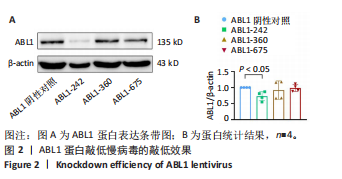
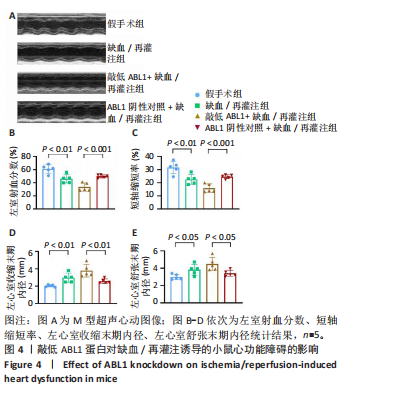
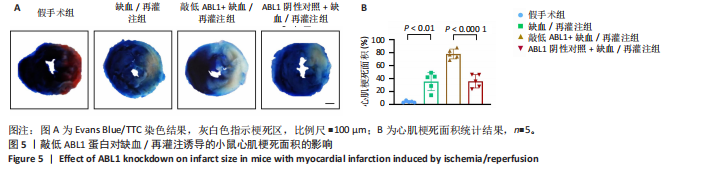
[1] HEUSCH G. Myocardial ischemia/reperfusion: Translational pathophysiology of ischemic heart disease. Med. 2024;5(1):10-31.
[2] CHANG AJ, LIANG Y, HAMILTON SA, et al. Medical Decision-Making and Revascularization in Ischemic Cardiomyopathy. Med Clin North Am. 2024; 108(3):553-566.
[3] MONTONE RA, CAMILLI M, CALVIERI C, et al. Exposome in ischaemic heart disease: beyond traditional risk factors. Eur Heart J. 2024;45(6):419-438.
[4] XIAO H, ZHANG M, WU H, et al. CIRKIL Exacerbates Cardiac Ischemia/Reperfusion Injury by Interacting With Ku70. Circ Res. 2022;130(5):e3-e17.
[5] WELT FGP, BATCHELOR W, SPEARS JR, et al. Reperfusion Injury in Patients With Acute Myocardial Infarction: JACC Scientific Statement.J Am Coll Cardiol. 2024;83(22):2196-2213.
[6] BAWASKAR P, THOMAS N, ISMAIL K, et al. Nonischemic or Dual Cardiomyopathy in Patients With Coronary Artery Disease. Circulation. 2024;149(11):807-821.
[7] PERERA D, CLAYTON T, O’KANE PD, et al. Percutaneous Revascularization for Ischemic Left Ventricular Dysfunction. N Engl J Med. 2022;387(15): 1351-1360.
[8] PANG H, HUANG G, XIE Z, et al. The role of regulated necrosis in diabetes and its complications. J Mol Med (Berl). 2024;102(4):495-505.
[9] WANG K, LI FH, ZHOU LY, et al. HNEAP Regulates Necroptosis of Cardiomyocytes by Suppressing the m(5) C Methylation of Atf7 mRNA. Adv Sci (Weinh). 2023;10(34):e2304329.
[10] GUO R, JIA X, DING Z, et al. Loss of MLKL ameliorates liver fibrosis by inhibiting hepatocyte necroptosis and hepatic stellate cell activation.Theranostics. 2022;12(11):5220-5236.
[11] MEIER P, LEGRAND AJ, ADAM D, et al. Immunogenic cell death in cancer: targeting necroptosis to induce antitumour immunity. Nat Rev Cancer. 2024;24(5):299-315.
[12] VRINGER E, TAIT SWG. Mitochondria and cell death-associated inflammation. Cell Death Differ. 2023;30(2):304-312.
[13] YIN H, GUO X, CHEN Y, et al. TAB2 deficiency induces dilated cardiomyopathy by promoting RIPK1-dependent apoptosis and necroptosis. J Clin Invest. 2022;132(4):e152297.
[14] GAO P, CAO M, JIANG X, et al. Cannabinoid Receptor 2-Centric Molecular Feedback Loop Drives Necroptosis in Diabetic Heart Injuries.Circulation. 2023;147(2):158-174.
[15] SONG Z, SONG H, LIU D, et al. Overexpression of MFN2 alleviates sorafenib-induced cardiomyocyte necroptosis via the MAM-CaMKIIδ pathway in vitro and in vivo.Theranostics. 2022;12(3):1267-1285.
[16] ZHU H, TAN Y, DU W, et al. Phosphoglycerate mutase 5 exacerbates cardiac ischemia-reperfusion injury through disrupting mitochondrial quality control.Redox Biol. 2021;38:101777.
