Chinese Journal of Tissue Engineering Research ›› 2018, Vol. 22 ›› Issue (7): 1072-1077.doi: 10.3969/j.issn.2095-4344.0119
Previous Articles Next Articles
Effects of different doses of microwave therapy on adjacent tissue of titanium alloy implants after fracture surgery
Wang Gang, Xu Yi-ming, Ye Dong-mei, Fu Teng-fei, Zou Yu-zhen, Feng Xian-xuan, Bai Yue-hong
- Department of Rehabilitation, Affiliated Sixth People’s Hospital of Shanghai Jiao Tong University, Shanghai 200233, China
-
Online:2018-03-08Published:2018-03-08 -
Contact:Bai Yue-hong, M.D., Professor, Doctoral supervisor, Department of Rehabilitation, Affiliated Sixth People’s Hospital of Shanghai Jiao Tong University, Shanghai 200233, China -
About author:Wang Gang, M.D., Attending physician, Department of Rehabilitation, Affiliated Sixth People’s Hospital of Shanghai Jiao Tong University, Shanghai 200233, China -
Supported by:the Scientific Research Program of Science and Technology Commission of Shanghai City, No. 13231202600
CLC Number:
Cite this article
Wang Gang, Xu Yi-ming, Ye Dong-mei, Fu Teng-fei, Zou Yu-zhen, Feng Xian-xuan, Bai Yue-hong. Effects of different doses of microwave therapy on adjacent tissue of titanium alloy implants after fracture surgery[J]. Chinese Journal of Tissue Engineering Research, 2018, 22(7): 1072-1077.
share this article
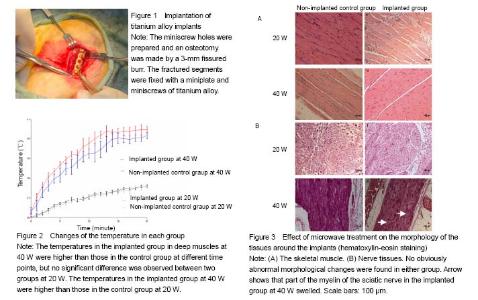
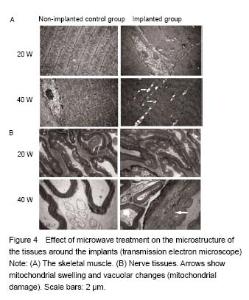
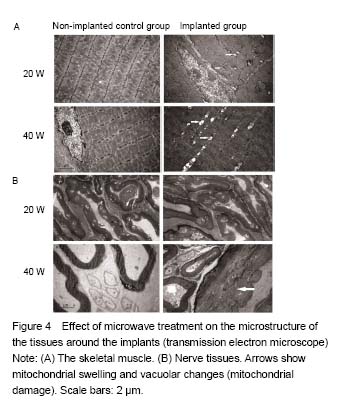
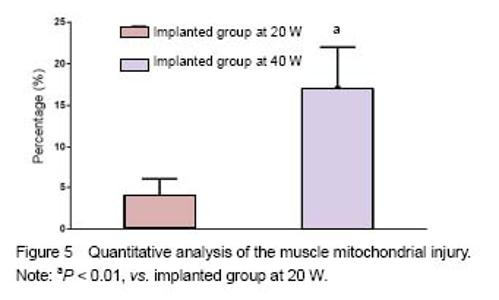
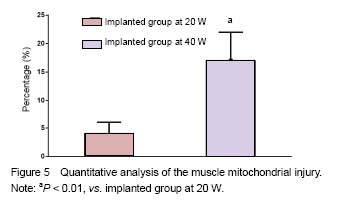
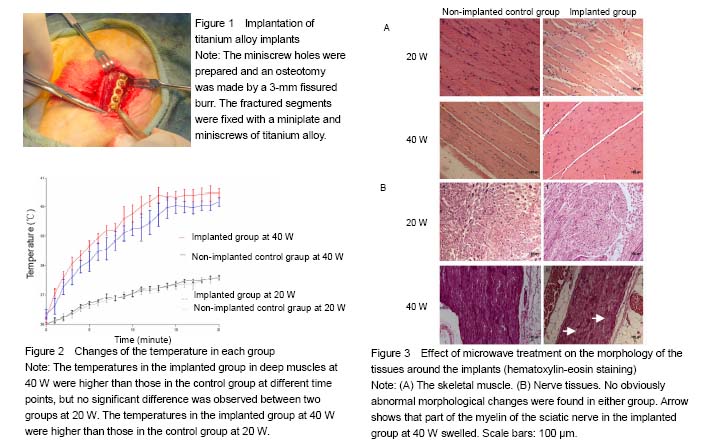
Quantitative analysis of animals None rabbits in the study dropped out, and all were involved in the result analysis. Temperature changes During microwave irradiation, we simultaneously measured the temperatures in deep muscle tissues. Temperature changes are shown in Figure 2. It was found that the temperature in the implanted group at 40 W was higher than that in the implanted group at 20 W (P < 0.000 1). In addition, the temperature in the implanted group at 40 W was higher than that in the control group at 40 W (P = 0.037 3). However, there was no significant