene and regulates obesity and insulin resistance by maintaining intestinal integrity. Nat Commun. 2024;15(1):5919.
[10] WAHLI W. A gut feeling of the PXR, PPAR and NF-kappaB connection. J Intern Med. 2008;263(6):613-619.
[11] FENG Y, SHEN J, LIN Z, et al. PXR Activation Relieves Deoxynivalenol-Induced Liver Oxidative Stress Via Malat1 LncRNA m6A Demethylation. Adv Sci (Weinh). 2024;11(25): e2308742.
[12] 王甜,赵哲仪,穆银贵,等.亚砷酸钠所致L-02人肝细胞损伤与p14ARF表达下调及MDM2、 p53表达增加有关[J].细胞与分子免疫学杂志,2020, 36(6):507-512.
[13] 田侦丽,张小旭,方兴艳,等.亚砷酸钠对人正常肝细胞脂质代谢及因子调控的作用[J].中国组织工程研究,2025,29(23):4956-4964.
[14] BEINSTEINER B, BILLAS IML, MORAS D. Structural insights into the HNF4 biology. Front Endocrinol (Lausanne). 2023;14:1197063.
[15] RADI SH, VEMURI K, MARTINEZ-LOMELI J, et al. HNF4α isoforms: the fraternal twin master regulators of liver function. Front Endocrinol (Lausanne). 2023;14: 1226173.
[16] FREDIANI JK, NAIOTI EA, VOS MB, et al. Arsenic exposure and risk of nonalcoholic fatty liver disease (NAFLD) among U.S. adolescents and adults: an association modified by race/ethnicity, NHANES 2005-2014. Environ Health. 2018;17(1):6.
[17] ABU EL-SAAD AM, AL-KAHTANI MA, ABDEL-MONEIM AM. N-acetylcysteine and meso-2,3-dimercaptosuccinic acid alleviate oxidative stress and hepatic dysfunction induced by sodium arsenite in male rats. Drug Des Devel Ther. 2016;10: 3425-3434.
[18] MOLAVINIA S, MOOSAVI M, HEJAZI S, et al. Metformin alleviates sodium arsenite-induced hepatotoxicity and glucose intolerance in mice by suppressing oxidative stress, inflammation, and apoptosis. J Trace Elem Med Biol. 2023;80: 127299.
[19] PHULL AR, NASIR B, HAQ IU, et al. Oxidative stress, consequences and ROS mediated cellular signaling in rheumatoid arthritis. Chem Biol Interact. 2018; 281:121-36.
[20] ZHANG B, PAN C, FENG C, et al. Role of mitochondrial reactive oxygen species in homeostasis regulation. Redox Rep. 2022;27(1): 45-52.
[21] SENA LA, CHANDEL NS. Physiological roles of mitochondrial reactive oxygen species. Mol Cell. 2012;48(2):158-167.
[22] LI Y, MA Q, LIU G, et al. Effects of donkey milk on oxidative stress and inflammatory response. J Food Biochem. 2022;46(4):e13935.
[23] WU KC, LIU J, KLAASSEN CD. Role of Nrf2 in preventing ethanol-induced oxidative stress and lipid accumulation. Toxicol Appl Pharmacol. 2012;262(3):321-329.
[24] LEE YH, KIM JH, SONG CH, et al. Ethanol Extract of Ganoderma lucidum Augments Cellular Anti-oxidant Defense through Activation of Nrf2/HO-1. J Pharmacopuncture. 2016;19(1): 59-69.
[25] ZINATIZADEH MR, SCHOCK B, CHALBATANI GM, et al. The Nuclear Factor Kappa B (NF-kB) signaling in cancer development and immune diseases. Genes Dis. 2021;8(3):287-297.
[26] LIU P, LI Y, WANG W, et al. Role and mechanisms of the NF-ĸB signaling pathway in various developmental processes. Biomed Pharmacother. 2022;153:113513.
[27] MOTOLANI A, MARTIN M, SUN M, et al. Phosphorylation of the Regulators, a Complex Facet of NF-κB Signaling in Cancer. Biomolecules. 2020;11(1):15.
[28] LIU F, ZHAO Y, PEI Y, et al. Role of the NF-kB signalling pathway in heterotopic ossification: biological and therapeutic significance. Cell Commun Signal. 2024;22(1):159.
[29] EBRAHIMI N, ABDULWAHID ARR, MANSOURI A, et al. Targeting the NF-κB pathway as a potential regulator of immune checkpoints in cancer immunotherapy. Cell Mol Life Sci. 2024;81(1):106.
[30] GUO Q, JIN Y, CHEN X, et al. NF-κB in biology and targeted therapy: new insights and translational implications. Signal Transduct Target Ther. 2024;9(1):53.
[31] KARPALE M, HUKKANEN J, HAKKOLA J. Nuclear Receptor PXR in Drug-Induced Hypercholesterolemia. Cells. 2022;11(3):313.