[17] LI Z, DAI R, CHEN M, et al. p55γ degrades RIP3 via MG53 to suppress ischaemia-induced myocardial necroptosis and mediates cardioprotection of preconditioning. Cardiovasc Res. 2023;119(14):2421-2440.
[18] SONG C, DONG Q, YAO Y, et al. Nonreceptor tyrosine kinase ABL1 regulates lysosomal acidification by phosphorylating the ATP6V1B2 subunit of the vacuolar-type H(+)-ATPase. Autophagy. 2025;11:1-20.
[19] MALNASSY G, KEATING CR, GAD S, et al. Inhibition of Abelson Tyrosine-Protein Kinase 2 Suppresses the Development of Alcohol-Associated Liver Disease by Decreasing PPARgamma Expression. Cell Mol Gastroenterol Hepatol. 2023;16(5):685-709.
[20] BLAKES AJM, GAUL E, LAM W, et al. Pathogenic variants causing ABL1 malformation syndrome cluster in a myristoyl-binding pocket and increase tyrosine kinase activity. Eur J Hum Genet. 2021;29(4):593-603.
[21] WANG F, HOU W, CHITSIKE L, et al. ABL1, Overexpressed in Hepatocellular Carcinomas, Regulates Expression of NOTCH1 and Promotes Development of Liver Tumors in Mice. Gastroenterology. 2020;159(1):289-305.e16.
[22] SOURBIER C, RICKETTS C J, MATSUMOTO S, et al. Targeting ABL1-mediated oxidative stress adaptation in fumarate hydratase-deficient cancer. Cancer cell. 2014;26(6):840-850.
[23] KERKELÄ R, GRAZETTE L, YACOBI R, et al. Cardiotoxicity of the cancer therapeutic agent imatinib mesylate. Nature medicine. 2006;12(8):908-916.
[24] MAHARSY W, ARIES A, MANSOUR O, et al. Ageing is a risk factor in imatinib mesylate cardiotoxicity. Eur J Heart Fail. 2014;16(4):367-376.
[25] YE Y, YANG K, LIU H, et al. SIRT2 counteracts primate cardiac aging via deacetylation of STAT3 that silences CDKN2B. Nat Aging. 2023; 3(10):1269-1287.
[26] LIU Y, ZHONG C, CHEN S, et al. Circulating exosomal mir-16-2-3p is associated with coronary microvascular dysfunction in diabetes through regulating the fatty acid degradation of endothelial cells. Cardiovasc Diabetol. 2024;23(1):60.
[27] SUN T, DING W, XU T, et al. Parkin Regulates Programmed Necrosis and Myocardial Ischemia/Reperfusion Injury by Targeting Cyclophilin-D.Antioxid Redox Signal. 2019;31(16):1177-1193.
[28] ALGOET M, JANSSENS S, HIMMELREICH U, et al. Myocardial ischemia-reperfusion injury and the influence of inflammation. Trends Cardiovasc Med. 2023;33(6):357-366.
[29] ZHANG M, LIU Q, MENG H, et al. Ischemia-reperfusion injury: molecular mechanisms and therapeutic targets. Signal Transduct Target Ther. 2024; 9(1):12.
[30] WU F, HUANG W, TAN Q, et al. ZFP36L2 regulates myocardial ischemia/reperfusion injury and attenuates mitochondrial fusion and fission by LncRNA PVT1. Cell Death Dis. 2021;12(6):614.
[31] SÁNCHEZ-PÉREZ P, MATA A, TORP MK, et al. Energy substrate metabolism, mitochondrial structure and oxidative stress after cardiac ischemia-reperfusion in mice lacking UCP3. Free Radic Biol Med. 2023;205:244-261.
[32] LINDSEY ML, BRUNT KR, KIRK JA, et al. Guidelines for in vivo mouse models of myocardial infarction. Am J Physiol Heart Circ Physiol. 2021; 21(6):H1056-H1073.
[33] WANG L, QIU S, LI X, et al. Myocardial-Targeting Tannic Cerium Nanocatalyst Attenuates Ischemia/Reperfusion Injury. Angew Chem Int Ed Engl. 2023; 62(39):e202305576.
[34] ZHANG L, LIU Y, LI JY, et al. Protective Effect of Rosamultin against H(2)O(2)-Induced Oxidative Stress and Apoptosis in H9c2 Cardiomyocytes. Oxid Med Cell Longev. 2018;2018:8415610.
[35] YU C, KIM B S, KIM E. FAF1 mediates regulated necrosis through PARP1 activation upon oxidative stress leading to dopaminergic neurodegeneration. Cell Death Differ. 2016;23(11):1873-1885.