difference between control and the implanted groups at 20 W (P = 0.547 9). The peak temperature of the deep muscles in the implanted group at 20 W was between 37.5 ℃ and 37.7 ℃. The peak temperature of the deep muscles in the implanted group at 40 W was between 40.3 ℃ and 40.6 ℃. Light microscopy Sections of the thigh muscle in each group were stained with hematoxylin and eosin after 30 days of microwave treatment. No abnormal morphological changes were found either in the control or implanted groups. Sections of sciatic nerve from each group were also stained with hematoxylin and eosin after the 30-day microwave treatment. The myelin sheaths of the sciatic nerves in the implanted group at 40 W were found to be swollen (Figure 3). Transmission electron microscopy Transmission electron microscope examinations of the skeletal muscle and sciatic nerves in each group were conducted after the 30-day microwave treatment. The muscles in the implanted group at 20 and 40 W showed mitochondrial swelling and vacuolization (mitochondrial damage). Myofilaments and myocommata were unclear. The myelin sheath of the sciatic nerves adjacent to the fractures at 20 W was normal. Part of the myelin sheath of myelinated nerve fibers showed lamellar separation, and ball-like changes in the pith were also observed. The mitochondria in the axons of the implanted group at 40 W were normal (Figure 4). No abnormal morphological changes in the sciatic nerve were discovered in the control group or the implanted group at 20 W. A KS400 image analysis system (Zeiss) was applied to calculate the percentage of the damaged mitochondrial area in each sample, to allow the quantitative analysis of mitochondrial damage in the muscle between two implanted groups, and found significant differences (Figure 5). The percentage of the damaged myelin sheaths of sciatic nerves adjacent to the fractures in the implanted group at 40 W was (31% ± 5%), while the myelin sheaths of sciatic nerves adjacent to the fractures in the implanted group at 20 W were normal. Thus, muscle and nerve injuries in the implanted group at 40 W were more serious than those at 20 W."