[32] YUAN T, LV S, ZHANG W, et al. PF-PLC micelles ameliorate cholestatic liver injury via regulating TLR4/MyD88/NF-κB and PXR/CAR/UGT1A1 signaling pathways in EE-induced rats. Int J Pharm. 2022;615:121480.
[33] LIANG HF, YANG X, LI HL, et al. Activation of pregnane X receptor protects against cholestatic liver injury by inhibiting hepatocyte pyroptosis. Acta Pharmacol Sin. 2025;46(1):147-158.
[34] DOU JY, ZHANG M, CEN H, et al. Salvia miltiorrhiza Bunge (Danshen) and Bioactive Compound Tanshinone IIA Alleviates Cisplatin-Induced Acute Kidney Injury Through Regulating PXR/NF-κB Signaling. Front Pharmacol. 2022;13:860383.
[35] OKAMURA M, SHIZU R, HOSAKA T, et al. Possible involvement of the competition for the transcriptional coactivator glucocorticoid receptor-interacting protein 1 in the inflammatory signal-dependent suppression of PXR-mediated CYP3A induction in vitro. Drug Metab Pharmacokinet. 2019;34(4):272-279.
[36] SHAO YY, GUO Y, FENG XJ, et al. Oridonin Attenuates TNBS-induced Post-inflammatory Irritable Bowel Syndrome via PXR/NF-κB Signaling . Inflammation. 2021;44(2):645-658.
[37] LI H, FU Y, GONG W, et al. Remission of copper-induced liver injury through the PXR/NF-kB signaling pathway: The effects of dietary curcumin supplementation in largemouth bass (Micropterus salmoides). Ecotoxicol Environ Saf. 2024;285: 117070.
[38] ZHANG X, MA Z, LIANG Q, et al. Tanshinone IIA exerts protective effects in a LCA-induced cholestatic liver model associated with participation of pregnane X receptor. J Ethnopharmacol. 2015;164: 357-367.
[39] NIU H, ZHOU X, LIU P, et al. Lactobacillus rhamnosus MN-431 Metabolic Tryptophan Alleviates Complementary Food-Induced Diarrhea through PXR-NF-κB Pathway and AHR-Th17 Cell Response Pathways. Mol Nutr Food Res. 2023;67(13):e2200530.
[40] DU L, JIANG W, ZHU X, et al. Rifaximin alleviates intestinal barrier disruption and systemic inflammation via the PXR/NFκB/MLCK pathway and modulates intestinal Lachnospiraceae abundance in heat-stroke mice. Int Immunopharmacol. 2024;143(Pt 2):113462.
[41] YU X, XU M, MENG X, et al. Nuclear receptor PXR targets AKR1B7 to protect mitochondrial metabolism and renal function in AKI. Sci Transl Med. 2020; 12(543):eaay7591.
[42] SUN M, CUI W, WOODY SK, et al. Pregnane X receptor modulates the inflammatory response in primary cultures of hepatocytes. Drug Metab Dispos. 2015;43(3):335-343.
[43] MULDER TAM, VAN EERDEN RAG, DE WITH M, et al. CYP3A4∗22 Genotyping in Clinical Practice: Ready for Implementation? Front Genet. 2021;12:711943.
[44] KONDŽA M, BRIZIĆ I, JOKIĆ S. Flavonoids as CYP3A4 Inhibitors In Vitro. Biomedicines. 2024;12(3):644.
[45] ZHAO M, MA J, LI M, et al. Cytochrome P450 Enzymes and Drug Metabolism in Humans. Int J Mol Sci. 2021;22(23):12808.
[46] BI G, LIANG F, WU T, et al. Pregnane X receptor activation induces liver enlargement and regeneration and simultaneously promotes the metabolic activity of CYP3A1/2 and CYP2C6/11 in rats. Basic Clin Pharmacol Toxicol. 2024;135(2):148-163.
[47] NOREAULT TL, KOSTRUBSKY VE, WOOD SG, et al. Arsenite decreases CYP3A4 and RXRalpha in primary human hepatocytes. Drug Metab Dispos. 2005;33(7): 993-1003.
[48] EL-GHIATY MA, EL-KADI AOS. Arsenic: Various species with different effects on cytochrome P450 regulation in humans. EXCLI J. 2021;20:1184-1242.
[49] JIANG W, SANG R, ZHANG C, et al. Application of small interfering RNA technology in cytochrome P450 gene modulation. Drug Metab Dispos. 2025;53(3):100040.
[50] YANG X, WEBER AA, MENNILLO E, et al. Oral arsenic administration to humanizedUDP-glucuronosyltransferase1 neonatal mice induces UGT1A1 through a dependence on Nrf2 and PXR. J Biol Chem. 2023;299(3):102955.
|