[36] JABBOUR E, KANTARJIAN H. Chronic myeloid leukemia: 2025 update on diagnosis, therapy, and monitoring. Am J Hematol. 2024;99(11):2191-2212.
[37] LEE H, SEO J, SHIN S, et al. Development and validation of sensitive BCR::ABL1 fusion gene quantitation using next-generation sequencing.Cancer Cell Int. 2023;23(1):106.
[38] HEFNI E, MENON D, MA T, et al. Angiopoietin-like 4 induces head and neck squamous cell carcinoma cell migration through the NRP1/ABL1/PXN pathway.Cell Signal. 2023;108:110697.
[39] JULIAN L, OLSON MF. Rho-associated coiled-coil containing kinases (ROCK): structure, regulation, and functions.Small GTPases. 2014;5:e29846.
[40] BALAJI RAGUNATHRAO VA, ANWAR M, AKHTER MZ, et al. Sphingosine-1-Phosphate Receptor 1 Activity Promotes Tumor Growth by Amplifying VEGF-VEGFR2 Angiogenic Signaling. Cell Rep. 2019;29(11):3472-3487.
[41] ZHOU X, CHENG J, CHEN Z, et al. Role of c-Abl in Ang II-induced aortic dissection formation: Potential regulatory efficacy on phenotypic transformation and apoptosis of VSMCs. Life Sci. 2020;256:117882.
[42] YAMANI A, WU D, WAGGONER L, et al. The vascular endothelial specific IL-4 receptor alpha-ABL1 kinase signaling axis regulates the severity of IgE-mediated anaphylactic reactions. J Allergy Clin Immunol. 2018;142(4):1159-1172.e5.
[43] QIU Z, CANG Y, GOFF SP. c-Abl tyrosine kinase regulates cardiac growth and development. Proc Natl Acad Sci U S A. 2010;107(3):1136-1141.
[44] GUO X, YIN H, LI L, et al. Cardioprotective Role of Tumor Necrosis Factor Receptor-Associated Factor 2 by Suppressing Apoptosis and Necroptosis. Circulation. 2017;136(8):729-742.
[45] GUPTA R, KUMARI S, TRIPATHI R, et al. Unwinding the modalities of necrosome activation and necroptosis machinery in neurological diseases. Ageing Res Rev. 2023;86:101855.
[46] DEL RE DP, AMGALAN D, LINKERMANN A, et al. Fundamental Mechanisms of Regulated Cell Death and Implications for Heart Disease. Physiol Rev. 2019;99(4):1765-1817.
[47] BONORA M, GIORGI C, PINTON P. Molecular mechanisms and consequences of mitochondrial permeability transition. Nat Rev Mol Cell Biol. 2022; 23(4):266-285.
[48] MENDOZA A, PATEL P, ROBICHAUX D, et al. Inhibition of the mPTP and Lipid Peroxidation Is Additively Protective Against I/R Injury. Circ Res. 2024;134(10):1292-1305.
[49] SAMANTA S, AKHTER F, ROY A, et al. New cyclophilin D inhibitor rescues mitochondrial and cognitive function in Alzheimer’s disease. Brain. 2024; 147(5):1710-1725.
[50] LU A, XU Z, ZHAO Z, et al. Double Braking Effects of Nanomedicine on Mitochondrial Permeability Transition Pore for Treating Idiopathic Pulmonary Fibrosis. Adv Sci (Weinh). 2024;11(47):e2405406.
[51] KUDO T, NAKAZAWA D, WATANABE-KUSUNOKI K, et al. Regulation of NETosis and Inflammation by Cyclophilin D in Myeloperoxidase-Positive Antineutrophil Cytoplasmic Antibody-Associated Vasculitis. Arthritis Rheumatol. 2023;75(1):71-83.
[52] HALECKOVA A, BENEK O, ZEMANOVÁ L, et al. Small-molecule inhibitors of cyclophilin D as potential therapeutics in mitochondria-related diseases. Med Res Rev. 2022;42(5):1822-1855.
[53] ZHOU C, KUANG M, TAO Y, et al. Nynrin preserves hematopoietic stem cell function by inhibiting the mitochondrial permeability transition pore opening. Cell Stem Cell. 2024;31(9):1359-1375.e8.
[54] DIKALOVA A, FEHRENBACH D, MAYOROV V, et al. Mitochondrial CypD Acetylation Promotes Endothelial Dysfunction and Hypertension. Circ Res. 2024;134(11):1451-1464.
[55] WANG K, AN T, ZHOU LY, et al. E2F1-regulated miR-30b suppresses Cyclophilin D and protects heart from ischemia/reperfusion injury and necrotic cell death.Cell Death Differ. 2015;22(5):743-754.
|