| [1] Perren SM. Fracture healing. The evolution of our understanding. Acta Chir Orthop Traumatol Cech. 2008;75(4):241-246.[2] Giombini A, Giovannini V, Di Cesare A, et al. Hyperthermia induced by microwave diathermy in the management of muscle and tendon injuries. Br Med Bull. 2007;83:379-396.[3] Schwan HP, Piersol GM. The absorption of electromagnetic energy in body tissues. Am J Phys Med. 1954;33(6):371-404.[4] Giovanella BC, Mondovi B. Selective heat sensitivity of cancer cells: Introduction. Recent Results Cancer Res. 1977;(59):1-6.[5] Lehmann JF, Guy AW, Stonebridge JB, et al. Temperature distribution produced in models by three microwave applicators at 433.92 megahertz. Arch Phys Med Rehabil. 1975;56(4):145-151.[6] deLateur BJ, Stonebridge JB, Lehmann JF. Fibrous muscular contractures: treatment with a new direct contact microwave applicator operating at 915 MHz. Arch Phys Med Rehabil. 1978;59(10):488-499.[7] Goats GC. Microwave diathermy. Br J Sports Med. 1990;24(4):212-218.[8] Chang WH, Sun JS, Chang SP, et al. Study of thermal effects of ultrasound stimulation on fracture healing. Bioelectromagnetics. 2002;23(4):256-263.[9] Leon SA, Asbell SO, Edelstein G, et al. Effects of hyperthermia on bone. I. Heating rate patterns induced by microwave irradiation in bone and muscle phantoms. Int J Hyperthermia. 1993;9(1):69-75.[10] Lubner MG, Brace CL, Hinshaw JL, et al. Microwave tumor ablation: mechanism of action, clinical results, and devices. J Vasc Interv Radiol. 2010;21(8 Suppl):S192-203.[11] Shields N, Gormley J, O'Hare N. Short-wave diathermy: current clinical and safety practices. Physiother Res Int. 2002;7(4):191-202.[12] Guy AW. Dosimetry associated with exposure to non-ionizing radiation: very low frequency to microwaves. Health Phys. 1987;53(6):569-584.[13] Ruggera PS, Witters DM, von Maltzahn G, et al. In vitro assessment of tissue heating near metallic medical implants by exposure to pulsed radio frequency diathermy. Phys Med Biol. 2003;48(17):2919-2928.[14] Virtanen H, Keshvari J, Lappalainen R. The effect of authentic metallic implants on the SAR distribution of the head exposed to 900, 1800 and 2450 MHz dipole near field. Phys Med Biol. 2007;52(5):1221-1236. [15] McIntosh RL, Anderson V, McKenzie RJ. A numerical evaluation of SAR distribution and temperature changes around a metallic plate in the head of a RF exposed worker. Bioelectromagnetics. 2005;26(5):377-388.[16] Virtanen H, Huttunen J, Toropainen A, et al. Interaction of mobile phones with superficial passive metallic implants. Phys Med Biol. 2005;50(11):2689-2700.[17] Seiger C, Draper DO. Use of pulsed shortwave diathermy and joint mobilization to increase ankle range of motion in the presence of surgical implanted metal: A case series. J Orthop Sports Phys Ther. 2006;36(9):669-677.[18] Wen CE, Yamada Y, Shimojima K, et al. Processing and mechanical properties of autogenous titanium implant materials. J Mater Sci Mater Med. 2002;13(4):397-401.[19] Muranaka H, Horiguchi T, Ueda Y, et al. Evaluation of RF heating on hip joint implant in phantom during MRI examinations. Nihon Hoshasen Gijutsu Gakkai Zasshi. 2010;66(7):725-733.[20] Huidobro C, Larson B, Mynderse S, et al. Characterizing Prostiva RF treatments of the prostate for BPH with gadolinium-enhanced MRI. ScientificWorldJournal. 2009;9:10-16.[21] Ye D, Xu Y, Zhang H, et al. Effects of low-dose microwave on healing of fractures with titanium alloy internal fixation: an experimental study in a rabbit model. PLoS One. 2013;8(9):e75756.[22] Danilov RK. Ultrastructure of skeletal muscle tissue of microwave damaged chick embryos. Arkh Anat Gistol Embriol. 1980;78(1):83-88.[23] Dwivedi RS, Dwivedi U, Chiang B. Low intensity microwave radiation effects on the ultrastructure of Chang liver cells. Exp Cell Res. 1989;180(1):253-265.[24] Zhao L, Peng RY, Wang SM, et al. Relationship between cognition function and hippocampus structure after long-term microwave exposure. Biomed Environ Sci. 2012;25(2):182-188.[25] Mileva K, Georgieva B, Radicheva N. About the biological effects of high and extremely high frequency electromagnetic fields. Acta Physiol Pharmacol Bulg. 2003;27(2-3):89-100.[26] Green DR, Reed JC. Mitochondria and apoptosis. Science. 1998;281(5381):1309-1312.[27] Mehdizadeh M, Kermanian F, Farjah G. Schwann cell injuries of radial nerve after lead (Pb) exposure in rats. Pathophysiology. 2008;15(1):13-17.[28] Barroso-Moguel R, Villeda Hernández J. Experimental neuropathy produced in rats with industrial solvents (thinner). Arch Invest Med (Mex). 1989;20(1):53-60.[29] Can B, Saray A, Caglikulekçi M, et al. Effects of obstructive jaundice on the peripheral nerve: an ultrastructural study in rats. Eur Surg Res. 2004;36(4):226-233.[30] Berry EM. Dietary fatty acids in the management of diabetes mellitus. Am J Clin Nutr. 1997;66(4 Suppl):991S-997S.[31] Chen Y, Yi Q, Liu G, et al. Cerebral white matter injury and damage to myelin sheath following whole-brain ischemia. Brain Res. 2013;1495:11-17.[32] Hoogeveen JF, Troost D, Wondergem J, et al. Hyperthermic injury versus crush injury in the rat sciatic nerve: a comparative functional, histopathological and morphometrical study. J Neurol Sci. 1992;108(1):55-64.[33] Vujaskovic Z, McChesney Gillette S, Powers BE, et al. Effects of intraoperative hyperthermia on canine sciatic nerve: histopathologic and morphometric studies. Int J Hyperthermia. 1994;10(6):845-855.[34] Dewey WC. Arrhenius relationships from the molecule and cell to the clinic. Int J Hyperthermia. 2009;25(1):3-20.[35] Ginzburg EL, Mashanski? VF, Krylenkov VA, et al. Correlation of mitochondrial adenosine triphosphatase activity with their size following exposure of cells to higher temperatures. Tsitologiia. 1976;18(5):600-604.[36] Funk RH, Nagel F, Wonka F, et al. Effects of heat shock on the functional morphology of cell organelles observed by video-enhanced microscopy. Anat Rec. 1999;255(4):458-464. |
| [1] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [2] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [3] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [4] | Li Xingping, Xiao Dongqin, Zhao Qiao, Chen Shuo, Bai Yiguang, Liu Kang, Feng Gang, Duan Ke. Preparation and properties of copper-loaded antibacterial functional film on titanium surface [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 553-557. |
| [5] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [6] | Shi Xiaoxiu, Mao Shilong, Liu Yang, Ma Xingshuang, Luo Yanfeng. Comparison of tantalum and titanium (alloy) as orthopedic materials: physical and chemical indexes, antibacterial and osteogenic ability [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 593-599. |
| [7] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| [8] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [9] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| [10] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| [11] | Chen Jiana, Qiu Yanling, Nie Minhai, Liu Xuqian. Tissue engineering scaffolds in repairing oral and maxillofacial soft tissue defects [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 644-650. |
| [12] | Xing Hao, Zhang Yonghong, Wang Dong. Advantages and disadvantages of repairing large-segment bone defect [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 426-430. |
| [13] | Chen Siqi, Xian Debin, Xu Rongsheng, Qin Zhongjie, Zhang Lei, Xia Delin. Effects of bone marrow mesenchymal stem cells and human umbilical vein endothelial cells combined with hydroxyapatite-tricalcium phosphate scaffolds on early angiogenesis in skull defect repair in rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3458-3465. |
| [14] | Wang Hao, Chen Mingxue, Li Junkang, Luo Xujiang, Peng Liqing, Li Huo, Huang Bo, Tian Guangzhao, Liu Shuyun, Sui Xiang, Huang Jingxiang, Guo Quanyi, Lu Xiaobo. Decellularized porcine skin matrix for tissue-engineered meniscus scaffold [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3473-3478. |
| [15] | Mo Jianling, He Shaoru, Feng Bowen, Jian Minqiao, Zhang Xiaohui, Liu Caisheng, Liang Yijing, Liu Yumei, Chen Liang, Zhou Haiyu, Liu Yanhui. Forming prevascularized cell sheets and the expression of angiogenesis-related factors [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3479-3486. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||